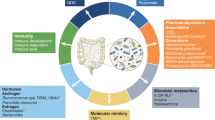
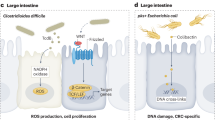
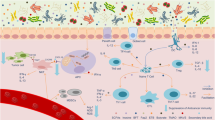
Abstract
Collectively known as the microbiota, the commensal bacteria and other microorganisms that colonize the epithelial surfaces of our body have been shown to produce small molecules and metabolites that have both local and systemic effects on cancer onset, progression and therapy response. To date, most studies focusing on the microbiome have used traditional preclinical mouse models and identified correlative relationships between microbial species and cancer phenotypes. Now, the profound influence of the microbiota on the efficacy of cancer treatments, such as immunotherapies, has begun to be extensively characterized in humans. Paramount to the development of microbiota-based therapeutics, the next challenge in microbiome research will be to identify individual microbial species that causally affect cancer phenotypes and unravel the underlying mechanisms. In this Viewpoint article, we asked four scientists working on the cancer microbiome for their opinions on the current state of the field, where the research is heading and how we can advance our understanding to rationally design microbial-based therapeutics to transform treatment strategies for patients with cancer.
The contributors
Eran Elinav is a professor in the Immunology Department, Weizmann Institute of Science, Israel, and the Director of the Cancer-Microbiome Division, Deutsches Krebsforschungszentrum (DKFZ), Germany. His research focuses on deciphering the molecular basis of host–microbiome interactions and their effects on health and disease, with a goal of personalizing medicine and nutrition.
Wendy S. Garrett is a professor of immunology and infectious diseases at Harvard and a medical oncologist in the Gastrointestinal Cancer Center at Dana-Farber Cancer Institute. She is co-director of the Harvard Chan Center for the Microbiome in Public Health. Her laboratory seeks to carry out mechanistic microbiome studies focused on cancer, inflammatory bowel disease and immune and epithelial cell development and function.
Giorgio Trinchieri is a US National Institutes of Health (NIH) Distinguished Investigator and Director of the Cancer and Inflammation Program, Center for Cancer Research (CCR), National Cancer Institute (NCI), NIH. His research has focused for many years on the interplay between inflammation, innate resistance and adaptive immunity and on the role of pro-inflammatory cytokines and interferons in the regulation of haematopoiesis, innate resistance and immunity against infections and tumours. The present focus of his laboratory is on the role of inflammation, innate resistance, immunity and the commensal microbiota in carcinogenesis, cancer progression and prevention or therapy of cancer.
Jennifer Wargo is an Associate Professor of Surgical Oncology and Genomic Medicine at the University of Texas MD Anderson Cancer Center and is a co-leader of the Melanoma Moon Shot programme. Her primary interests are in response and resistance to targeted therapy for melanoma and other cancers, neoadjuvant strategies and the influence of the gut and tumour microbiome on immunity and antitumour immune responses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gopalakrishnan, V. et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 359, 97–103 (2018).
Matson, V. et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 359, 104–108 (2018).
Pauli, C. et al. Personalized in vitro and in vivo cancer models to guide precision medicine. Cancer Discov. 7, 462–477 (2017).
Sontheimer-Phelps, A., Hassell, B. A. & Ingber, D. E. Modelling cancer in microfluidic human organs-on-chips. Nat. Rev. Cancer 19, 65–81 (2019).
Zeevi, D. et al. Personalized nutrition by prediction of glycemic responses. Cell 163, 1079–1094 (2015).
Bruynseels, K., Santoni de Sio, F. & van den Hoven, J. Digital twins in health care: ethical implications of an emerging engineering paradigm. Front. Genet. 9, 31 (2018).
Drost, J. & Clevers, H. Organoids in cancer research. Nat. Rev. Cancer 18, 407–418 (2018).
Fujii, M., Clevers, H. & Sato, T. Modeling human digestive diseases with CRISPR-Cas9-modified organoids. Gastroenterology 156, 562–576 (2019).
Tanoue, T. et al. A defined commensal consortium elicits CD8 T cells and anti-cancer immunity. Nature 565, 600–605 (2019).
Routy, B. et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 359, 91–97 (2018).
