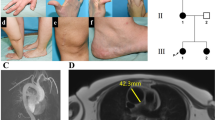
Abstract
The Ehlers–Danlos syndromes (EDS) are a heterogeneous group of hereditary disorders of connective tissue, with common features including joint hypermobility, soft and hyperextensible skin, abnormal wound healing and easy bruising. Fourteen different types of EDS are recognized, of which the molecular cause is known for 13 types. These types are caused by variants in 20 different genes, the majority of which encode the fibrillar collagen types I, III and V, modifying or processing enzymes for those proteins, and enzymes that can modify glycosaminoglycan chains of proteoglycans. For the hypermobile type of EDS, the molecular underpinnings remain unknown. As connective tissue is ubiquitously distributed throughout the body, manifestations of the different types of EDS are present, to varying degrees, in virtually every organ system. This can make these disorders particularly challenging to diagnose and manage. Management consists of a care team responsible for surveillance of major and organ-specific complications (for example, arterial aneurysm and dissection), integrated physical medicine and rehabilitation. No specific medical or genetic therapies are available for any type of EDS.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ehlers, E. Cutis laxa, neigung zu haemorrhagien in der haut, lockerung meherer artikulationen [German]. Dermatol. Z. 8, 173–174 (1901).
Danlos, M. Un cas de cutis laxa avec tumeurs par contusion chronique des coudes et des genoux (xanthome juvénile pseudo-diabétique de MM. Hallopeau et Macé de Lépinay) [French]. Bull. Soc. Fr. Dermatol. Syphiligr. 19, 70–72 (1908).
Chernogubow, A. N. Uber einen Fall von Cutis Laxa [German]. Jahresber. Ges. Med. 27, 562 (1892).
Weber, F. P. Ehlers–Danlos syndrome. Proc. R. Soc. Med. 30, 30–31 (1936).
Beighton, P. et al. International nosology of heritable disorders of connective tissue, Berlin, 1986. Am. J. Med. Genet. 29, 581–594 (1988).
Beighton, P., De Paepe, A., Steinmann, B., Tsipouras, P. & Wenstrup, R. J. Ehlers–Danlos syndromes: revised nosology, Villefranche, 1997. Ehlers–Danlos National Foundation (USA) and Ehlers–Danlos support group (UK). Am. J. Med. Genet. 77, 31–37 (1998).
Malfait, F. et al. The 2017 international classification of the Ehlers–Danlos syndromes. Am. J. Med. Genet. Part. C, Semin. Med. Genetics 175, 8–26 (2017). This paper presents the 2017 revised classification of EDS with major and minor clinical diagnostic criteria and strategies for molecular testing.
Blackburn, P. R. et al. Bi-allelic alterations in AEBP1 lead to defective collagen assembly and connective tissue structure resulting in a variant of Ehlers–Danlos syndrome. Am. J. Hum. Genet. 102, 696–705 (2018). This paper adds a 14th type of EDS to the EDS classification by delineating a novel EDS type caused by genetic defects in AEBP1, thereby expanding the list of EDS-associated genes to 20.
Steinmann, B., Royce, P. M. & Superti-Furga, A. in Connective Tissue and its Heritable Disorders (eds Royce, P. M. & Steinmann, B.) 431−523 (Wiley-Liss, 2002). This is a very comprehensive, detailed and accurate book on the biochemistry, genetics, clinics and pathology of connective tissue, including a chapter on EDS.
Pepin, M. G. et al. Survival is affected by mutation type and molecular mechanism in vascular Ehlers–Danlos syndrome (EDS type IV). Genet. Med. 16, 881–888 (2014). The largest retrospective review of clinical and molecular data of >1,200 patients with vEDS provides insights into the natural history of vEDS and genotype–phenotype correlations.
Symoens, S. et al. Comprehensive molecular analysis demonstrates type V collagen mutations in over 90% of patients with classic EDS and allows to refine diagnostic criteria. Hum. Mutat. 33, 1485–1493 (2012).
Brady, A. F. et al. The Ehlers–Danlos syndromes, rare types. Am. J. Med. Genet. C Semin. Med. Genet. 175, 70–115 (2017).
Byers, P. H. et al. Diagnosis, natural history, and management in vascular Ehlers–Danlos syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 175, 40–47 (2017).
Rare Disease Day. What is a rare disease? Rare Disease Day https://www.rarediseaseday.org/article/what-is-a-rare-disease (2020).
Grahame, R., Bird, H. A. & Child, A. The revised (Brighton 1998) criteria for the diagnosis of benign joint hypermobility syndrome (BJHS). J. Rheumatol. 27, 1777–1779 (2000).
Tinkle, B. T. et al. The lack of clinical distinction between the hypermobility type of Ehlers–Danlos syndrome and the joint hypermobility syndrome (a.k.a. hypermobility syndrome). Am. J. Med. Genet. A 149A, 2368–2370 (2009).
Hakim, A. J. & Sahota, A. Joint hypermobility and skin elasticity: the hereditary disorders of connective tissue. Clin. Dermatol. 24, 521–533 (2006).
Tinkle, B. et al. Hypermobile Ehlers–Danlos syndrome (a.k.a. Ehlers–Danlos syndrome type III and Ehlers–Danlos syndrome hypermobility type): clinical description and natural history. Am. J. Med. Genet. C Semin. Med. Genet. 175, 48–69 (2017).
Castori, M. et al. A framework for the classification of joint hypermobility and related conditions. Am. J. Med. Genet. C Semin. Med. Genet. 175, 148–157 (2017). This paper provides a simplified categorization of genetic syndromes featuring joint hypermobility and introduces the term ‘hypermobility spectrum disorders’.
Burgeson, R. E., El Adli, F. A., Kaitila, I. I. & Hollister, D. W. Fetal membrane collagens: identification of two new collagen alpha chains. Proc. Natl Acad. Sci. USA 73, 2579–2583 (1976).
Gay, S., Rhodes, R. K., Gay, R. E. & Miller, E. J. Collagen molecules comprised of alpha 1(V)-chains (B-chains): an apparent localization in the exocytoskeleton. Coll. Relat. Res. 1, 53–58 (1981).
Imamura, Y., Scott, I. C. & Greenspan, D. S. The pro-alpha3(V) collagen chain. Complete primary structure, expression domains in adult and developing tissues, and comparison to the structures and expression domains of the other types V and XI procollagen chains. J. Biol. Chem. 275, 8749–8759 (2000).
Birk, D. E. Type V collagen: heterotypic type I/V collagen interactions in the regulation of fibril assembly. Micron 32, 223–237 (2001).
Birk, D. E., Fitch, J. M., Babiarz, J. P. & Linsenmayer, T. F. Collagen type I and type V are present in the same fibril in the avian corneal stroma. J. Cell Biol. 106, 999–1008 (1988).
Wenstrup, R. J., Florer, J. B., Cole, W. G., Willing, M. C. & Birk, D. E. Reduced type I collagen utilization: a pathogenic mechanism in COL5A1 haplo-insufficient Ehlers–Danlos syndrome. J. Cell. Biochem. 92, 113–124 (2004).
