Abstract
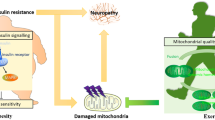
Neurological and mental illnesses account for a considerable proportion of the global burden of disease. Exercise has many beneficial effects on brain health, contributing to decreased risks of dementia, depression and stress, and it has a role in restoring and maintaining cognitive function and metabolic control. The fact that exercise is sensed by the brain suggests that muscle-induced peripheral factors enable direct crosstalk between muscle and brain function. Muscle secretes myokines that contribute to the regulation of hippocampal function. Evidence is accumulating that the myokine cathepsin B passes through the blood–brain barrier to enhance brain-derived neurotrophic factor production and hence neurogenesis, memory and learning. Exercise increases neuronal gene expression of FNDC5 (which encodes the PGC1α-dependent myokine FNDC5), which can likewise contribute to increased brain-derived neurotrophic factor levels. Serum levels of the prototype myokine, IL-6, increase with exercise and might contribute to the suppression of central mechanisms of feeding. Exercise also increases the PGC1α-dependent muscular expression of kynurenine aminotransferase enzymes, which induces a beneficial shift in the balance between the neurotoxic kynurenine and the neuroprotective kynurenic acid, thereby reducing depression-like symptoms. Myokine signalling, other muscular factors and exercise-induced hepatokines and adipokines are implicated in mediating the exercise-induced beneficial impact on neurogenesis, cognitive function, appetite and metabolism, thus supporting the existence of a muscle–brain endocrine loop.
Key points
-
Exercise can indirectly be sensed by the brain via adipose tissue (adiponectin) or the liver (fibroblast growth factor 21 and insulin-like growth factor 1).
-
Myokines mediate muscle–organ crosstalk to the liver, gut, pancreas, adipose tissue, bone, vascular bed, skin and brain.
-
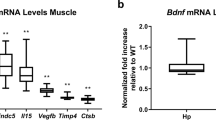
Cathepsin B is an exercise-induced myokine required for exercise-induced improvement in memory and adult neurogenesis.
-
Exercise enhances neuronal gene expression of FNDC5, the protein product of which might stimulate brain-derived neurotrophic factor in the hippocampus.
-
Serum levels of the myokine IL-6 increase with exercise, and this myokine might regulate central mechanisms for food intake.
-
Exercise increases muscular expression of kynurenine aminotransferases, which convert blood levels of neurotoxic kynurenine to the neuroprotective kynurenic acid, thereby reducing depression-like symptoms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Nietzsche, F. W. Beyond Good and Evil Aphorism 34 (Cambridge Univ. Press, 2002).
Kierkegaard, S. Kierkegaard’s Writings, XXV, Volume 25: Letters and Documents (ed. Rosenmeier, H.) letter 150, 1847 (Princeton Univ. Press, 2009).
Gros, F. A Philosophy of Walking (Verso Books, 2014).
Noakes, T. & Spedding, M. Olympics: run for your life. Nature 487, 295–296 (2012).
Booth, F. W., Roberts, C. K., Thyfault, J. P., Ruegsegger, G. N. & Toedebusch, R. G. Role of inactivity in chronic diseases: evolutionary insight and pathophysiological mechanisms. Physiol. Rev. 97, 1351–1402 (2017).
Cotman, C. W., Berchtold, N. C. & Christie, L. A. Exercise builds brain health: key roles of growth factor cascades and inflammation. Trends Neurosci. 30, 464–472 (2007).
Mattson, M. P. Energy intake and exercise as determinants of brain health and vulnerability to injury and disease. Cell Metab. 16, 706–722 (2012).
Arem, H. et al. Leisure time physical activity and mortality: a detailed pooled analysis of the dose-response relationship. JAMA Intern. Med. 175, 959–967 (2015).
Santos-Lozano, A. et al. Physical activity and Alzheimer disease: a protective association. Mayo Clin. Proc. 91, 999–1020 (2016).
Aarsland, D., Sardahaee, F. S., Anderssen, S. & Ballard, C. Is physical activity a potential preventive factor for vascular dementia? A systematic review. Aging Ment. Health 14, 386–395 (2010).
