Abstract
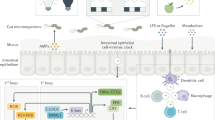
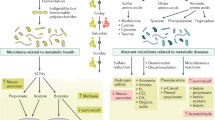
The metabolic syndrome is prevalent in developed nations and accounts for the largest burden of non-communicable diseases worldwide. The metabolic syndrome has direct effects on health and increases the risk of developing cancer. Lifestyle factors that are known to promote the metabolic syndrome generally cause pro-inflammatory alterations in microbiota communities in the intestine. Indeed, alterations to the structure and function of intestinal microbiota are sufficient to promote the metabolic syndrome, inflammation and cancer. Among the lifestyle factors that are associated with the metabolic syndrome, disruption of the circadian system, known as circadian dysrhythmia, is increasingly common. Disruption of the circadian system can alter microbiome communities and can perturb host metabolism, energy homeostasis and inflammatory pathways, which leads to the metabolic syndrome. This Perspective discusses the role of intestinal microbiota and microbial metabolites in mediating the effects of disruption of circadian rhythms on human health.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
World Health Organization. Global Health Estimates 2016: Disease burden by Cause, Age, Sex, by Country and by Region, 2000–2016 (WHO, 2018).
Scholze, J. et al. Epidemiological and economic burden of metabolic syndrome and its consequences in patients with hypertension in Germany, Spain and Italy; a prevalence-based model. BMC Public. Health 10, 529 (2010).
Pothiwala, P., Jain, S. K. & Yaturu, S. Metabolic syndrome and cancer. Metab. Syndr. Relat. Disord. 7, 279–288 (2009).
Feigin, V. L. et al. Global burden of stroke and risk factors in 188 countries, during 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet Neurol. 15, 913–924 (2016).
Ogden, C. L., Carroll, M. D., Fryar, C. D. & Flegal, K. M. Prevalence of obesity among adults and youth: United States, 2011–2014. NCHS Data Brief 219, 1–8 (2015).
Misra, A. & Khurana, L. Obesity and the metabolic syndrome in developing countries. J. Clin. Endocrinol. Metab. 93, S9–S30 (2008).
Moreira, G. C., Cipullo, J. P., Ciorlia, L. A., Cesarino, C. B. & Vilela-Martin, J. F. Prevalence of metabolic syndrome: association with risk factors and cardiovascular complications in an urban population. PLoS One 9, e105056 (2014).
Zhang, R., Lahens, N. F., Ballance, H. I., Hughes, M. E. & Hogenesch, J. B. A circadian gene expression atlas in mammals: implications for biology and medicine. Proc. Natl Acad. Sci. USA 111, 16219–16224 (2014).
Mohawk, J. A., Green, C. B. & Takahashi, J. S. Central and peripheral circadian clocks in mammals. Annu. Rev. Neurosci. 35, 445–462 (2012).
Stokkan, K. A., Yamazaki, S., Tei, H., Sakaki, Y. & Menaker, M. Entrainment of the circadian clock in the liver by feeding. Science. 291, 490–493 (2001).
Hoogerwerf, W. A. et al. Clock gene expression in the murine gastrointestinal tract: endogenous rhythmicity and effects of a feeding regimen. Gastroenterology 133, 1250–1260 (2007).
Bishehsari, F., Levi, F., Turek, F. W. & Keshavarzian, A. Circadian rhythms in gastrointestinal health and diseases. Gastroenterology 151, e1–e5 (2016).
Touitou, Y., Touitou, D. & Reinberg, A. Disruption of adolescents’ circadian clock: the vicious circle of media use, exposure to light at night, sleep loss and risk behaviors. J. Physiol. Paris 110, 467–479 (2016).
Mota, M. C., Silva, C. M., Balieiro, L. C. T., Fahmy, W. M. & Crispim, C. A. Social jetlag and metabolic control in non-communicable chronic diseases: a study addressing different obesity statuses. Sci. Rep. 7, 6358 (2017).
Kervezee, L., Kosmadopoulos, A. & Boivin, D. B. Metabolic and cardiovascular consequences of shift work: the role of circadian disruption and sleep disturbances. Eur. J. Neurosci. 51, 396–412 (2020).
Moossavi, S. & Bishehsari, F. Microbes: possible link between modern lifestyle transition and the rise of metabolic syndrome. Obes. Rev. 20, 407–419 (2019).