Wenstrup, R. J. et al. Type V collagen controls the initiation of collagen fibril assembly. J. Biol. Chem. 279, 53331–53337 (2004).
Emanuel, B. S., Cannizzaro, L. A., Seyer, J. M. & Myers, J. C. Human alpha 1(III) and alpha 2(V) procollagen genes are located on the long arm of chromosome 2. Proc. Natl Acad. Sci. USA 82, 3385–3389 (1985).
Gelse, K., Poschl, E. & Aigner, T. Collagens — structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 55, 1531–1546 (2003).
Keene, D. R., Sakai, L. Y., Bachinger, H. P. & Burgeson, R. E. Type III collagen can be present on banded collagen fibrils regardless of fibril diameter. J. Cell Biol. 105, 2393–2402 (1987).
Romanic, A. M., Adachi, E., Kadler, K. E., Hojima, Y. & Prockop, D. J. Copolymerization of pNcollagen III and collagen I. pNcollagen III decreases the rate of incorporation of collagen I into fibrils, the amount of collagen I incorporated, and the diameter of the fibrils formed. J. Biol. Chem. 266, 12703–12709 (1991).
Liu, X., Wu, H., Byrne, M., Krane, S. & Jaenisch, R. Type III collagen is crucial for collagen I fibrillogenesis and for normal cardiovascular development. Proc. Natl Acad. Sci. USA 94, 1852–1856 (1997).
D’Hondt, S. et al. Type III collagen affects dermal and vascular collagen fibrillogenesis and tissue integrity in a mutant Col3a1 transgenic mouse model. Matrix Biol. 70, 72–83 (2018).
Marini, J. C. et al. Osteogenesis imperfecta. Nat. Rev. Dis. Prim. 3, 17052 (2017).
Schwarze, U., Atkinson, M., Hoffman, G. G., Greenspan, D. S. & Byers, P. H. Null alleles of the COL5A1 gene of type V collagen are a cause of the classical forms of Ehlers–Danlos syndrome (types I and II). Am. J. Hum. Genet. 66, 1757–1765 (2000).
Wenstrup, R. J. et al. COL5A1 haploinsufficiency is a common molecular mechanism underlying the classical form of EDS. Am. J. Hum. Genet. 66, 1766–1776 (2000).
Malfait, F. & De Paepe, A. Molecular genetics in classic Ehlers–Danlos syndrome. Am. J. Med. Genet. C Semin. Med Genet. 139C, 17–23 (2005).
Ritelli, M. et al. Clinical and molecular characterization of 40 patients with classic Ehlers–Danlos syndrome: identification of 18 COL5A1 and 2 COL5A2 novel mutations. Orphanet J. Rare Dis. 8, 58 (2013).
Wenstrup, R. J. et al. Murine model of the Ehlers–Danlos syndrome. col5a1 haploinsufficiency disrupts collagen fibril assembly at multiple stages. J. Biol. Chem. 281, 12888–12895 (2006).
Chanut-Delalande, H. et al. Development of a functional skin matrix requires deposition of collagen V heterotrimers. Mol. Cell Biol. 24, 6049–6057 (2004).
Bowen, J. M. et al. Ehlers–Danlos syndrome, classical type. Am. J. Med. Genet. C Semin. Med. Genet. 175, 27–39 (2017).
Symoens, S. et al. COL5A1 signal peptide mutations interfere with protein secretion and cause classic Ehlers–Danlos syndrome. Hum. Mutat. 30, E395–E403 (2009).
Wenstrup, R. J., Langland, G. T., Willing, M. C., D’Souza, V. N. & Cole, W. G. A splice-junction mutation in the region of COL5A1 that codes for the carboxyl propeptide of pro alpha 1(V) chains results in the gravis form of the Ehlers–Danlos syndrome (type I). Hum. Mol. Genet. 5, 1733–1736 (1996).
De Paepe, A., Nuytinck, L., Hausser, I., Anton-Lamprecht, I. & Naeyaert, J. M. Mutations in the COL5A1 gene are causal in the Ehlers–Danlos syndromes I and II. Am. J. Hum. Genet. 60, 547–554 (1997).
Zoppi, N., Gardella, R., De Paepe, A., Barlati, S. & Colombi, M. Human fibroblasts with mutations in COL5A1 and COL3A1 genes do not organize collagens and fibronectin in the extracellular matrix, down-regulate alpha2beta1 integrin, and recruit alphavbeta3 instead of alpha5beta1 integrin. J. Biol. Chem. 279, 18157–18168 (2004).
Viglio, S. et al. Rescue of migratory defects of Ehlers–Danlos syndrome fibroblasts in vitro by type V collagen but not insulin-like binding protein-1. J. Investig. Dermatol. 128, 1915–1919 (2008).
Chiarelli, N., Carini, G., Zoppi, N., Ritelli, M. & Colombi, M. Molecular insights in the pathogenesis of classical Ehlers–Danlos syndrome from transcriptome-wide expression profiling of patients’ skin fibroblasts. PLoS One 14, e0211647 (2019).
Chiarelli, N., Ritelli, M., Zoppi, N. & Colombi, M. Cellular and molecular mechanisms in the pathogenesis of classical, vascular, and hypermobile Ehlers–Danlos syndromes. Genes 10, 609 (2019).
Vogel, A., Holbrook, K. A., Steinmann, B., Gitzelmann, R. & Byers, P. H. Abnormal collagen fibril structure in the gravis form (type I) of Ehlers–Danlos syndrome. Lab. Invest. 40, 201–206 (1979).
Hausser, I. & Anton-Lamprecht, I. Differential ultrastructural aberrations of collagen fibrils in Ehlers–Danlos syndrome types I–IV as a means of diagnostics and classification. Hum. Genet. 93, 394–407 (1994).
Kirschner, J. et al. Ullrich congenital muscular dystrophy: connective tissue abnormalities in the skin support overlap with Ehlers–Danlos syndromes. Am. J. Med. Genet. A 132A, 296–301 (2005).
Holbrook, K. A. & Byers, P. H. Structural abnormalities in the dermal collagen and elastic matrix from the skin of patients with inherited connective tissue disorders. J. Invest. Dermatol. 79, 7s–16s (1982).
Byers, P. H., Holbrook, K. A., Barsh, G. S., Smith, L. T. & Bornstein, P. Altered secretion of type III procollagen in a form of type IV Ehlers–Danlos syndrome. Biochemical studies in cultured fibroblasts. Lab. Invest. 44, 336–341 (1981).
Schwarze, U. et al. Haploinsufficiency for one COL3A1 allele of type III procollagen results in a phenotype similar to the vascular form of Ehlers–Danlos syndrome, Ehlers–Danlos syndrome type IV. Am. J. Hum. Genet. 69, 989–1001 (2001).
Frank, M. et al. The type of variants at the COL3A1 gene associates with the phenotype and severity of vascular Ehlers–Danlos syndrome. Eur. J. Hum. Genet. 23, 1657–1664 (2015).
Ghali, N. et al. Atypical COL3A1 variants (glutamic acid to lysine) cause vascular Ehlers–Danlos syndrome with a consistent phenotype of tissue fragility and skin hyperextensibility. Genet. Med. 21, 2081–2091 (2019).