Williams, J. W., Plassman, B. L., Burke, J. & Benjamin, S. Preventing Alzheimer’s disease and cognitive decline. Evid. Rep. Technol. Assess. (Full Rep.) 193, 1–727 (2010).
Pedersen, B. K. & Saltin, B. Exercise as medicine - evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand. J. Med. Sci. Sports 25 (Suppl. 3), 1–72 (2015).
Voss, M. W., Nagamatsu, L. S., Liu-Ambrose, T. & Kramer, A. F. Exercise, brain, and cognition across the life span. J. Appl. Physiol. 111, 1505–1513 (2011).
Cotman, C. W. & Berchtold, N. C. Exercise: a behavioral intervention to enhance brain health and plasticity. Trends Neurosci. 25, 295–301 (2002).
Smith, P. J. et al. Aerobic exercise and neurocognitive performance: a meta-analytic review of randomized controlled trials. Psychosom. Med. 72, 239–252 (2010).
Snowden, M. et al. Effect of exercise on cognitive performance in community-dwelling older adults: review of intervention trials and recommendations for public health practice and research. J. Am. Geriatr. Soc. 59, 704–716 (2011).
Gaser, C. & Schlaug, G. Brain structures differ between musicians and non-musicians. J. Neurosci. 23, 9240–9245 (2003).
Aberg, M. A. et al. Cardiovascular fitness is associated with cognition in young adulthood. Proc. Natl Acad. Sci. USA 106, 20906–20911 (2009).
Kelley, G. A. & Kelley, K. S. Exercise and sleep: a systematic review of previous meta-analyses. J. Evid. Based Med. 10, 26–36 (2017).
Blundell, J. E., Gibbons, C., Caudwell, P., Finlayson, G. & Hopkins, M. Appetite control and energy balance: impact of exercise. Obes. Rev. 16 (Suppl. 1), 67–76 (2015).
Crush, E. A., Frith, E. & Loprinzi, P. D. Experimental effects of acute exercise duration and exercise recovery on mood state. J. Affect. Disord. 229, 282–287 (2018).
Ruiz, J. R. et al. Resistance training does not have an effect on cognition or related serum biomarkers in nonagenarians: a randomized controlled trial. Int. J. Sports Med. 36, 54–60 (2015).
Gordon, B. R. et al. Association of efficacy of resistance exercise training with depressive symptoms: meta-analysis and meta-regression analysis of randomized clinical trials. JAMA Psychiatry 75, 566–576 (2018).
Batouli, S. A. H. & Saba, V. At least eighty percent of brain grey matter is modifiable by physical activity: a review study. Behav. Brain Res. 332, 204–217 (2017).
Kobilo, T. et al. Running is the neurogenic and neurotrophic stimulus in environmental enrichment. Learn. Mem. 18, 605–609 (2011).
Febbraio, M. A. & Pedersen, B. K. Contraction-induced myokine production and release: is skeletal muscle an endocrine organ? Exerc. Sport Sci. Rev. 33, 114–119 (2005).
Giudice, J. & Taylor, J. M. Muscle as a paracrine and endocrine organ. Curr. Opin. Pharmacol. 34, 49–55 (2017).
Karstoft, K. & Pedersen, B. K. Skeletal muscle as a gene regulatory endocrine organ. Curr. Opin. Clin. Nutr. Metab. Care 19, 270–275 (2016).
Pedersen, B. K. Muscle as a secretory organ. Compr. Physiol. 3, 1337–1362 (2013).
Pedersen, B. K. & Febbraio, M. A. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 8, 457–465 (2012).
Hoffmann, C. & Weigert, C. Skeletal muscle as an endocrine organ: the role of myokines in exercise adaptations. Cold Spring Harb. Perspect. Med. 7, a029793 (2017).
Schnyder, S. & Handschin, C. Skeletal muscle as an endocrine organ: PGC-1α, myokines and exercise. Bone 80, 115–125 (2015).
Raschke, S. & Eckel, J. Adipo-myokines: two sides of the same coin—mediators of inflammation and mediators of exercise. Mediators. Inflamm. 2013, 320724 (2013).