Bass, J. & Takahashi, J. S. Circadian integration of metabolism and energetics. Science 330, 1349–1354 (2010).
Panda, S. et al. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell 109, 307–320 (2002).
Rudic, R. D. et al. BMAL1 and CLOCK, two essential components of the circadian clock, are involved in glucose homeostasis. PLoS Biol. 2, e377 (2004).
Marcheva, B. et al. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature 466, 627–631 (2010).
Turek, F. W. et al. Obesity and metabolic syndrome in circadian clock mutant mice. Science 308, 1043–1045 (2005).
Cho, H. et al. Regulation of circadian behaviour and metabolism by REV-ERB-α and REV-ERB-β. Nature 485, 123–127 (2012).
Gale, J. E. et al. Disruption of circadian rhythms accelerates development of diabetes through pancreatic beta-cell loss and dysfunction. J. Biol. Rhythm. 26, 423–433 (2011).
Qian, J., Block, G. D., Colwell, C. S. & Matveyenko, A. V. Consequences of exposure to light at night on the pancreatic islet circadian clock and function in rats. Diabetes 62, 3469–3478 (2013).
Pappa, K. I. et al. Circadian clock gene expression is impaired in gestational diabetes mellitus. Gynecol. Endocrinol. 29, 331–335 (2013).
Garcia-Rios, A. et al. Beneficial effect of CLOCK gene polymorphism rs1801260 in combination with low-fat diet on insulin metabolism in the patients with metabolic syndrome. Chronobiol. Int. 31, 401–408 (2014).
Bracci, M. et al. Rotating-shift nurses after a day off: peripheral clock gene expression, urinary melatonin, and serum 17-β-estradiol levels. Scand. J. Work. Env. Health 40, 295–304 (2014).
Wehrens, S. M. T. et al. Meal timing regulates the human circadian system. Curr. Biol. 27, 1768–1775 (2017).
Leproult, R., Holmback, U. & Van, C. E. Circadian misalignment augments markers of insulin resistance and inflammation, independently of sleep loss. Diabetes 63, 1860–1869 (2014).
Morikawa, Y. et al. Effect of shift work on body mass index and metabolic parameters. Scand. J. Work. Env. Health 33, 45–50 (2007).
Bo, S. et al. Consuming more of daily caloric intake at dinner predisposes to obesity. A 6-year population-based prospective cohort study. PLoS ONE 9, e108467 (2014).
McHill, A. W. et al. Caloric and macronutrient intake differ with circadian phase and between lean and overweight young adults. Nutrients 11, 587 (2019).
McHill, A. W. et al. Later circadian timing of food intake is associated with increased body fat. Am. J. Clin. Nutr. 106, 1213–1219 (2017).
McMullan, C. J., Curhan, G. C., Schernhammer, E. S. & Forman, J. P. Association of nocturnal melatonin secretion with insulin resistance in nondiabetic young women. Am. J. Epidemiol. 178, 231–238 (2013).
Bass, J. Circadian topology of metabolism. Nature 491, 348–356 (2012).
Panda, S. Circadian physiology of metabolism. Science 354, 1008–1015 (2016).
Faria, J. A. et al. Melatonin acts through MT1/MT2 receptors to activate hypothalamic Akt and suppress hepatic gluconeogenesis in rats. Am. J. Physiol. Endocrinol. Metab. 305, E230–E242 (2013).
Bazwinsky-Wutschke, I., Wolgast, S., Muhlbauer, E., Albrecht, E. & Peschke, E. Phosphorylation of cyclic AMP-response element-binding protein (CREB) is influenced by melatonin treatment in pancreatic rat insulinoma β-cells (INS-1). J. Pineal Res. 53, 344–357 (2012).
Bonnefond, A. et al. Rare MTNR1B variants impairing melatonin receptor 1B function contribute to type 2 diabetes. Nat. Genet. 44, 297–301 (2012).
Masri, S. Sirtuin-dependent clock control: new advances in metabolism, aging and cancer. Curr. Opin. Clin. Nutr. Metab. Care 18, 521–527 (2015).
Global Burden of Disease Cancer Collaboration. et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2016: a systematic analysis for the global burden of disease study. JAMA Oncol. 4, 1553–1568 (2018).
Bishehsari, F., Mahdavinia, M., Vacca, M., Malekzadeh, R. & Mariani-Costantini, R. Epidemiological transition of colorectal cancer in developing countries: environmental factors, molecular pathways, and opportunities for prevention. World J. Gastroenterol. 20, 6055–6072 (2014).