Jorgensen, A. et al. Vascular Ehlers–Danlos syndrome in siblings with biallelic COL3A1 sequence variants and marked clinical variability in the extended family. Eur. J. Hum. Genet. 23, 796–802 (2015).
Plancke, A. et al. Homozygosity for a null allele of COL3A1 results in recessive Ehlers–Danlos syndrome. Eur. J. Hum. Genet. 17, 1411–1416 (2009).
Horn, D. et al. Biallelic COL3A1 mutations result in a clinical spectrum of specific structural brain anomalies and connective tissue abnormalities. Am. J. Med. Genet. A 173, 2534–2538 (2017).
Vandervore, L. et al. Bi-allelic variants in COL3A1 encoding the ligand to GPR56 are associated with cobblestone-like cortical malformation, white matter changes and cerebellar cysts. J. Med. Genet. 54, 432–440 (2017).
Loeys, B. L. et al. Aneurysm syndromes caused by mutations in the TGF-beta receptor. N. Engl. J. Med. 355, 788–798 (2006).
Chiarelli, N., Carini, G., Zoppi, N., Ritelli, M. & Colombi, M. Transcriptome analysis of skin fibroblasts with dominant negative COL3A1 mutations provides molecular insights into the etiopathology of vascular Ehlers–Danlos syndrome. PLoS One 13, e0191220 (2018).
Holbrook, K. A. & Byers, P. H. Ultrastructural characteristics of the skin in a form of the Ehlers–Danlos syndrome type IV. Storage in the rough endoplasmic reticulum. Lab. Invest. 44, 342–350 (1981).
Smith, L. T., Schwarze, U., Goldstein, J. & Byers, P. H. Mutations in the COL3A1 gene result in the Ehlers–Danlos syndrome type IV and alterations in the size and distribution of the major collagen fibrils of the dermis. J. Investig. Dermatol. 108, 241–247 (1997).
Byers, P. H. et al. Ehlers–Danlos syndrome type VIIA and VIIB result from splice-junction mutations or genomic deletions that involve exon 6 in the COL1A1 and COL1A2 genes of type I collagen. Am. J. Med. Genet. 72, 94–105 (1997).
Chiodo, A. A., Hockey, A. & Cole, W. G. A base substitution at the splice acceptor site of intron 5 of the COL1A2 gene activates a cryptic splice site within exon 6 and generates abnormal type I procollagen in a patient with Ehlers–Danlos syndrome type VII. J. Biol. Chem. 267, 6361–6369 (1992).
Colige, A. et al. Human Ehlers–Danlos syndrome type VII C and bovine dermatosparaxis are caused by mutations in the procollagen I N-proteinase gene. Am. J. Hum. Genet. 65, 308–317 (1999).
Shi, L. et al. Comprehensive population screening in the Ashkenazi Jewish population for recurrent disease-causing variants. Clin. Genet. 91, 599–604 (2017).
Van Damme, T. et al. Expanding the clinical and mutational spectrum of the Ehlers–Danlos syndrome, dermatosparaxis type. Genet. Med. 18, 882–891 (2016).
Fernandes, R. J. et al. Procollagen II amino propeptide processing by ADAMTS-3. Insights on dermatosparaxis. J. Biol. Chem. 276, 31502–31509 (2001).
Colige, A. et al. Cloning and characterization of ADAMTS-14, a novel ADAMTS displaying high homology with ADAMTS-2 and ADAMTS-3. J. Biol. Chem. 277, 5756–5766 (2002).
Smith, L. T. et al. Human dermatosparaxis: a form of Ehlers–Danlos syndrome that results from failure to remove the amino-terminal propeptide of type I procollagen. Am. J. Hum. Genet. 51, 235–244 (1992).
Nusgens, B. V. et al. Evidence for a relationship between Ehlers–Danlos type VII C in humans and bovine dermatosparaxis. Nat. Genet. 1, 214–217 (1992).
Le Goff, C. et al. Regulation of procollagen amino-propeptide processing during mouse embryogenesis by specialization of homologous ADAMTS proteases: insights on collagen biosynthesis and dermatosparaxis. Development 133, 1587–1596 (2006).
Colige, A. et al. Domains and maturation processes that regulate the activity of ADAMTS-2, a metalloproteinase cleaving the aminopropeptide of fibrillar procollagens types I–III and V. J. Biol. Chem. 280, 34397–34408 (2005).
Schwarze, U. et al. Rare autosomal recessive cardiac valvular form of Ehlers–Danlos syndrome results from mutations in the COL1A2 gene that activate the nonsense-mediated RNA decay pathway. Am. J. Hum. Genet. 74, 917–930 (2004).
Malfait, F. et al. Total absence of the alpha2(I) chain of collagen type I causes a rare form of Ehlers–Danlos syndrome with hypermobility and propensity to cardiac valvular problems. J. Med. Genet. 43, e36 (2006).
Sasaki, T. et al. Ehlers–Danlos syndrome. A variant characterized by the deficiency of pro alpha 2 chain of type I procollagen. Arch. Dermatol. 123, 76–79 (1987).
Kojima, T., Shinkai, H., Fujita, M., Morita, E. & Okamoto, S. Case report and study of collagen metabolism in Ehlers–Danlos syndrome type II. J. Dermatol. 15, 155–160 (1988).
Guarnieri, V. et al. Cardiac valvular Ehlers–Danlos syndrome is a well-defined condition due to recessive null variants in COL1A2. Am. J. Med. Genet. A 179, 846–851 (2019).
Nicholls, A. C. et al. The clinical features of homozygous alpha 2(I) collagen deficient osteogenesis imperfecta. J. Med. Genet. 21, 257–262 (1984).
Pihlajaniemi, T. et al. Osteogenesis imperfecta: cloning of a pro-alpha 2(I) collagen gene with a frameshift mutation. J. Biol. Chem. 259, 12941–12944 (1984).
Byers, P. H. & Murray, M. L. Ehlers–Danlos syndrome: a showcase of conditions that lead to understanding matrix biology. Matrix Biol. 33, 10–15 (2014).
Malfait, F. et al. Three arginine to cysteine substitutions in the pro-alpha (I)-collagen chain cause Ehlers–Danlos syndrome with a propensity to arterial rupture in early adulthood. Hum. Mutat. 28, 387–395 (2007).
Adham, S. et al. Classical Ehlers–Danlos syndrome with a propensity to arterial events: a new report on a French family with a COL1A1 p.(Arg312Cys) variant. Clin. Genet. 97, 357–361 (2019).
Cabral, W. A. et al. Y-position cysteine substitution in type I collagen (alpha1(I) R888C/p.R1066C) is associated with osteogenesis imperfecta/Ehlers–Danlos syndrome phenotype. Hum. Mutat. 28, 396–405 (2007).
Gaines, R. et al. Spontaneous ruptured dissection of the right common iliac artery in a patient with classic Ehlers–Danlos syndrome phenotype. Ann. Vasc. Surg. 29, 595.e11–595.e14 (2015).
