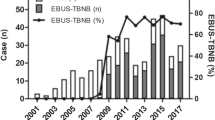
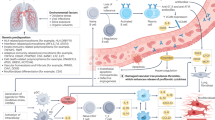
Abstract
Sarcoidosis is an inflammatory disorder of unknown cause that is characterized by granuloma formation in affected organs, most often in the lungs. Patients frequently suffer from cough, shortness of breath, chest pain and pronounced fatigue and are at risk of developing lung fibrosis or irreversible damage to other organs. The disease develops in genetically predisposed individuals with exposure to an as-yet unknown antigen. Genetic factors affect not only the risk of developing sarcoidosis but also the disease course, which is highly variable and difficult to predict. The typical T cell accumulation, local T cell immune response and granuloma formation in the lungs indicate that the inflammatory response in sarcoidosis is induced by specific antigens, possibly including self-antigens, which is consistent with an autoimmune involvement. Diagnosis can be challenging for clinicians because of the potential for almost any organ to be affected. As the aetiology of sarcoidosis is unknown, no specific treatment and no pathognomic markers exist. Thus, improved biomarkers to determine disease activity and to identify patients at risk of developing fibrosis are needed. Corticosteroids still constitute the first-line treatment, but new treatment strategies, including those targeting quality-of-life issues, are being evaluated and should yield appropriate, personalized and more effective treatments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
Change history
16 July 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Grunewald, J. & Eklund, A. Sex-specific manifestations of Lofgren’s syndrome. Am. J. Respir. Crit. Care Med. 175, 40–44 (2007).
Löfgren, S. Erythema nodosum: studies on etiology and pathogenesis in 185 adult cases. Acta Med. Scand. 124, 1–197 (1946).
Dubaniewicz, A. Mycobacterium tuberculosis heat shock proteins and autoimmunity in sarcoidosis. Autoimmun. Rev. 9, 419–424 (2010).
Judson, M. A., Boan, A. D. & Lackland, D. T. The clinical course of sarcoidosis: presentation, diagnosis, and treatment in a large white and black cohort in the United States. Sarcoidosis Vasc. Diffuse Lung Dis. 29, 119–127 (2012).
Arkema, E. V., Grunewald, J., Kullberg, S., Eklund, A. & Askling, J. Sarcoidosis incidence and prevalence: a nationwide register-based assessment in Sweden. Eur. Respir. J. 48, 1690–1699 (2016).
Byg, K. E., Milman, N. & Hansen, S. Sarcoidosis in Denmark 1980–1994. A registry-based incidence study comprising 5536 patients. Sarcoidosis Vasc. Diffuse Lung Dis. 20, 46–52 (2003).
Milman, N. & Selroos, O. Pulmonary sarcoidosis in the Nordic countries 1950–1982. Epidemiology and clinical picture. Sarcoidosis 7, 50–57 (1990).
Baughman, R. P. et al. Sarcoidosis in America. Analysis based on health care use. Ann. Am. Thorac. Soc. 13, 1244–1252 (2016).
Cozier, Y. C. et al. Sarcoidosis in black women in the United States: data from the Black Women’s Health Study. Chest 139, 144–150 (2011).
Dumas, O., Abramovitz, L., Wiley, A. S., Cozier, Y. C. & Camargo, C. A. Jr Epidemiology of sarcoidosis in a prospective cohort study of U.S. women. Ann. Am. Thorac. Soc. 13, 67–71 (2016).
Morimoto, T. et al. Epidemiology of sarcoidosis in Japan. Eur. Respir. J. 31, 372–379 (2008).
Yoon, H. Y., Kim, H. M., Kim, Y. J. & Song, J. W. Prevalence and incidence of sarcoidosis in Korea: a nationwide population-based study. Respir. Res. 19, 158 (2018).
Beghe, D. et al. Sarcoidosis in an Italian province. Prevalence and environmental risk factors. PLOS ONE 12, e0176859 (2017).
Kowalska, M., Niewiadomska, E. & Zejda, J. E. Epidemiology of sarcoidosis recorded in 2006–2010 in the Silesian voivodeship on the basis of routine medical reporting. Ann. Agric. Environ. Med. 21, 55–58 (2014).
Deubelbeiss, U., Gemperli, A., Schindler, C., Baty, F. & Brutsche, M. H. Prevalence of sarcoidosis in Switzerland is associated with environmental factors. Eur. Respir. J. 35, 1088–1097 (2010).
Arkema, E. V. & Cozier, Y. C. Epidemiology of sarcoidosis: current findings and future directions. Ther. Adv. Chronic Dis. 9, 227–240 (2018).
Rabin, D. L. et al. Sarcoidosis: social predictors of severity at presentation. Eur. Respir. J. 24, 601–608 (2004).
Rabin, D. L., Richardson, M. S., Stein, S. R. & Yeager, H. Jr Sarcoidosis severity and socioeconomic status. Eur. Respir. J. 18, 499–506 (2001).
Baughman, R. P. et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am. J. Respir. Crit. Care Med. 164, 1885–1889 (2001).
Grunewald, J. & Eklund, A. Lofgren’s syndrome: human leukocyte antigen strongly influences the disease course. Am. J. Respir. Crit. Care Med. 179, 307–312 (2009).
Rivera, N. V. et al. High-density genetic mapping identifies new susceptibility variants in sarcoidosis phenotypes and shows genomic-driven phenotypic differences. Am. J. Respir. Crit. Care Med. 193, 1008–1022 (2016).
Schurmann, M. et al. Results from a genome-wide search for predisposing genes in sarcoidosis. Am. J. Respir. Crit. Care Med. 164, 840–846 (2001).
Grunewald, J., Spagnolo, P., Wahlstrom, J. & Eklund, A. Immunogenetics of disease-causing inflammation in sarcoidosis. Clin. Rev. Allergy Immunol. 49, 19–35 (2015).
Rossides, M. et al. Familial aggregation and heritability of sarcoidosis: a Swedish nested case-control study. Eur. Respir. J. 52, 1800385 (2018).
Rybicki, B. A. et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am. J. Respir. Crit. Care Med. 164, 2085–2091 (2001).
Sverrild, A. et al. Heredity in sarcoidosis: a registry-based twin study. Thorax 63, 894–896 (2008).
Headings, V. E., Weston, D., Young, R. C. Jr & Hackney, R. L. Jr Familial sarcoidosis with multiple occurrences in eleven families: a possible mechanism of inheritance. Ann. NY Acad. Sci. 278, 377–385 (1976).
Newman, L. S. et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am. J. Respir. Crit. Care Med. 170, 1324–1330 (2004).
Vihlborg, P., Bryngelsson, I. L., Andersson, L. & Graff, P. Risk of sarcoidosis and seropositive rheumatoid arthritis from occupational silica exposure in Swedish iron foundries: a retrospective cohort study. BMJ Open 7, e016839 (2017).
Kern, D. G., Neill, M. A., Wrenn, D. S. & Varone, J. C. Investigation of a unique time-space cluster of sarcoidosis in firefighters. Am. Rev. Respir. Dis. 148, 974–980 (1993).
Prezant, D. J. et al. The incidence, prevalence, and severity of sarcoidosis in New York City firefighters. Chest 116, 1183–1193 (1999).