Henningsen, J., Pedersen, B. K. & Kratchmarova, I. Quantitative analysis of the secretion of the MCP family of chemokines by muscle cells. Mol. Biosyst. 7, 311–321 (2011).
Henningsen, J., Rigbolt, K. T., Blagoev, B., Pedersen, B. K. & Kratchmarova, I. Dynamics of the skeletal muscle secretome during myoblast differentiation. Mol. Cell. Proteomics 9, 2482–2496 (2010).
Benatti, F. B. & Pedersen, B. K. Exercise as an anti-inflammatory therapy for rheumatic diseases-myokine regulation. Nat. Rev. Rheumatol. 11, 86–97 (2015).
Hojman, P., Gehl, J., Christensen, J. F. & Pedersen, B. K. Molecular mechanisms linking exercise to cancer prevention and treatment. Cell Metab. 27, 10–21 (2018).
Pedersen, L. et al. Voluntary running suppresses tumor growth through epinephrine- and IL-6-dependent NK cell mobilization and redistribution. Cell Metab. 23, 554–562 (2016).
Rai, M. & Demontis, F. Systemic nutrient and stress signaling via myokines and myometabolites. Annu. Rev. Physiol. 78, 85–107 (2016).
Makarova, J. A. et al. Exercise immunology meets MiRNAs. Exerc. Immunol. Rev. 20, 135–164 (2014).
Agudelo, L. Z. et al. Skeletal muscle PGC-1α1 modulates kynurenine metabolism and mediates resilience to stress-induced depression. Cell 159, 33–45 (2014).
Safdar, A., Saleem, A. & Tarnopolsky, M. A. The potential of endurance exercise-derived exosomes to treat metabolic diseases. Nat. Rev. Endocrinol. 12, 504–517 (2016).
Whitham, M. et al. Extracellular vesicles provide a means for tissue crosstalk during exercise. Cell Metab. 27, 237–251 (2018).
Safdar, A. & Tarnopolsky, M. A. Exosomes as mediators of the systemic adaptations to endurance exercise. Cold Spring Harb. Perspect. Med. 8, a029827 (2018).
Loprinzi, P. D. & Frith, E. A brief primer on the mediational role of BDNF in the exercise-memory link. Clin. Physiol. Funct. Imaging 39, 9–14 (2019).
Neeper, S. A., Gomez-Pinilla, F., Choi, J. & Cotman, C. Exercise and brain neurotrophins. Nature 373, 109 (1995).
Liu, P. Z. & Nusslock, R. Exercise-mediated neurogenesis in the hippocampus via BDNF. Front. Neurosci. 12, 52 (2018).
Oliff, H. S., Berchtold, N. C., Isackson, P. & Cotman, C. W. Exercise-induced regulation of brain-derived neurotrophic factor (BDNF) transcripts in the rat hippocampus. Brain Res. Mol. Brain Res. 61, 147–153 (1998).
Van Hoomissen, J. D., Chambliss, H. O., Holmes, P. V. & Dishman, R. K. Effects of chronic exercise and imipramine on mRNA for BDNF after olfactory bulbectomy in rat. Brain Res. 974, 228–235 (2003).
Farmer, J. et al. Effects of voluntary exercise on synaptic plasticity and gene expression in the dentate gyrus of adult male Sprague-Dawley rats in vivo. Neuroscience 124, 71–79 (2004).
Adlard, P. A., Perreau, V. M. & Cotman, C. W. The exercise-induced expression of BDNF within the hippocampus varies across life-span. Neurobiol. Aging 26, 511–520 (2005).
Li, X. et al. Blocking of BDNF-TrkB signaling inhibits the promotion effect of neurological function recovery after treadmill training in rats with spinal cord injury. Spinal Cord 57, 65–74 (2019).
Vaynman, S., Ying, Z. & Gomez-Pinilla, F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur. J. Neurosci. 20, 2580–2590 (2004).
Vaynman, S., Ying, Z. & Gomez-Pinilla, F. Exercise induces BDNF and synapsin I to specific hippocampal subfields. J. Neurosci. Res. 76, 356–362 (2004).