Nuytinck, L. et al. Classical Ehlers–Danlos syndrome caused by a mutation in type I collagen. Am. J. Hum. Genet. 66, 1398–1402 (2000).
Duong, J. et al. A family with classical Ehlers–Danlos syndrome (cEDS), mild bone fragility and without vascular complications, caused by the p.Arg312Cys mutation in COL1A1. Eur. J. Med. Genet. 63, 103730 (2019).
Colombi, M. et al. Delineation of Ehlers–Danlos syndrome phenotype due to the c.934C>T, p.(Arg312Cys) mutation in COL1A1: report on a three-generation family without cardiovascular events, and literature review. Am. J. Med. Genet. A 173, 524–530 (2017).
Lund, A. et al. A novel arginine-to-cysteine substitution in the triple helical region of the alpha1(I) collagen chain in a family with an osteogenesis imperfecta/Ehlers–Danlos phenotype. Clin. Genet. 73, 97–101 (2008).
Pinnell, S. R., Krane, S. M., Kenzora, J. E. & Glimcher, M. J. A heritable disorder of connective tissue. Hydroxylysine-deficient collagen disease. N. Engl. J. Med. 286, 1013–1020 (1972). This study presents the first heritable disorder of collagen biosynthesis in humans.
Yeowell, H. N. & Walker, L. C. Mutations in the lysyl hydroxylase 1 gene that result in enzyme deficiency and the clinical phenotype of Ehlers–Danlos syndrome type VI. Mol. Genet. Metab. 71, 212–224 (2000).
Yeowell, H. N. & Steinmann, B. in GeneReviews (eds Adam, M. P. et al.) (University of Washington, 1993).
Giunta, C., Randolph, A. & Steinmann, B. Mutation analysis of the PLOD1 gene: an efficient multistep approach to the molecular diagnosis of the kyphoscoliotic type of Ehlers–Danlos syndrome (EDS VIA). Mol. Genet. Metab. 86, 269–276 (2005).
Baumann, M. et al. Mutations in FKBP14 cause a variant of Ehlers–Danlos syndrome with progressive kyphoscoliosis, myopathy, and hearing loss. Am. J. Hum. Genet. 90, 201–216 (2012).
Giunta, C. et al. A cohort of 17 patients with kyphoscoliotic Ehlers–Danlos syndrome caused by biallelic mutations in FKBP14: expansion of the clinical and mutational spectrum and description of the natural history. Genet. Med. 20, 42–54 (2018).
Dordoni, C. et al. Further delineation of FKBP14-related Ehlers–Danlos syndrome: a patient with early vascular complications and non-progressive kyphoscoliosis, and literature review. Am. J. Med. Genet. A 170, 2031–2038 (2016).
Murray, M. L., Yang, M., Fauth, C. & Byers, P. H. FKBP14-related Ehlers–Danlos syndrome: expansion of the phenotype to include vascular complications. Am. J. Med. Genet. A 164A, 1750–1755 (2014).
Boudko, S. P., Ishikawa, Y., Nix, J., Chapman, M. S. & Bachinger, H. P. Structure of human peptidyl-prolyl cis-trans isomerase FKBP22 containing two EF-hand motifs. Protein Sci. 23, 67–75 (2014).
Ishikawa, Y., Mizuno, K. & Bachinger, H. P. Ziploc-ing the structure 2.0: endoplasmic reticulum-resident peptidyl prolyl isomerases show different activities toward hydroxyproline. J. Biol. Chem. 292, 9273–9282 (2017).
Ishikawa, Y. & Bachinger, H. P. A substrate preference for the rough endoplasmic reticulum resident protein FKBP22 during collagen biosynthesis. J. Biol. Chem. 289, 18189–18201 (2014).
Gjaltema, R. A., van der Stoel, M. M., Boersema, M. & Bank, R. A. Disentangling mechanisms involved in collagen pyridinoline cross-linking: the immunophilin FKBP65 is critical for dimerization of lysyl hydroxylase 2. Proc. Natl Acad. Sci. USA 113, 7142–7147 (2016).
Burch, G. H. et al. Tenascin-X Deficiency is associated with Ehlers–Danlos syndrome. Nat. Genet. 17, 104–108 (1997).
Schalkwijk, J. et al. A recessive form of the Ehlers–Danlos syndrome caused by tenascin-X deficiency. N. Engl. J. Med. 345, 1167–1175 (2001).
Narasimhan, M. L. & Khattab, A. Genetics of congenital adrenal hyperplasia and genotype-phenotype correlation. Fertil. Steril. 111, 24–29 (2019).
Zweers, M. C. et al. Haploinsufficiency of TNXB is associated with hypermobility type of Ehlers–Danlos syndrome. Am. J. Hum. Genet. 73, 214–217 (2003).
Zweers, M. C., Kucharekova, M. & Schalkwijk, J. Tenascin-X: a candidate gene for benign joint hypermobility syndrome and hypermobility type Ehlers–Danlos syndrome? Ann. Rheum. Dis. 64, 504–505 (2005).
Hicks, D. et al. Mutations in the collagen XII gene define a new form of extracellular matrix-related myopathy. Hum. Mol. Genet. 23, 2353–2363 (2014).
Zou, Y. et al. Recessive and dominant mutations in COL12A1 cause a novel EDS/myopathy overlap syndrome in humans and mice. Hum. Mol. Genet. 23, 2339–2352 (2014).
Delbaere, S. et al. Novel defects in collagen XII and VI expand the mixed myopathy/Ehlers–Danlos syndrome spectrum and lead to variant-specific alterations in the extracellular matrix. Genet. Med. 22, 112–123 (2020).
Veit, G. et al. Collagen XII interacts with avian tenascin-X through its NC3 domain. J. Biol. Chem. 281, 27461–27470 (2006).
Valcourt, U., Alcaraz, L. B., Exposito, J. Y., Lethias, C. & Bartholin, L. Tenascin-X: beyond the architectural function. Cell Adhes. Migr. 9, 154–165 (2015).
Koch, M. et al. Large and small splice variants of collagen XII: differential expression and ligand binding. J. Cell Biol. 130, 1005–1014 (1995).
Keene, D. R., Lunstrum, G. P., Morris, N. P., Stoddard, D. W. & Burgeson, R. E. Two type XII-like collagens localize to the surface of banded collagen fibrils. J. Cell Biol. 113, 971–978 (1991).
Font, B., Eichenberger, D., Rosenberg, L. M. & van der Rest, M. Characterization of the interactions of type XII collagen with two small proteoglycans from fetal bovine tendon, decorin and fibromodulin. Matrix Biol. 15, 341–348 (1996).
Bristow, J., Carey, W., Egging, D. & Schalkwijk, J. Tenascin-X, collagen, elastin, and the Ehlers–Danlos syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 139C, 24–30 (2005).
Hernandez, A. et al. A distinct variant of the Ehlers–Danlos syndrome. Clin. Genet. 16, 335–339 (1979).
Hernandez, A. et al. Ehlers–Danlos features with progeroid facies and mild mental retardation. Further delineation of the syndrome. Clin. Genet. 30, 456–461 (1986).