Crowley, L. E. et al. “Sarcoid like” granulomatous pulmonary disease in World Trade Center disaster responders. Am. J. Ind. Med. 54, 175–184 (2011).
Izbicki, G. et al. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. Chest 131, 1414–1423 (2007).
Jordan, H. T. et al. Sarcoidosis diagnosed after September 11, 2001, among adults exposed to the World Trade Center disaster. J. Occup. Environ. Med. 53, 966–974 (2011).
Esteves, T., Aparicio, G. & Garcia-Patos, V. Is there any association between Sarcoidosis and infectious agents? A systematic review and meta-analysis. BMC Pulm. Med. 16, 165 (2016).
Carlens, C. et al. Smoking, use of moist snuff, and risk of chronic inflammatory diseases. Am. J. Respir. Crit. Care Med. 181, 1217–1222 (2010).
Ungprasert, P., Crowson, C. S. & Matteson, E. L. Smoking, obesity and risk of sarcoidosis: A population-based nested case-control study. Respir. Med. 120, 87–90 (2016).
Valeyre, D. et al. Smoking and pulmonary sarcoidosis: effect of cigarette smoking on prevalence, clinical manifestations, alveolitis, and evolution of the disease. Thorax 43, 516–524 (1988).
Cozier, Y. C. et al. Obesity and weight gain in relation to incidence of sarcoidosis in US black women: data from the Black Women’s Health Study. Chest 147, 1086–1093 (2015).
Dumas, O., Boggs, K. M., Cozier, Y. C., Stampfer, M. J. & Camargo, C. A. Jr Prospective study of body mass index and risk of sarcoidosis in US women. Eur. Respir. J. 50, 1701397 (2017).
Versini, M., Jeandel, P. Y., Rosenthal, E. & Shoenfeld, Y. Obesity in autoimmune diseases: not a passive bystander. Autoimmun. Rev. 13, 981–1000 (2014).
Cozier, Y. C. et al. Reproductive and hormonal factors in relation to incidence of sarcoidosis in US Black women: the Black Women’s Health Study. Am. J. Epidemiol. 176, 635–641 (2012).
Gerke, A. K., Judson, M. A., Cozier, Y. C., Culver, D. A. & Koth, L. L. Disease burden and variability in sarcoidosis. Ann. Am. Thorac. Soc. 14, S421–S428 (2017).
Gribbin, J. et al. Incidence and mortality of idiopathic pulmonary fibrosis and sarcoidosis in the UK. Thorax 61, 980–985 (2006).
Park, J. E. et al. Prevalence, incidence, and mortality of sarcoidosis in Korea, 2003-2015: A nationwide population-based study. Respir. Med. 144S, S28–S34 (2018).
Rossides, M. et al. Sarcoidosis mortality in Sweden: a population-based cohort study. Eur. Respir. J. 51, 1701815 (2018).
Tukey, M. H. et al. Mortality among African American women with sarcoidosis: data from the Black Women’s Health Study. Sarcoidosis Vasc. Diffuse Lung Dis. 30, 128–133 (2013).
Ungprasert, P. et al. Epidemiology of sarcoidosis 1946-2013: a population-based study. Mayo Clin. Proc. 91, 183–188 (2016).
Nardi, A. et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur. Respir. J. 38, 1368–1373 (2011).
Ungprasert, P., Crowson, C. S. & Matteson, E. L. Sarcoidosis increases risk of hospitalized infection. A population-based study, 1976–2013. Ann. Am. Thorac. Soc. 14, 676–681 (2017).
Crawshaw, A. P., Wotton, C. J., Yeates, D. G., Goldacre, M. J. & Ho, L. P. Evidence for association between sarcoidosis and pulmonary embolism from 35-year record linkage study. Thorax 66, 447–448 (2011).
Ungprasert, P., Crowson, C. S. & Matteson, E. L. Risk of cardiovascular disease among patients with sarcoidosis: a population-based retrospective cohort study, 1976–2013. Eur. Respir. J. 49, 1601290 (2017).
Yaqoob, Z. J., Al-Kindi, S. G. & Zein, J. G. Sarcoidosis and risk of VTE: validation with big data. Chest 151, 1398–1399 (2017).
Ungprasert, P., Crowson, C. S. & Matteson, E. L. Association of sarcoidosis with increased risk of VTE: a population-based study, 1976 to 2013. Chest 151, 425–430 (2017).
Wu, C. H. et al. Comorbid autoimmune diseases in patients with sarcoidosis: a nationwide case-control study in Taiwan. J. Dermatol. 44, 423–430 (2017).
Bonifazi, M. et al. Sarcoidosis and cancer risk: systematic review and meta-analysis of observational studies. Chest 147, 778–791 (2015).
Hunninghake, G. W. et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc. Diffuse Lung Dis. 16, 149–173 (1999).
Facco, M. et al. Sarcoidosis is a Th1/Th17 multisystem disorder. Thorax 66, 144–150 (2011).
Ramstein, J. et al. Interferon-gamma-producing Th17.1 Cells are increased in sarcoidosis and more prevalent than Th1 cells. Am. J. Respir. Crit. Care Med. 193, 1281–1291 (2016).
Broos, C. E. et al. Increased T-helper 17.1 cells in sarcoidosis mediastinal lymph nodes. Eur. Respir. J. 51, 1701124 (2018).
Miedema, J. R. et al. Th17-lineage cells in pulmonary sarcoidosis and Lofgren’s syndrome: friend or foe? J. Autoimmun. 87, 82–96 (2018).
Ostadkarampour, M. et al. Higher levels of interleukin IL-17 and antigen-specific IL-17 responses in pulmonary sarcoidosis patients with Lofgren’s syndrome. Clin. Exp. Immunol. 178, 342–352 (2014).
Kaiser, Y. et al. Expanded lung T-bet+RORgammaT+ CD4+ T cells in sarcoidosis patients with a favourable disease phenotype. Eur. Respir. J. 48, 484–494 (2016).
Miyara, M. et al. The immune paradox of sarcoidosis and regulatory T cells. J. Exp. Med. 203, 359–370 (2006).
Taflin, C. et al. FoxP3+ regulatory T cells suppress early stages of granuloma formation but have little impact on sarcoidosis lesions. Am. J. Pathol. 174, 497–508 (2009).
Prasse, A. et al. Inhaled vasoactive intestinal peptide exerts immunoregulatory effects in sarcoidosis. Am. J. Respir. Crit. Care Med. 182, 540–548 (2010).
Li, H., Zhao, X., Wang, J., Zong, M. & Yang, H. Bioinformatics analysis of gene expression profile data to screen key genes involved in pulmonary sarcoidosis. Gene 596, 98–104 (2017).
Rosenbaum, J. T. et al. Hypothesis: sarcoidosis is a STAT1-mediated disease. Clin. Immunol. 132, 174–183 (2009).
Zhou, T. et al. Identification of Jak-STAT signaling involvement in sarcoidosis severity via a novel microRNA-regulated peripheral blood mononuclear cell gene signature. Sci. Rep. 7, 4237 (2017).
Zhou, T. et al. Peripheral blood gene expression as a novel genomic biomarker in complicated sarcoidosis. PLOS ONE 7, e44818 (2012).
