Abstract
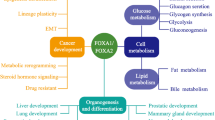
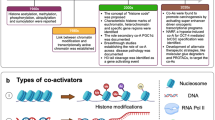
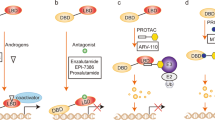
FOXO proteins are transcription factors that are involved in numerous physiological processes and in various pathological conditions, including cardiovascular disease, cancer, diabetes and chronic neurological diseases. For example, FOXO proteins are context-dependent tumour suppressors that are frequently inactivated in human cancers, and FOXO3 is the second most replicated gene associated with extreme human longevity. Therefore, pharmacological manipulation of FOXO proteins is a promising approach to developing therapeutics for cancer and for healthy ageing. In this Review, we overview the role of FOXO proteins in health and disease and discuss the pharmacological approaches to modulate FOXO function.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Myatt, S. S. & Lam, E. W. F. The emerging roles of forkhead box (Fox) proteins in cancer. Nat. Rev. Cancer 7, 847–859 (2007).
Carter, M. E. & Brunet, A. FOXO transcription factors. Curr. Biol. 17, R113–R114 (2007).
Santo, E. E. & Paik, J. A splice junction-targeted CRISPR approach (spJCRISPR) reveals human FOXO3B to be a protein-coding gene. Gene 673, 95–101 (2018).
Eijkelenboom, A. & Burgering, B. M. FOXOs: signalling integrators for homeostasis maintenance. Nat. Rev. Mol. Cell Biol. 14, 83–97 (2013).
van der Horst, A. & Burgering, B. M. Stressing the role of FoxO proteins in lifespan and disease. Nat. Rev. Mol. Cell Biol. 8, 440–450 (2007).
Brown, A. K. & Webb, A. E. in Current Topics in Developmental Biology Vol. 127 165–192 (Academic Press Inc., 2018).
Weigel, D., Jürgens, G., Küttner, F., Seifert, E. & Jäckle, H. The homeotic gene fork head encodes a nuclear protein and is expressed in the terminal regions of the Drosophila embryo. Cell 57, 645–658 (1989).
Link, W. Introduction to FOXO biology. Methods Mol. Biol. 1890, 1–9 (2019).
Morris, B. J., Willcox, D. C., Donlon, T. A. & Willcox, B. J. FOXO3: a major gene for human longevity - a mini-review. Gerontology 61, 515–525 (2015).
Singh, P. P., Demmitt, B. A., Nath, R. D. & Brunet, A. The genetics of aging: a vertebrate perspective. Cell 177, 200–220 (2019).
Baar, M. P. et al. Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 169, 132–147.e16 (2017). This study reports a peptide that perturbs the FOXO4 interaction with p53, which can selectively target senescent cells and restore fitness in fast and naturally aged mice.
Cheng, Z. The FoxO–autophagy axis in health and disease. Trends Endocrinol. Metab. 30, 658–671 (2019).
Liang, R. & Ghaffari, S. in Current Topics in Developmental Biology Vol. 127 23–47 (Academic Press Inc., 2018).
Grossi, V. et al. The longevity SNP rs2802292 uncovered: HSF1 activates stress-dependent expression of FOXO3 through an intronic enhancer. Nucleic Acids Res. 46, 5587–5600 (2018).
Flachsbart, F. et al. Identification and characterization of two functional variants in the human longevity gene FOXO3. Nat. Commun. 8, 2063 (2017).
Sun, Y. et al. Integrated discovery of FOXO1-DNA stabilizers from marine natural products to restore chemosensitivity to anti-EGFR-based therapy for metastatic lung cancer. Mol. Biosyst. 13, 330–337 (2017). This study reports the discovery of small-molecule compounds capable of stabilizing the DNA-bound conformation of FOXO1.
Salcher, S. et al. A drug library screen identifies carbenoxolone as novel FOXO inhibitor that overcomes FOXO3-mediated chemoprotection in high-stage neuroblastoma. Oncogene 39, 1080–1097 (2019). This paper reports a drug-repurposing approach to identify compounds that bind to the FOXO3 DNA-binding domain and inhibit the transcriptional activity of FOXO3.
Hagenbuchner, J. et al. Modulating FOXO3 transcriptional activity by small, DBD-binding molecules. eLife 8, e48876 (2019).
Dansen, T. B. & Burgering, B. M. Unravelling the tumor-suppressive functions of FOXO proteins. Trends Cell Biol. 18, 421–429 (2008).
Paik, J. H. et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell 128, 309–323 (2007). This seminal paper established FOXO proteins as tumour suppressors, demonstrating that somatic deletion of Foxo1, Foxo3 and Foxo4 genes results in lineage-restricted tumour phenotypes.
Hornsveld, M., Dansen, T. B., Derksen, P. W. & Burgering, B. M. T. Re-evaluating the role of FOXOs in cancer. Semin. Cancer Biol. 50, 90–100 (2018).
Nagashima, T. et al. Discovery of novel forkhead box O1 inhibitors for treating type 2 diabetes: improvement of fasting glycemia in diabetic db/db mice. Mol. Pharmacol. 78, 961–970 (2010). This article describes the discovery and characterization of a compound that binds to FOXO1 and inhibits its transactivation, which leads to a drastic decrease in fasting plasma glucose levels via the inhibition of hepatic gluconeogenic genes.
Langlet, F. et al. Selective inhibition of FOXO1 activator/repressor balance modulates hepatic glucose handling. Cell 171, 824–835.e18 (2017). This article reports selective targeting of the activator and repressor functions of FOXO1, providing evidence that different functions of FOXO proteins can be independently modulated by small-molecule compounds.
Zhang, Y. et al. Metformin induces FOXO3-dependent fetal hemoglobin production in human primary erythroid cells. Blood 132, 321–333 (2018).
Alvarez-Garcia, O. et al. FOXO are required for intervertebral disk homeostasis during aging and their deficiency promotes disk degeneration. Aging Cell 17, e12800 (2018).
Rajendran, N. K., Kumar, S. S. D., Houreld, N. N. & Abrahamse, N. Understanding the perspectives of forkhead transcription factors in delayed wound healing. J. Cell Commun. Signal. 13, 151–162 (2019).
Xin, Z. et al. FOXOs in the impaired heart: new therapeutic targets for cardiac diseases. Biochim. Biophys. Acta Mol. Basis Dis. 1863, 486–498 (2017).
Hu, W. et al. Roles of forkhead box O (FoxO) transcription factors in neurodegenerative diseases: a panoramic view. Prog. Neurobiol. https://doi.org/10.1016/j.pneurobio.2019.101645 (2019).
Obsil, T. & Obsilova, V. Structure/function relationships underlying regulation of FOXO transcription factors. Oncogene 27, 2263–2275 (2008).
Boura, E., Rezabkova, L., Brynda, J., Obsilova, V. & Obsil, T. Structure of the human FOXO4-DBD-DNA complex at 1.9 Å resolution reveals new details of FOXO binding to the DNA. Acta Crystallogr. Sect. D. Biol. Crystallogr. 66, 1351–1357 (2010).
Brent, M. M., Anand, R. & Marmorstein, R. Structural basis for DNA recognition by FoxO1 and its regulation by posttranslational modification. Structure 16, 1407–1416 (2008).
Tsai, K. L. et al. Crystal structure of the human FOXO3a-DBD/DNA complex suggests the effects of post-translational modification. Nucleic Acids Res. 35, 6984–6994 (2007).
Clark, K. L., Halay, E. D., Lai, E. & Burley, S. K. Co-crystal structure of the HNF-3/fork head DNA-recognition motif resembles histone H5. Nature 364, 412–420 (1993).