Wege, A. K., Melkus, M. W., Denton, P. W., Estes, J. D. & Garcia, J. V. Functional and phenotypic characterization of the humanized BLT mouse model. Curr. Top. Microbiol. Immunol. 324, 149–165 (2008).
Young, G. R. et al. Resurrection of endogenous retroviruses in antibody-deficient mice. Nature 491, 774–778 (2012).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03341143 (2019).
Vetizou, M. et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350, 1079–1084 (2015).
Gharaibeh, R. Z. & Jobin, C. Microbiota and cancer immunotherapy: in search of microbial signals. Gut https://doi.org/10.1136/gutjnl-2018-317220 (2018).
Knight, R. et al. Best practices for analysing microbiomes. Nat. Rev. Microbiol. 16, 410–422 (2018).
Blank, C. U., Haanen, J. B., Ribas, A. & Schumacher, T. N. The “cancer immunogram”. Science 352, 658–660 (2016).
Cogdill, A. P., Andrews, M. C. & Wargo, J. A. Hallmarks of response to immune checkpoint blockade. Br. J. Cancer 117, 1–7 (2017).
Zmora, N., Soffer, E. & Elinav, E. Transforming medicine with the microbiome. Sci. Transl Med. 11, eaaw1815 (2019).
Shin, W. & Kim, H. J. Intestinal barrier dysfunction orchestrates the onset of inflammatory host-microbiome cross-talk in a human gut inflammation-on-a-chip. Proc. Natl Acad. Sci. USA 115, E10539–E10547 (2018).
Rubinstein, M. R. et al. Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/beta-catenin signaling via its FadA adhesin. Cell Host Microbe 14, 195–206 (2013).
Tahara, T. et al. Fusobacterium in colonic flora and molecular features of colorectal carcinoma. Cancer Res. 74, 1311–1318 (2014).
Bashiardes, S., Tuganbaev, T., Federici, S. & Elinav, E. The microbiome in anti-cancer therapy. Semin. Immunol. 32, 74–81 (2017).
Geller, L. T. et al. Potential role of intratumor bacteria in mediating tumor resistance to the chemotherapeutic drug gemcitabine. Science 357, 1156–1160 (2017).
Zhang, X. et al. Fecal fusobacterium nucleatum for the diagnosis of colorectal tumor: a systematic review and meta-analysis. Cancer Med. 8, 480–491 (2019).
Forbes, N. S. et al. White paper on microbial anti-cancer therapy and prevention. J. Immunother. Cancer 6, 78 (2018).
Iida, N. et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science 342, 967–970 (2013).
Viaud, S. et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science 342, 971–976 (2013).
He, Y. et al. Regional variation limits applications of healthy gut microbiome reference ranges and disease models. Nat. Med. 24, 1532–1535 (2018).
Vedanta Biosciences. Bristol-Myers Squibb and Vedanta Biosciences announce a new clinical collaboration to evaluate OPDIVO® (nivolumab) and VE800 in patients with advanced or metastatic cancers. Vedanta Biosciences https://www.vedantabio.com/news-media/press-releases/detail/2492 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03595683 (2018).
Frankel, A. E. et al. Metagenomic shotgun sequencing and unbiased metabolomic profiling identify specific human gut microbiota and metabolites associated with immune checkpoint therapy efficacy in melanoma patients. Neoplasia 19, 848–855 (2017).
Chaput, N. et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann. Oncol. 28, 1368–1379 (2017).
Wang, Y. et al. Fecal microbiota transplantation for refractory immune checkpoint inhibitor-associated colitis. Nat. Med. 24, 1804–1808 (2018).
McQuade, J. L., Daniel, C. R., Helmink, B. A. & Wargo, J. A. Modulating the microbiome to improve therapeutic response in cancer. Lancet Oncol. 20, e77–e91 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03353402 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03817125 (2019).
Byrd, A. L., Belkaid, Y. & Segre, J. A. The human skin microbiome. Nat. Rev. Microbiol. 16, 143–155 (2018).
Sethi, V. et al. Gut microbiota promotes tumor growth in mice by modulating immune response. Gastroenterology 155, 33–37 (2018).
van der Velden, W. J. et al. Role of the mycobiome in human acute graft-versus-host disease. Biol. Blood Marrow Transplant. 19, 329–332 (2013).