Celada, L. J. et al. PD-1 up-regulation on CD4+ T cells promotes pulmonary fibrosis through STAT3-mediated IL-17A and TGF-beta1 production. Sci. Transl Med. 10, eaar8356 (2018).
Damsky, W., Thakral, D., Emeagwali, N., Galan, A. & King, B. Tofacitinib treatment and molecular analysis of cutaneous sarcoidosis. N. Engl. J. Med. 379, 2540–2546 (2018).
Rotenberg, C. et al. Dramatic response of refractory sarcoidosis under ruxolitinib in a patient with associated JAK2-mutated polycythemia. Eur. Respir. J. 52, 1801482 (2018).
Vega, L. E. & Espinoza, L. R. HIV infection and its effects on the development of autoimmune disorders. Pharmacol. Res. 129, 1–9 (2018).
Foulon, G. et al. Sarcoidosis in HIV-infected patients in the era of highly active antiretroviral therapy. Clin. Infect. Dis. 38, 418–425 (2004).
Morris, D. G. et al. Sarcoidosis following HIV infection: evidence for CD4+ lymphocyte dependence. Chest 124, 929–935 (2003).
Linke, M. et al. Chronic signaling via the metabolic checkpoint kinase mTORC1 induces macrophage granuloma formation and marks sarcoidosis progression. Nat. Immunol. 18, 293–302 (2017).
Dubaniewicz, A. Microbial and human heat shock proteins as ‘danger signals’ in sarcoidosis. Hum. Immunol. 74, 1550–1558 (2013).
Wiken, M., Grunewald, J., Eklund, A. & Wahlstrom, J. Higher monocyte expression of TLR2 and TLR4, and enhanced pro-inflammatory synergy of TLR2 with NOD2 stimulation in sarcoidosis. J. Clin. Immunol. 29, 78–89 (2009).
Wiken, M. et al. No evidence of altered alveolar macrophage polarization, but reduced expression of TLR2, in bronchoalveolar lavage cells in sarcoidosis. Respir. Res. 11, 121 (2010).
Chen, E. S. & Moller, D. R. Sarcoidosis—scientific progress and clinical challenges. Nat. Rev. Rheumatol. 7, 457–467 (2011).
Chen, E. S. et al. Serum amyloid A regulates granulomatous inflammation in sarcoidosis through Toll-like receptor-2. Am. J. Respir. Crit. Care Med. 181, 360–373 (2010).
Katchar, K., Wahlstrom, J., Eklund, A. & Grunewald, J. Highly activated T cell receptor AV2S3+ CD4+ lung T cell expansions in pulmonary sarcoidosis. Am. J. Respir. Crit. Care Med. 163, 1540–1545 (2001).
Muller-Quernheim, J., Kronke, M., Strausz, J., Schykowski, M. & Ferlinz, R. Interleukin-2 receptor gene expression by bronchoalveolar lavage lymphocytes in pulmonary sarcoidosis. Am. Rev. Respir. Dis. 140, 82–88 (1989).
Karakaya, B., Kaiser, Y., van Moorsel, C. H. M. & Grunewald, J. Lofgren’s syndrome: diagnosis, management, and disease pathogenesis. Semin. Respir. Crit. Care Med. 38, 463–476 (2017).
Sakthivel, P., Grunewald, J., Eklund, A., Bruder, D. & Wahlstrom, J. Pulmonary sarcoidosis is associated with high-level inducible co-stimulator (ICOS) expression on lung regulatory T cells–possible implications for the ICOS/ICOS-ligand axis in disease course and resolution. Clin. Exp. Immunol. 183, 294–306 (2015).
Idali, F. et al. Reduced Th1 response in the lungs of HLA-DRB1*0301 patients with pulmonary sarcoidosis. Eur. Respir. J. 27, 451–459 (2006).
Idali, F. et al. Altered expression of T cell immunoglobulin-mucin (TIM) molecules in bronchoalveolar lavage CD4+ T cells in sarcoidosis. Respir. Res. 10, 42 (2009).
Chen, E. S. & Moller, D. R. Etiology of sarcoidosis. Clin. Chest Med. 29, 365–377 (2008).
Grunewald, J. et al. Restricted V alpha 2.3 gene usage by CD4+ T lymphocytes in bronchoalveolar lavage fluid from sarcoidosis patients correlates with HLA-DR3. Eur. J. Immunol. 22, 129–135 (1992).
Moller, D. R., Konishi, K., Kirby, M., Balbi, B. & Crystal, R. G. Bias toward use of a specific T cell receptor beta-chain variable region in a subgroup of individuals with sarcoidosis. J. Clin. Invest. 82, 1183–1191 (1988).
Grunewald, J. et al. T cell receptor variable region gene usage by CD4+ and CD8+ T cells in bronchoalveolar lavage fluid and peripheral blood of sarcoidosis patients. Proc. Natl Acad. Sci. USA 91, 4965–4969 (1994).
Grunewald, J., Berlin, M., Olerup, O. & Eklund, A. Lung T-helper cells expressing T cell receptor AV2S3 associate with clinical features of pulmonary sarcoidosis. Am. J. Respir. Crit. Care Med. 161, 814–818 (2000).
Ahlgren, K. M., Ruckdeschel, T., Eklund, A., Wahlstrom, J. & Grunewald, J. T cell receptor-Vbeta repertoires in lung and blood CD4+ and CD8+ T cells of pulmonary sarcoidosis patients. BMC Pulm. Med. 14, 50 (2014).
Grunewald, J. et al. T cell receptor-HLA-DRB1 associations suggest specific antigens in pulmonary sarcoidosis. Eur. Respir. J. 47, 898–909 (2016).
Mitchell, A. M. et al. Shared alphabeta TCR usage in lungs of sarcoidosis patients with Lofgren’s syndrome. J. Immunol. 199, 2279–2290 (2017).
Heckmann, J. G., Stefan, H., Heuss, D., Hopp, P. & Neundorfer, B. Isolated muscular sarcoidosis. Eur. J. Neurol. 8, 365–366 (2001).
Heyder, T. et al. Approach for identifying human leukocyte antigen (HLA)-DR bound peptides from scarce clinical samples. Mol. Cell. Proteomics 15, 3017–3029 (2016).
Kveim, A. A new and specific cutaneous reaction in Boeck’s sarcoid [Norwegian]. Nord. Med. 9, 169 (1941).
Siltzbach, L. The Kveim test in sarcoidosis: a study of 750 patients. JAMA 178, 476–482 (1961).
Chase, M. The preparation and standardization of Kveim testing antigen. Am. Rev. Respir. Dis. 84, 86–88 (1961).
Eberhardt, C. et al. Proteomic analysis of Kveim reagent identifies targets of cellular immunity in sarcoidosis. PLOS ONE 12, e0170285 (2017).
Klein, J. T. et al. Selection of oligoclonal V beta-specific T cells in the intradermal response to Kveim-Siltzbach reagent in individuals with sarcoidosis. J. Immunol. 154, 1450–1460 (1995).
Coulombe, P. A. & Wong, P. Cytoplasmic intermediate filaments revealed as dynamic and multipurpose scaffolds. Nat. Cell Biol. 6, 699–706 (2004).