Feng, R. M., Zong, Y. N., Cao, S. M. & Xu, R. H. Current cancer situation in China: good or bad news from the 2018 global cancer statistics? Cancer Commun. 39, 22 (2019).
Renehan, A. G., Tyson, M., Egger, M., Heller, R. F. & Zwahlen, M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet 371, 569–578 (2008).
Schernhammer, E. S. et al. Night-shift work and risk of colorectal cancer in the Nurses’ Health Study. J. Natl Cancer Inst. 95, 825–828 (2003).
Carter, B. D., Diver, W. R., Hildebrand, J. S., Patel, A. V. & Gapstur, S. M. Circadian disruption and fatal ovarian cancer. Am. J. Prev. Med. 46, S34–S41 (2014).
Altman, B. J. Cancer clocks out for lunch: disruption of circadian rhythm and metabolic oscillation in cancer. Front. Cell Dev. Biol. 4, 62 (2016).
Kettner, N. M. et al. Circadian homeostasis of liver metabolism suppresses hepatocarcinogenesis. Cancer Cell 30, 909–924 (2016).
Bishehsari, F. et al. Light/dark shifting promotes alcohol-induced colon carcinogenesis: possible role of intestinal inflammatory milieu and microbiota. Int. J. Mol. Sci. 17, 2017 (2016).
Bishehsari, F. et al. Abnormal eating patterns cause circadian disruption and promote alcohol-associated colon carcinogenesis. Cell. Mol. Gastroenterol. Hepatol. 9, 219–237 (2020).
Masri, S., Kinouchi, K. & Sassone-Corsi, P. Circadian clocks, epigenetics, and cancer. Curr. Opin. Oncol. 27, 50–56 (2015).
Sulli, G. et al. Pharmacological activation of REV-ERBs is lethal in cancer and oncogene-induced senescence. Nature 553, 351–355 (2018).
Papagiannakopoulos, T. et al. Circadian rhythm disruption promotes lung tumorigenesis. Cell Metab. 24, 324–331 (2016).
Altman, B. J. et al. MYC disrupts the circadian clock and metabolism in cancer cells. Cell Metab. 22, 1009–1019 (2015).
Sonnenburg, J. L. & Sonnenburg, E. D. Vulnerability of the industrialized microbiota. Science 366, eaaw9255 (2019).
Bishehsari, F. & Keshavarzian, A. Microbes help to track time. Science 365, 1379–1380 (2019).
Furuya, S. & Yugari, Y. Daily rhythmic change of L-histidine and glucose absorptions in rat small intestine in vivo. Biochim. Biophys. Acta 343, 558–564 (1974).
Zarrinpar, A., Chaix, A., Yooseph, S. & Panda, S. Diet and feeding pattern affect the diurnal dynamics of the gut microbiome. Cell Metab. 20, 1006–1017 (2014).
Thaiss, C. A. et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell 159, 514–529 (2014).
Paulose, J. K., Wright, J. M., Patel, A. G. & Cassone, V. M. Human gut bacteria are sensitive to melatonin and express endogenous circadian rhythmicity. PLoS ONE 11, e0146643 (2016).
Thaiss, C. A. et al. Microbiota diurnal rhythmicity programs host transcriptome oscillations. Cell 167, 1495–1510 (2016).
Paulose, J. K., Cassone, C. V., Graniczkowska, K. B. & Cassone, V. M. Entrainment of the circadian clock of the enteric bacterium Klebsiella aerogenes by temperature cycles. iScience 19, 1202–1213 (2019).
Liang, X., Bushman, F. D. & FitzGerald, G. A. Rhythmicity of the intestinal microbiota is regulated by gender and the host circadian clock. Proc. Natl Acad. Sci. USA 112, 10479–10484 (2015).
Chaix, A., Zarrinpar, A., Miu, P. & Panda, S. Time-restricted feeding is a preventative and therapeutic intervention against diverse nutritional challenges. Cell Metab. 20, 991–1005 (2014).
Chaix, A., Lin, T., Le, H. D., Chang, M. W. & Panda, S. Time-restricted feeding prevents obesity and metabolic syndrome in mice lacking a circadian clock. Cell Metab. 29, 303–319 (2019).
Nobs, S. P., Tuganbaev, T. & Elinav, E. Microbiome diurnal rhythmicity and its impact on host physiology and disease risk. EMBO Rep. 20, e47129 (2019).
