Abstract
Most older individuals develop inflammageing, a condition characterized by elevated levels of blood inflammatory markers that carries high susceptibility to chronic morbidity, disability, frailty, and premature death. Potential mechanisms of inflammageing include genetic susceptibility, central obesity, increased gut permeability, changes to microbiota composition, cellular senescence, NLRP3 inflammasome activation, oxidative stress caused by dysfunctional mitochondria, immune cell dysregulation, and chronic infections. Inflammageing is a risk factor for cardiovascular diseases (CVDs), and clinical trials suggest that this association is causal. Inflammageing is also a risk factor for chronic kidney disease, diabetes mellitus, cancer, depression, dementia, and sarcopenia, but whether modulating inflammation beneficially affects the clinical course of non-CVD health problems is controversial. This uncertainty is an important issue to address because older patients with CVD are often affected by multimorbidity and frailty — which affect clinical manifestations, prognosis, and response to treatment — and are associated with inflammation by mechanisms similar to those in CVD. The hypothesis that inflammation affects CVD, multimorbidity, and frailty by inhibiting growth factors, increasing catabolism, and interfering with homeostatic signalling is supported by mechanistic studies but requires confirmation in humans. Whether early modulation of inflammageing prevents or delays the onset of cardiovascular frailty should be tested in clinical trials.
Key points
-
High levels of pro-inflammatory markers in the blood and other tissues are often detected in older individuals and predict the risk of cardiovascular diseases, frailty, multimorbidity, and decline of physical and cognitive function.
-
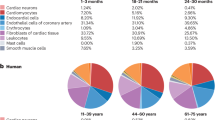
In individuals with obesity, visceral fat produces pro-inflammatory and chemotactic compounds and is infiltrated by macrophages, lymphocytes, and senescent cells with a senescence-associated secretory phenotype that contributes to inflammageing.
-
Mechanisms potentially underlying inflammageing include genomic instability, cell senescence, mitochondria dysfunction, microbiota composition changes, NLRP3 inflammasome activation, primary dysregulation of immune cells, and chronic infections.
-
Clinical trials suggest that modulating inflammation prevents cardiovascular diseases, but studies to explore the effects on other chronic diseases, frailty, and disability are scarce and controversial.
-
Inflammageing can complicate the clinical features of cardiovascular disease in older individuals by causing an energetic imbalance towards catabolism and interfering with homeostatic signalling, leading to frailty.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bektas, A., Schurman, S. H., Sen, R. & Ferrucci, L. Aging, inflammation and the environment. Exp. Gerontol. https://doi.org/10.1016/j.exger.2017.12.015 (2018).
Franceschi, C. et al. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. NY Acad. Sci. 908, 244–254 (2000).
Ferrucci, L. et al. Proinflammatory state, hepcidin, and anemia in older persons. Blood 115, 3810–3816 (2010).
Fulop, T. et al. Immunosenescence and inflamm-aging as two sides of the same coin: friends or foes? Frontiers Immunol. 8, 1960 (2018).
Cohen, H. J., Pieper, C. F., Harris, T., Rao, K. M. & Currie, M. S. The association of plasma IL-6 levels with functional disability in community-dwelling elderly. J. Gerontol. A. Biol. Sci. Med. Sci. 52, M201–M208 (1997).
Newman, A. B. et al. Trajectories of function and biomarkers with age: the CHS All Stars Study. Int. J. Epidemiol. 45, 1135–1145 (2016).
Gerli, R. et al. Chemokines, sTNF-Rs and sCD30 serum levels in healthy aged people and centenarians. Mech. Ageing Dev. 121, 37–46 (2000).
Franceschi, C., Garagnani, P., Vitale, G., Capri, M. & Salvioli, S. Inflammaging and ‘garb-aging’. Trends Endocrinol. Metab 28, 199–212 (2017).
Ruparelia, N., Chai, J. T., Fisher, E. A. & Choudhury, R. P. Inflammatory processes in cardiovascular disease: a route to targeted therapies. Nat. Rev. Cardiol. 14, 133–144 (2017).
Leonardi, G. C., Accardi, G., Monastero, R., Nicoletti, F. & Libra, M. Ageing: from inflammation to cancer. Immun. Ageing 15, 1 (2018).
Salimi, S. et al. Inflammation and trajectory of renal function in community-dwelling older adults. J. Am. Geriatr. Soc. 66, 804–811 (2018).
Gorelick, P. B. Role of inflammation in cognitive impairment: results of observational epidemiological studies and clinical trials. Ann. NY Acad. Sci. 1207, 155–162 (2010).
Miller, A. H. & Raison, C. L. The role of inflammation in depression: From evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 16, 22–34 (2016).
Fabbri, E. et al. Aging and the burden of multimorbidity: associations with inflammatory and anabolic hormonal biomarkers. J. Gerontol. A. Biol. Sci. Med. Sci. 70, 63–70 (2015).
Ferrucci, L. et al. Serum IL-6 level and the development of disability in older persons. J. Am. Geriatr. Soc. 47, 639–646 (1999).
Kuo, H., Bean, J. F., Yen, C. & Leveille, S. G. Linking C-reactive protein to late-life disability in the National Health and Nutrition Examination Survey (NHANES) 1999-2002. J. Gerontol. A. Biol. Sci. Med. Sci. 61, 380–387 (2006).
Schaap, L. A. et al. Higher inflammatory marker levels in older persons: associations with 5-year change in muscle mass and muscle strength. J. Gerontol. A. Biol. Sci. Med. Sci. 64, 1183–1189 (2009).
Soysal, P. et al. Inflammation and frailty in the elderly: a systematic review and meta-analysis. Ageing Res. Rev. 31, 1–8 (2016).
Volpato, S. et al. Cardiovascular disease, interleukin-6, and risk of mortality in older women: the women’s health and aging study. Circulation 103, 947–953 (2001).
Hodes, R. J. et al. Disease drivers of aging. Ann. NY Acad. Sci. 1386, 45–68 (2016).
Smith, A. J. P. & Humphries, S. E. Cytokine and cytokine receptor gene polymorphisms and their functionality. Cytokine Growth Factor Rev. 20, 43–59 (2009).
Rafiq, S. et al. Common genetic variation in the gene encoding interleukin-1-receptor antagonist (IL-1RA) is associated with altered circulating IL-1RA levels. Genes Immun. 8, 344–351 (2007).
Sarwar, N. et al. Interleukin-6 receptor pathways in coronary heart disease: a collaborative meta-analysis of 82 studies. Lancet 379, 1205–1213 (2012).
Swerdlow, D. I. et al. The interleukin-6 receptor as a target for prevention of coronary heart disease: a mendelian randomisation analysis. Lancet 379, 1214–1224 (2012).
Dehghan, A. et al. Meta-analysis of genome-wide association studies in >80 000 subjects identifies multiple loci for C-reactive protein levels. Circulation 123, 731–738 (2011).
Reiner, A. P. et al. Polymorphisms of the IL1-receptor antagonist gene (IL1RN) are associated with multiple markers of systemic inflammation. Arterioscler. Thromb. Vasc. Biol. 28, 1407–1412 (2008).
Carrol, E. D. et al. The IL1RN promoter rs4251961 correlates with IL-1 receptor antagonist concentrations in human infection and is differentially regulated by GATA-1. J. Immunol. 186, 2329–2335 (2011).