Rasmussen, P. et al. Evidence for a release of brain-derived neurotrophic factor from the brain during exercise. Exp. Physiol. 94, 1062–1069 (2009).
Seifert, T. et al. Endurance training enhances BDNF release from the human brain. Am. J. Physiol. Regul. Integr. Comp. Physiol. 298, R372–R377 (2010).
Pajonk, F. G. et al. Hippocampal plasticity in response to exercise in schizophrenia. Arch. Gen. Psychiatry 67, 133–143 (2010).
Wrann, C. D. et al. Exercise induces hippocampal BDNF through a PGC-1α/FNDC5 pathway. Cell Metab. 18, 649–659 (2013).
de Assis, G. G. & de Almondes, K. M. Exercise-dependent BDNF as a modulatory factor for the executive processing of individuals in course of cognitive decline. a systematic review. Front. Psychol. 8, 584 (2017).
Matthews, V. B. et al. Brain-derived neurotrophic factor is produced by skeletal muscle cells in response to contraction and enhances fat oxidation via activation of AMP-activated protein kinase. Diabetologia 52, 1409–1418 (2009).
Moon, H. Y. et al. Running-induced systemic cathepsin B secretion is associated with memory function. Cell Metab. 24, 332–340 (2016).
Martinowich, K., Manji, H. & Lu, B. New insights into BDNF function in depression and anxiety. Nat. Neurosci. 10, 1089–1093 (2007).
Suzuki, W. A. How body affects brain. Cell Metab. 24, 192–193 (2016).
Olesen, J., Kiilerich, K. & Pilegaard, H. PGC-1α-mediated adaptations in skeletal muscle. Pflugers Arch. 460, 153–162 (2010).
Spiegelman, B. M. Transcriptional control of mitochondrial energy metabolism through the PGC1 coactivators. Novartis Found. Symp. 287, 60–63 (2007).
Bostrom, P. et al. A PGC1α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 481, 463–468 (2012).
Ding, Q., Vaynman, S., Akhavan, M., Ying, Z. & Gomez-Pinilla, F. Insulin-like growth factor I interfaces with brain-derived neurotrophic factor-mediated synaptic plasticity to modulate aspects of exercise-induced cognitive function. Neuroscience 140, 823–833 (2006).
Albrecht, E. et al. Irisin - a myth rather than an exercise-inducible myokine. Sci. Rep. 5, 8889 (2015).
Pekkala, S. et al. Are skeletal muscle FNDC5 gene expression and irisin release regulated by exercise and related to health? J. Physiol. 591, 5393–5400 (2013).
Eaton, M. et al. Impact of a single bout of high-intensity interval exercise and short-term interval training on interleukin-6, FNDC5, and METRNL mRNA expression in human skeletal muscle. J. Sport Health Sci. 7, 191–196 (2018).
Jedrychowski, M. P. et al. Detection and quantitation of circulating human irisin by tandem mass spectrometry. Cell Metab. 22, 734–740 (2015).
Wrann, C. D. FNDC5/irisin - their role in the nervous system and as a mediator for beneficial effects of exercise on the brain. Brain Plast. 1, 55–61 (2015).
Lourenco, M. V. et al. Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models. Nat. Med. 5, 165–175 (2019).
Koppel, I. & Timmusk, T. Differential regulation of Bdnf expression in cortical neurons by class-selective histone deacetylase inhibitors. Neuropharmacology 75, 106–115 (2013).
Mitchell, G. A. et al. Medical aspects of ketone body metabolism. Clin. Invest. Med. 18, 193–216 (1995).
Sleiman, S. F. et al. Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body beta-hydroxybutyrate. eLife 5, e15092 (2016).
Lim, S. et al. D-β-hydroxybutyrate is protective in mouse models of Huntington’s disease. PLOS ONE 6, e24620 (2011).
Kashiwaya, Y. et al. D-β-hydroxybutyrate protects neurons in models of Alzheimer’s and Parkinson’s disease. Proc. Natl Acad. Sci. USA 97, 5440–5444 (2000).
Sothmann, M. S., Hart, B. A. & Horn, T. S. Sympathetic nervous system and behavioral responses to stress following exercise training. Physiol. Behav. 51, 1097–1103 (1992).