Tahara, Y. et al. Gut microbiota-derived short chain fatty acids induce circadian clock entrainment in mouse peripheral tissue. Sci. Rep. 8, 1395 (2018).
Leone, V. et al. Effects of diurnal variation of gut microbes and high-fat feeding on host circadian clock function and metabolism. Cell Host Microbe 17, 681–689 (2015).
Parkar, S. G., Kalsbeek, A. & Cheeseman, J. F. Potential role for the gut microbiota in modulating host circadian rhythms and metabolic health. Microorganisms 7, 41 (2019).
Kuang, Z. et al. The intestinal microbiota programs diurnal rhythms in host metabolism through histone deacetylase 3. Science 365, 1428–1434 (2019).
Wang, Z. et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 472, 57–63 (2011).
Tang, W. H. et al. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 368, 1575–1584 (2013).
Wu, X. et al. Regulation of circadian rhythms by NEAT1 mediated TMAO-induced endothelial proliferation: a protective role of asparagus extract. Exp. Cell Res. 382, 111451 (2019).
Ley, R. E. et al. Obesity alters gut microbial ecology. Proc. Natl Acad. Sci. USA 102, 11070–11075 (2005).
Backhed, F. et al. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl Acad. Sci. USA 101, 15718–15723 (2004).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Verdam, F. J. et al. Human intestinal microbiota composition is associated with local and systemic inflammation in obesity. Obesity 21, E607–E615 (2013).
Karlsson, F. H. et al. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature 498, 99–103 (2013).
Fei, N. & Zhao, L. An opportunistic pathogen isolated from the gut of an obese human causes obesity in germfree mice. ISME J. 7, 880–884 (2013).
Cortes-Martin, A., Iglesias-Aguirre, C. E., Meoro, A., Selma, M. V. & Espin, J. C. There is no distinctive gut microbiota signature in the metabolic syndrome: contribution of cardiovascular disease risk factors and associated medication. Microorganisms 8, 416 (2020).
Liou, A. P. et al. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci. Transl Med. 5, 178ra41 (2013).
Vrieze, A. et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology 143, 913–916 (2012).
Kootte, R. S. et al. Improvement of insulin sensitivity after lean donor feces in metabolic syndrome is driven by baseline intestinal microbiota composition. Cell Metab. 26, 611–619 (2017).
Fujisaka, S. et al. Antibiotic effects on gut microbiota and metabolism are host dependent. J. Clin. Invest. 126, 4430–4443 (2016).
Janssen, A. W. & Kersten, S. The role of the gut microbiota in metabolic health. FASEB J. 29, 3111–3123 (2015).
Zhang, X. et al. Human gut microbiota changes reveal the progression of glucose intolerance. PLoS ONE 8, e71108 (2013).
Yassour, M. et al. Sub-clinical detection of gut microbial biomarkers of obesity and type 2 diabetes. Genome Med. 8, 17 (2016).
Li, J. et al. Gut microbiota dysbiosis contributes to the development of hypertension. Microbiome 5, 14 (2017).
Depommier, C. et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat. Med. 25, 1096–1103 (2019).
Plovier, H. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 23, 107–113 (2017).
Voigt, R. M. et al. Circadian disorganization alters intestinal microbiota. PLoS ONE 9, e97500 (2014).
Rosselot, A. E., Hong, C. I. & Moore, S. R. Rhythm and bugs: circadian clocks, gut microbiota, and enteric infections. Curr. Opin. Gastroenterol. 32, 7–11 (2016).
Voigt, R. M., Forsyth, C. B. & Keshavarzian, A. Circadian disruption: potential implications in inflammatory and metabolic diseases associated with alcohol. Alcohol Res. Curr. Rev. 35, 87–96 (2013).
Voigt, R. M. et al. The circadian clock mutation promotes intestinal dysbiosis. Alcohol. Clin. Exp. Res. 40, 335–347 (2016).
Ramanan, D. & Cadwell, K. Intrinsic defense mechanisms of the intestinal epithelium. Cell Host Microbe 19, 434–441 (2016).
Henao-Mejia, J. et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 482, 179–185 (2012).
Purohit, J. S. et al. The effects of NOD activation on adipocyte differentiation. Obesity 21, 737–747 (2013).
Tremaroli, V. & Backhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 489, 242–249 (2012).
Mukherji, A., Kobiita, A., Ye, T. & Chambon, P. Homeostasis in intestinal epithelium is orchestrated by the circadian clock and microbiota cues transduced by TLRs. Cell 153, 812–827 (2013).