Herder, C. et al. Genetic determinants of circulating interleukin-1 receptor antagonist levels and their association with glycemic traits. Diabetes 63, 4343–4359 (2014).
Wu, X. et al. IL-1 receptor antagonist gene as a predictive biomarker of progression of knee osteoarthritis in a population cohort. Osteoarthr. Cartil. 21, 930–938 (2013).
Dai, L., Liu, D., Guo, H., Wang, Y. & Bai, Y. Association between polymorphism in the promoter region of interleukin 6 (-174 G/C) and risk of Alzheimer’s disease: a meta-analysis. Neurol. J. 259, 414–419 (2012).
Hou, H. et al. Association of interleukin-6 gene polymorphism with coronary artery disease: an updated systematic review and cumulative meta-analysis. Inflamm. Res. 64, 707–720 (2015).
Testa, R. et al. Interleukin-6-174 G > C polymorphism affects the association between IL-6 plasma levels and insulin resistance in type 2 diabetic patients. Diabetes Res. Clin. Pract. 71, 299–305 (2006).
Moffett, S. P. et al. Association of the G-174C variant in the interleukin-6 promoter region with bone loss and fracture risk in older women. J. Bone Miner. Res. 19, 1612–1618 (2004).
Fishman, D. et al. The effect of novel polymorphisms in the interleukin-6 (IL-6) gene on IL-6 transcription and plasma IL-6 levels, and an association with systemic-onset juvenile chronic arthritis. J. Clin. Invest. 102, 1369–1376 (1998).
Zeng, Y. et al. Novel loci and pathways significantly associated with longevity. Sci. Rep. 6, 21243 (2016).
Khandaker, G. M., Zammit, S., Burgess, S., Lewis, G. & Jones, P. B. Association between a functional interleukin 6 receptor genetic variant and risk of depression and psychosis in a population-based birth cohort. Brain. Behav. Immun. 69, 264–272 (2017).
Ferreira, R. C. et al. Functional IL6R 358Ala allele impairs classical IL-6 receptor signaling and influences risk of diverse inflammatory diseases. PLoS Genet. 9, e1003444 (2013).
Lange, L. A. et al. Association of polymorphisms in the CRP gene with circulating C-reactive protein levels and cardiovascular events. JAMA 296, 2703 (2006).
Peters, M. J. et al. The transcriptional landscape of age in human peripheral blood. Nat. Commun. 6, 8570 (2015).
Pilling, L. C. et al. Gene expression markers of age-related inflammation in two human cohorts. Exp. Gerontol. 70, 37–45 (2015).
Fabian, M. R., Sonenberg, N. & Filipowicz, W. Regulation of mRNA translation and stability by microRNAs. Annu. Rev. Biochem. 79, 351–379 (2010).
Noren Hooten, N. et al. microRNA expression patterns reveal differential expression of target genes with age. PLoS ONE 5, e10724 (2010).
Lai, C.-Y. et al. Modulated expression of human peripheral blood microRNAs from infancy to adulthood and its role in aging. Aging Cell 13, 679–689 (2014).
Olivieri, F. et al. Age-related differences in the expression of circulating microRNAs: miR-21 as a new circulating marker of inflammaging. Mech. Ageing Dev. 133, 675–685 (2012).
Freedman, J. E. et al. Diverse human extracellular RNAs are widely detected in human plasma. Nat. Commun. 7, 11106 (2016).
Noren Hooten, N. et al. Age-related changes in microRNA levels in serum. Aging 5, 725–740 (2013).
Ameling, S. et al. Associations of circulating plasma microRNAs with age, body mass index and sex in a population-based study. BMC Med. Genomics 8, 61 (2015).
Zhang, H. et al. Investigation of microRNA expression in human serum during the aging process. J. Gerontol. A. Biol. Sci. Med. Sci. 70, 102–109 (2015).
Dluzen, D. F., Noren Hooten, N. & Evans, M. K. Extracellular. RNA in aging. WIREs RNA. https://doi.org/10.1002/wrna.1385 (2017).
Olivieri, F. et al. Age- and glycemia-related miR-126-3p levels in plasma and endothelial cells. Aging (Albany. NY). 6, 771–787 (2014).
Olivieri, F. et al. DNA damage response (DDR) and senescence: shuttled inflamma-miRNAs on the stage of inflamm-aging. Oncotarget 6, 35509–35521 (2015).
Frasca, D., Blomberg, B. B. & Paganelli, R. Aging, obesity, and inflammatory age-related diseases. Front. Immunol. 8, 1–10 (2017).
Rocha, V. Z. & Libby, P. Obesity, inflammation, and atherosclerosis. Nat. Rev. Cardiol. 6, 399–409 (2009).
Vandanmagsar, B. et al. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 17, 179–189 (2011).
Frasca, D. & Blomberg, B. B. Adipose tissue inflammation induces B cell inflammation and decreases B cell function in aging. Front. Immunol. 8, 1003 (2017).
Lee-Chang, C. et al. Accumulation of 4-1BBL+ B cells in the elderly induces the generation of granzyme-B+ CD8+ T cells with potential antitumor activity. Blood 124, 1450–1459 (2014).
Weisberg, S. P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Panagiotakos, D. B., Pitsavos, C., Yannakoulia, M., Chrysohoou, C. & Stefanadis, C. The implication of obesity and central fat on markers of chronic inflammation: The ATTICA study. Atherosclerosis 183, 308–315 (2005).
Clément, K. et al. Weight loss regulates inflammation-related genes in white adipose tissue of obese subjects. FASEB J. 18, 1657–1669 (2004).
Nicklas, B. J., You, T. & Pahor, M. Behavioural treatments for chronic systemic inflammation: effects of dietary weight loss and exercise training. Can. Med. Assoc. J. 172, 1199–1209 (2005).
Illán-Gómez, F. et al. Obesity and inflammation: change in adiponectin, C-reactive protein, tumour necrosis factor-alpha and interleukin-6 after bariatric surgery. Obes. Surg. 22, 950–955 (2012).
Meydani, S. N. et al. Long-term moderate calorie restriction inhibits inflammation without impairing cell-mediated immunity: a randomized controlled trial in non-obese humans. Aging 8, 1416–1431 (2016).
Zomer, E. et al. Interventions that cause weight loss and the impact on cardiovascular risk factors: a systematic review and meta-analysis. Obes. Rev. 17, 1001–1011 (2016).
Ma, C. et al. Effects of weight loss interventions for adults who are obese on mortality, cardiovascular disease, and cancer: systematic review and meta-analysis. BMJ 359, j4849 (2017).
Villareal, D. T. et al. Aerobic or resistance exercise, or both, in dieting obese older adults. N. Engl. J. Med. 376, 1943–1955 (2017).
Mariat, D. et al. The Firmicutes/Bacteroidetes ratio of the human microbiota changes with age. BMC Microbiol. 9, 123 (2009).
Biagi, E. et al. Gut microbiota and extreme longevity. Curr. Biol. 26, 1480–1485 (2016).
Shapiro, H., Thaiss, C. A., Levy, M. & Elinav, E. The cross talk between microbiota and the immune system: metabolites take center stage. Curr. Opin. Immunol. 30, 54–62 (2014).
Rampelli, S. et al. Functional metagenomic profiling of intestinal microbiome in extreme ageing. Aging 5, 902–912 (2013).
Biagi, E. et al. Through ageing, and beyond: gut microbiota and inflammatory status in seniors and centenarians. PLoS ONE 5, e10667 (2010).