Mor-Vaknin, N., Punturieri, A., Sitwala, K. & Markovitz, D. M. Vimentin is secreted by activated macrophages. Nat. Cell Biol. 5, 59–63 (2003).
Wahlstrom, J. et al. Identification of HLA-DR-bound peptides presented by human bronchoalveolar lavage cells in sarcoidosis. J. Clin. Invest. 117, 3576–3582 (2007).
Wahlstrom, J. et al. Autoimmune T cell responses to antigenic peptides presented by bronchoalveolar lavage cell HLA-DR molecules in sarcoidosis. Clin. Immunol. 133, 353–363 (2009).
Kinloch, A. J. et al. In situ humoral immunity to vimentin in HLA-DRB1*03+ patients with pulmonary sarcoidosis. Front. Immunol. 9, 1516 (2018).
Wegner, N. et al. Autoimmunity to specific citrullinated proteins gives the first clues to the etiology of rheumatoid arthritis. Immunol. Rev. 233, 34–54 (2010).
Kinloch, A. J. et al. Vimentin is a dominant target of in situ humoral immunity in human lupus tubulointerstitial nephritis. Arthritis Rheumatol. 66, 3359–3370 (2014).
Kaiser, Y., Eklund, A. & Grunewald, J. Moving target — shifting the focus to pulmonary sarcoidosis as an autoimmune spectrum disorder. Eur. Respir. J. https://doi.org/10.1183/13993003.021532018 (2019).
Chiang, C. H. & Lai, F. J. Sarcoidosis on the injection sites following treatment of interferon-alpha and ribavirin for hepatitis C. J. Formos. Med. Assoc. 113, 981–982 (2014).
Jeon, E. K. et al. First reported case of interferon-alpha-induced sarcoidosis in an Asian patient with malignant melanoma. Asia Pac. J. Clin. Oncol. 12, e347–e349 (2016).
Viana de Andrade, A. C. et al. Development of systemic sarcoidosis and xanthoma planum during multiple sclerosis treatment with interferon-beta 1a: case report. Int. J. Dermatol. 54, e140–e145 (2015).
Berthod, G. et al. Pulmonary sarcoid-like granulomatosis induced by ipilimumab. J. Clin. Oncol. 30, e156–e159 (2012).
Spain, L., Diem, S. & Larkin, J. Management of toxicities of immune checkpoint inhibitors. Cancer Treat. Rev. 44, 51–60 (2016).
Abdel-Wahab, N., Shah, M. & Suarez-Almazor, M. E. Adverse events associated with immune checkpoint blockade in patients with cancer: a systematic review of case reports. PLOS ONE 11, e0160221 (2016).
Chopra, A., Nautiyal, A., Kalkanis, A. & Judson, M. A. Drug-induced sarcoidosis-like reactions. Chest 154, 664–677 (2018).
Broos, C. E. et al. Decreased cytotoxic T-lymphocyte antigen 4 expression on regulatory T cells and Th17 cells in sarcoidosis: double trouble? Am. J. Respir. Crit. Care Med. 192, 763–765 (2015).
Celada, L. J. et al. Programmed death-1 inhibition of phosphatidylinositol 3-kinase/AKT/mechanistic target of rapamycin signaling impairs sarcoidosis CD4+ T cell proliferation. Am. J. Respir. Cell. Mol. Biol. 56, 74–82 (2017).
Palmer, B. E. et al. Up-regulation of programmed death-1 expression on beryllium-specific CD4+ T cells in chronic beryllium disease. J. Immunol. 180, 2704–2712 (2008).
Mack, D. G. et al. 4-1BB enhances proliferation of beryllium-specific T cells in the lung of subjects with chronic beryllium disease. J. Immunol. 181, 4381–4388 (2008).
Birnbaum, M. R. et al. Nivolumab-related cutaneous sarcoidosis in a patient with lung adenocarcinoma. JAAD Case Rep. 3, 208–211 (2017).
Danlos, F. X. et al. Nivolumab-induced sarcoid-like granulomatous reaction in a patient with advanced melanoma. Chest 149, e133–e136 (2016).
Montaudie, H., Pradelli, J., Passeron, T., Lacour, J. P. & Leroy, S. Pulmonary sarcoid-like granulomatosis induced by nivolumab. Br. J. Dermatol. 176, 1060–1063 (2017).
Suozzi, K. C. et al. Immune-related sarcoidosis observed in combination ipilimumab and nivolumab therapy. JAAD Case Rep. 2, 264–268 (2016).
Vogel, W. V. et al. Ipilimumab-induced sarcoidosis in a patient with metastatic melanoma undergoing complete remission. J. Clin. Oncol. 30, e7–e10 (2012).
Marijon, E., Mirabel, M., Celermajer, D. S. & Jouven, X. Rheumatic heart disease. Lancet 379, 953–964 (2012).
Demirkok, S. S., Basaranoglu, M. & Akbilgic, O. Seasonal variation of the onset of presentations in stage 1 sarcoidosis. Int. J. Clin. Pract. 60, 1443–1450 (2006).
Glennas, A. et al. Acute sarcoid arthritis: occurrence, seasonal onset, clinical features and outcome. Br. J. Rheumatol. 34, 45–50 (1995).
Wilsher, M. L. Seasonal clustering of sarcoidosis presenting with erythema nodosum. Eur. Respir. J. 12, 1197–1199 (1998).
Heyll, A. et al. Possible transmission of sarcoidosis via allogeneic bone marrow transplantation. Bone Marrow Transplant. 14, 161–164 (1994).
Gupta, D., Agarwal, R., Aggarwal, A. N. & Jindal, S. K. Molecular evidence for the role of mycobacteria in sarcoidosis: a meta-analysis. Eur. Respir. J. 30, 508–516 (2007).
Chen, E. S. et al. T cell responses to mycobacterial catalase-peroxidase profile a pathogenic antigen in systemic sarcoidosis. J. Immunol. 181, 8784–8796 (2008).
Song, Z. et al. Mycobacterial catalase-peroxidase is a tissue antigen and target of the adaptive immune response in systemic sarcoidosis. J. Exp. Med. 201, 755–767 (2005).
Drake, W. P. et al. Cellular recognition of Mycobacterium tuberculosis ESAT-6 and KatG peptides in systemic sarcoidosis. Infect. Immun. 75, 527–530 (2007).
Oswald-Richter, K. et al. Mycobacterial ESAT-6 and katG are recognized by sarcoidosis CD4+ T cells when presented by the American sarcoidosis susceptibility allele, DRB1*1101. J. Clin. Immunol. 30, 157–166 (2009).
Eishi, Y. et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J. Clin. Microbiol. 40, 198–204 (2002).
Ishige, I. et al. Propionibacterium acnes is the most common bacterium commensal in peripheral lung tissue and mediastinal lymph nodes from subjects without sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 22, 33–42 (2005).
Tebben, P. J., Singh, R. J. & Kumar, R. Vitamin D-mediated hypercalcemia: mechanisms, diagnosis, and treatment. Endocr. Rev. 37, 521–547 (2016).