Obsil, T. & Obsilova, V. Structural basis for DNA recognition by FOXO proteins. Biochim. Biophys. Acta Mol. Cell Res. 1813, 1946–1953 (2011).
Fu, Z. & Tindall, D. J. FOXOs, cancer and regulation of apoptosis. Oncogene 27, 2312–2319 (2008).
Van Der Heide, L. P., Hoekman, M. F. & Smidt, M. P. The ins and outs of FoxO shuttling: mechanisms of FoxO translocation and transcriptional regulation. Biochem. J. 380, 297–309 (2004).
Brownawell, A. M., Kops, G. J. P. L., Macara, I. G. & Burgering, B. M. T. Inhibition of nuclear import by protein kinase B (Akt) regulates the subcellular distribution and activity of the forkhead transcription factor AFX. Mol. Cell. Biol. 21, 3534–3546 (2001).
Zhang, X. et al. Phosphorylation of serine 256 suppresses transactivation by FKHR (FOXO1) by multiple mechanisms. J. Biol. Chem. 277, 45276–45284 (2002).
Zhao, X. et al. Multiple elements regulate nuclear/cytoplasmic shuttling of FOXO1: characterization of phosphorylation- and 14-3-3-dependent and -independent mechanisms. Biochem. J. 378, 839–849 (2004).
Wang, F., Marshall, C. B. & Ikura, M. Forkhead followed by disordered tail: the intrinsically disordered regions of FOXO3a. Intrinsically Disord. Proteins 3, e1056906 (2015).
Wang, F. et al. Structures of KIX domain of CBP in complex with two FOXO3a transactivation domains reveal promiscuity and plasticity in coactivator recruitment. Proc. Natl Acad. Sci. USA 109, 6078–6083 (2012).
Eijkelenboom, A. et al. Genome-wide analysis of FOXO3 mediated transcription regulation through RNA polymerase II profiling. Mol. Syst. Biol. 9, 638 (2013).
Ramaswamy, S., Nakamura, N., Sansal, I., Bergeron, L. & Sellers, W. R. A novel mechanism of gene regulation and tumor suppression by the transcription factor FKHR. Cancer Cell 2, 81–91 (2002).
van der Vos, K. E. & Coffer, P. J. FOXO-binding partners: it takes two to tango. Oncogene 27, 2289–2299 (2008).
Link, W. & Fernandez-Marcos, P. J. FOXO transcription factors at the interface of metabolism and cancer. Int. J. Cancer 141, 2379–2391 (2017).
Essaghir, A., Dif, N., Marbehant, C. Y., Coffer, P. J. & Demoulin, J. B. The transcription of FOXO genes is stimulated by FOXO3 and repressed by growth factors. J. Biol. Chem. 284, 10334–10342 (2009).
Kannike, K., Sepp, M., Zuccato, C., Cattaneo, E. & Timmusk, T. Forkhead transcription factor FOXO3a levels are increased in Huntington disease because of overactivated positive autofeedback loop. J. Biol. Chem. 289, 32845–32857 (2014).
Lützner, N., Kalbacher, H., Krones-Herzig, A. & Rösl, F. FOXO3 is a glucocorticoid receptor target and regulates LKB1 and Its own expression based on cellular AMP levels via a positive autoregulatory loop. PLoS ONE 7, e42166 (2012).
Guttilla, I. K. & White, B. A. Coordinate regulation of FOXO1 by miR-27a, miR-96, and miR-182 in breast cancer cells. J. Biol. Chem. 284, 23204–23216 (2009).
Myatt, S. S. et al. Definition of microRNAs that repress expression of the tumor suppressor gene FOXO1 in endometrial cancer. Cancer Res. 70, 367–377 (2010).
Segura, M. F. et al. Aberrant miR-182 expression promotes melanoma metastasis by repressing FOXO3 and microphthalmia-associated transcription factor. Proc. Natl Acad. Sci. USA 106, 1814–1819 (2009).
Yang, W., Du, W. W., Li, X., Yee, A. J. & Yang, B. B. Foxo3 activity promoted by non-coding effects of circular RNA and Foxo3 pseudogene in the inhibition of tumor growth and angiogenesis. Oncogene 35, 3919–3931 (2016).
Lin, H. et al. Unregulated miR-96 induces cell proliferation in human breast cancer by downregulating transcriptional factor FOXO3a. PLoS ONE 5, e15797 (2010).
Jeong, O.-S. et al. Long noncoding RNA linc00598 regulates CCND2 transcription and modulates the G1 checkpoint. Sci. Rep. 6, 32172 (2016).
Xi, J., Feng, J., Li, Q., Li, X. & Zeng, S. The long non-coding RNA lncFOXO1 suppresses growth of human breast cancer cells through association with BAP1. Int. J. Oncol. 50, 1663–1670 (2017).
Du, W. W. et al. Foxo3 circular RNA retards cell cycle progression via forming ternary complexes with p21 and CDK2. Nucleic Acids Res. 44, 2846–2858 (2016).
Du, W. W. et al. Foxo3 circular RNApromotes cardiac senescence by modulating multiple factors associated with stress and senescence responses. Eur. Heart J. 38, 1402–1412 (2017).
Brunet, A. et al. Protein kinase SGK mediates survival signals by phosphorylating the forkhead transcription factor FKHRL1 (FOXO3a). Mol. Cell. Biol. 21, 952–965 (2001).
Yang, J. Y. et al. ERK promotes tumorigenesis by inhibiting FOXO3a via MDM2-mediated degradation. Nat. Cell Biol. 10, 138–148 (2008).
Brown, A. K. & Webb, A. E. Regulation of FOXO factors in mammalian cells. Curr. Top. Dev. Biol. 127, 165–192 (2018).
Ho, K.-K. et al. Phosphorylation of FOXO3a on Ser-7 by p38 promotes Its nuclear localization in response to doxorubicin. J. Biol. Chem. 287, 1545–1555 (2012).
Woods, Y. L. et al. The kinase DYRK1A phosphorylates the transcription factor FKHR at Ser329 in vitro, a novel in vivo phosphorylation site. Biochem. J. 355, 597–607 (2001).
Soundararajan, M. et al. Structures of Down syndrome kinases, DYRKs, reveal mechanisms of kinase activation and substrate recognition. Structure 21, 986–996 (2013).
Hong, S.-H. et al. Minibrain/Dyrk1a regulates food intake through the Sir2-FOXO-sNPF/NPY Pathway in drosophila and mammals. PLoS Genet. 8, e1002857 (2012).
Hu, M. C. T. et al. IkappaB kinase promotes tumorigenesis through inhibition of forkhead FOXO3a. Cell 117, 225–237 (2004).
Huang, H., Regan, K. M., Lou, Z., Chen, J. & Tindall, D. J. CDK2-dependent phosphorylation of FOXO1 as an apoptotic response to DNA damage. Science 314, 294–297 (2006).
Yuan, Z. et al. Activation of FOXO1 by Cdk1 in cycling cells and postmitotic neurons. Science 319, 1665–1668 (2008).
Liu, P., Kao, T. P. & Huang, H. CDK1 promotes cell proliferation and survival via phosphorylation and inhibition of FOXO1 transcription factor. Oncogene 27, 4733–4744 (2008).
Flotow, H. et al. Phosphate groups as substrate determinants for casein kinase I action. J. Biol. Chem. 265, 14264–14269 (1990).
Rena, G. et al. Two novel phosphorylation sites on FKHR that are critical for its nuclear exclusion. EMBO J. 21, 2263–2271 (2002).
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999).
Greer, E. L. et al. The energy sensor AMP-activated protein kinase directly regulates the mammalian FOXO3 transcription factor. J. Biol. Chem. 282, 30107–30119 (2007).