Zapata, H. J. & Quagliarello, V. J. The microbiota and microbiome in aging: potential implications in health and age-related diseases. Am. J. Geriatr. Soc 63, 776–781 (2015).
Thevaranjan, N. et al. Age-associated microbial dysbiosis promotes intestinal permeability, systemic inflammation, and macrophage dysfunction. Cell Host Microbe 21, 455–466.e4 (2017).
Picca, A. et al. Gut dysbiosis and muscle aging: searching for novel targets against sarcopenia. Mediators Inflamm. 2018, 7026198 (2018).
van Tongeren, S. P., Slaets, J. P. J., Harmsen, H. J. M. & Welling, G. W. Fecal microbiota composition and frailty. Appl. Environ. Microbiol. 71, 6438–6442 (2005).
O’Toole, P. W. & Jeffery, I. B. Gut microbiota and aging. Science 350, 1214–1215 (2015).
Mello, A. M., Paroni, G., Daragjati, J. & Pilotto, A. Gastrointestinal microbiota and their contribution to healthy aging. Dig. Dis. 34, 194–201 (2016).
Barrios, C. et al. Gut-microbiota-metabolite axis in early renal function decline. PLoS ONE 10, e0134311 (2015).
Everard, A. et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl Acad. Sci. USA 110, 9066–9071 (2013).
Turroni, F. et al. Molecular dialogue between the human gut microbiota and the host: a Lactobacillus and Bifidobacterium perspective. Cell. Mol. Life Sci. 71, 183–203 (2014).
Ott, B. et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci. Rep. 7, 11955 (2017).
Westfall, S. et al. Microbiome, probiotics and neurodegenerative diseases: deciphering the gut brain axis. Cell. Mol. Life Sci. 74, 3769–3787 (2017).
Kim, Y. A., Keogh, J. B. & Clifton, P. M. Probiotics, prebiotics, synbiotics and insulin sensitivity. Nutr. Res. Rev. 31, 35–51 (2018).
Liu, Y., Gibson, G. R. & Walton, G. E. An in vitro approach to study effects of prebiotics and probiotics on the faecal microbiota and selected immune parameters relevant to the elderly. PLoS ONE 11, e0162604 (2016).
Nagpal, R. et al. Gut microbiota in health and disease: an overview focused on metabolic inflammation. Benef. Microbes 7, 181–194 (2016).
Turchet, P., Laurenzano, M., Auboiron, S. & Antoine, J. M. Effect of fermented milk containing the probiotic Lactobacillus casei DN-114001 on winter infections in free-living elderly subjects: a randomised, controlled pilot study. J. Nutr. Health Aging 7, 75–77 (2003).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Kennedy, B. K. et al. Geroscience: linking aging to chronic disease. Cell 159, 709–713 (2014).
Campisi, J. & D’Adda Di Fagagna, F. Cellular senescence: when bad things happen to good cells. Nat. Rev. Mol. Cell. Biol. 8, 729–740 (2007).
Sharpless, N. E. & Sherr, C. J. Forging a signature of in vivo senescence. Nat. Rev. Cancer 15, 397–408 (2015).
Bernardes de Jesus, B. & Blasco, M. A. Assessing cell and organ senescence biomarkers. Circ. Res. 111, 97–109 (2012).
Jeck, W. R., Siebold, A. P. & Sharpless, N. E. Review: a meta-analysis of GWAS and age-associated diseases. Aging Cell 11, 727–731 (2012).
Johnson, S. C., Dong, X., Vijg, J. & Suh, Y. Genetic evidence for common pathways in human age-related diseases. Aging Cell 14, 809–817 (2015).
Melzer, D. et al. A common variant of the p16INK4a genetic region is associated with physical function in older people. Mech. Ageing Dev. 128, 370–377 (2007).
van Deursen, J. M. The role of senescent cells in ageing. Nature 509, 439–446 (2014).
Hernandez-Segura, A. et al. Unmasking transcriptional heterogeneity in senescent cells. Curr. Biol. 27, 2652–2660.e4 (2017).
Borodkina, A. V., Deryabin, P. I., Giukova, A. A. & Nikolsky, N. N. ‘Social life’ of senescent sells: what is SASP and why study it? Acta Naturae 10, 4–14 (2018).
Herbig, U., Ferreira, M., Condel, L., Carey, D. & Sedivy, J. M. Cellular senescence in aging primates. Science 311, 1257 (2006).
Waaijer, M. E. C. et al. The number of p16INK4a positive cells in human skin reflects biological age. Aging Cell 11, 722–725 (2012).
Liu, Y. et al. Expression of p16INK4a in peripheral blood T-cells is a biomarker of human aging. Aging Cell 8, 439–448 (2009).
Koppelstaetter, C. et al. Markers of cellular senescence in zero hour biopsies predict outcome in renal transplantation. Aging Cell 7, 491–497 (2008).
Helman, A. et al. p16Ink4a-induced senescence of pancreatic beta cells enhances insulin secretion. Nat. Med. 22, 412–420 (2016).
Rossman, M. J. et al. Endothelial cell senescence with aging in healthy humans: prevention by habitual exercise and relation to vascular endothelial function. Am. J. Physiol. Heart Circ. Physiol. 313, H890–H895 (2017).
Diekman, B. O. et al. Expression of p16INK4a is a biomarker of chondrocyte aging but does not cause osteoarthritis. Aging Cell. https://doi.org/10.1111/acel.12771 (2018).
Chimenti, C. et al. Senescence and death of primitive cells and myocytes lead to premature cardiac aging and heart failure. Circ. Res. 93, 604–613 (2003).
Kajstura, J. et al. Myocyte turnover in the aging human heart. Circ. Res. 107, 1374–1386 (2010).
Gregor, M. F. & Hotamisligil, G. S. Inflammatory mechanisms in obesity. Annu. Rev. Immunol. 29, 415–445 (2011).
Ogrodnik, M. et al. Cellular senescence drives age-dependent hepatic steatosis. Nat. Commun. 8, 15691 (2017).
Klenerman, P. & Oxenius, A. T cell responses to cytomegalovirus. Nat. Rev. Immunol. 16, 367–377 (2016).
Sansoni, P. et al. New advances in CMV and immunosenescence. Exp. Gerontol. 55, 54–62 (2014).
Fulop, T., Larbi, A. & Pawelec, G. Human T cell aging and the impact of persistent viral infections. Front. Immunol. 4, 271 (2013).
Baker, D. J. et al. Clearance of p16 Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479, 232–236 (2011).
Baker, D. J. et al. Naturally occurring p16 Ink4a-positive cells shorten healthy lifespan. Nature 530, 184–189 (2016).
Medina, C. B. & Ravichandran, K. S. Do not let death do us part: ‘find-me’ signals in communication between dying cells and the phagocytes. Cell Death Differ. 23, 979–989 (2016).
Youm, Y. H. et al. Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab. 18, 519–532 (2013).
Goldberg, E. L. & Dixit, V. D. Drivers of age-related inflammation and strategies for healthspan extension. Immunol. Rev. 265, 63–74 (2015).
Medzhitov, R. & Janeway, C. A. J. Decoding the patterns of self and nonself by the innate immune system. Science 296, 298–300 (2002).
Kepp, O., Galluzzi, L. & Kroemer, G. Mitochondrial control of the NLRP3 inflammasome. Nat. Immunol. 12, 199–200 (2011).