Hernandez, A., Aguirre-Negrete, M. G., Liparoli, J. C. & Cantu, J. M. Third case of a distinct variant of the Ehlers–Danlos syndrome (EDS). Clin. Genet. 20, 222–224 (1981).
Kresse, H. et al. Glycosaminoglycan-free small proteoglycan core protein is secreted by fibroblasts from a patient with a syndrome resembling progeroid. Am. J. Hum. Genet. 41, 436–453 (1987).
Quentin, E., Gladen, A., Roden, L. & Kresse, H. A genetic defect in the biosynthesis of dermatan sulfate proteoglycan: galactosyltransferase I deficiency in fibroblasts from a patient with a progeroid syndrome. Proc. Natl Acad. Sci. USA 87, 1342–1346 (1990). This article is the first to link a genetic defect in GAG biosynthesis to EDS.
Okajima, T., Fukumoto, S., Furukawa, K. & Urano, T. Molecular basis for the progeroid variant of Ehlers–Danlos syndrome. Identification and characterization of two mutations in galactosyltransferase I gene. J. Biol. Chem. 274, 28841–28844 (1999).
Malfait, F. et al. Defective initiation of glycosaminoglycan synthesis due to B3GALT6 mutations causes a pleiotropic Ehlers–Danlos-syndrome-like connective tissue disorder. Am. J. Hum. Genet. 92, 935–945 (2013).
Nakajima, M. et al. Mutations in B3GALT6, which encodes a glycosaminoglycan linker region enzyme, cause a spectrum of skeletal and connective tissue disorders. Am. J. Hum. Genet. 92, 927–934 (2013).
Seidler, D. G. et al. Defective glycosylation of decorin and biglycan, altered collagen structure, and abnormal phenotype of the skin fibroblasts of an Ehlers–Danlos syndrome patient carrying the novel Arg270Cys substitution in galactosyltransferase I (beta4GalT-7). J. Mol. Med. 84, 583–594 (2006).
Van Damme, T. et al. Biallelic B3GALT6 mutations cause spondylodysplastic Ehlers–Danlos syndrome. Hum. Mol. Genet. 27, 3475–3487 (2018).
Ritelli, M. et al. Insights in the etiopathology of galactosyltransferase II (GalT-II) deficiency from transcriptome-wide expression profiling of skin fibroblasts of two sisters with compound heterozygosity for two novel B3GALT6 mutations. Mol. Genet. Metab. Rep. 2, 1–15 (2015).
Dündar, M. et al. Loss of dermatan-4-sulfotransferase 1 function results in adducted thumb-clubfoot syndrome. Am. J. Hum. Genet. 85, 873–882 (2009).
Müller, T. et al. Loss of dermatan sulfate epimerase (DSE) function results in musculocontractural Ehlers–Danlos syndrome. Hum. Mol. Genet. 22, 3761–3772 (2013).
Miyake, N. et al. Loss-of-function mutations of CHST14 in a new type of Ehlers–Danlos syndrome. Hum. Mutat. 31, 966–974 (2010).
Syx, D. et al. Genetic heterogeneity and clinical variability in musculocontractural Ehlers–Danlos syndrome caused by impaired dermatan sulfate biosynthesis. Hum. Mutat. 36, 535–547 (2015).
Malfait, F. et al. Musculocontractural Ehlers–Danlos syndrome (former EDS type VIB) and adducted thumb clubfoot syndrome (ATCS) represent a single clinical entity caused by mutations in the dermatan-4-sulfotransferase 1 encoding CHST14 gene. Hum. Mutat. 31, 1233–1239 (2010).
Mizumoto, S. et al. Defect in dermatan sulfate in urine of patients with Ehlers–Danlos syndrome caused by a CHST14/D4ST1 deficiency. Clin. Biochem. 50, 670–677 (2017).
Nomura, Y. Structural change in decorin with skin aging. Connect. Tissue Res. 47, 249–255 (2006).
Schirwani, S. et al. DSE associated musculocontractural EDS, a milder phenotype or phenotypic variability. Eur. J. Med. Genet. 63, 103798 (2019).
Hirose, T. et al. Structural alteration of glycosaminoglycan side chains and spatial disorganization of collagen networks in the skin of patients with mcEDS-CHST14. Biochim. Biophys. Acta Gen. Subj. 1863, 623–631 (2019).
Watanabe, T. et al. Ring-mesh model of proteoglycan glycosaminoglycan chains in tendon based on three-dimensional reconstruction by focused ion beam scanning electron microscopy. J. Biol. Chem. 291, 23704–23708 (2016).
Giunta, C. et al. Spondylocheiro dysplastic form of the Ehlers–Danlos syndrome — an autosomal-recessive entity caused by mutations in the zinc transporter gene SLC39A13. Am. J. Hum. Genet. 82, 1290–1305 (2008).
Fukada, T. et al. The zinc transporter SLC39A13/ZIP13 is required for connective tissue development: its involvement in BMP/TGF-beta signaling pathways. PLoS One 3, e3642 (2008).
Dusanic, M. et al. Novel nonsense mutation in SLC39A13 initially presenting as myopathy: case report and review of the literature. Mol. Syndromol. 9, 100–109 (2018).
Jeong, J. & Eide, D. J. The SLC39 family of zinc transporters. Mol. Asp. Med. 34, 612–619 (2013).
Jeong, J. et al. Promotion of vesicular zinc efflux by ZIP13 and its implications for spondylocheiro dysplastic Ehlers–Danlos syndrome. Proc. Natl Acad. Sci. USA 109, E3530–E3538 (2012).
Xiao, G., Wan, Z., Fan, Q., Tang, X. & Zhou, B. The metal transporter ZIP13 supplies iron into the secretory pathway in Drosophila melanogaster. eLife 3, e03191 (2014).
Cameron, J. A. Corneal abnormalities in Ehlers–Danlos syndrome type VI. Cornea 12, 54–59 (1993).
Royce PM, S. B., Vogel, A., Steinhorst, U. & Kohlschuetter, A. Brittle cornea syndrome: an heritable connective tissue disorder distinct from Ehlers–Danlos syndrome type VI and fragilitas oculi, with spontaneous perforations of the eye, blue sclerae, red hair, and normal collagen lysyl hydroxylation. Eur. J. Pediatr. 149, 465–469 (1990).
Al-Hussain H, Z. S., Huber, P. R., Giunta, C. & Steinmann, B. Brittle cornea syndrome and its delineation from the kyphoscoliotic type of Ehlers–Danlos syndrome (EDS VI): report on 23 patients and review of the literature. Am. J. Med. Genet. A 124, 28–34 (2004).
Abu A, F. M. et al. Deleterious mutations in the zinc-finger 469 gene cause brittle cornea syndrome. Am. J. Hum. Genet. 82, 1217–1222 (2008).
Burkitt Wright, E. M. et al. Mutations in PRDM5 in brittle cornea syndrome identify a pathway regulating extracellular matrix development and maintenance. Am. J. Hum. Genet. 88, 767–777 (2011).
Meani, N., Pezzimenti, F., Deflorian, G., Mione, M. & Alcalay, M. The tumor suppressor PRDM5 regulates Wnt signaling at early stages of zebrafish development. PLoS One 4, e4273 (2009).