Essers, M. A. et al. FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J. 23, 4802–4812 (2004).
Sunayama, J., Tsuruta, F., Masuyama, N. & Gotoh, Y. JNK antagonizes Akt-mediated survival signals by phosphorylating 14-3-3. J. Cell Biol. 170, 295–304 (2005).
Lehtinen, M. K. et al. A conserved MST-FOXO signaling pathway mediates oxidative-stress responses and extends life span. Cell 125, 987–1001 (2006).
Singh, A. et al. Protein phosphatase 2A reactivates FOXO3a through a dynamic interplay with 14-3-3 and AKT. Mol. Biol. Cell 21, 1140–1152 (2010).
Yan, L. et al. PP2A regulates the pro-apoptotic activity of FOXO1. J. Biol. Chem. 283, 7411–7420 (2008).
Manning, B. D. & Toker, A. AKT/PKB signaling: navigating the network. Cell 169, 381–405 (2017).
van der Horst, A. et al. FOXO4 is acetylated upon peroxide stress and deacetylated by the longevity protein hSir2(SIRT1). J. Biol. Chem. 279, 28873–28879 (2004).
Bertaggia, E., Coletto, L. & Sandri, M. Posttranslational modifications control FoxO3 activity during denervation. Am. J. Physiol. Cell Physiol. 302, 587–596 (2012).
Motta, M. C. et al. Mammalian SIRT1 represses forkhead transcription factors. Cell 116, 551–563 (2004).
Brunet, A. et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303, 2011–2015 (2004).
Kobayashi, Y. et al. SIRT1 is critical regulator of FOXO-mediated transcription in response to oxidative stress. Int. J. Mol. Med. 16, 237–243 (2005).
Matsuzaki, H. et al. Acetylation of Foxo1 alters its DNA-binding ability and sensitivity to phosphorylation. Proc. Natl Acad. Sci. USA 102, 11278–11283 (2005).
Mahmud, Z. et al. EP300 and SIRT1/6 Co-regulate lapatinib sensitivity via modulating FOXO3-acetylation and activity in breast cancer. Cancers 11, 1067 (2019).
Aimjongjun, S. et al. Lapatinib sensitivity in nasopharyngeal carcinoma is modulated by SIRT2-mediated FOXO3 deacetylation. BMC Cancer 19, 1106 (2019).
Sun, W. et al. Overexpression of Sirt1 in mesenchymal stem cells protects against bone loss in mice by FOXO3a deacetylation and oxidative stress inhibition. Metabolism 88, 61–71 (2018).
Qiang, L., Banks, A. S. & Accili, D. Uncoupling of acetylation from phosphorylation regulates FoxO1 function independent of its subcellular localization. J. Biol. Chem. 285, 27396–27401 (2010).
Huang, H. et al. Skp2 inhibits FOXO1 in tumor suppression through ubiquitin-mediated degradation. Proc. Natl Acad. Sci. USA 102, 1649–1654 (2005).
Li, F. et al. C terminus of Hsc70-interacting protein promotes smooth muscle cell proliferation and survival through ubiquitin-mediated degradation of FoxO1. J. Biol. Chem. 284, 20090–20098 (2009).
Kato, S., Ding, J., Pisck, E., Jhala, U. S. & Du, K. COP1 functions as a FoxO1 ubiquitin E3 ligase to regulate FoxO1-mediated gene expression. J. Biol. Chem. 283, 35464–35473 (2008).
van der Horst, A. et al. FOXO4 transcriptional activity is regulated by monoubiquitination and USP7/HAUSP. Nat. Cell Biol. 8, 1064–1073 (2006).
Yamagata, K. et al. Arginine methylation of FOXO transcription factors inhibits their phosphorylation by Akt. Mol. Cell 32, 221–231 (2008).
Takahashi, Y. et al. Asymmetric arginine dimethylation determines life span in C. elegans by regulating forkhead transcription factor DAF-16. Cell Metab. 13, 505–516 (2011).
Xie, Q. et al. Lysine methylation of FOXO3 regulates oxidative stress-induced neuronal cell death. EMBO Rep. 13, 371–377 (2012).
Calnan, D. R. et al. Methylation by Set9 modulates FoxO3 stability and transcriptional activity. Aging 4, 462–479 (2012).
Wang, F. et al. Deacetylation of FOXO3 by SIRT1 or SIRT2 leads to Skp2-mediated FOXO3 ubiquitination and degradation. Oncogene 31, 1546–1557 (2012).
Daitoku, H., Sakamaki, J. & Fukamizu, A. Regulation of FoxO transcription factors by acetylation and protein–protein interactions. Biochim. Biophys. Acta 1813, 1954–1960 (2011).
Zhao, H. H. et al. Forkhead homologue in rhabdomyosarcoma functions as a bifunctional nuclear receptor-interacting protein with both coactivator and corepressor functions. J. Biol. Chem. 276, 27907–27912 (2001).
Li, P. et al. AKT-independent protection of prostate cancer cells from apoptosis mediated through complex formation between the androgen receptor and FKHR. Mol. Cell. Biol. 23, 104–118 (2003).
Fan, W. et al. Insulin-like growth factor 1/insulin signaling activates androgen signaling through direct interactions of Foxo1 with androgen receptor. J. Biol. Chem. 282, 7329–7338 (2007).
Schuur, E. R. et al. Ligand-dependent interaction of estrogen receptor-alpha with members of the forkhead transcription factor family. J. Biol. Chem. 276, 33554–33560 (2001).
von Groote-Bidlingmaier, F. et al. DYRK1 is a co-activator of FKHR (FOXO1a)-dependent glucose-6-phosphatase gene expression. Biochem. Biophys. Res. Commun. 300, 764–769 (2003).
Dowell, P., Otto, T. C., Adi, S. & Lane, M. D. Convergence of peroxisome proliferator-activated receptor γ and foxo1 signaling pathways. J. Biol. Chem. 278, 45485–45491 (2003).
Armoni, M. et al. FOXO1 represses peroxisome proliferator-activated receptor-gamma1 and -gamma2 gene promoters in primary adipocytes. A novel paradigm to increase insulin sensitivity. J. Biol. Chem. 281, 19881–19891 (2006).
Puigserver, P. et al. Insulin-regulated hepatic gluconeogenesis through FOXO1–PGC-1α interaction. Nature 423, 550–555 (2003).
Sandri, M. et al. PGC-1 protects skeletal muscle from atrophy by suppressing FoxO3 action and atrophy-specific gene transcription. Proc. Natl Acad. Sci. USA 103, 16260–16265 (2006).
Seoane, J., Le, H.-V., Shen, L., Anderson, S. A. & Massagué, J. Integration of Smad and forkhead pathways in the control of neuroepithelial and glioblastoma cell proliferation. Cell 117, 211–223 (2004).
Qiao, L. & Shao, J. SIRT1 regulates adiponectin gene expression through Foxo1-C/enhancer-binding protein alpha transcriptional complex. J. Biol. Chem. 281, 39915–39924 (2006).
Grinius, L., Kessler, C., Schroeder, J. & Handwerger, S. Forkhead transcription factor FOXO1A is critical for induction of human decidualization. J. Endocrinol. 189, 179–187 (2006).
Hoogeboom, D. et al. Interaction of FOXO with beta-catenin inhibits beta-catenin/T cell factor activity. J. Biol. Chem. 283, 9224–9230 (2008).
Tenbaum, S. P. et al. beta-catenin confers resistance to PI3K and AKT inhibitors and subverts FOXO3a to promote metastasis in colon cancer. Nat. Med. 18, 892–901 (2012).
Sakamaki, J., Daitoku, H., Yoshimochi, K., Miwa, M. & Fukamizu, A. Regulation of FOXO1-mediated transcription and cell proliferation by PARP-1. Biochem. Biophys. Res. Commun. 382, 497–502 (2009).