Ferrucci, L. et al. The origins of age-related proinflammatory state. Blood 105, 2294–2299 (2005).
Sies, H., Berndt, C. & Jones, D. P. Oxidative stress. Annu. Rev. Biochem. 86, 715–748 (2017).
Bektas, A. et al. Age-associated alterations in inducible gene transcription in human CD4+ T lymphocytes. Aging 5, 18–36 (2013).
Bektas, A. et al. Age-associated changes in basal NF-κB function in human CD4+ T lymphocytes via dysregulation of PI3 kinase. Aging 6, 957–974 (2014).
Cannon, M. J., Schmid, D. S. & Hyde, T. B. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev. Med. Virol. 20, 202–213 (2010).
Vescovini, R. et al. Massive load of functional effector CD4+ and CD8+ T cells against cytomegalovirus in very old subjects. J. Immunol. 179, 4283–4291 (2007).
Simon, C. O. et al. CD8 T cells control cytomegalovirus latency by epitope-specific sensing of transcriptional reactivation. J. Virol. 80, 10436–10456 (2006).
Roberts, E. T., Haan, M. N., Dowd, J. B. & Aiello, A. E. Cytomegalovirus antibody levels, inflammation, and mortality among elderly Latinos over 9 years of follow-up. Am. J. Epidemiol. 172, 363–371 (2010).
Spyridopoulos, I. et al. CMV seropositivity and T-cell senescence predict increased cardiovascular mortality in octogenarians: results from the Newcastle 85+ study. Aging Cell 15, 389–392 (2016).
Adriaensen, W. et al. CD4:8 ratio above 5 is associated with all-cause mortality in CMV-seronegative very old women: results from the BELFRAIL study. J. Gerontol. A. Biol. Sci. Med. Sci. 72, 1155–1162 (2017).
Brodin, P. et al. Variation in the human immune system is largely driven by non-heritable influences. Cell 160, 37–47 (2015).
Aiello, A. E., Chiu, Y.-L. & Frasca, D. How does cytomegalovirus factor into diseases of aging and vaccine responses, and by what mechanisms? GeroScience 39, 261–271 (2017).
Goldeck, D. et al. No strong correlations between serum cytokine levels, CMV serostatus and hand-grip strength in older subjects in the Berlin BASE-II cohort. Biogerontology 17, 189–198 (2016).
Nasi, M. et al. Ageing and inflammation in patients with HIV infection. Clin. Exp. Immunol. 187, 44–52 (2017).
Neuhaus, J. et al. Markers of inflammation, coagulation, and renal function are elevated in adults with HIV infection. J. Infect. Dis. 201, 1788–1795 (2010).
Losina, E. et al. Projecting 10-year, 20-year, and lifetime risks of cardiovascular disease in persons living with human immunodeficiency virus in the United States. Clin. Infect. Dis. 65, 1266–1271 (2017).
Brothers, T. D. et al. Frailty in people aging with human immunodeficiency virus (HIV) infection. J. Infect. Dis. 210, 1170–1179 (2014).
Stein, J. H. & Hsue, P. Y. Inflammation, immune activation, and CVD risk in individuals with HIV infection. JAMA 308, 405–406 (2012).
Brenchley, J. M. et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat. Med. 12, 1365–1371 (2006).
Grunfeld, C. et al. Association of upper trunk and visceral adipose tissue volume with insulin resistance in control and HIV-infected subjects in the FRAM study. J. Acquir. Immune Def. Syndr. 46, 283–290 (2007).
Odegaard, A. O. et al. Oxidative stress, inflammation, endothelial dysfunction and incidence of type 2 diabetes. Cardiovasc. Diabetol. 15, 51 (2016).
Lai, K. S. P. et al. Peripheral inflammatory markers in Alzheimer’s disease: a systematic review and meta-analysis of 175 studies. J. Neurol. Neurosurg. Psychiatry 88, 876–882 (2017).
Iseme, R. A. et al. Is osteoporosis an autoimmune mediated disorder? Bone Rep. 7, 121–131 (2017).
Dalle, S., Rossmeislova, L. & Koppo, K. The role of inflammation in age-related sarcopenia. Front. Physiol. 8, 1045 (2017).
Amdur, R. L. et al. Inflammation and progression of CKD: the CRIC study. Clin. J. Am. Soc. Nephrol. 11, 1546–1556 (2016).
Kohler, O., Krogh, J., Mors, O. & Eriksen Benros, M. Inflammation in depression and the potential for anti-inflammatory treatment. Curr. Neuropharmacol. 14, 732–742 (2016).
Libby, P., Ridker, P. M. & Hansson, G. K. Progress and challenges in translating the biology of atherosclerosis. Nature 473, 317–325 (2011).
Andreou, D. E. & Andreadou, I. Atherosclerosis: an inflammatory disease. Pharmakeftiki 22, 83–96 (2009).
Hansson, G. K. Inflammation, atherosclerosis and coronary artery disease. N. Engl. J. Med. 352, 1685–1695 (2005).
Libby, P., Ridker, P. M. & Hansson, G. K. Inflammation in atherosclerosis. from pathophysiology to practice. Am. J. Coll. Cardiol. 54, 2129–2138 (2009).
De Caterina, R., D’Ugo, E. & Libby, P. Inflammation and thrombosis – Testing the hypothesis with anti-inflammatory drug trials. Thromb. Haemost. 116, 1012–1021 (2016).
Libby, P., Okamoto, Y., Rocha, V. Z. & Folco, E. Inflammation in atherosclerosis. Circ. J. 74, 213–220 (2010).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010).
Warnatsch, A., Ioannou, M., Wang, Q. & Papayannopoulos, V. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science 349, 316–320 (2015).
Wang, M., Kim, S. H., Monticone, R. E. & Lakatta, E. G. Matrix metalloproteinases promote arterial remodeling in aging, hypertension, and atherosclerosis. Hypertension 65, 698–703 (2015).
Matthews, C. et al. Vascular smooth muscle cells undergo telomere-based senescence in human atherosclerosis: effects of telomerase and oxidative stress. Circ. Res. 99, 156–164 (2006).
Grootaert, M. O. J. et al. Vascular smooth muscle cell death, autophagy and senescence in atherosclerosis. Cardiovasc. Res. 114, 622–634 (2018).
Ketelhuth, D. F. J. & Hansson, G. K. Adaptive response of T and B cells in atherosclerosis. Circ. Res. 118, 668–678 (2016).
Feinberg, M. W. & Moore, K. J. MicroRNA regulation of atherosclerosis. Circ. Res. 118, 703–720 (2016).
Ridker, P. M., Cushman, M., Stampfer, M. J., Tracy, R. P. & Hennekens, C. H. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N. Engl. J. Med. 336, 973–979 (1997).
Ridker, P. M., Hennekens, C. H., Buring, J. E. & Rifai, N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N. Engl. J. Med. 342, 836–843 (2000).
Cushman, M. et al. C-reactive protein and the 10-year incidence of coronary heart disease in older men and women: the cardiovascular health study. Circulation 112, 25–31 (2005).
Cesari, M. et al. Inflammatory markers and onset of cardiovascular events: results from the Health ABC study. Circulation 108, 2317–2322 (2003).
Levinson, S. S. Rosuvastatin to prevent vascular events in men and women with elevated C.-reactive protein – an analysis. Clin. J. Ligand Assay 31, 25–28 (2008).