Gleeson, L. C., Ryan, K. J., Griffin, E. W., Connor, T. J. & Harkin, A. The β2-adrenoceptor agonist clenbuterol elicits neuroprotective, anti-inflammatory and neurotrophic actions in the kainic acid model of excitotoxicity. Brain Behav. Immun. 24, 1354–1361 (2010).
Cooke, J. D., Grover, L. M. & Spangler, P. R. Venlafaxine treatment stimulates expression of brain-derived neurotrophic factor protein in frontal cortex and inhibits long-term potentiation in hippocampus. Neuroscience 162, 1411–1419 (2009).
Mohamed-Ali, V. et al. Subcutaneous adipose tissue releases interleukin-6, but not tumor necrosis factor-alpha, in vivo. J. Clin. Endocrinol. Metab. 82, 4196–4200 (1997).
Carey, A. L. et al. Interleukin-6 and tumor necrosis factor-alpha are not increased in patients with type 2 diabetes: evidence that plasma IL-6 is related to fat mass and not insulin responsiveness. Diabetologia 47, 1029–1037 (2004).
Wedell-Neergaard, A. S. et al. Cardiorespiratory fitness and the metabolic syndrome: roles of inflammation and abdominal obesity. PLOS ONE 13, e0194991 (2018).
Kim, H. J. et al. Differential effects of interleukin-6 and -10 on skeletal muscle and liver insulin action in vivo. Diabetes 53, 1060–1067 (2004).
Cai, D. et al. Local and systemic insulin resistance resulting from hepatic activation of IKK-β and NF-κB. Nat. Med. 11, 183–190 (2005).
Matthews, V. B. et al. Interleukin-6-deficient mice develop hepatic inflammation and systemic insulin resistance. Diabetologia 53, 2431–2441 (2010).
Wallenius, V. et al. Interleukin-6-deficient mice develop mature-onset obesity. Nat. Med. 8, 75–79 (2002).
Ellingsgaard, H. et al. Interleukin-6 enhances insulin secretion by increasing glucagon-like peptide-1 secretion from L cells and alpha cells. Nat. Med. 17, 1481–1489 (2011).
Mauer, J. et al. Signaling by IL-6 promotes alternative activation of macrophages to limit endotoxemia and obesity-associated resistance to insulin. Nat. Immunol. 15, 423–430 (2014).
Mauer, J., Denson, J. L. & Bruning, J. C. Versatile functions for IL-6 in metabolism and cancer. Trends Immunol. 36, 92–101 (2015).
Carey, A. L. et al. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes 55, 2688–2697 (2006).
van Hall, G. et al. Interleukin-6 stimulates lipolysis and fat oxidation in humans. J. Clin. Endocrinol. Metab. 88, 3005–3010 (2003).
Lang Lehrskov, L. et al. Interleukin-6 delays gastric emptying in humans with direct effects on glycemic control. Cell Metab. 27, 1201–1211 (2018).
Starkie, R., Ostrowski, S. R., Jauffred, S., Febbraio, M. & Pedersen, B. K. Exercise and IL-6 infusion inhibit endotoxin-induced TNF-alpha production in humans. FASEB. J. 17, 884–886 (2003).
Steensberg, A., Fischer, C. P., Keller, C., Moller, K. & Pedersen, B. K. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am. J. Physiol. Endocrinol. Metab. 285, E433–E437 (2003).
Pedersen, B. K. & Febbraio, M. A. Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol. Rev. 88, 1379–1406 (2008).
Febbraio, M. A. & Pedersen, B. K. Muscle-derived interleukin-6: mechanisms for activation and possible biological roles. FASEB J. 16, 1335–1347 (2002).
Keller, C., Hellsten, Y., Steensberg, A. & Pedersen, B. K. Differential regulation of IL-6 and TNF-α via calcineurin in human skeletal muscle cells. Cytokine 36, 141–147 (2006).
Keller, C. et al. Transcriptional activation of the IL-6 gene in human contracting skeletal muscle: influence of muscle glycogen content. FASEB J. 15, 2748–2750 (2001).