Li, J., Du, W., Maynard, S., Andreassen, P. R. & Pang, Q. Oxidative stress-specific interaction between FANCD2 and FOXO3a. Blood 115, 1545–1548 (2010).
Wang, F. et al. Biochemical and structural characterization of an intramolecular interaction in FOXO3a and its binding with p53. J. Mol. Biol. 384, 590–603 (2008).
Tsai, W.-B., Chung, Y. M., Takahashi, Y., Xu, Z. & Hu, M. C.-T. Functional interaction between FOXO3a and ATM regulates DNA damage response. Nat. Cell Biol. 10, 460–467 (2008).
Zhao, Y. et al. Cytosolic FoxO1 is essential for the induction of autophagy and tumour suppressor activity. Nat. Cell Biol. 12, 665–675 (2010).
You, H., Yamamoto, K. & Mak, T. W. Regulation of transactivation-independent proapoptotic activity of p53 by FOXO3a. Proc. Natl Acad. Sci. USA 103, 9051–9056 (2006).
Galili, N. et al. Fusion of a fork head domain gene to PAX3 in the solid tumour alveolar rhabdomyosarcoma. Nat. Genet. 5, 230–235 (1993).
Davis, R. J., D’Cruz, C. M., Lovell, M. A., Biegel, J. A. & Barr, F. G. Fusion of PAX7 to FKHR by the Variant t(l;13)(p36;ql4) translocation in alveolar rhabdomyosarcoma. Cancer Res. 54, 2869–2872 (1994).
Slany, R. K. The molecular biology of mixed lineage leukemia. Haematologica 94, 984–993 (2009).
Hennessy, B. T., Smith, D. L., Ram, P. T., Lu, Y. & Mills, G. B. Exploiting the PI3K/AKT pathway for cancer drug discovery. Nat. Rev. Drug Discov. 4, 988–1004 (2005).
Hill, R. et al. A novel phosphatidylinositol 3-kinase (PI3K) inhibitor directs a potent FOXO-dependent, p53-independent cell cycle arrest phenotype characterized by the differential induction of a subset of FOXO-regulated genes. Breast Cancer Res. 16, 482 (2014).
Chapuis, N. et al. IκB kinase overcomes PI3K/Akt and ERK/MAPK to control FOXO3a activity in acute myeloid leukemia. Blood 116, 4240–4250 (2010).
Yang, H., Zhao, R., Yang, H.-Y. & Lee, M.-H. Constitutively active FOXO4 inhibits Akt activity, regulates p27 Kip1 stability, and suppresses HER2-mediated tumorigenicity. Oncogene 24, 1924–1935 (2005).
Hu, T. et al. Reprogramming ovarian and breast cancer cells into non-cancerous cells by low-dose metformin or SN-38 through FOXO3 activation. Sci. Rep. 4, 5810 (2014).
Habashy, H. O. et al. FOXO3a nuclear localisation is associated with good prognosis in luminal-like breast cancer. Breast Cancer Res. Treat. 129, 11–21 (2011).
Jiang, Y., Zou, L., Lu, W.-Q., Zhang, Y. & Shen, A.-G. Foxo3a expression is a prognostic marker in breast cancer. PLoS ONE 8, e70746 (2013).
Fei, M. et al. Low expression of Foxo3a is associated with poor prognosis in ovarian cancer patients. Cancer Invest. 27, 52–59 (2009).
Schmitt-Ney, M. The FOXO’s advantages of being a family: considerations on function and evolution. Cell 9, 787 (2020).
Chung, S. Y. et al. FoxO6 and PGC-1α form a regulatory loop in myogenic cells. Biosci. Rep. 33, 485–500 (2020).
Kitamura, T. The role of FOXO1 in β-cell failure and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 9, 615–623 (2013).
Sykes, S. M. et al. AKT/FOXO signaling enforces reversible differentiation blockade in myeloid leukemias. Cell 146, 697–708 (2011).
Wang, J.-H. et al. Elevated FOXO6 expression correlates with progression and prognosis in gastric cancer. Oncotarget 8, 31682–31691 (2017).
Qinyu, L. et al. FOXO6 promotes gastric cancer cell tumorigenicity via upregulation of C-myc. FEBS Lett. 587, 2105–2111 (2013).
Beretta, G. L. et al. Role of FoxO proteins in cellular response to antitumor agents. Cancers 11, 90 (2019).
Jiramongkol, Y. & Lam, E. W.-F. FOXO transcription factor family in cancer and metastasis. Cancer Metastasis Rev. 39, 681–709 (2020).
Wang, J. et al. Thioredoxin 1 upregulates FOXO1 transcriptional activity in drug resistance in ovarian cancer cells. Biochim. Biophys. Acta Mol. Basis Dis. 1852, 395–405 (2015).
Fernández de Mattos, S., Villalonga, P., Clardy, J. & Lam, E. W.-F. FOXO3a mediates the cytotoxic effects of cisplatin in colon cancer cells. Mol. Cancer Ther. 7, 3237–3246 (2008).
Han, C.-Y., Cho, K.-B., Choi, H.-S., Han, H.-K. & Kang, K.-W. Role of FoxO1 activation in MDR1 expression in adriamycin-resistant breast cancer cells. Carcinogenesis 29, 1837–1844 (2008).
Kajihara, T. et al. Differential expression of FOXO1 and FOXO3a confers resistance to oxidative cell death upon endometrial decidualization. Mol. Endocrinol. 20, 2444–2455 (2006).
Alasiri, G. et al. Regulation of PERK expression by FOXO3: a vulnerability of drug-resistant cancer cells. Oncogene 38, 6382–6398 (2019).
Hui, R. C.-Y. et al. Doxorubicin activates FOXO3a to induce the expression of multidrug resistance gene ABCB1 (MDR1) in K562 leukemic cells. Mol. Cancer Ther. 7, 670–678 (2008).
Hui, R. C.-Y. et al. The forkhead transcription factor FOXO3a increases phosphoinositide-3 kinase/Akt activity in drug-resistant leukemic cells through induction of PIK3CA expression. Mol. Cell. Biol. 28, 5886–5898 (2008).
Chen, C. C. et al. FoxOs inhibit mTORC1 and activate Akt by inducing the expression of Sestrin3 and rictor. Dev. Cell 18, 592–604 (2010).
Hoekman, M. F. M., Jacobs, F. M. J., Smidt, M. P. & Burbach, J. P. H. Spatial and temporal expression of FoxO transcription factors in the developing and adult murine brain. Gene Expr. Patterns 6, 134–140 (2006).
Santo, E. E. & Paik, J. FOXO in neural cells and diseases of the nervous system. in Current Topics in Developmental Biology Vol. 127 105–118 (Academic Press Inc., 2018).
Renault, V. M. et al. FoxO3 regulates neural stem cell homeostasis. Cell Stem Cell 5, 527–539 (2009).
Hwang, I. et al. FOXO protects against age-progressive axonal degeneration. Aging Cell 17, e12701 (2018).
Salih, D. A. M. et al. FoxO6 regulates memory consolidation and synaptic function. Genes Dev. 26, 2780–2801 (2012).
Sanphui, P. & Biswas, S. C. FoxO3a is activated and executes neuron death via Bim in response to β-amyloid. Cell Death Dis. https://doi.org/10.1038/cddis.2013.148 (2013).
Pino, E. et al. FOXO3 determines the accumulation of alpha-synuclein and controls the fate of dopaminergic neurons in the substantia nigra. Hum. Mol. Genet. 23, 1435–1452 (2014).
Vidal, R. L. et al. Targeting the UPR transcription factor XBP1 protects against Huntington’s disease through the regulation of FoxO1 and autophagy. Hum. Mol. Genet. 21, 2245–2262 (2012).