Porter, L. F. et al. Bruch’s membrane abnormalities in PRDM5-related brittle cornea syndrome. Orphanet. J. Rare Dis. 10, 145 (2015).
Rohrbach, M. et al. ZNF469 frequently mutated in the brittle cornea syndrome (BCS) is a single exon gene possibly regulating the expression of several extracellular matrix components. Mol. Genet. Metab. 109, 289–295 (2013).
Stewart, R. E., Hollister, D. W. & Rimoin, D. L. A new variant of Ehlers–Danlos syndrome: an autosomal dominant disorder of fragile skin, abnormal scarring, and generalized periodontitis. Birth Defects Orig. Artic. Ser. 13, 85–93 (1977).
Rahman, N. et al. Ehlers–Danlos syndrome with severe early-onset periodontal disease (EDS-VIII) is a distinct, heterogeneous disorder with one predisposition gene at chromosome 12p13. Am. J. Hum. Genet. 73, 198–204 (2003).
Kapferer-Seebacher, I. et al. Periodontal Ehlers–Danlos syndrome is caused by mutations in C1R and C1S, which encode subcomponents C1r and C1s of complement. Am. J. Hum. Genet. 99, 1005–1014 (2016). The identification of genetic defects in C1r and C1s in pEDS opens a connection between the inflammatory classical complement pathway and connective tissue homeostasis.
Cooper, N. R. The classical complement pathway: activation and regulation of the first complement component. Adv. Immunol. 37, 151–216 (1985).
Arlaud, G. J., Colomb, M. G. & Gagnon, J. A functional model of the human C1 complex: emergence of a functional model. Immunol. Today 8, 106–111 (1987).
Arlaud, G. J. et al. Structural and functional studies on C1r and C1s: new insights into the mechanisms involved in C1 activity and assembly. Immunobiology 199, 303–316 (1998).
Arlaud, G. J. et al. Structural biology of C1: dissection of a complex molecular machinery. Immunol. Rev. 180, 136–145 (2001).
Bally, I. et al. Identification of the C1q-binding sites of human C1r and C1s: a refined three-dimensional model of the C1 complex of complement. J. Biol. Chem. 284, 19340–19348 (2009).
Bork, P. & Beckmann, G. The CUB domain. A widespread module in developmentally regulated proteins. J. Mol. Biol. 231, 539–545 (1993).
Vadon-Le Goff, S. et al. Procollagen C-proteinase enhancer stimulates procollagen processing by binding to the C-propeptide region only. J. Biol. Chem. 286, 38932–38938 (2011).
Steiglitz, B. M., Keene, D. R. & Greenspan, D. S. PCOLCE2 encodes a functional procollagen C-proteinase enhancer (PCPE2) that is a collagen-binding protein differing in distribution of expression and post-translational modification from the previously described PCPE1. J. Biol. Chem. 277, 49820–49830 (2002).
Grobner, R. et al. C1R mutations trigger constitutive complement 1 activation in periodontal Ehlers–Danlos syndrome. Front. Immunol. 10, 2537 (2019).
Layne, M. D. et al. Impaired abdominal wall development and deficient wound healing in mice lacking aortic carboxypeptidase-like protein. Mol. Cell. Biol. 21, 5256–5261 (2001).
Ith, B., Wei, J., Yet, S. F., Perrella, M. A. & Layne, M. D. Aortic carboxypeptidase-like protein is expressed in collagen-rich tissues during mouse embryonic development. Gene Expr. Patterns 5, 533–537 (2005).
Schissel, S. L. et al. Aortic carboxypeptidase-like protein is expressed in fibrotic human lung and its absence protects against bleomycin-induced lung fibrosis. Am. J. Pathol. 174, 818–828 (2009).
Tumelty, K. E., Smith, B. D., Nugent, M. A. & Layne, M. D. Aortic carboxypeptidase-like protein (ACLP) enhances lung myofibroblast differentiation through transforming growth factor beta receptor-dependent and -independent pathways. J. Biol. Chem. 289, 2526–2536 (2014).
Teratani, T. et al. Aortic carboxypeptidase-like protein, a WNT ligand, exacerbates nonalcoholic steatohepatitis. J. Clin. Invest. 128, 1581–1596 (2018).
Ritelli, M. et al. Expanding the clinical and mutational spectrum of recessive AEBP1-related classical-like Ehlers–Danlos syndrome. Genes 10, 135 (2019).
Beighton, P., Solomon, L. & Soskolne, C. L. Articular mobility in an African population. Ann. Rheum. Dis. 32, 413–418 (1973).
Remvig, L., Jensen, D. V. & Ward, R. C. Are diagnostic criteria for general joint hypermobility and benign joint hypermobility syndrome based on reproducible and valid tests? A review of the literature. J. Rheumatol. 34, 798–803 (2007).
Juul-Kristensen, B., Schmedling, K., Rombaut, L., Lund, H. & Engelbert, R. H. Measurement properties of clinical assessment methods for classifying generalized joint hypermobility — a systematic review. Am. J. Med. Genet. C Semin. Med. Genet. 175, 116–147 (2017).
Steinmann, B., Eyre, D. R. & Shao, P. Urinary pyridinoline cross-links in Ehlers–Danlos syndrome type VI. Am. J. Hum. Genet. 57, 1505–1508 (1995).
Rohrbach, M. et al. Phenotypic variability of the kyphoscoliotic type of Ehlers–Danlos syndrome (EDS VIA): clinical, molecular and biochemical delineation. Orphanet J. Rare Dis. 6, 46 (2011).
Legrand, A. et al. Frequency of de novo variants and parental mosaicism in vascular Ehlers–Danlos syndrome. Genet. Med. 21, 1568–1575 (2019).
Sulli, A. et al. Ehlers–Danlos syndromes: state of the art on clinical practice guidelines. RMD Open 4, e000790 (2018).
Kosho, T. et al. A new Ehlers–Danlos syndrome with craniofacial characteristics, multiple congenital contractures, progressive joint and skin laxity, and multisystem fragility-related manifestations. Am. J. Med. Genet. A 152, 1333–1346 (2010).
Mast, K. J., Nunes, M. E., Ruymann, F. B. & Kerlin, B. A. Desmopressin responsiveness in children with Ehlers–Danlos syndrome associated bleeding symptoms. Br. J. Haematol. 144, 230–233 (2009).
Stine, K. C. & Becton, D. L. DDAVP therapy controls bleeding in Ehlers–Danlos syndrome. J. Pediatr. Hematol. Oncol. 19, 156–158 (1997).
Engelbert, R. H. et al. The evidence-based rationale for physical therapy treatment of children, adolescents, and adults diagnosed with joint hypermobility syndrome/hypermobile Ehlers Danlos syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 175, 158–167 (2017).
Levy, H. P. in GeneReviews (eds Adam, M. P. et al.) (University of Washington, 1993).
Bathen, T., Hangmann, A. B., Hoff, M., Andersen, L. O. & Rand-Hendriksen, S. Multidisciplinary treatment of disability in Ehlers–Danlos syndrome hypermobility type/hypermobility syndrome: a pilot study using a combination of physical and cognitive-behavioral therapy on 12 women. Am. J. Med. Genet. A 161A, 3005–3011 (2013).