Noren Hooten, N., Ejiogu, N., Zonderman, A. B. & Evans, M. K. Association of oxidative DNA damage and C-reactive protein in women at risk for cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 32, 2776–2784 (2012).
Ridker, P. M. From C-reactive protein to interleukin-6 to interleukin-1: moving upstream to identify novel targets for atheroprotection. Circ. Res. 118, 145–156 (2016).
Elliott, P. et al. Genetic loci associated with C-reactive protein levels and risk of coronary heart disease. JAMA - J. Am. Med. Assoc. 302, 37–48 (2009).
McInnes, I. B. et al. Effect of interleukin-6 receptor blockade on surrogates of vascular risk in rheumatoid arthritis: MEASURE, a randomised, placebo-controlled study. Ann. Rheum. Dis. 74, 694–702 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/show/NCT01331837 (2011).
Libby, P. Interleukin-1 β as a target for atherosclerosis therapy: biological basis of CANTOS and beyond. J. Am. Coll. Cardiol. 70, 2278–2289 (2017).
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Ridker, P. M. et al. Effects of interleukin-1β inhibition with canakinumab on hemoglobin A1c, lipids, C-reactive protein, interleukin-6, and fibrinogen. Circulation 126, 2739–2748 (2012).
Everett, B. M. et al. Rationale and design of the cardiovascular inflammation reduction trial: a test of the inflammatory hypothesis of atherothrombosis. Am. Heart J. 166, 199–207.e15 (2013).
Daniels, L. B. Pretenders and contenders: inflammation, C-reactive protein and interleukin-6. Am. J. Heart Assoc. 6, e007490 (2017).
Nidorf, S. M., Mbbs, J. W. E., Hons, C. A. B. & Thompson, P. L. Low-dose colchicine for secondary prevention of cardiovascular disease. Am. J. Coll. Cardiol. 61, 404–410 (2013).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/show/NCT02551094 (2015).
Cigolle, C. T., Blaum, C. S. & Halter, J. B. Diabetes and cardiovascular disease prevention in older adults. Clin. Geriatr. Med. 25, 607–641 (2009).
Stout, M. B., Justice, J. N., Nicklas, B. J. & Kirkland, J. L. Physiological aging: Links among adipose tissue dysfunction, diabetes, and frailty. Physiology 32, 9–19 (2017).
Halter, J. B. et al. Diabetes and cardiovascular disease in older adults: current status and future directions. Diabetes 63, 2578–2589 (2014).
Nahrendorf, M. & Swirski, F. K. Immunology. Neutrophil-macrophage communication in inflammation and atherosclerosis. Science 349, 237–238 (2015).
Gimbrone, M. A. J. & Garcia-Cardena, G. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ. Res. 118, 620–636 (2016).
Shakeri, H., Lemmens, K., Gevaert, A. B., De Meyer, G. R. Y. & Segers, V. Cellular senescence links aging and diabetes in cardiovascular disease. Am. J. Physiol. Heart Circ. Physiol. https://doi.org/10.1152/ajpheart.00287.2018 (2018).
Aryan, Z. et al. Baseline high-sensitivity C-reactive protein predicts macrovascular and microvascular complications of type 2 diabetes: a population-pased study. Ann. Nutr. Metab. 72, 287–295 (2018).
Eguchi, S., Kawai, T., Scalia, R. & Rizzo, V. Understanding Angiotensin II type 1 receptor signaling in vascular pathophysiology. Hypertension 71, 804–810 (2018).
Jacobsson, L. T. et al. Treatment with tumor necrosis factor blockers is associated with a lower incidence of first cardiovascular events in patients with rheumatoid arthritis. J. Rheumatol. 32, 1213–1218 (2005).
Greenberg, J. D. et al. Tumour necrosis factor antagonist use and associated risk reduction of cardiovascular events among patients with rheumatoid arthritis. Ann. Rheum. Dis. 70, 576–582 (2011).
Solomon, D. H. et al. Cardiovascular risk in rheumatoid arthritis: comparing TNF-α blockade with nonbiologic DMARDs. Am. J. Med. 126, 730.e9–730.e17 (2013).
Bili, A. et al. Tumor necrosis factor α inhibitor use and decreased risk for incident coronary events in rheumatoid arthritis. Arthritis Care Res. 66, 355–363 (2014).
Roubille, C. et al. The effects of tumour necrosis factor inhibitors, methotrexate, non-steroidal anti-inflammatory drugs and corticosteroids on cardiovascular events in rheumatoid arthritis, psoriasis and psoriatic arthritis: a systematic review and meta-analysis. Ann. Rheum. Dis. 74, 480–489 (2015).
Low, A. S. L. et al. Relationship between exposure to tumour necrosis factor inhibitor therapy and incidence and severity of myocardial infarction in patients with rheumatoid arthritis. Ann. Rheum. Dis. 76, 654–660 (2017).
Shaaban, D. & Al-Mutairi, N. The effect of tumor necrosis factor inhibitor therapy on the incidence of myocardial infarction in patients with psoriasis: a retrospective study. J. Dermatolog. Treat. 29, 3–7 (2018).
Yang, Z., Lin, N., Li, L. & Li, Y. The effect of TNF inhibitors on cardiovascular events in psoriasis and psoriatic arthritis: an updated meta-analysis. Clin. Rev. Allergy Immunol. 51, 240–247 (2016).
Anker, S. D. Inflammatory mediators in chronic heart failure: an overview. Heart 90, 464–470 (2004).
Levine, B., Kalman, J., Mayer, L., Fillit, H. M. & Packer, M. Elevated circulating levels of tumor necrosis factor in severe chronic heart failure. N. Engl. J. Med. 323, 236–241 (1990).
Torre-Amione, G. et al. Proinflammatory cytokine levels in patients with depressed left ventricular ejection fraction: A report from the studies of left ventricular dysfunction (SOLVD). J. Am. Coll. Cardiol. 27, 1201–1206 (1996).
Mann, D. L. et al. Targeted anticytokine therapy in patients with chronic heart failure: results of the randomized etanercept worldwide evaluation (RENEWAL). Circulation 109, 1594–1602 (2004).
Chung, E. S. Randomized, double-blind, placebo-controlled, pilot trial of infliximab, a chimeric monoclonal antibody to tumor necrosis factor-α, in patients with moderate-to-severe heart failure: results of the anti-TNF therapy against congestive heart failure. Circulation 107, 3133–3140 (2003).
Forman, D. E. et al. Multimorbidity in older adults with cardiovascular disease. J. Am. Coll. Cardiol. 71, 2149–2161 (2018).
Brandenberger, C. & Muhlfeld, C. Mechanisms of lung aging. Cell Tissue Res. 367, 469–480 (2017).
Tisminetzky, M., Goldberg, R. & Gurwitz, J. H. Magnitude and impact of multimorbidity on clinical outcomes in older adults with cardiovascular aisease: a literature review. Clin. Geriatr. Med. 32, 227–246 (2016).
Fried, L. P. et al. Frailty in older adults: evidence for a phenotype. J. Gerontol. A. Biol. Sci. Med. Sci. 56, M146–M156 (2001).
Bergman, H. et al. Frailty: An emerging research and clinical paradigm — issues and controversies. J. Gerontol. A. Biol. Sci. Med. Sci. 62, 731–737 (2007).
Afilalo, J. et al. Frailty assessment in the cardiovascular care of older adults. J. Am. Coll. Cardiol. 63, 747–762 (2014).