Korenromp, I. H., Grutters, J. C., van den Bosch, J. M. & Heijnen, C. J. Post-inflammatory fatigue in sarcoidosis: personality profiles, psychological symptoms and stress hormones. J. Psychosom. Res. 72, 97–102 (2012).
Braam, A. W. et al. Influence of repeated maximal exercise testing on biomarkers and fatigue in sarcoidosis. Brain Behav. Immun. 33, 57–64 (2013).
Korenromp, I. H. et al. Reduced Th2 cytokine production by sarcoidosis patients in clinical remission with chronic fatigue. Brain Behav. Immun. 25, 1498–1502 (2011).
Korenromp, I. H. E., Heijnen, C. J., Vogels, O. J. M., van den Bosch, J. M. M. & Grutters, J. C. Characterization of chronic fatigue in patients with sarcoidosis in clinical remission. Chest 140, 441–447 (2011).
Strookappe, B. et al. Predictors of fatigue in sarcoidosis: the value of exercise testing. Respir. Med. 116, 49–54 (2016).
Valeyre, D. et al. Sarcoidosis. Lancet 383, 1155–1167 (2014).
Wells, A. U. Sarcoidosis: a benign disease or a culture of neglect? Respir. Med. 144S, S1–S2 (2018).
Heerfordt, C. Uber eine febris uveo-parotidea subchronica. Albrecht Von Graefes Arch. Ophthalmol. 70, 254–258 (1909).
Scadding, J. G. Sarcoidosis, with special reference to lung changes. Br. Med. J. 1, 745–753 (1950).
Scadding, J. G. Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Br. Med. J. 2, 1165–1172 (1961).
Abehsera, M. et al. Sarcoidosis with pulmonary fibrosis: CT patterns and correlation with pulmonary function. Am. J. Roentgenol. 174, 1751–1757 (2000).
Schupp, J. C. et al. Phenotypes of organ involvement in sarcoidosis. Eur. Respir. J. 51, 1700991 (2018).
Sayah, D. M., Bradfield, J. S., Moriarty, J. M., Belperio, J. A. & Lynch, J. P. 3rd Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin. Respir. Crit. Care Med. 38, 477–498 (2017).
Birnie, D. H., Nery, P. B., Ha, A. C. & Beanlands, R. S. Cardiac sarcoidosis. J. Am. Coll. Cardiol. 68, 411–421 (2016).
Chau, E. M., Fan, K. Y. & Chow, W. H. Cardiac sarcoidosis: a potentially fatal but treatable form of infiltrative heart disease. Hong Kong Med. J. 12, 65–67 (2006).
Culver, D. A., Ribeiro Neto, M. L., Moss, B. P. & Willis, M. A. Neurosarcoidosis. Semin. Respir. Crit. Care Med. 38, 499–513 (2017).
Tabuena, R. P. et al. Diabetes insipidus from neurosarcoidosis: long-term follow-up for more than eight years. Intern. Med. 43, 960–966 (2004).
Hoitsma, E. et al. Small fibre neuropathy in sarcoidosis. Lancet 359, 2085–2086 (2002).
Saygin, D., Karunamurthy, A., English, J. & Aggarwal, R. Tattoo reaction as a presenting manifestation of systemic sarcoidosis. Rheumatology 58, 927 (2018).
Fallahi, P. et al. The association of other autoimmune diseases in patients with autoimmune thyroiditis: review of the literature and report of a large series of patients. Autoimmun. Rev. 15, 1125–1128 (2016).
Costabel, U. & Hunninghake, G. W. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur. Respir. J. 14, 735–737 (1999).
Bargagli, E. & Prasse, A. Sarcoidosis: a review for the internist. Intern. Emerg. Med. 13, 325–331 (2018).
James, W. E. et al. Clinical features of extrapulmonary sarcoidosis without lung involvement. Chest 154, 349–356 (2018).
Judson, M. A. et al. Two year prognosis of sarcoidosis: the ACCESS experience. Sarcoidosis Vasc. Diffuse Lung Dis. 20, 204–211 (2003).
Gilbert, S., Steinbrech, D. S., Landas, S. K. & Hunninghake, G. W. Amounts of angiotensin-converting enzyme mRNA reflect the burden of granulomas in granulomatous lung disease. Am. Rev. Respir. Dis. 148, 483–486 (1993).
Berlin, M., Fogdell-Hahn, A., Olerup, O., Eklund, A. & Grunewald, J. HLA-DR predicts the prognosis in Scandinavian patients with pulmonary sarcoidosis. Am. J. Respir. Crit. Care Med. 156, 1601–1605 (1997).
Huitema, M. P. et al. Pulmonary artery diameter to predict pulmonary hypertension in pulmonary sarcoidosis. Eur. Respir. J. 47, 673–676 (2016).
Walsh, S. L. et al. An integrated clinicoradiological staging system for pulmonary sarcoidosis: a case-cohort study. Lancet Respir. Med. 2, 123–130 (2014).
Soussan, M. et al. Clinical value of a high-fat and low-carbohydrate diet before FDG-PET/CT for evaluation of patients with suspected cardiac sarcoidosis. J. Nucl. Cardiol. 20, 120–127 (2013).
Vorselaars, A. D. et al. Effectiveness of infliximab in refractory FDG PET-positive sarcoidosis. Eur. Respir. J. 46, 175–185 (2015).
Yatsynovich, Y. et al. Updates on the role of imaging in cardiac sarcoidosis. Curr. Treat. Opt. Cardiovasc. Med. 20, 74 (2018).
Adams, H., Keijsers, R. G., Korenromp, I. H. & Grutters, J. C. FDG PET for gauging of sarcoid disease activity. Semin. Respir. Crit. Care Med. 35, 352–361 (2014).
Goktalay, T. et al. The role of endobronchial biopsy in the diagnosis of pulmonary sarcoidosis. Turk. Thorac. J. 17, 22–27 (2016).
Heron, M. et al. Evaluation of CD103 as a cellular marker for the diagnosis of pulmonary sarcoidosis. Clin. Immunol. 126, 338–344 (2008).
Bonifazi, M. et al. Conventional versus ultrasound-guided transbronchial needle aspiration for the diagnosis of hilar/mediastinal lymph adenopathies: a randomized controlled trial. Respiration 94, 216–223 (2017).
von Bartheld, M. B. et al. Endosonography versus conventional bronchoscopy for the diagnosis of sarcoidosis: the GRANULOMA randomized clinical trial. JAMA 309, 2457–2464 (2013).
Baughman, R. P. et al. Defining the clinical outcome status (COS) in sarcoidosis: results of WASOG Task Force. Sarcoidosis Vasc. Diffuse Lung Dis. 28, 56–64 (2011).
Pereira, C. A., Dornfeld, M. C., Baughman, R. & Judson, M. A. Clinical phenotypes in sarcoidosis. Curr. Opin. Pulm. Med. 20, 496–502 (2014).
Prasse, A. et al. Phenotyping sarcoidosis from a pulmonary perspective. Am. J. Respir. Crit. Care Med. 177, 330–336 (2008).
James, W. E. & Baughman, R. Treatment of sarcoidosis: grading the evidence. Expert Rev. Clin. Pharmacol. 11, 677–687 (2018).