Pajvani, U. B. & Accili, D. The new biology of diabetes. Diabetologia 58, 2459–2468 (2015).
Nakae, J. et al. Regulation of insulin action and pancreatic β-cell function by mutated alleles of the gene encoding forkhead transcription factor Foxo1. Nat. Genet. 32, 245–253 (2002).
Talchai, C., Xuan, S., Kitamura, T., DePinho, R. A. & Accili, D. Generation of functional insulin-producing cells in the gut by Foxo1 ablation. Nat. Genet. 44, 406–412 (2012).
Bouchi, R. et al. FOXO1 inhibition yields functional insulin-producing cells in human gut organoid cultures. Nat. Commun. 5, 4242 (2014). This article shows that genetic FOXO1 inhibition promotes the generation of insulin-positive cells in gut organoids derived from human induced pluripotent stem cells.
Yu, F. et al. FoxO1 inhibition promotes differentiation of human embryonic stem cells into insulin producing cells. Exp. Cell Res. 362, 227–234 (2018).
Haeusler, R. A. et al. Integrated control of hepatic lipogenesis versus glucose production requires FoxO transcription factors. Nat. Commun. 5, 5109 (2014).
Talchai, C., Xuan, S., Lin, H. V., Sussel, L. & Accili, D. Pancreatic β cell dedifferentiation as a mechanism of diabetic β cell failure. Cell 150, 1223–1234 (2012).
Puig, O. & Tjian, R. Transcriptional feedback control of insulin receptor by dFOXO/FOXO1. Genes Dev. 19, 2435–2446 (2005).
O’Neill, B. T. et al. FoxO transcription factors are critical regulators of diabetes-related muscle atrophy. Diabetes 68, 556–570 (2019).
Battiprolu, P. K. et al. Metabolic stress-induced activation of FoxO1 triggers diabetic cardiomyopathy in mice. J. Clin. Invest. 122, 1109–1118 (2012).
Qi, Y. et al. Activation of Foxo1 by insulin resistance promotes cardiac dysfunction and β–myosin heavy chain gene expression. Circ. Hear. Fail. 8, 198–208 (2015).
Palomer, X., Salvadó, L., Barroso, E. & Vázquez-Carrera, M. An overview of the crosstalk between inflammatory processes and metabolic dysregulation during diabetic cardiomyopathy. Int. J. Cardiol. 168, 3160–3172 (2013).
Hariharan, N. et al. Autophagy plays an essential role in mediating regression of hypertrophy during unloading of the heart. PLoS ONE 8, e51632 (2013).
Cao, D. J. et al. Mechanical unloading activates FoxO3 to trigger Bnip3-dependent cardiomyocyte atrophy. J. Am. Heart Assoc. 2, e000016 (2013).
Wei, X. et al. Myocardial hypertrophic preconditioning attenuates cardiomyocyte hypertrophy and slows progression to heart failure through upregulation of S100A8/A9. Circulation 131, 1506–1517 (2015).
Sundaresan, N. R. et al. Sirt3 blocks the cardiac hypertrophic response by augmenting Foxo3a-dependent antioxidant defense mechanisms in mice. J. Clin. Invest. 119, 2758–2771 (2009).
Sengupta, A., Molkentin, J. D. & Yutzey, K. E. FoxO transcription factors promote autophagy in cardiomyocytes. J. Biol. Chem. 284, 28319–28331 (2009).
Ni, Y. G. et al. Foxo transcription factors blunt cardiac hypertrophy by inhibiting calcineurin signaling. Circulation 114, 1159–1168 (2006).
Tucka, J. et al. Akt1 regulates vascular smooth muscle cell apoptosis through FoxO3a and Apaf1 and protects against arterial remodeling and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 34, 2421–2428 (2014).
Schips, T. G. et al. FoxO3 induces reversible cardiac atrophy and autophagy in a transgenic mouse model. Cardiovasc. Res. 91, 587–597 (2011).
Zhang, M. & Zhang, X. The role of PI3K/AKT/FOXO signaling in psoriasis. Arch. Dermatol. Res. 311, 83–91 (2019).
Liu, Y., Luo, W. & Chen, S. Comparison of gene expression profiles reveals aberrant expression of FOXO1, Aurora A/B and EZH2 in lesional psoriatic skins. Mol. Biol. Rep. 38, 4219–4224 (2011).
WO2014157380A1 - Therapeutic agent for wounds or fibrosis - Google Patents. Google https://patents.google.com/patent/WO2014157380A1/en?oq=FOXO1 (2014).
Zhang, C. et al. FOXO1 differentially regulates both normal and diabetic wound healing. J. Cell Biol. 209, 289–303 (2015).
Xu, F. et al. Foxo1 inhibits diabetic mucosal wound healing but enhances healing of normoglycemic Wounds. Diabetes 64, 243–256 (2015).
Kenyon, C., Chang, J., Gensch, E., Rudner, A. & Tabtiang, R. A C. elegans mutant that lives twice as long as wild type. Nature 366, 461–464 (1993).
Kimura, K. D., Tissenbaum, H. A., Liu, Y. & Ruvkun, G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans. Science 277, 942–946 (1997).
Boehm, A. M. et al. FoxO is a critical regulator of stem cell maintenance in immortal Hydra. Proc. Natl Acad. Sci. USA 109, 19697–19702 (2012).
Kenyon, C. J. The genetics of ageing. Nature 464, 504–512 (2010).
Fontana, L., Partridge, L. & Longo, V. D. Extending healthy life span–from yeast to humans. Science 328, 321–326 (2010).
Birnbaum, A., Wu, X., Tatar, M., Liu, N. & Bai, H. Age-dependent changes in transcription factor FoxO targeting in female Drosophila. Front. Genet. 10, 321 (2019).
Tothova, Z. et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell 128, 325–339 (2007).
Shimokawa, I. et al. The life-extending effect of dietary restriction requires Foxo3 in mice. Aging Cell 14, 707–709 (2015).
Yamaza, H. et al. FoxO1 is involved in the antineoplastic effect of calorie restriction. Aging Cell 9, 372–382 (2010).
Martins, R., Lithgow, G. J. & Link, W. Long live FOXO: unraveling the role of FOXO proteins in aging and longevity. Aging Cell 15, 196–207 (2016).
Willcox, B. J. et al. FOXO3A genotype is strongly associated with human longevity. Proc. Natl Acad. Sci. USA 105, 13987–13992 (2008). This paper is the first report of a polymorphism in the FOXO3 gene associated with human longevity.
Soerensen, M. et al. Association study of FOXO3A SNPs and aging phenotypes in Danish oldest-old individuals. Aging Cell 14, 60–66 (2015).
Brooks-Wilson, A. R. Genetics of healthy aging and longevity. Hum. Genet. 132, 1323–1338 (2013).
Donlon, T. A. et al. FOXO3 gene variants and human aging: coding variants may not be key players. J. Gerontol. A Biol. Sci. Med. Sci. 67, 1132–1139 (2012).
Murabito, J. M., Yuan, R. & Lunetta, K. L. The search for longevity and healthy aging genes: insights from epidemiological studies and samples of long-lived individuals. J. Gerontol. A Biol. Sci. Med. Sci. 67, 470–479 (2012).
Li, Y. et al. Genetic association of FOXO1A and FOXO3A with longevity trait in Han Chinese populations. Hum. Mol. Genet. 18, 4897–4904 (2009).
Willcox, B. J. et al. The FoxO3 gene and cause-specific mortality. Aging Cell 15, 617–624 (2016).
Patel, M. N., Halling-Brown, M. D., Tym, J. E., Workman, P. & Al-Lazikani, B. Objective assessment of cancer genes for drug discovery. Nat. Rev. Drug Discov. 12, 35–50 (2013).