Rogero, M. M. & Calder, P. C. Obesity, inflammation, toll-like receptor 4 and fatty acids. Nutrients 10, 432 (2018).
Semlali, A. et al. Expression and polymorphism of toll-like receptor 4 and effect on NF-κB mediated inflammation in colon cancer patients. PLoS ONE 11, e0146333 (2016).
Himes, R. W. & Smith, C. W. Tlr2 is critical for diet-induced metabolic syndrome in a murine model. FASEB J. 24, 731–739 (2010).
Kim, F. et al. Toll-like receptor-4 mediates vascular inflammation and insulin resistance in diet-induced obesity. Circ. Res. 100, 1589–1596 (2007).
Saberi, M. et al. Hematopoietic cell-specific deletion of toll-like receptor 4 ameliorates hepatic and adipose tissue insulin resistance in high-fat-fed mice. Cell Metab. 10, 419–429 (2009).
Vijay-Kumar, M. et al. Metabolic syndrome and altered gut microbiota in mice lacking Toll-like receptor 5. Science 328, 228–231 (2010).
Ahmad, R. et al. Elevated expression of the toll like receptors 2 and 4 in obese individuals: its significance for obesity-induced inflammation. J. Inflamm. 9, 48 (2012).
Masters, S. L. et al. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nat. Immunol. 11, 897–904 (2010).
Vandanmagsar, B. et al. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 17, 179–188 (2011).
Byrne, C. S., Chambers, E. S., Morrison, D. J. & Frost, G. The role of short chain fatty acids in appetite regulation and energy homeostasis. Int. J. Obes. 39, 1331–1338 (2015).
Ganapathy, V., Thangaraju, M., Prasad, P. D., Martin, P. M. & Singh, N. Transporters and receptors for short-chain fatty acids as the molecular link between colonic bacteria and the host. Curr. Opin. Pharmacol. 13, 869–874 (2013).
Tan, J. et al. The role of short-chain fatty acids in health and disease. Adv. Immunol. 121, 91–119 (2014).
Frost, G. et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 5, 3611 (2014).
Hong, Y. H. et al. Acetate and propionate short chain fatty acids stimulate adipogenesis via GPCR43. Endocrinology 146, 5092–5099 (2005).
Zaibi, M. S. et al. Roles of GPCR41 and GPCR43 in leptin secretory responses of murine adipocytes to short chain fatty acids. FEBS Lett. 584, 2381–2386 (2010).
Teichman, E. M., O’Riordan, K. J., Gahan, C. G. M., Dinan, T. G. & Cryan, J. F. When rhythms meet the blues: circadian interactions with the microbiota-gut-brain axis. Cell Metab. 31, 448–471 (2020).
Gao, Z. et al. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 58, 1509–1517 (2009).
Bellahcene, M. et al. Male mice that lack the G-protein-coupled receptor GPCR41 have low energy expenditure and increased body fat content. Br. J. Nutr. 109, 1755–1764 (2013).
Kimura, I. et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPCR43. Nat. Commun. 4, 1829 (2013).
Yaribeygi, H., Sathyapalan, T. & Sahebkar, A. Molecular mechanisms by which GLP-1 RA and DPP-4i induce insulin sensitivity. Life Sci. 234, 116776 (2019).
Takiishi, T., Fenero, C. I. M. & Camara, N. O. S. Intestinal barrier and gut microbiota: shaping our immune responses throughout life. Tissue Barriers 5, e1373208 (2017).
Hotamisligil, G. S. Inflammation, metaflammation and immunometabolic disorders. Nature 542, 177–185 (2017).
Fechner, A., Kiehntopf, M. & Jahreis, G. The formation of short-chain fatty acids is positively associated with the blood lipid-lowering effect of lupin kernel fiber in moderately hypercholesterolemic adults. J. Nutr. 144, 599–607 (2014).
Talati, R., Baker, W. L., Pabilonia, M. S., White, C. M. & Coleman, C. I. The effects of barley-derived soluble fiber on serum lipids. Ann. Fam. Med. 7, 157–163 (2009).
Ferrell, J. M. & Chiang, J. Y. Short-term circadian disruption impairs bile acid and lipid homeostasis in mice. Cell. Mol. Gastroenterol. Hepatol. 1, 664–677 (2015).