Schutt, A. C., Bullington, W. M. & Judson, M. A. Pharmacotherapy for pulmonary sarcoidosis: a Delphi consensus study. Respir. Med. 104, 717–723 (2010).
Atkins, C. & Wilson, A. M. Managing fatigue in sarcoidosis — a systematic review of the evidence. Chron. Respir. Dis. 14, 161–173 (2017).
Dekhuijzen, P. N. & Decramer, M. Steroid-induced myopathy and its significance to respiratory disease: a known disease rediscovered. Eur. Respir. J. 5, 997–1003 (1992).
Schreiber, T. & Windisch, W. Respiratory muscle involvement in sarcoidosis. Expert Rev. Respir. Med. 12, 545–548 (2018).
O’Connell, K. et al. Neurosarcoidosis: clinical presentations and changing treatment patterns in an Irish Caucasian population. Ir. J. Med. Sci. 186, 759–766 (2017).
Padala, S. K., Peaslee, S., Sidhu, M. S., Steckman, D. A. & Judson, M. A. Impact of early initiation of corticosteroid therapy on cardiac function and rhythm in patients with cardiac sarcoidosis. Int. J. Cardiol. 227, 565–570 (2017).
Baughman, R. P., Winget, D. B. & Lower, E. E. Methotrexate is steroid sparing in acute sarcoidosis: results of a double blind, randomized trial. Sarcoidosis Vasc. Diffuse Lung Dis. 17, 60–66 (2000).
Müller-Quernheim, J., Kienast, K., Held, M., Pfeifer, S. & Costabel, U. Treatment of chronic sarcoidosis with an azathioprine/prednisolone regimen. Eur. Respir. J. 14, 1117–1122 (1999).
Hunninghake, G. W. et al. Outcome of the treatment of sarcoidosis. Am. J. Respir. Crit. Care Med. 149, 893–898 (1994).
Gibson, G. J. et al. British Thoracic Society Sarcoidosis study: effects of long term corticosteroid treatment. Thorax 51, 238–247 (1996).
Drent, M. et al. Does the cellular bronchoalveolar lavage fluid profile reflect the severity of sarcoidosis? Eur. Respir. J. 13, 1338–1344 (1999).
Ward, K., O’Connor, C., Odlum, C. & Fitzgerald, M. X. Prognostic value of bronchoalveolar lavage in sarcoidosis: the critical influence of disease presentation. Thorax 44, 6–12 (1989).
Ziegenhagen, M. W., Rothe, M. E., Schlaak, M. & Müller-Quernheim, J. Bronchoalveolar and serological parameters reflecting the severity of sarcoidosis. Eur. Respir. J. 21, 407–413 (2003).
Paramothayan, S. & Lasserson, T. Treatments for pulmonary sarcoidosis. Respir. Med. 102, 1–9 (2008).
Broos, C. E. et al. No evidence found for an association between prednisone dose and FVC change in newly-treated pulmonary sarcoidosis. Respir. Med. 138S, S31–S37 (2018).
Conron, M., Young, C. & Beynon, H. L. Calcium metabolism in sarcoidosis and its clinical implications. Rheumatology 39, 707–713 (2000).
Baughman, R. P., Janovcik, J., Ray, M., Sweiss, N. & Lower, E. E. Calcium and vitamin D metabolism in sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 30, 113–120 (2013).
Burke, R. R., Rybicki, B. A. & Rao, D. S. Calcium and vitamin D in sarcoidosis: how to assess and manage. Semin. Respir. Crit. Care Med. 31, 474–484 (2010).
Baughman, R. P., Nunes, H., Sweiss, N. J. & Lower, E. E. Established and experimental medical therapy of pulmonary sarcoidosis. Eur. Respir. J. 41, 1424–1438 (2013).
Baughman, R. P., Barney, J. B., O’Hare, L. & Lower, E. E. A retrospective pilot study examining the use of Acthar gel in sarcoidosis patients. Respir. Med. 110, 66–72 (2016).
Ziegenhagen, M. W., Rothe, M. E., Zissel, G. & Muller-Quernheim, J. Exaggerated TNFalpha release of alveolar macrophages in corticosteroid resistant sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 19, 185–190 (2002).
Baughman, R. P., Judson, M. A., Teirstein, A. S., Moller, D. R. & Lower, E. E. Thalidomide for chronic sarcoidosis. Chest 122, 227–232 (2002).
Giv, M. J., Yoosuff, A. & Bazargan, A. Use of lenalidomide in 5q-myelodysplastic syndrome provides novel treatment prospects in management of pulmonary sarcoidosis. Chest 148, e35–e37 (2015).
Moller, D. R. et al. Inhibition of human interleukin-12 production by pentoxifylline. Immunology 91, 197–203 (1997).
Park, M. K. et al. Steroid-sparing effects of pentoxifylline in pulmonary sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 26, 121–131 (2009).
Gomes, J. P., Watad, A. & Shoenfeld, Y. Nicotine and autoimmunity: the lotus’ flower in tobacco. Pharmacol. Res. 128, 101–109 (2018).
Julian, M. W. et al. Nicotine treatment improves Toll-like receptor 2 and Toll-like receptor 9 responsiveness in active pulmonary sarcoidosis. Chest 143, 461–470 (2013).
Saketkoo, L. A. & Baughman, R. P. Biologic therapies in the treatment of sarcoidosis. Expert Rev. Clin. Immunol. 12, 817–825 (2016).
Baughman, R. P. et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am. J. Respir. Crit. Care Med. 174, 795–802 (2006).
Judson, M. A. et al. Safety and efficacy of ustekinumab or golimumab in patients with chronic sarcoidosis. Eur. Respir. J. 44, 1296–1307 (2014).
Loza, M. J. et al. Inflammatory profile and response to anti-tumor necrosis factor therapy in patients with chronic pulmonary sarcoidosis. Clin. Vaccine Immunol. 18, 931–939 (2011).
Judson, M. A. et al. Efficacy of infliximab in extrapulmonary sarcoidosis: results from a randomised trial. Eur. Respir. J. 31, 1189–1196 (2008).
Hostettler, K. E., Studler, U., Tamm, M. & Brutsche, M. H. Long-term treatment with infliximab in patients with sarcoidosis. Respiration 83, 218–224 (2012).
Perez-Alvarez, R., Perez-de-Lis, M. & Ramos-Casals, M. Biologics-induced autoimmune diseases. Curr. Opin. Rheumatol. 25, 56–64 (2013).
Crouser, E. D. Role of imbalance between Th17 and regulatory T cells in sarcoidosis. Curr. Opin. Pulm. Med. 24, 521–526 (2018).
Culver, D. A. et al. Cibinetide improves corneal nerve fiber abundance in patients with sarcoidosis-associated small nerve fiber loss and neuropathic pain. Invest. Ophthalmol. Vis. Sci. 58, BIO52–BIO60 (2017).
Lingner, H. et al. Short-term effects of a multimodal 3-week inpatient pulmonary rehabilitation programme for patients with sarcoidosis: the ProKaSaRe Study. Respiration 95, 343–353 (2018).
Shlobin, O. A. & Nathan, S. D. Management of end-stage sarcoidosis: pulmonary hypertension and lung transplantation. Eur. Respir. J. 39, 1520–1533 (2012).