Ericson, W. B. Jr & Wolman, R. Orthopaedic management of the Ehlers–Danlos syndromes. Am. J. Med. Genet. C Semin. Med. Genet. 175, 188–194 (2017).
Chopra, P. et al. Pain management in the Ehlers–Danlos syndromes. Am. J. Med. Genet. C Semin. Med. Genet. 175, 212–219 (2017).
Syx, D., De Wandele, I., Rombaut, L. & Malfait, F. Hypermobility, the Ehlers–Danlos syndromes and chronic pain. Clin. Exp. Rheumatol. 35, 116–122 (2017).
Castori, M. et al. Re-writing the natural history of pain and related symptoms in the joint hypermobility syndrome/Ehlers–Danlos syndrome, hypermobility type. Am. J. Med. Genet. A 161A, 2989–3004 (2013).
Rauser-Foltz, K. K., Starr, L. J. & Yetman, A. T. Utilization of echocardiography in Ehlers–Danlos syndrome. Congenit. Heart Dis. 14, 864–867 (2019).
Oderich, G. S. et al. The spectrum, management and clinical outcome of Ehlers–Danlos syndrome type IV: a 30-year experience. J. Vasc. Surg. 42, 98–106 (2005).
Shalhub, S. et al. A multi-institutional experience in vascular Ehlers–Danlos syndrome. J. Vasc. Surg. 71, 149–157 (2020).
Ong, K. T. et al. Effect of celiprolol on prevention of cardiovascular events in vascular Ehlers–Danlos syndrome: a prospective randomised, open, blinded-endpoints trial. Lancet 376, 1476–1484 (2010). This article presents the first and only clinical pharmacological trial on EDS.
Frank, M. et al. Vascular Ehlers–Danlos syndrome: long-term observational study. J. Am. Coll. Cardiol. 73, 1948–1957 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02597361 (2018).
Speake, D., Dvorkin, L., Vaizey, C. J. & Carlson, G. L. Management of colonic complications of type IV Ehlers–Danlos syndrome: a systematic review and evidence-based management strategy. Colorectal Dis. 22, 129–135 (2020).
Adham, S., Finzindohoué, F. M., Jeunemaitre, X. & Frank, M. Natural history and surgical management of colonic perforations in vascular Ehlers–Danlos syndrome: a retrospective review. Dis. Colon Rectum 62, 859–866 (2019).
Kosho, T. CHST14/D4ST1 deficiency: new form of Ehlers–Danlos syndrome. Pediatr. Int. 58, 88–99 (2016).
Nee, J. et al. Prevalence of functional GI diseases and pelvic floor symptoms in Marfan syndrome and Ehlers–Danlos syndrome: a national cohort study. J. Clin. Gastroenterol. 53, 653–659 (2019).
Fikree, A., Chelimsky, G., Collins, H., Kovacic, K. & Aziz, Q. Gastrointestinal involvement in the Ehlers–Danlos syndromes. Am. J. Med. Genet. C Semin. Med. Genet. 175, 181–187 (2017).
Biswas, M. et al. Prescription pattern & adverse drug reactions of prokinetics. Indian J. Med. Res. 149, 748–754 (2019).
Malfait, F. et al. Helical mutations in type I collagen that affect the processing of the amino-propeptide result in an osteogenesis imperfecta/Ehlers–Danlos syndrome overlap syndrome. Orphanet J. Rare Dis. 8, 78 (2013).
Morlino, S. et al. COL1-related overlap disorder: a novel connective tissue disorder incorporating the osteogenesis imperfecta/Ehlers–Danlos syndrome overlap. Clin. Genet. 97, 396–406 (2020).
Marom, R., Lee, Y. C., Grafe, I. & Lee, B. Pharmacological and biological therapeutic strategies for osteogenesis imperfecta. Am. J. Med. Genet. C Semin. Med. Genet. 172, 367–383 (2016).
Murray, M. L., Pepin, M., Peterson, S. & Byers, P. H. Pregnancy-related deaths and complications in women with vascular Ehlers–Danlos syndrome. Genet. Med. 16, 874–880 (2014).
Berglund, B., Pettersson, C., Pigg, M. & Kristiansson, P. Self-reported quality of life, anxiety and depression in individuals with Ehlers–Danlos syndrome (EDS): a questionnaire study. BMC Musculoskelet. Disord. 16, 89 (2015).
Johannessen, E. C., Reiten, H. S., Lovaas, H., Maeland, S. & Juul-Kristensen, B. Shoulder function, pain and health related quality of life in adults with joint hypermobility syndrome/Ehlers–Danlos syndrome-hypermobility type. Disabil. Rehabil. 38, 1382–1390 (2016).
Bovet, C., Carlson, M. & Taylor, M. Quality of life, unmet needs, and iatrogenic injuries in rehabilitation of patients with Ehlers–Danlos syndrome hypermobility type/joint hypermobility Syndrome. Am. J. Med. Genet. A 170, 2044–2051 (2016).
Scheper, M. C. et al. Disability in adolescents and adults diagnosed with hypermobility-related disorders: a meta-analysis. Arch. Phys. Med. Rehabil. 97, 2174–2187 (2016).
Zeitoun, J. D. et al. Functional digestive symptoms and quality of life in patients with Ehlers–Danlos syndromes: results of a national cohort study on 134 patients. PLoS One 8, e80321 (2013).
Pacey, V., Tofts, L., Adams, R. D., Munns, C. F. & Nicholson, L. L. Quality of life prediction in children with joint hypermobility syndrome. J. Paediatr. Child. Health 51, 689–695 (2015).
Muriello, M. et al. Pain and sleep quality in children with non-vascular Ehlers–Danlos syndromes. Am. J. Med. Genet. A 176, 1858–1864 (2018).
Domany, K. A. et al. Sleep disorders and their management in children with Ehlers–Danlos syndrome referred to sleep clinics. J. Clin. Sleep Med. 14, 623–629 (2018).
Scheper, M. C., Nicholson, L. L., Adams, R. D., Tofts, L. & Pacey, V. The natural history of children with joint hypermobility syndrome and Ehlers–Danlos hypermobility type: a longitudinal cohort study. Rheumatology 56, 2073–2083 (2017).
Chiarelli, N. et al. Transcriptome-wide expression profiling in skin fibroblasts of patients with joint hypermobility syndrome/Ehlers–Danlos syndrome hypermobility type. PLoS One 11, e0161347 (2016).
Barabas, A. P. Heterogeneity of the Ehlers–Danlos syndrome: description of three clinical types and a hypothesis to explain the basic defect(s). Br. Med. J. 2, 612–613 (1967).
Beighton, P. Ehlers–Danlos syndrome. Ann. Rheum. Dis. 29, 332–333 (1970).
Morissette, R. et al. Transforming growth factor-beta and inflammation in vascular (type IV) Ehlers–Danlos syndrome. Circ. Cardiovasc. Genet. 7, 80–88 (2014). This study presents the first evidence for a pre-inflammatory state in vEDS and for changes in serum biomarker profiles.