Zhai, H. et al. Takeda G protein-coupled receptor 5-mechanistic target of rapamycin complex 1 signaling contributes to the increment of glucagon-like peptide-1 production after Roux-en-Y gastric bypass. EBioMedicine 32, 201–214 (2018).
Pathak, P. et al. Intestine farnesoid X receptor agonist and the gut microbiota activate G-protein bile acid receptor-1 signaling to improve metabolism. Hepatology 68, 1574–1588 (2018).
Chavez-Talavera, O., Tailleux, A., Lefebvre, P. & Staels, B. Bile acid control of metabolism and inflammation in obesity, type 2 diabetes, dyslipidemia, and nonalcoholic fatty liver disease. Gastroenterology 152, 1679–1694 (2017).
Albaugh, V. L. et al. Role of bile acids and GLP-1 in mediating the metabolic improvements of bariatric surgery. Gastroenterology 156, 1041–1051 (2019).
Tang, G., Zhang, L., Yang, G., Wu, L. & Wang, R. Hydrogen sulfide-induced inhibition of L-type Ca2+ channels and insulin secretion in mouse pancreatic beta cells. Diabetologia 56, 533–541 (2013).
Szabo, C. Roles of hydrogen sulfide in the pathogenesis of diabetes mellitus and its complications. Antioxid. Redox Signal. 17, 68–80 (2012).
Moore, S. R. et al. Robust circadian rhythms in organoid cultures from PERIOD2::LUCIFERASE mouse small intestine. Dis. Model. Mech. 7, 1123–1130 (2014).
Jovel, J. et al. Characterization of the gut microbiome using 16S or shotgun metagenomics. Front. Microbiol. 7, 459 (2016).
Heintz-Buschart, A. & Wilmes, P. Human gut microbiome: function matters. Trends Microbiol. 26, 563–574 (2018).
Wang, Y. et al. The intestinal microbiota regulates body composition through NFIL3 and the circadian clock. Science 357, 912–916 (2017).
Nguyen, T. L., Vieira-Silva, S., Liston, A. & Raes, J. How informative is the mouse for human gut microbiota research? Dis. Model. Mech. 8, 1–16 (2015).
Vargason, A. M. & Anselmo, A. C. Clinical translation of microbe-based therapies: current clinical landscape and preclinical outlook. Bioeng. Transl. Med. 3, 124–137 (2018).
Baydoun, M. et al. An interphase microfluidic culture system for the study of ex vivo intestinal tissue. Micromachines 11, 150 (2020).
Brouwer, A. et al. Light therapy for better mood and insulin sensitivity in patients with major depression and type 2 diabetes: a randomised, double-blind, parallel-arm trial. BMC Psychiatry 15, 169 (2015).
Brouwer, A. et al. Effects of light therapy on mood and insulin sensitivity in patients with type 2 diabetes and depression: results from a randomized placebo-controlled trial. Diabetes Care 42, 529–538 (2019).
Acknowledgements
The authors acknowledge the support provided by NIAAA AA025387 and Rush Translational Sciences Consortium/Swim Across America Organization to F.B., NIAAA AA026801 to A.K. and R.M.V., NIAAA AA023417 and AA026801 to A.K, and NIA AG056653 to R.M.V.. The authors are grateful for the support of the Brinson Foundation, Barbara and Larry Field, Ellen and Philip Glass, and Marcia and Silas Keehn.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Endocrinology thanks J. Cryan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bishehsari, F., Voigt, R.M. & Keshavarzian, A. Circadian rhythms and the gut microbiota: from the metabolic syndrome to cancer. Nat Rev Endocrinol 16, 731–739 (2020). https://doi.org/10.1038/s41574-020-00427-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-020-00427-4
This article is cited by
-
Triglyceride-glucose index is a risk factor for breast cancer in China: a cross-sectional study
Lipids in Health and Disease (2024)
-
Association of systemic immunity-inflammation index with metabolic syndrome in U.S. adult: a cross-sectional study
BMC Geriatrics (2024)
-
Correlation analysis between occupational stress and metabolic syndrome in workers of a petrochemical enterprise: based on two assessment models of occupational stress
BMC Public Health (2024)
-
Liver ACSM3 deficiency mediates metabolic syndrome via a lauric acid-HNF4α-p38 MAPK axis
The EMBO Journal (2024)
-
Exploring reciprocal causation: bidirectional mendelian randomization study of gut microbiota composition and thyroid cancer
Journal of Cancer Research and Clinical Oncology (2024)