Baughman, R. P. et al. Bosentan for sarcoidosis associated pulmonary hypertension: A double-blind placebo controlled randomized trial. Chest 145, 810–817 (2013).
Drent, M., Strookappe, B., Hoitsma, E. & De Vries, J. Consequences of sarcoidosis. Clin. Chest Med. 36, 727–737 (2015).
Borrell-Carrio, F., Suchman, A. L. & Epstein, R. M. The biopsychosocial model 25 years later: principles, practice, and scientific inquiry. Ann. Fam. Med. 2, 576–582 (2004).
Judson, M. A. Strategies for identifying pulmonary sarcoidosis patients at risk for severe or chronic disease. Expert Rev. Respir. Med. 11, 111–118 (2017).
Judson, M. A., Chaudhry, H., Louis, A., Lee, K. & Yucel, R. The effect of corticosteroids on quality of life in a sarcoidosis clinic: the results of a propensity analysis. Respir. Med. 109, 526–531 (2015).
Saketkoo, L. A. et al. Feasibility, utility and symptom impact of modified mindfulness training in sarcoidosis. ERJ Open Res. 4, 00085-2017 (2018).
Shires, A., Sharpe, L. & Newton John, T. The relative efficacy of mindfulness versus distraction: the moderating role of attentional bias. Eur. J. Pain 23, 727–738 (2018).
Moor, C. C. et al. Needs, perceptions and education in sarcoidosis: a live interactive survey of patients and partners. Lung 196, 569–575 (2018).
Smets, E. M., Garssen, B., Bonke, B. & De Haes, J. C. The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. J. Psychosom. Res. 39, 315–325 (1995).
Minnock, P., Kirwan, J. & Bresnihan, B. Fatigue is a reliable, sensitive and unique outcome measure in rheumatoid arthritis. Rheumatology 48, 1533–1536 (2009).
Petri, M. A., Martin, R. S., Scheinberg, M. A. & Furie, R. A. Assessments of fatigue and disease activity in patients with systemic lupus erythematosus enrolled in the Phase 2 clinical trial with blisibimod. Lupus 26, 27–37 (2017).
Abad, S. et al. Association of peripheral multifocal choroiditis with sarcoidosis: a study of thirty-seven patients. Arthritis Rheum. 51, 974–982 (2004).
De Boer, S. & Wilsher, M. L. Validation of the Sarcoidosis Health Questionnaire in a non-US population. Respirology 17, 519–524 (2012).
De Vries, J., Michielsen, H., Van Heck, G. L. & Drent, M. Measuring fatigue in sarcoidosis: the Fatigue Assessment Scale (FAS). Br. J. Health Psychol. 9, 279–291 (2004).
Wilsher, M. L. Psychological stress in sarcoidosis. Curr. Opin. Pulm. Med. 18, 524–527 (2012).
Benn, B. S. et al. Sleep disturbance and symptom burden in sarcoidosis. Respir. Med. 144S, S35–S40 (2018).
Bosse-Henck, A., Wirtz, H. & Hinz, A. Subjective sleep quality in sarcoidosis. Sleep Med. 16, 570–576 (2015).
Hinz, A., Geue, K., Zenger, M., Wirtz, H. & Bosse-Henck, A. Daytime sleepiness in patients diagnosed with sarcoidosis compared with the general population. Can. Respir. J. 2018, 6853948 (2018).
Lal, C., Medarov, B. I. & Judson, M. A. Interrelationship between sleep-disordered breathing and sarcoidosis. Chest 148, 1105–1114 (2015).
Chang, B. et al. Depression in sarcoidosis. Am. J. Respir. Crit. Care Med. 163, 329–334 (2001).
Ireland, J. & Wilsher, M. Perceptions and beliefs in sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 27, 36–42 (2010).
Elwyn, G., Cochran, N. & Pignone, M. Shared decision making — the importance of diagnosing preferences. JAMA Intern. Med. 177, 1239–1240 (2017).
Dubovsky, A. N., Arvikar, S., Stern, T. A. & Axelrod, L. The neuropsychiatric complications of glucocorticoid use: steroid psychosis revisited. Psychosomatics 53, 103–115 (2012).
Rees, J., O’Boyle, C. & MacDonagh, R. Quality of life: impact of chronic illness on the partner. J. R. Soc. Med. 94, 563–566 (2001).
Sklenarova, H. et al. When do we need to care about the caregiver? Supportive care needs, anxiety, and depression among informal caregivers of patients with cancer and cancer survivors. Cancer 121, 1513–1519 (2015).
Saketkoo, L. A. et al. Reconciling healthcare professional and patient perspectives in the development of disease activity and response criteria in connective tissue disease-related interstitial lung diseases. J. Rheumatol. 41, 792–798 (2014).
Arkema, E. V., Eklund, A., Grunewald, J. & Bruze, G. Work ability before and after sarcoidosis diagnosis in Sweden. Respir. Med. 144S, S7–S12 (2018).
Kawalec, P. P. & Malinowski, K. P. The indirect costs of systemic autoimmune diseases, systemic lupus erythematosus, systemic sclerosis and sarcoidosis: a summary of 2012 real-life data from the Social Insurance Institution in Poland. Expert Rev. Pharmacoecon. Outcomes Res. 15, 667–673 (2015).
Rice, J. B. et al. Economic burden of sarcoidosis in a commercially-insured population in the United States. J. Med. Econ. 20, 1048–1055 (2017).
Borgh, M., Eek, F., Wagman, P. & Hakansson, C. Organisational factors and occupational balance in working parents in Sweden. Scand. J. Public Health 46, 409–416 (2018).
Markham, S. E. & Markham, I. S. Biometeorological effects on worker absenteeism. Int. J. Biometeorol. 49, 317–324 (2005).
Muller, C. California State University catastrophic (Cat) leave donation program: demographics, economic security, and social equity. J. Health Hum. Serv. Adm. 38, 108–159 (2015).
Alexanderson, H. et al. Resistive home exercise in patients with recent-onset polymyositis and dermatomyositis — a randomized controlled single-blinded study with a 2-year followup. J. Rheumatol. 41, 1124–1132 (2014).
Bernardi, E., Pomidori, L., Cassutti, F. & Cogo, A. Home-based, moderate-intensity exercise training using a metronome improves the breathing pattern and oxygen saturation during exercise in patients with COPD. J. Cardiopulm. Rehabil. Prev. 38, E16–E18 (2018).
Russell, A. M. et al. Daily home spirometry: an effective tool for detecting progression in idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 194, 989–997 (2016).
Tuckson, R. V., Edmunds, M. & Hodgkins, M. L. Telehealth. N. Engl. J. Med. 377, 1585–1592 (2017).
Bolat, M. S., Celik, B., Celik, H. K. & Akdeniz, E. The impact of thoracotomy on psychological and sexual function in men with lung cancer. Rev. Int. Androl. https://doi.org/10.1016/j.androl.2018.05.002 (2018).
Hassanin, A. M., Ismail, N. N., El Guindi, A. & Sowailam, H. A. The emotional burden of chronic skin disease dominates physical factors among women, adversely affecting quality of life and sexual function. J. Psychosom. Res. 115, 53–57 (2018).