Steensberg, A. et al. Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J. Physiol. 537, 633–639 (2001).
Li, G. et al. Induction of uncoupling protein 1 by central interleukin-6 gene delivery is dependent on sympathetic innervation of brown adipose tissue and underlies one mechanism of body weight reduction in rats. Neuroscience 115, 879–889 (2002).
Hidalgo, J. et al. Transgenic mice with astrocyte-targeted production of interleukin-6 are resistant to high-fat diet-induced increases in body weight and body fat. Brain Behav. Immun. 24, 119–126 (2010).
Senaris, R. M. et al. Interleukin-6 regulates the expression of hypothalamic neuropeptides involved in body weight in a gender-dependent way. J. Neuroendocrinol. 23, 675–686 (2011).
Schele, E. et al. Inter-relation between interleukin (IL)-1, IL-6 and body fat regulating circuits of the hypothalamic arcuate nucleus. J. Neuroendocrinol. 25, 580–589 (2013).
Benrick, A. et al. Interleukin-6 gene knockout influences energy balance regulating peptides in the hypothalamic paraventricular and supraoptic nuclei. J. Neuroendocrinol. 21, 620–628 (2009).
Molinero, A. et al. Role of muscle IL-6 in gender-specific metabolism in mice. PLOS ONE 12, e0173675 (2017).
Timper, K. et al. IL-6 improves energy and glucose homeostasis in obesity via enhanced central IL-6 trans-signaling. Cell Rep. 19, 267–280 (2017).
Pedersen, M. et al. Does the aging skeletal muscle maintain its endocrine function? Exerc. Immunol. Rev. 10, 42–55 (2004).
Scheele, C. et al. Satellite cells derived from obese humans with type 2 diabetes and differentiated into myocytes in vitro exhibit abnormal response to IL-6. PLOS ONE 7, e39657 (2012).
Bunprajun, T., Henriksen, T. I., Scheele, C., Pedersen, B. K. & Green, C. J. Lifelong physical activity prevents aging-associated insulin resistance in human skeletal muscle myotubes via increased glucose transporter expression. PLOS ONE 8, e66628 (2013).
Global Burden of Disease Study 2013 Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 386, 743–800 (2015).
Canli, T. & Lesch, K. P. Long story short: the serotonin transporter in emotion regulation and social cognition. Nat. Neurosci. 10, (1103–1109 (2007).
Muller, N. & Schwarz, M. J. The immune-mediated alteration of serotonin and glutamate: towards an integrated view of depression. Mol. Psychiatry 12, 988–1000 (2007).
Beal, M. F. et al. Replication of the neurochemical characteristics of Huntington’s disease by quinolinic acid. Nature 321, 168–171 (1986).
Wu, W. et al. Expression of tryptophan 2,3-dioxygenase and production of kynurenine pathway metabolites in triple transgenic mice and human Alzheimer’s disease brain. PLOS ONE 8, e59749 (2013).
Claes, S. et al. The kynurenine pathway in major depression: haplotype analysis of three related functional candidate genes. Psychiatry Res. 188, 355–360 (2011).
Myint, A. M. & Kim, Y. K. Network beyond IDO in psychiatric disorders: revisiting neurodegeneration hypothesis. Prog. Neuropsychopharmacol. Biol. Psychiatry 48, 304–313 (2014).
Schwarcz, R., Bruno, J. P., Muchowski, P. J. & Wu, H. Q. Kynurenines in the mammalian brain: when physiology meets pathology. Nat. Rev. Neurosci. 13, 465–477 (2012).
Arany, Z. PGC-1 coactivators and skeletal muscle adaptations in health and disease. Curr. Opin. Genet. Dev. 18, 426–434 (2008).
White, Z. et al. Voluntary resistance wheel exercise from mid-life prevents sarcopenia and increases markers of mitochondrial function and autophagy in muscles of old male and female C57BL/6J mice. Skelet. Muscle 6, 45 (2016).
Lin, J. et al. Transcriptional co-activator PGC-1α drives the formation of slow-twitch muscle fibres. Nature 418, 797–801 (2002).
Lewis, G. D. et al. Metabolic signatures of exercise in human plasma. Sci. Transl Med. 2, 33ra37 (2010).