Infante, P. et al. Gli1/DNA interaction is a druggable target for Hedgehog-dependent tumors. EMBO J. 34, 200–217 (2015).
Moellering, R. E. et al. Direct inhibition of the NOTCH transcription factor complex. Nature 462, 182–188 (2009).
Lambert, M. et al. Targeting transcription factors for cancer treatment. Molecules 23, 1479 (2018).
Biggs III, W. H., Meisenhelder, J., Hunter, T., Cavenee, W. K. & Arden, K. C. Protein kinase B/Akt-mediated phosphorylation promotes nuclear exclusion of the winged helix transcription factor FKHR1. Proc. Natl Acad. Sci. USA 96, 7421–7426 (1999).
Putker, M. et al. Redox-dependent control of FOXO/DAF-16 by transportin-1. Mol. Cell 49, 730–742 (2013).
Kudo, N. et al. Leptomycin B inactivates CRM1/exportin 1 by covalent modification at a cysteine residue in the central conserved region. Proc. Natl Acad. Sci. USA 96, 9112–9117 (1999).
Kudo, N. et al. Leptomycin B inhibition of signal-mediated nuclear export by direct binding to CRM1. Exp. Cell Res. 242, 540–547 (1998).
Newlands, E. S., Rustin, G. J. & Brampton, M. H. Phase I trial of elactocin. Br. J. Cancer 74, 648–649 (1996).
Jans, D. A., Martin, A. J. & Wagstaff, K. M. Inhibitors of nuclear transport. Curr. Opin. Cell Biol. 58, 50–60 (2019).
Parikh, K., Cang, S., Sekhri, A. & Liu, D. Selective inhibitors of nuclear export (SINE)-A novel class of anti-cancer agents. J. Hematol. Oncol. 7, 78 (2014).
Kau, T. R. et al. A chemical genetic screen identifies inhibitors of regulated nuclear export of a Forkhead transcription factor in PTEN-deficient tumor cells. Cancer Cell 4, 463–476 (2003). This study reports the first screening of a compound library based on the subcellular localization of FOXO1.
Zanella, F. et al. An HTS approach to screen for antagonists of the nuclear export machinery using high content cell-based assays. Assay Drug Dev. Technol. 5, 333–341 (2007).
Link, W. et al. Chemical interrogation of FOXO3a nuclear translocation identifies potent and selective inhibitors of phosphoinositide 3-kinases. J. Biol. Chem. 284, 28392–28400 (2009). This study reports the identification and optimization of a PI3K inhibitor using a high content screening approach based on FOXO3 subcellular localization for the first time.
Abedinpour, P. et al. Plumbagin improves the efficacy of androgen deprivation therapy in prostate cancer: A pre-clinical study. Prostate 77, 1550–1562 (2017).
Cautain, B. et al. High-content screening of natural products reveals novel nuclear export inhibitors. J. Biomol. Screen. 19, 57–65 (2014).
Zhang, Z. et al. Octahydrocurcumin, a final hydrogenated metabolite of curcumin, possesses superior anti-tumor activity through induction of cellular apoptosis. Food Funct. 9, 2005–2014 (2018).
Das, A., Wei, G., Parikh, K. & Liu, D. Selective inhibitors of nuclear export (SINE) in hematological malignancies. Exp. Hematol. Oncol. 4, 7 (2015).
Chari, A. et al. Oral selinexor–dexamethasone for triple-class refractory multiple myeloma. N. Engl. J. Med. 381, 727–738 (2019).
Corno, C. et al. FoxO-1 contributes to the efficacy of the combination of the XPO1 inhibitor selinexor and cisplatin in ovarian carcinoma preclinical models. Biochem. Pharmacol. 147, 93–103 (2018).
Yasgar, A., Jadhav, A., Simeonov, A. & Coussens, N. P. in Methods in Molecular Biology Vol. 1439 77–98 (Humana Press Inc., 2016).
Fraser, J. E. et al. A nuclear transport inhibitor that modulates the unfolded protein response and provides in vivo protection against lethal dengue virus infection. J. Infect. Dis. 210, 1780–1791 (2014).
Wagstaff, K. M. et al. Molecular dissection of an inhibitor targeting the HIV integrase dependent preintegration complex nuclear import. Cell. Microbiol. 21, e12953 (2019).
Nakae, J., Park, B. C. & Accili, D. Insulin stimulates phosphorylation of the forkhead transcription factor FKHR on serine 253 through a wortmannin-sensitive pathway. J. Biol. Chem. 274, 15982–15985 (1999).
Zanella, F., Rosado, A., Garcia, B., Carnero, A. & Link, W. Chemical genetic analysis of FOXO nuclear-cytoplasmic shuttling by using image-based cell screening. Chembiochem 9, 2229–2237 (2008).
Granda, T. G. et al. Biological characterization of ETP-46321 a selective and efficacious inhibitor of phosphoinositide-3-kinases. Invest. N. Drugs 31, 66–76 (2013).
Martinez Gonzalez, S. et al. Imidazo[1,2-a]pyrazines as novel PI3K inhibitors. Bioorg Med. Chem. Lett. 22, 1874–1878 (2012).
Martinez Gonzalez, S. et al. Identification of ETP-46321, a potent and orally bioavailable PI3K alpha, delta inhibitor. Bioorg Med. Chem. Lett. 22, 3460–3466 (2012).
Saxton, R. A. & Sabatini, D. M. mTOR Signaling in Growth, Metabolism, and Disease. Cell 168, 960–976 (2017).
Feehan, R. P., Nelson, A. M. & Shantz, L. M. Inhibition of mTORC2 enhances UVB-induced apoptosis in keratinocytes through a mechanism dependent on the FOXO3a transcriptional target NOXA but independent of TRAIL. Cell. Signal. 52, 35–47 (2018).
Janes, M. R. et al. Efficacy of the investigational mTOR kinase inhibitor MLN0128/INK128 in models of B-cell acute lymphoblastic leukemia. Leukemia 27, 586–594 (2013).
Zheng, B. et al. Pre-clinical evaluation of AZD-2014, a novel mTORC1/2 dual inhibitor, against renal cell carcinoma. Cancer Lett. 357, 468–475 (2015).
Sarbassov, D. D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005).
Davies, B. R. et al. Preclinical pharmacology of AZD5363, an inhibitor of AKT: Pharmacodynamics, antitumor activity, and correlation of monotherapy activity with genetic background. Mol. Cancer Ther. 11, 873–887 (2012).
Ching, T.-T., Chiang, W.-C., Chen, C.-S. & Hsu, A.-L. Celecoxib extends C. elegans lifespan via inhibition of insulin-like signaling but not cyclooxygenase-2 activity. Aging Cell 10, 506 (2011).
Sunters, A. et al. Paclitaxel-induced nuclear translocation of FOXO3a in breast cancer cells is mediated by c-Jun NH 2 -terminal kinase and Akt. Cancer Res. 66, 212–220 (2006).
Zhou, G. et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 (2001).
Hou, X. et al. Metformin reduces intracellular reactive oxygen species levels by upregulating expression of the antioxidant thioredoxin via the AMPK-FOXO3 pathway. Biochem. Biophys. Res. Commun. 396, 199–205 (2010).
Takayama, H. et al. Metformin suppresses expression of the selenoprotein P gene via an AMP-activated kinase (AMPK)/FoxO3a pathway in H4IIEC3 hepatocytes. J. Biol. Chem. 289, 335–345 (2014).
Scott, D. E., Bayly, A. R., Abell, C. & Skidmore, J. Small molecules, big targets: Drug discovery faces the protein-protein interaction challenge. Nat. Rev. Drug Discov. 15, 533–550 (2016).