Woods, N. F. et al. Frailty: emergence and consequences in women aged 65 and older in the Women’s Health Initiative Observational Study. J. Am. Geriatr. Soc. 53, 1321–1330 (2005).
Corti, M. C., Salive, M. E. & Guralnik, J. M. Serum albumin and physical function as predictors of coronary heart disease mortality and incidence in older persons. Clin. J. Epidemiol. 49, 519–526 (1996).
McDermott, M. M. et al. Decline in functional performance predicts later increased mobility loss and mortality in peripheral arterial disease. J. Am. Coll. Cardiol. 57, 962–970 (2011).
Walker, K. A. et al. Midlife systemic inflammation is associated with frailty in later life: the ARIC Study. J. Gerontol. A Biol. Sci. Med. Sci. https://doi.org/10.1093/gerona/gly045 (2018).
Abbatecola, A. M. & Paolisso, G. Is there a relationship between insulin resistance and frailty syndrome? Curr. Pharm. Des. 14, 405–410 (2008).
Barzilay, J. I. et al. Insulin resistance and inflammation as precursors of frailty: the Cardiovascular Health Study. Arch. Intern. Med. 167, 635–641 (2007).
Walston, J. et al. Frailty and activation of the inflammation and coagulation systems with and without clinical comorbidities: results from the Cardiovascular Health Study. Arch. Intern. Med. 162, 2333–2341 (2002).
Afilalo, J., Karunananthan, S., Eisenberg, M. J., Alexander, K. P. & Bergman, H. Role of frailty in patients with cardiovascular disease. Am. J. Cardiol. 103, 1616–1621 (2009).
Gupta, J. et al. Association between albuminuria, kidney function, and inflammatory biomarker profile in CKD in CRIC. Clin. J. Am. Soc. Nephrol. 7, 1938–1946 (2012).
Mc Causland, F. R. et al. C-reactive protein and risk of ESRD: results from the trial to reduce cardiovascular events with aranesp therapy (TREAT). Am. J. Kidney Dis. 68, 873–881 (2016).
Grivennikov, S. I., Greten, F. R. & Karin, M. Immunity. inflammation, and cancer. Cell 140, 883–899 (2010).
Todoric, J., Antonucci, L. & Karin, M. Targeting inflammation in cancer prevention and therapy. Cancer Prev. Res. 9, 895–905 (2016).
Howren, M. B., Lamkin, D. M. & Suls, J. Associations of depression with C-reactive protein, IL-1, and IL-6: a meta-analysis. Psychosom. Med. 71, 171–186 (2009).
Miller, A. H., Maletic, V. & Raison, C. L. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 65, 732–741 (2009).
Matthews, K. A. et al. Are there bi-directional associations between depressive symptoms and C-reactive protein in mid-life women? Brain. Behav. Immun. 24, 96–101 (2010).
Zalli, A., Jovanova, O., Hoogendijk, W. J. G., Tiemeier, H. & Carvalho, L. A. Low-grade inflammation predicts persistence of depressive symptoms. Psychopharmacology 233, 1669–1678 (2016).
Lamers, F., Milaneschi, Y., de Jonge, P., Giltay, E. J. & Penninx, B. W. J. H. Metabolic and inflammatory markers: associations with individual depressive symptoms. Psychol. Med. 48, 1102–1110 (2018).
Heneka, M. T. et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 14, 388–405 (2015).
Eikelenboom, P. et al. Innate immunity and the etiology of late-onset Alzheimer’s disease. Neurodegener. Dis. 10, 271–273 (2012).
Hansen, P. R. Chronic inflammatory diseases and atherosclerotic cardiovascular disease: Innocent bystanders or partners in crime? Curr. Pharm. Des. 24, 281–290 (2018).
Goldfine, A. B. & Shoelson, S. E. Therapeutic approaches targeting inflammation for diabetes and associated cardiovascular risk. J. Clin. Invest. 127, 83–93 (2017).
Zitvogel, L., Pietrocola, F. & Kroemer, G. Nutrition, inflammation and cancer. Nat. Immunol. 18, 843–850 (2017).
Morgan, A. R. et al. The correlation between inflammatory biomarkers and polygenic risk score in Alzheimer’s Disease. J. Alzheimers. Dis. 56, 25–36 (2017).
Schlegel, T. F., Hawkins, R. J., Lewis, C. W., Motta, T. & Turner, A. S. The effects of augmentation with swine small intestine submucosa on tendon healing under tension: histologic and mechanical evaluations in sheep. Am. J. Sports Med. 34, 275–280 (2006).
Ferrucci, L. et al. Change in muscle strength explains accelerated decline of physical function in older women with high interleukin-6 serum levels. J. Am. Geriatr. Soc. 50, 1947–1954 (2002).
Visser, M. et al. Relationship of interleukin-6 and tumor necrosis factor-α with muscle nass and muscle strength in elderly men and women: the Health ABC Study. J. Gerontol. A Biol. Sci. Med. Sci. 57, M326–M332 (2002).
Cesari, M. et al. Inflammatory markers and physical performance in older persons: the InCHIANTI Study. J. Gerontol. A Biol. Sci. Med. Sci. 59, M242–M248 (2004).
Santos-Eggimann, B., Cuénoud, P., Spagnoli, J. & Junod, J. Prevalence of frailty in middle-aged and older community-dwelling Europeans living in 10 countries. J. Gerontol. A Biol. Sci. Med. Sci. 64, 675–681 (2009).
Walston, J. et al. Research agenda for frailty in older adults: toward a better understanding of physiology and etiology: summary from the American Geriatrics Society/National Institute on Aging research conference on frailty in older adults. J. Am. Geriatr. Soc. 54, 991–1001 (2006).
Stepanova, M., Rodriguez, E., Birerdinc, A. & Baranova, A. Age-independent rise of inflammatory scores may contribute to accelerated aging in multi-morbidity. Oncotarget 6, 1414–1421 (2015).
Friedman, E. M., Montez, J. K., Sheehan, C. M. D., Guenewald, T. L. & Seeman, T. E. Childhood adversities and adult cardiometabolic health: does the quantity, timing, and type of adversity matter? J. Aging Health 27, 1311–1338 (2015).
Hubbard, R. E., O’Mahony, M. S., Savva, G. M., Calver, B. L. & Woodhouse, K. W. Inflammation and frailty measures in older people. J. Cell Mol. Med. 13, 3103–3109 (2009).
Newman, A. B. et al. Weight change in old age and its association with mortality. J. Am. Geriatr. Soc. 49, 1309–1318 (2001).
Higashi, Y. et al. Insulin-like growth factor-1 receptor deficiency in macrophages accelerates atherosclerosis and induces an unstable plaque phenotype in apolipoprotein E-deficient mice. Circulation 133, 2263–2278 (2016).
Lazarus, D. D., Moldawer, L. L. & Lowry, S. F. Insulin-like growth factor-1 activity is inhibited by interleukin-1α, tumor necrosis factor-α, and interleukin-6. Lymphokine Cytokine Res. 12, 219–223 (1993).
Barbieri, M. et al. Chronic inflammation and the effect of IGF-I on muscle strength and power in older persons. Am. J. Physiol. Metab. 284, E481–E487 (2003).
Cappola, A. R. et al. Insulin-like growth factor I and interleukin-6 contribute synergistically to disability and mortality in older women. J. Clin. Endocrinol. Metab. 88, 2019–2025 (2003).