Eckardt, K., Taube, A. & Eckel, J. Obesity-associated insulin resistance in skeletal muscle: role of lipid accumulation and physical inactivity. Rev. Endocr. Metab. Disord. 12, 163–172 (2011).
Roy, T. & Lloyd, C. E. Epidemiology of depression and diabetes: a systematic review. J. Affect. Disord. 142, S8–S21 (2012).
Pouwer, F. Should we screen for emotional distress in type 2 diabetes mellitus? Nat. Rev. Endocrinol. 5, 665–671 (2009).
Fallarini, S., Magliulo, L., Paoletti, T., de Lalla, C. & Lombardi, G. Expression of functional GPR35 in human iNKT cells. Biochem. Biophys. Res. Commun. 398, 420–425 (2010).
Agudelo, L. Z. et al. Kynurenic acid and Gpr35 regulate adipose tissue energy homeostasis and inflammation. Cell Metab. 27, 378–392 (2018).
Dai, Y., Pang, J., Gong, H., Fan, W. & Zhang, T. M. Roles and tissue source of adiponectin involved in lifestyle modifications. J. Gerontol. A Biol. Sci. Med. Sci. 68, 117–128 (2013).
Yau, S. Y. et al. Physical exercise-induced hippocampal neurogenesis and antidepressant effects are mediated by the adipocyte hormone adiponectin. Proc. Natl Acad. Sci. USA 111, 15810–15815 (2014).
Li, A., Yau, S. Y., Machado, S., Yuan, T. F. & So, K. F. Adult neurogenic and antidepressant effects of adiponectin: a potential replacement for exercise? CNS Neurol. Disord. Drug Targets 14, 1129–1144 (2015).
Cooper, C., Moon, H. Y. & van Praag, H. On the run for hippocampal plasticity. Cold Spring Harb. Perspect. Med. 8, a029736 (2018).
Stefan, N. & Haring, H. U. The role of hepatokines in metabolism. Nat. Rev. Endocrinol. 9, 144–152 (2013).
Markan, K. R. et al. Circulating FGF21 is liver derived and enhances glucose uptake during refeeding and overfeeding. Diabetes 63, 4057–4063 (2014).
Gaich, G. et al. The effects of LY2405319, an FGF21 analog, in obese human subjects with type 2 diabetes. Cell Metab. 18, 333–340 (2013).
Hansen, J. S. et al. Exercise-induced secretion of FGF21 and follistatin are blocked by pancreatic clamp and impaired in type 2 diabetes. J. Clin. Endocrinol. Metab. 101, 2816–2825 (2016).
Liang, Q. et al. FGF21 maintains glucose homeostasis by mediating the cross talk between liver and brain during prolonged fasting. Diabetes 63, 4064–4075 (2014).
von Holstein-Rathlou, S. et al. FGF21 mediates endocrine control of simple sugar intake and sweet taste preference by the liver. Cell Metab. 23, 335–343 (2016).
Talukdar, S. et al. FGF21 regulates sweet and alcohol preference. Cell Metab. 23, 344–349 (2016).
Soberg, S. et al. FGF21 is a sugar-induced hormone associated with sweet intake and preference in humans. Cell Metab. 25, 1045–1053 (2017).
Sa-Nguanmoo, P. et al. FGF21 improves cognition by restored synaptic plasticity, dendritic spine density, brain mitochondrial function and cell apoptosis in obese-insulin resistant male rats. Horm. Behav. 85, 86–95 (2016).
Hamrick, M. W. A role for myokines in muscle-bone interactions. Exerc. Sport Sci. Rev. 39, 43–47 (2011).
Berg, U. & Bang, P. Exercise and circulating insulin-like growth factor I. Horm. Res. 62, (Suppl. 1), 50–58 (2004).
Chen, M. J. & Russo-Neustadt, A. A. Running exercise- and antidepressant-induced increases in growth and survival-associated signaling molecules are IGF-dependent. Growth Factors 25, 118–131 (2007).
Duzel, E., van Praag, H. & Sendtner, M. Can physical exercise in old age improve memory and hippocampal function? Brain 139, 662–673 (2016).