Bushweller, J. H. Targeting transcription factors in cancer — from undruggable to reality. Nat. Rev. Cancer https://doi.org/10.1038/s41568-019-0196-7 (2019).
Bouhlel, M., Lambert, M. & David-Cordonnier, M.-H. Targeting transcription factor binding to DNA by competing with DNA binders as an approach for controlling gene expression. Curr. Top. Med. Chem. 15, 1323–1358 (2015).
Bouck, D. C., Shu, P., Cui, J., Shelat, A. & Chen, T. A high-content screen identifies inhibitors of nuclear export of forkhead transcription factors. J. Biomol. Screen. 16, 394–404 (2011).
Erkizan, H. V. et al. A small molecule blocking oncogenic protein EWS-FLI1 interaction with RNA helicase A inhibits growth of Ewing’s sarcoma. Nat. Med. 15, 750–756 (2009).
Zhang, Z. et al. Chemical perturbation of an intrinsically disordered region of TFIID distinguishes two modes of transcription initiation. eLife 4, e07777 (2015).
Mori, M. et al. Discovery of 14-3-3 Protein-protein interaction inhibitors that sensitize multidrug-resistant cancer cells to doxorubicin and the Akt inhibitor GSK690693. ChemMedChem 9, 973–983 (2014).
Burslem, G. M. & Crews, C. M. Proteolysis-targeting chimeras as therapeutics and tools for biological discovery. Cell https://doi.org/10.1016/j.cell.2019.11.031 (2020).
Potente, M. et al. Involvement of Foxo transcription factors in angiogenesis and postnatal neovascularization. J. Clin. Invest. 115, 2382–2392 (2005).
Keniry, M. et al. Survival factor NFIL3 restricts FOXO-induced gene expression in cancer. Genes Dev. 27, 916–927 (2013).
Hosaka, T. et al. Disruption of forkhead transcription factor (FOXO) family members in mice reveals their functional diversification. Proc. Natl Acad. Sci. USA 101, 2975–2980 (2004).
Schroeder, F. C., Kau, T. R., Silver, P. A. & Clardy, J. The psammaplysenes, specific inhibitors of FOXO1a nuclear export. J. Nat. Prod. 68, 574–576 (2005).
Liu, X. et al. CRM1/XPO1 is associated with clinical outcome in glioma and represents a therapeutic target by perturbing multiple core pathways. J. Hematol. Oncol. 9, 108 (2016).
Serra, V. et al. NVP-BEZ235, a Dual PI3K/mTOR inhibitor, prevents PI3K signaling and inhibits the growth of cancer cells with activating PI3K mutations. Cancer Res. 68, 8022–8030 (2008).
Frescas, D., Valenti, L. & Accili, D. Nuclear trapping of the forkhead transcription factor FoxO1 via sirt-dependent deacetylation promotes expression of glucogenetic genes. J. Biol. Chem. 280, 20589–20595 (2005).
Chen, X., Luo, X. & Cheng, Y. Trifluoperazine prevents FOXO1 nuclear excretion and reverses doxorubicin-resistance in the SHG44/DOX drug-resistant glioma cell line. Int. J. Mol. Med. 42, 3300–3308 (2018).
Carbajo-Pescador, S. et al. Melatonin induces transcriptional regulation of Bim by FoxO3a in HepG2 cells. Br. J. Cancer 108, 442–449 (2013).
Liu, Z. et al. Piperlongumine-induced nuclear translocation of the FOXO3A transcription factor triggers BIM-mediated apoptosis in cancer cells. Biochem. Pharmacol. 163, 101–110 (2019).
Park, S.-H. et al. Pharmacological activation of FOXO3 suppresses triple-negative breast cancer in vitro and in vivo. Oncotarget 7, 42110–42125 (2016).
Jagani, Z. et al. Proteasome inhibition causes regression of leukemia and abrogates BCR-ABL-induced evasion of apoptosis in part through regulation of forkhead tumor suppressors. Cancer Res. 69, 6546–6555 (2009).
Pan, Q., Xie, X., Guo, Y. & Wang, H. Simvastatin promotes cardiac microvascular endothelial cells proliferation, migration and survival by phosphorylation of p70 S6K and FoxO3a. Cell Biol. Int. 38, 599–609 (2014).
Polter, A. et al. Forkhead box, class O transcription factors in brain: regulation and behavioral manifestation. Biol. Psychiatry 65, 150–159 (2009).
Wang, H. et al. The role of Akt/FoxO3a in the protective effect of venlafaxine against corticosterone-induced cell death in PC12 cells. Psychopharmacology 228, 129–141 (2013).
Burgering, B. M. T. A brief introduction to FOXOlogy. Oncogene 27, 2258–2262 (2008).
Jacobs, F. M. J. et al. FoxO6, a novel member of the FoxO class of transcription factors with distinct shuttling dynamics. J. Biol. Chem. 278, 35959–35967 (2003).
Furuyama, T., Nakazawa, T., Nakano, I. & Mori, N. Identification of the differential distribution patterns of mRNAs and consensus binding sequences for mouse DAF-16 homologues. Biochem. J. 349, 629 (2000).
Kops, G. J. P. L. et al. Control of cell cycle exit and entry by protein kinase B-regulated forkhead transcription factors. Mol. Cell. Biol. 22, 2025–2036 (2002).
Schmidt, M. et al. Cell cycle inhibition by FoxO forkhead transcription factors involves downregulation of cyclin D. Mol. Cell. Biol. 22, 7842–7852 (2002).
Martínez-Gac, L., Marqués, M., García, Z., Campanero, M. R. & Carrera, A. C. Control of cyclin G2 mRNA expression by forkhead transcription factors: novel mechanism for cell cycle control by phosphoinositide 3-kinase and forkhead. Mol. Cell. Biol. 24, 2181–2189 (2004).
Medema, R. H., Kops, G. J. P. L., Bos, J. L. & Burgering, B. M. T. AFX-like Forkhead transcription factors mediate cell-cycle regulation by Ras and PKB through p27kip1. Nature 404, 782–787 (2000).
Dijkers, P. F., Medema, R. H., Lammers, J.-W. J., Koenderman, L. & Coffer, P. J. Expression of the pro-apoptotic Bcl-2 family member Bim is regulated by the forkhead transcription factor FKHR-L1. Curr. Biol. 10, 1201–1204 (2000).
Dijkers, P. F. et al. FKHR-L1 can act as a critical effector of cell death induced by cytokine withdrawal. J. Cell Biol. 156, 531–542 (2002).
Hornsveld, M. et al. Restraining FOXO3-dependent transcriptional BMF activation underpins tumour growth and metastasis of E-cadherin-negative breast cancer. Cell Death Differ. 23, 1483–1492 (2016).
Tang, T. T.-L. et al. The forkhead transcription factor AFX activates apoptosis by induction of the BCL-6 transcriptional repressor. J. Biol. Chem. 277, 14255–14265 (2002).
Su, D. et al. FoxO1 links insulin resistance to proinflammatory cytokine IL-1beta production in macrophages. Diabetes 58, 2624–2633 (2009).
Fan, W. et al. FoxO1 regulates Tlr4 inflammatory pathway signalling in macrophages. EMBO J. 29, 4223–4236 (2010).
Zhang, W. et al. Integrated regulation of hepatic lipid and glucose metabolism by adipose triacylglycerol lipase and FoxO proteins. Cell Rep. 15, 349–359 (2016).
Fan, W. et al. FOXO1 transrepresses peroxisome proliferator-activated receptor gamma transactivation, coordinating an insulin-induced feed-forward response in adipocytes. J. Biol. Chem. 284, 12188–12197 (2009).