Timmerman, K. L. et al. Pharmacological vasodilation improves insulin-stimulated muscle protein anabolism but not glucose utilization in older adults. Diabetes 59, 2764–2771 (2010).
Moaddel, R. et al. Plasma biomarkers of poor muscle quality in older men and women from the Baltimore Longitudinal Study of Aging. J. Gerontol. A Biol. Sci. Med. Sci. 71, 1266–1272 (2016).
Fichtlscherer, S. et al. Elevated C-reactive protein levels and impaired endothelial vasoreactivity in patients with coronary artery disease. Circulation 102, 1000–1006 (2000).
Bar-Shai, M., Carmeli, E. & Reznick, A. Z. The role of NF-κB in protein breakdown in immobilization, aging, and exercise: From basic processes to promotion of health. Ann. NY Acad. Sci. 1057, 431–447 (2005).
Justice, J. N. et al. Cellular senescence biomarker p16INK4a+ cell burden in thigh adipose is associated with poor physical function in older women. J. Gerontol. A Biol. Sci. Med. Sci. 73, 939–945 (2017).
Roth, S. M., Metter, E. J., Ling, S. & Ferrucci, L. Inflammatory factors in age-related muscle wasting. Curr. Opin. Rheumatol. 18, 625–630 (2006).
Jo, E., Lee, S.-R., Park, B.-S. & Kim, J.-S. Potential mechanisms underlying the role of chronic inflammation in age-related muscle wasting. Aging Clin. Exp. Res. 24, 412–422 (2012).
Walston, J. D. Connecting age-related biological decline to frailty and late-life vulnerability. Nestle Nutr. Inst. Workshop Ser. 83, 1–10 (2015).
Wang, J. et al. Vascular smooth muscle cell senescence promotes atherosclerosis and features of plaque vulnerability. Circulation 132, 1909–1919 (2015).
Ntanasi, E. et al. Adherence to mediterranean diet and frailty. J. Am. Med. Dir. Assoc. 19, 315–322.e2 (2017).
Talegawkar, S. A. et al. A higher adherence to a mediterranean-style diet is inversely associated with the development of frailty in community-dwelling elderly men and women. J. Nutr. 142, 2161–2166 (2012).
Rosato, V. et al. Mediterranean diet and cardiovascular disease: a systematic review and meta-analysis of observational studies. Eur. J. Nutr. https://doi.org/10.1007/s00394-017-1582-0 (2017).
Dinu, M., Pagliai, G., Casini, A. & Sofi, F. Mediterranean diet and multiple health outcomes: an umbrella review of meta-analyses of observational studies and randomized trials. Nutr. Metab. Cardiovasc. Dis. 27, e21 (2017).
Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 324, 71–86 (2002).
De Caterina, R. [Aspirin for primary cardiovascular disease prevention - an update]. G. Ital. Cardiol. 18, 1–6 (2017).
Landi, F. et al. Nonsteroidal anti-inflammatory drug (NSAID) use and sarcopenia in older people: results from the ilsirente study. J. Am. Med. Dir. Assoc. 14, 626.e9–626.e13 (2013).
Wang, C.-P., Lorenzo, C., Habib, S. L., Jo, B. & Espinoza, S. E. Differential effects of metformin on age related comorbidities in older men with type 2 diabetes. J. Diabetes Complications 31, 679–686 (2017).
Laksmi, P. W., Setiati, S., Tamin, T. Z. & Soewondo, P. Effect of metformin on handgrip strength, gait speed, myostatin serum level, and health-related quality of life: a double blind randomized controlled trial among non-diabetic pre-frail elderly patients. Acta Med. Indones. 49, 118–127 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02570672 (2017).
Manini, T. M. et al. ENabling reduction of low-grade inflammation in SEniors pilot study: concept, rationale, and design. J. Am. Geriatr. Soc. 65, 1961–1968 (2017).
Golpanian, S. et al. Allogeneic human mesenchymal stem cell infusions for aging frailty. J. Gerontol. A. Biol. Sci. Med. Sci. 72, 1505–1512 (2017).
Tompkins, B. A. et al. Allogeneic mesenchymal stem cells ameliorate aging frailty: a phase II randomized, double-blind, placebo-controlled clinical trial. J. Gerontol. A. Biol. Sci. Med. Sci. 72, 1513–1522 (2017).
LaCroix, A. Z. et al. Statin use and incident frailty in women aged 65 years or older: prospective findings from the Women’s Health Initiative Observational Study. J. Gerontol. A. Biol. Sci. Med. Sci. 63, 369–375 (2008).
Barzilai, N., Huffman, D. M., Muzumdar, R. H. & Bartke, A. The critical role of metabolic pathways in aging. Diabetes 61, 1315–1322 (2012).
Fontana, L. Neuroendocrine factors in the regulation of inflammation: excessive adiposity and calorie restriction. Exp. Gerontol. 44, 41–45 (2009).
Kim, H. J. et al. Modulation of redox-sensitive transcription factors by calorie restriction during aging. Mech. Ageing Dev. 123, 1589–1595 (2002).
LaRocca, T. J., Martens, C. R. & Seals, D. R. Nutrition and other lifestyle influences on arterial aging. Ageing Res. Rev. 39, 106–119 (2017).
Johnson, S. C., Rabinovitch, P. S. & Kaeberlein, M. MTOR is a key modulator of ageing and age-related disease. Nature 493, 338–345 (2013).
Li, J., Kim, S. G. & Blenis, J. Rapamycin: one drug, many effects. Cell Metab. 19, 373–379 (2014).
Harries, L. W. et al. Advancing age is associated with gene expression changes resembling mTOR inhibition: evidence from two human populations. Mech. Ageing Dev. 133, 556–562 (2012).
Blagosklonny, M. V. From rapalogs to anti-aging formula. Oncotarget 8, 35492–35507 (2017).
Halloran, J. et al. Chronic inhibition of mammalian target of rapamycin by rapamycin modulates cognitive and non-cognitive components of behavior throughout lifespan in mice. Neuroscience 223, 102–113 (2012).
Zhang, Y. et al. Rapamycin extends life and health in C57BL/6 mice. J. Gerontol. A. Biol. Sci. Med. Sci. 69A, 119–130 (2014).
Harrison, D. E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
Liao, C. Y. et al. Rapamycin reverses metabolic deficits in lamin A/C-deficient mice. Cell Rep. 17, 2542–2552 (2016).
Strong, R. et al. Nordihydroguaiaretic acid and aspirin increase lifespan of genetically heterogeneous male mice. Aging Cell 7, 641–650 (2008).
Saisho, Y. Metformin and inflammation: Its potential beyond glucose-lowering effect. Endocr. Metab. Immune Disord. Drug Targets 15, 196–205 (2015).
Martin-Montalvo, A. et al. Metformin improves healthspan and lifespan in mice. Nat. Commun. 4, 2192 (2013).
Campbell, J. M., Bellman, S. M., Stephenson, M. D. & Lisy, K. Metformin reduces all-cause mortality and diseases of ageing independent of its effect on diabetes control: a systematic review and meta-analysis. Ageing Res. Rev. 40, 31–44 (2017).
Barzilai, N., Crandall, J. P., Kritchevsky, S. B. & Espeland, M. A. Metformin as a tool to target aging. Cell Metab. 23, 1060–1065 (2016).