Krishnan, V. S. et al. A neurogenic perspective of sarcopenia: time course study of sciatic nerves from aging mice. J. Neuropathol. Exp. Neurol. 75, 464–478 (2016).
Krishnan, V. S., Shavlakadze, T., Grounds, M. D., Hodgetts, S. I. & Harvey, A. R. Age-related loss of VGLUT1 excitatory, but not VGAT inhibitory, immunoreactive terminals on motor neurons in spinal cords of old sarcopenic male mice. Biogerontology 19, 385–399 (2018).
Snyder-Warwick, A. K., Satoh, A., Santosa, K. B., Imai, S. I. & Jablonka-Shariff, A. Hypothalamic Sirt1 protects terminal Schwann cells and neuromuscular junctions from age-related morphological changes. Aging Cell 17, e12776 (2018).
Spalding, K. L. et al. Dynamics of hippocampal neurogenesis in adult humans. Cell 153, 1219–1227 (2013).
Acknowledgements
The Centre for Physical Activity Research (CFAS) is supported by a grant from TrygFonden. The author also thanks Alzheimer-forskningsfonden for support.
Reviewer information
Nature Reviews Endocrinology thanks M. Grounds, S. Schiaffino and other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Physical activity
-
Any bodily movement produced by skeletal muscles that requires energy expenditure.
- Moderate-to-vigorous physical activity
-
Any activity with an energy expenditure of ≥3 metabolic equivalents (for example, brisk walking); the WHO minimum recommendations are 150 min of moderate-to-vigorous physical activity each week (or 20 min or 10,000 steps on most days of the week) for adults and 60 min of active playing on most days of the week for children and adolescents.
- Aerobic exercise
-
Exercise involving dynamic movements and large muscle groups that predominantly rely on aerobic metabolism for fuelling muscle contractions; examples include jogging, running, swimming and rowing.
- Resistance training
-
Movement performed against a specific external force that is regularly increased during training; examples include weightlifting and exercises using resistance machines.
- Exercise training
-
A subset of physical activity that is planned, structured and repetitive and has a final or intermediate objective of improving or maintaining physical fitness. The terms ‘exercise’ and ‘exercise training’ are used interchangeably to refer to the cardiovascular adaptations produced by this specific type of physical activity; a single bout of exercise is referred to as ‘acute exercise’.
- Myokines
-
Cytokines or peptides produced by skeletal muscle cells and subsequently released into the circulation, where they exert autocrine, paracrine or endocrine effects in other cells, tissues or organs.
- Browning
-
Browning can be defined as any substantial increase in the expression of uncoupling protein 1 (UCP1) at the mRNA level occurring in what is normally considered as a white adipose tissue depot. The resulting brown adipocytes that express UCP1 and appear in white adipose tissue are referred to as beige, brite, convertible, ectopic, inducible or recruitable.
- Sarcopenia
-
Derived from the Greek sarx (‘flesh’) and penia (‘loss’), sarcopenia is the age-induced loss of muscle mass and function that typically manifests as reduced gait speed.
Rights and permissions
About this article
Cite this article
Pedersen, B.K. Physical activity and muscle–brain crosstalk. Nat Rev Endocrinol 15, 383–392 (2019). https://doi.org/10.1038/s41574-019-0174-x
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-019-0174-x
This article is cited by
-
Exercise mimetics: a novel strategy to combat neuroinflammation and Alzheimer’s disease
Journal of Neuroinflammation (2024)
-
Resistance training's impact on blood biomarkers and cognitive function in older adults with low and high risk of mild cognitive impairment: a randomized controlled trial
European Review of Aging and Physical Activity (2024)
-
The bidirectional associations between sarcopenia-related traits and cognitive performance
Scientific Reports (2024)
-
Resistance exercise effects on hippocampus subfield volumes and biomarkers of neuroplasticity and neuroinflammation in older adults with low and high risk of mild cognitive impairment: a randomized controlled trial
GeroScience (2024)
-
Tai chi, irisin and cognitive performance: a clinical and biological investigation in older adults
Aging Clinical and Experimental Research (2024)