Lindsay, M. E. & Dietz, H. C. Lessons on the pathogenesis of aneurysm from heritable conditions. Nature 473, 308–316 (2011).
Milewicz, D. M., Prakash, S. K. & Ramirez, F. Therapeutics targeting drivers of thoracic aortic aneurysms and acute aortic dissections: insights from predisposing genes and mouse models. Annu. Rev. Med. 68, 51–67 (2017).
Briest, W. et al. Doxycycline ameliorates the susceptibility to aortic lesions in a mouse model for the vascular type of Ehlers–Danlos syndrome. J. Pharmacol. Exp. Ther. 337, 621–627 (2011).
Tae, H. J. et al. Chronic treatment with a broad-spectrum metalloproteinase inhibitor, doxycycline, prevents the development of spontaneous aortic lesions in a mouse model of vascular Ehlers–Danlos syndrome. J. Pharmacol. Exp. Ther. 343, 246–251 (2012).
Dubacher, N. et al. Celiprolol but not losartan improves the biomechanical integrity of the aorta in a mouse model of vascular Ehlers–Danlos syndrome. Cardiovasc. Res. 116, 457–465 (2020).
Bowen, C. J. et al. Targetable cellular signaling events mediate vascular pathology in vascular Ehlers–Danlos syndrome. J. Clin. Invest. 130, 686–698 (2020).
Muller, G. A. et al. Allele-specific siRNA knockdown as a personalized treatment strategy for vascular Ehlers–Danlos syndrome in human fibroblasts. FASEB J. 26, 668–677 (2012).
Kadler, K. E., Hill, A. & Canty-Laird, E. G. Collagen fibrillogenesis: fibronectin, integrins, and minor collagens as organizers and nucleators. Curr. Opin. Cell Biol. 20, 495–501 (2008). A comprehensive review on collagen biosynthesis and the role of organizers, nucleators and regulators in this process.
Kadler, K. E., Holmes, D. F., Trotter, J. A. & Chapman, J. A. Collagen fibril formation. Biochem. J. 316, 1–11 (1996).
Canty, E. G. & Kadler, K. E. Procollagen trafficking, processing and fibrillogenesis. J. Cell Sci. 118, 1341–1353 (2005).
Syx, D. et al. Bi-allelic AEBP1 mutations in two patients with Ehlers–Danlos syndrome. Hum. Mol. Genet. 28, 1853–1864 (2019).
Castori, M. & Hakim, A. Contemporary approach to joint hypermobility and related disorders. Curr. Opin. Pediatr. 29, 640–649 (2017).
Merke, D. P. et al. Tenascin-X haploinsufficiency associated with Ehlers–Danlos syndrome in patients with congenital adrenal hyperplasia. J. Clin. Endocrinol. Metab. 98, E379–E387 (2013).
Hakim, A. J. & Grahame, R. A simple questionnaire to detect hypermobility: an adjunct to the assessment of patients with diffuse musculoskeletal pain. Int. J. Clin. Pract. 57, 163–166 (2003).
Dolan, A. L., Mishra, M. B., Chambers, J. B. & Grahame, R. Clinical and echocardiographic survey of the Ehlers–Danlos syndrome. Br. J. Rheumatol. 36, 459–462 (1997).
McDonnell, N. B. et al. Echocardiographic findings in classical and hypermobile Ehlers–Danlos syndromes. Am. J. Med. Genet. A 140, 129–136 (2006).
Atzinger, C. L., Meyer, R. A., Khoury, P. R., Gao, Z. & Tinkle, B. T. Cross-sectional and longitudinal assessment of aortic root dilation and valvular anomalies in hypermobile and classic Ehlers–Danlos syndrome. J. Pediatr. 158, 826–830.e1 (2011).
Camerota, F. et al. Heart rate, conduction and ultrasound abnormalities in adults with joint hypermobility syndrome/Ehlers–Danlos syndrome, hypermobility type. Clin. Rheumatol. 33, 981–987 (2014).
Kozanoglu, E., Coskun Benlidayi, I., Eker Akilli, R. & Tasal, A. Is there any link between joint hypermobility and mitral valve prolapse in patients with fibromyalgia syndrome? Clin. Rheumatol. 35, 1041–1044 (2016).
Zilocchi, M. et al. Vascular Ehlers–Danlos syndrome: imaging findings. AJR Am. J. Roentgenol. 189, 712–719 (2007).
Shalhub, S. et al. Molecular diagnosis in vascular Ehlers–Danlos syndrome predicts pattern of arterial involvement and outcomes. J. Vasc. Surg. 60, 160–169 (2014).
Acknowledgements
F.M. is partly supported by the Research Foundation, Flanders, Belgium. M.C. is partly supported by the Ricerca Corrente program 2020. C.A.F. is partly supported by the Ehlers–Danlos Society as the Director of the Center for Ehlers–Danlos Syndromes at Indiana University Health. T.K. is supported by the Japan Society for the Promotion of Science (grant-in-aid for scientific research), the Ministry of Health, Labour and Welfare, Japan (Research on Rare and Intractable Diseases), and the Japan Agency for Medical Research Development (AMED) (the Practical Research Project for Rare/Intractable Diseases, Initiative on Rare and Intractable Diseases, and Program for an Integrated Database of Clinical and Genomic Information).
Author information
Authors and Affiliations
Contributions
Introduction (F.M. and P.H.B.); Epidemiology (F.M., M.C. and P.H.B.); Mechanisms/pathophysiology (F.M., C.G., T.K. and P.H.B.); Diagnosis, screening and prevention (F.M., M.C. and P.H.B.); Management (F.M., M.C., C.A.F. and P.H.B.); Quality of life (F.M., C.A.F. and P.H.B.); Outlook (F.M. and P.H.B.); Overview of the Primer (F.M.).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Ehlers–Danlos Society: https://www.Ehlers–Danlos.com
European Reference Networks for Rare Diseases: https://vascern.eu and https://reconnet.ern-net.eu
The International EDS Consortium: https://www.ehlers-danlos.com/international-consortium
Rights and permissions
About this article
Cite this article
Malfait, F., Castori, M., Francomano, C.A. et al. The Ehlers–Danlos syndromes. Nat Rev Dis Primers 6, 64 (2020). https://doi.org/10.1038/s41572-020-0194-9
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-020-0194-9
This article is cited by
-
In silico analysis of mutation spectrum of Ehlers–Danlos, osteogenesis imperfecta, and cutis laxa overlapping phenotypes in Iranian population
Egyptian Journal of Medical Human Genetics (2024)
-
Echocardiographic abnormalities and joint hypermobility in Chinese patients with Osteogenesis imperfecta
Orphanet Journal of Rare Diseases (2024)
-
Psychological interventions for individuals with Ehlers-Danlos syndrome and hypermobility spectrum disorder: a scoping review
Orphanet Journal of Rare Diseases (2023)
-
Structure, extraction, processing, and applications of collagen as an ideal component for biomaterials - a review
Collagen and Leather (2023)
-
Pathogenic mechanisms of osteogenesis imperfecta, evidence for classification
Orphanet Journal of Rare Diseases (2023)