Robbins, P. D. & Niedernhofer, L. J. Advances in therapeutic approaches to extend healthspan: a perspective from the 2nd Scripps symposium on the biology of aging. Aging Cell 16, 610–614 (2017).
Walter, E. & Scott, M. The life & work of Rudolf Virchow 1821–1902: ‘Cell theory, thrombosis and the sausage duel’. J. Intensive Care Soc. 18, 234–235 (2017).
Ferrucci, L. et al. Proinflammatory state and circulating erythropoietin in persons with and without anemia. Am. J. Med. 118, 1288.e11–1288.e19 (2005).
de Luca, C. & Olefsky, J. M. Inflammation and insulin resistance. FEBS Lett. 582, 97–105 (2008).
Abbatecola, A. M. et al. Diverse effect of inflammatory markers on insulin resistance and insulin-resistance syndrome in the elderly. J. Am. Geriatr. Soc. 52, 399–404 (2004).
Hotamisligil, G. S. The role of TNFα and TNF receptors in obesity and insulin resistance. J. Intern. Med. 245, 621–625 (1999).
Shimobayashi, M. et al. Insulin resistance causes inflammation in adipose tissue. J. Clin. Invest. 128, 1538–1550 (2018).
Abdelmagid, S. M., Barbe, M. F. & Safadi, F. F. Role of inflammation in the aging bones. Life Sci. 123, 25–34 (2015).
Goldring, S. R. Pathogenesis of bone erosions in rheumatoid arthritis. Curr. Opin. Rheumatol. 14, 406–410 (2002).
Hahn, W. S. et al. Proinflammatory cytokines differentially regulate adipocyte mitochondrial metabolism, oxidative stress, and dynamics. Am. J. Physiol. Endocrinol. Metab. 306, E1033–E1045 (2014).
Lezi, E., Burns, J. M. & Swerdlow, R. H. Effect of high-intensity exercise on aged mouse brain mitochondria, neurogenesis, and inflammation. Neurobiol. Aging 35, 2574–2583 (2014).
Borsini, A. et al. Interferon-α reduces human hippocampal neurogenesis and increases apoptosis via activation of distinct STAT1-dependent mechanisms. Int. J. Neuropsychopharmacol. 21, 187–200 (2018).
Li, Y. P. & Stashenko, P. Proinflammatory cytokines tumor necrosis factor-α and IL-6, but not IL-1, down-regulate the osteocalcin gene promoter. J. Immunol. 148, 788–794 (1992).
Ginaldi, L., Di Benedetto, M. C. & De Martinis, M. Osteoporosis, inflammation and ageing. Immun. Ageing 2, 14 (2005).
Audet, M.-C. & Anisman, H. Interplay between pro-inflammatory cytokines and growth factors in depressive illnesses. Front. Cell. Neurosci. 7, 68 (2013).
Ridker, P. M. et al. Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: a secondary analysis from the CANTOS randomised controlled trial. Lancet 391, 319–328 (2017).
EU Clinical Trial Register. ClinicalTrialsRegister.eu https://www.clinicaltrialsregister.eu/ctr-search/search?query=LoDoCo2 (2016).
Navarro-Gonzalez, J. F. et al. Effect of pentoxifylline on renal function and urinary albumin excretion in patients with diabetic kidney disease: the PREDIAN trial. J. Am. Soc. Nephrol. 26, 220–229 (2015).
Voelker, J. et al. Anti–TGF-β 1 antibody therapy in patients with diabetic nephropathy. J. Am. Soc. Nephrol. 28, 953–962 (2017).
Flossmann, E. & Rothwell, P. M. Effect of aspirin on long-term risk of colorectal cancer: consistent evidence from randomised and observational studies. Lancet 369, 1603–1613 (2007).
Rothwell, P. M. et al. Long-term effect of aspirin on colorectal cancer incidence and mortality: 20-year follow-up of five randomised trials. Lancet 376, 1741–1750 (2010).
Iyengar, R. L. et al. NSAIDs are associated with lower depression scores in patients with osteoarthritis. Am. J. Med. 126, 1017.e11–1017.e18 (2013).
Fields, C., Drye, L., Vaidya, V. & Lyketsos, C. Celecoxib or naproxen treatment does not benefit depressive symptoms in persons age 70 and older: findings from a randomized controlled trial. Am. J. Geriatr. Psychiatry 20, 505–513 (2012).
Raison, C. L. et al. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: the role of baseline inflammatory biomarkers. Arch. Gen. Psychiatry 70, 31–41 (2013).
Menter, A. et al. The effect of adalimumab on reducing depression symptoms in patients with moderate to severe psoriasis: a randomized clinical trial. J. Am. Acad. Dermatol. 62, 812–818 (2010).
Tyring, S. et al. Etanercept and clinical outcomes, fatigue, and depression in psoriasis: double-blind placebo-controlled randomised phase III trial. Lancet 367, 29–35 (2006).
Aisen, P. S., Schmeidler, J. & Pasinetti, G. M. Randomized pilot study of nimesulide treatment in alzheimer’s disease. Neurology 58, 1050–1054 (2002).
Aisen, P. S. et al. Effects of rofecoxib or naproxen vs placebo on Alzheimer disease progression. JAMA 289, 2819 (2003).
Reines, S. A. et al. Rofecoxib: No effect on Alzheimer’s disease in a 1-year, randomized, blinded, controlled study. Neurology 62, 66–71 (2004).
Scharf, S., Mander, A., Ugoni, A., Vajda, F. & Christophidis, N. A double-blind, placebo-controlled trial of diclofenac/misoprostol in Alzheimer’s disease. Neurology 53, 197–197 (1999).
Martin, B. K. et al. Cognitive function over time in the Alzheimer’s disease anti-inflammatory prevention trial (ADAPT): results of a randomized, controlled trial of naproxen and celecoxib. Arch. Neurol. 65, 896–905 (2008).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/show/NCT02284906 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/show/NCT01931566 (2013).
Acknowledgements
The authors received support from the Intramural Research Program of the National Institute on Aging, NIH, Baltimore, MD, USA. The authors thank A. Cornish (National Institute on Aging) for help in editing the manuscript and for the many suggestions that greatly improved the quality of this work, in particular the microbiota section.
Review criteria
The information in this Review is based on a search of the scientific literature published since 2008 using the Medline database and the search terms: “inflammaging”, “inflammation and cardiovascular disease and aging”, “inflammation and frailty”, or “cardiovascular disease and frailty”. The authors reviewed all 3,377 relevant abstracts and selected the manuscripts for which information is reported in this Review. Of note, some articles >10 years old were also cited because their content was considered critical for the topic addressed.
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, discussed its content, wrote the manuscript, and reviewed and edited it before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ferrucci, L., Fabbri, E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol 15, 505–522 (2018). https://doi.org/10.1038/s41569-018-0064-2
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-018-0064-2
This article is cited by
-
Inflammatory markers and frailty in home-dwelling elderly, a cross-sectional study
BMC Geriatrics (2024)
-
Assessing causality between inflammatory bowel diseases with frailty index and sarcopenia: a bidirectional Mendelian randomization study
European Journal of Medical Research (2024)
-
Bazi Bushen ameliorates age-related energy metabolism dysregulation by targeting the IL-17/TNF inflammatory pathway associated with SASP
Chinese Medicine (2024)
-
The role of NLRP3 inflammasome in aging and age-related diseases
Immunity & Ageing (2024)
-
Inflammatory markers and physical frailty: towards clinical application
Immunity & Ageing (2024)