Ostlund, G., Bjork, M., Valtersson, E. & Sverker, A. Lived experiences of sex life difficulties in men and women with early RA — the Swedish TIRA Project. Musculoskeletal Care 13, 248–257 (2015).
Fourie, S., Jackson, D. & Aveyard, H. Living with inflammatory bowel disease: a review of qualitative research studies. Int. J. Nurs. Stud 87, 149–156 (2018).
Mittoo, S. et al. Patient perspectives in OMERACT provide an anchor for future metric development and improved approaches to healthcare delivery in connective tissue disease related interstitial lung disease (CTD-ILD). Curr. Respir. Med. Rev. 11, 175–183 (2015).
Cox, C. E., Donohue, J. F., Brown, C. D., Kataria, Y. P. & Judson, M. A. The Sarcoidosis Health Questionnaire: a new measure of health-related quality of life. Am. J. Respir. Crit. Care Med. 8, 8 (2003).
Patel, A. S. et al. The development and validation of the King’s Sarcoidosis Questionnaire for the assessment of health status. Thorax 68, 57–65 (2013).
Judson, M. A. et al. Validation and important differences for the Sarcoidosis Assessment Tool. A new patient-reported outcome measure. Am. J. Respir. Crit. Care Med. 191, 786–795 (2015).
Hendriks, C., Drent, M., Elfferich, M. & De Vries, J. The Fatigue Assessment Scale: quality and availability in sarcoidosis and other diseases. Curr. Opin. Pulm. Med. 24, 495–503 (2018).
Fauchon, C. et al. Does an observer’s empathy influence my pain? Effect of perceived empathetic or unempathetic support on a pain test. Eur. J. Neurosci. 46, 2629–2637 (2017).
Nixon, J. et al. Communicating actively responding empathically (CARE): comparison of communication training workshops for health professionals working in cancer care. J. Cancer Educ. https://doi.org/10.1007/s13187-018-1439-0 (2018).
Buckley, L. et al. 2017 American College of Rheumatology Guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Rheumatol. 69, 1521–1537 (2017).
Singh, J. A. et al. 2015 American College of Rheumatology Guideline for the treatment of rheumatoid arthritis. Arthritis Rheumatol. 68, 1–26 (2016).
Strookappe, B. et al. Physical activity and training in sarcoidosis: review and experience-based recommendations. Expert Rev. Respir. Med. 10, 1057–1068 (2016).
Spagnolo, P. et al. Pulmonary sarcoidosis. Lancet Respir. Med. 6, 389–402 (2018).
Moller, D. R. et al. Rationale and design of the genomic research in alpha-1 antitrypsin deficiency and sarcoidosis (GRADS) study. Sarcoidosis protocol. Ann. Am. Thorac. Soc. 12, 1561–1571 (2015).
Kaiser, Y. et al. Mass cytometry identifies distinct lung CD4+ T cell patterns in Lofgren’s syndrome and non-Lofgren’s syndrome sarcoidosis. Front. Immunol. 8, 1130 (2017).
Moller, D. R. et al. Genetic, immunologic, and environmental basis of sarcoidosis. Ann. Am. Thorac. Soc. 14, S429–S436 (2017).
Crouser, E. D. et al. Application of “omics” and systems biology to sarcoidosis research. Ann. Am. Thorac. Soc. 14, S445–S451 (2017).
Cinetto, F., Compagno, N., Scarpa, R., Malipiero, G. & Agostini, C. Rituximab in refractory sarcoidosis: a single centre experience. Clin. Mol. Allergy 13, 19 (2015).
Sweiss, N. J. et al. Rituximab in the treatment of refractory pulmonary sarcoidosis. Eur. Respir. J. 43, 1525–1528 (2014).
Birring, S. S. et al. The Leicester Cough Monitor: preliminary validation of an automated cough detection system in chronic cough. Eur. Respir. J. 31, 1013–1018 (2008).
Birring, S. S. et al. Treatment of interstitial lung disease associated cough: CHEST Guideline and Expert Panel Report. Chest 154, 904–917 (2018).
Judson, M. A. Quality of life in sarcoidosis. Semin. Respir. Crit. Care Med. 38, 546–558 (2017).
Judson, M. A. The diagnosis of sarcoidosis. Clin. Chest Med. 29, 415–427 (2008).
Judson, M. A. The clinical features of sarcoidosis: a comprehensive review. Clin. Rev. Allergy Immunol. 49, 63–78 (2015).
Acknowledgements
The authors thank A. Eklund and Y. Kaiser for their help with this article as well as research nurses G. de Forest, M. Dahl and H. Blomqvist and biomedical analyst B. Dahlberg (all at Respiratory Medicine Unit, Karolinska University Hospital, Solna, Sweden) for skilful assistance in bronchoscopy and lavage and in sample preparation and processing. The authors thank C. A. Seldenrijk (Department of Pathology), R. G. M. Keijsers (Department of Nuclear Medicine) and H. W. van Es (Department of Radiology), all at St Antonius Hospital, Nieuwegein, Netherlands, for providing the histopathology, PET and CT images, respectively. J.G. is supported by the Swedish Heart Lung Foundation (HLF 20160354 and 20160300), the Swedish Research Council (2016–01209) and the US NIH (R01HL136137) through the regional agreement on medical training and clinical research (ALF) between the Stockholm County Council and the Karolinska Institutet, The King Gustaf V and Queen Victoria’s Freemasons’ Foundation and the Karolinska Institutet.
Author information
Authors and Affiliations
Contributions
Introduction (J.G.); Epidemiology (E.V.A.); Mechanisms/pathophysiology (J.G. and D.R.M.); Diagnosis, screening and prevention (J.C.G.); Management (J.M.-Q.); Quality of life (L.A.S.); Outlook (J.G., J.C.G., E.V.A., L.A.S., D.R.M. and J.M.-Q.); Overview of the Primer (J.G.)
Corresponding author
Ethics declarations
Competing interests
D.R.M. is the Chairman and Chief Technology Officer of Sarcoidosis Diagnostic Testing, LLC. J.M.-Q. is supported by the German Research Foundation (Mu692/12) and Bristol-Myers-Squibb for investigator-initiated trials in sarcoidosis. All other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
MESARGEN: https://mesargen.wordpress.com
Rights and permissions
About this article
Cite this article
Grunewald, J., Grutters, J.C., Arkema, E.V. et al. Sarcoidosis. Nat Rev Dis Primers 5, 45 (2019). https://doi.org/10.1038/s41572-019-0096-x
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-019-0096-x
This article is cited by
-
Choroidal manifestations of non-ocular sarcoidosis: an enhanced depth imaging OCT study
BMC Ophthalmology (2024)
-
Hepatic Sarcoidosis—An Unusual Cause of Jaundice: A Case Report
SN Comprehensive Clinical Medicine (2024)
-
Menopausal hormone therapy and risk of sarcoidosis: a population-based nested case–control study in Sweden
European Journal of Epidemiology (2024)
-
A Case of Unilateral Vision Loss Due to Optic Disc Infiltration by Sarcoidosis
Journal of General Internal Medicine (2024)
-
Chronic constrictive pericarditis: a rare cardiac involvement in primary Sjögren’s syndrome
BMC Cardiovascular Disorders (2023)