Kops, G. J. et al. Forkhead transcription factor FOXO3a protects quiescent cells from oxidative stress. Nature 419, 316–321 (2002).
Nemoto, S. & Finkel, T. Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science 295, 2450–2452 (2002).
Dansen, T. B. et al. Regulation of sterol carrier protein gene expression by the forkhead transcription factor FOXO3a. J. Lipid Res. 45, 81–88 (2004).
Tamura, R. E. et al. GADD45 proteins: central players in tumorigenesis. Curr. Mol. Med. 12, 634–651 (2012).
Tran, H. et al. DNA repair pathway stimulated by the forkhead transcription factor FOXO3a through the Gadd45 protein. Science 296, 530–534 (2002).
Webb, A. E. & Brunet, A. FOXO transcription factors: key regulators of cellular quality control. Trends Biochem. Sci. 39, 159–169 (2014).
Milan, G. et al. Regulation of autophagy and the ubiquitin-proteasome system by the FoxO transcriptional network during muscle atrophy. Nat. Commun. 6, 6670 (2015).
Annis, D. A., Nickbarg, E., Yang, X., Ziebell, M. R. & Whitehurst, C. E. Affinity selection-mass spectrometry screening techniques for small molecule drug discovery. Curr. Opin. Chem. Biol. 11, 518–526 (2007).
Lin, K., Hsin, H., Libina, N. & Kenyon, C. Regulation of the caenorhabditis elegans longevity protein DAF-16 by insulin/IGF-1 and germline signaling. Nat. Genet. 28, 139–145 (2001).
Cautain, B. et al. Discovery of a novel, isothiazolonaphthoquinone-based small molecule activator of FOXO nuclear-cytoplasmic shuttling. PLoS ONE 11, e0167491 (2016).
Zanella, F. et al. Human TRIB2 is a repressor of FOXO that contributes to the malignant phenotype of melanoma cells. Oncogene 29, 2973–2982 (2010).
Zanella, F., Rosado, A., Garcia, B., Carnero, A. & Link, W. Using multiplexed regulation of luciferase activity and GFP translocation to screen for FOXO modulators. BMC Cell Biol. 10, 14 (2009).
Acknowledgements
This work was supported by the Spanish Ministry of Science, Innovation and Universities through Grant RTI2018-094629-B-I00 and by the European commission (INTERREG VA POCTEP-055 ALGARED_PLUS_5E) to W.L. E.W.-F.L.’s work is supported by MRC (MR/N012097/1), CRUK (C37/A12011) and Breast Cancer Now (2012MayPR070; 2012NovPhD016; 2014NovPhD326).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
W.L. is scientific co-founder of a company and is required by his institution to state so in his publications. G.C. and E.W.-F.L. declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Tissue homeostasis
-
A process that ensures the maintenance of an internal steady state of a specific tissue of an organism and includes the control of cell proliferation, cell death and metabolic functions.
- Senolytics
-
A class of drugs that selectively clears senescent cells, which are one of the hallmarks of ageing. Senescent cells are non-responsive to growth-promoting stimuli and are therefore unable to re-enter the cell cycle.
- Transactivation domain
-
(TAD). A protein domain present in many transcription factors that contain binding sites for regulatory proteins, such as transcriptional co-repressors or co-activators.
- DAF-16 binding element
-
A DNA regulatory element with the consensus sequence 5′-GTAAACAA-3′ that binds to FOXO transcription factors and is involved in the regulation of FOXO target gene expression. It is named after the FOXO orthologue in Caenorhabditis elegans, Daf-16.
- Insulin-responsive element
-
A DNA regulatory element with the consensus sequence 5′-(C/A)(A/C)AAA(C/T)AA-3′ that is present in several gene promoters, including the insulin-like growth factor-binding protein promoter, and that is responsible for the insulin-mediated inhibition of transcription. Insulin-responsive elements can bind to members of the forkhead family of transcription factors.
- Histone acetyltransferases
-
(HATs). A class of enzymes that transfer acetyl groups onto lysine residues of cellular proteins, including histones.
- Histone deacetylases
-
(HDACs). A class of enzymes that remove acetyl groups from ε-amino groups on lysine residues of the amino-terminal tails of histones. They also deacetylate non-histone proteins.
- Reactive oxygen species
-
(ROS). Oxygen-containing molecules with one or more unpaired electrons. ROS are free radicals that can cause damage to DNA, RNA and proteins. ROS-mediated damage has been implicated in ageing.
- Nigral neurons
-
A collection of nerve cells that form the substantia nigra located in the midbrain. The name is based on the dark colour of the cells due to the presence of neuromelanin. These neurons produce dopamine and their loss leads to the motor deficits observed in Parkinson disease.
- Embryonic stem cells
-
Stem cells isolated from the inner cell mass of an embryo at a very early developmental stage when they are able to give rise to any cell type of the body.
- Induced pluripotent stem cells
-
Pluripotent stem cells derived from adult somatic cells through reprogramming by inducing genes. They resemble embryonic stem cells in many aspects.
- β-cells
-
Cells found in the islets of Langerhans of the pancreas and responsible for the production of insulin. The number of β-cells is decreased in patients with type I diabetes, resulting in insufficient insulin secretion and hyperglycaemia.
- Cardiac hypertrophy
-
An increase in cardiac muscle mass that occurs as an adaptive response to physiological or pathophysiological events and results from increased cardiomyocyte size and alterations in other heart muscle components. This condition most commonly affects the left ventricle and is associated with many forms of heart disease.
- Fetal haemoglobin
-
A haemoglobin variant that predominates during fetal life and in the blood of a newborn, which binds to oxygen more strongly than to adult haemoglobin, allowing for the prenatal transfer of oxygen from mother to fetus. While adult haemoglobin is composed of two α-subunits and two β-subunits, fetal haemoglobin is composed of two α-subunits and two γ-subunits.
- Nucleus pulposus
-
The soft, gelatinous, inner core of the vertebral disc, which is an elastic structure consisting of water and collagen fibres that allows the disc to resist compression and torsion. It can herniate, leaking out of the disc space and inflaming the nerve roots.
- Cartilaginous endplates
-
Thin layers of hyaline cartilage that cover the top and bottom of intervertebral discs and provide structural support and regulate nutrient and metabolic exchange in the disc.
- Dietary restriction
-
A dietary regimen, also referred to as caloric restriction, that reduces calorie intake without incurring malnutrition and has been shown to delay ageing in several model organisms.
- Single-nucleotide polymorphisms
-
(SNPs). A DNA sequence variation in the genome of members of a species or between paired chromosomes in an individual that consists of the alteration of a single nucleotide. While mutations are a rare sequence variation, SNPs are present in more than 1% of a population.
Rights and permissions
About this article
Cite this article
Calissi, G., Lam, E.WF. & Link, W. Therapeutic strategies targeting FOXO transcription factors. Nat Rev Drug Discov 20, 21–38 (2021). https://doi.org/10.1038/s41573-020-0088-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41573-020-0088-2
This article is cited by
-
Forkhead box O proteins: steering the course of stem cell fate
Cell Regeneration (2024)
-
FOXO transcription factors as mediators of stress adaptation
Nature Reviews Molecular Cell Biology (2024)
-
Vascular smooth muscle-specific LRRC8A knockout ameliorates angiotensin II-induced cerebrovascular remodeling by inhibiting the WNK1/FOXO3a/MMP signaling pathway
Acta Pharmacologica Sinica (2024)
-
High clonal diversity and spatial genetic admixture in early prostate cancer and surrounding normal tissue
Nature Communications (2024)
-
A Phase 1 randomized study on the safety and pharmacokinetics of OCS-05, a neuroprotective disease modifying treatment for Acute Optic Neuritis and Multiple Sclerosis
Scientific Reports (2023)