Mukherjee, P. K. et al. Bacteriome and mycobiome associations in oral tongue cancer. Oncotarget 8, 97273–97289 (2017).
Coker, O. O. et al. Enteric fungal microbiota dysbiosis and ecological alterations in colorectal cancer. Gut 68, 654–662 (2018).
Brindley, P. J., Costa, J. & Sripa, B. Why does infection with some helminths cause cancer? Trends Cancer 1, 174–182 (2015).
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Biological agents Volume 100 B. A review of human carcinogens. IARC Monogr. Eval. Carcinog. Risks Hum. 100, 1–441 (2012).
Zhu, F. et al. Autoreactive T cells and chronic fungal infection drive esophageal carcinogenesis. Cell Host Microbe 21, 478–493 (2017).
Draper, L. A. et al. Long-term colonisation with donor bacteriophages following successful faecal microbial transplantation. Microbiome 6, 220 (2018).
Ott, S. J. et al. Efficacy of sterile fecal filtrate transfer for treating patients with Clostridium difficile infection. Gastroenterology 152, 799–811 (2017).
Acknowledgements
E.E. thanks all the students, postdocs and employees at the Elinav laboratory at Weizmann Institute of Science and Deutsches Krebsforschungszentrum (DKFZ) for their continued partnership. E.E. is a senior fellow of the Canadian Institute of Advanced Research (CIFAR) and an international scholar of the Bill and Melinda Gates Foundation and Howard Hughes Medical Institute (HHMI). W.S.G. thanks members of her laboratory and collaborators for stimulating discussions as well as the US National Institutes of Health (NIH) National Cancer Institute (NCI) and Cancer Research UK for research support. J.W. thanks the patients who contributed to these studies, as well as the faculty, staff, students and postdoctoral fellows at the University of Texas MD Anderson Cancer Center who contributed to this work (which was supported by the Melanoma Moon Shot programme). J.W. also thanks worldwide collaborators for providing critical insight and opportunities to learn together. J.W. is a member investigator of the Parker Institute for Cancer Immunotherapy and is also an awardee of an Innovative Research Grant from Stand Up to Cancer–American Association for Cancer Research. J.W.’s research efforts are also supported by a Sabin Family Fellowship, the Melanoma Research Alliance and the NIH (1R011CA219896-01A1).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
E.E. is a paid consultant at DayTwo and BiomX. E.E. states that none of the issues covered in this piece are related to, funded or endorsed by, shared or discussed with or licensed to these commercial entities. W.S.G. is a member of the scientific advisory boards for Evelo Biosciences, Kintai Therapeutics and Leap Therapeutics. G.T. declares no competing financial interests. J.W. is an advisory board member and consultant and adviser for AstraZeneca, Bristol-Myers Squibb, F. Hoffman–La Roche–Genentech, GlaxoSmithKline, Merck Sharp & Dohme, MicrobiomeDX, Biothera Pharmaceuticals and Novartis Pharmaceuticals Corporation, receives grant, clinical and research support from Bristol-Myers Squibb, F. Hoffman–La Roche–Genentech, GlaxoSmithKline and Novartis Pharmaceuticals Corporation, is a speakers bureau participant with Bristol-Myers Squibb, Dava Oncology, Gilead Sciences, Illumina, Imedex, MedImmune and Omniprex and receives other financial or material support from a US patent application (PCT/US17/53.717) submitted by the University of Texas MD Anderson Cancer Center that covers methods to enhance immune checkpoint blockade responses by modulating the microbiome.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elinav, E., Garrett, W.S., Trinchieri, G. et al. The cancer microbiome. Nat Rev Cancer 19, 371–376 (2019). https://doi.org/10.1038/s41568-019-0155-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41568-019-0155-3
This article is cited by
-
Co-enrichment of cancer-associated bacterial taxa is correlated with immune cell infiltrates in esophageal tumor tissue
Scientific Reports (2024)
-
Drug repurposing for cancer therapy
Signal Transduction and Targeted Therapy (2024)
-
Intratumoural microbiota: a new frontier in cancer development and therapy
Signal Transduction and Targeted Therapy (2024)
-
Exploring the microbiome of oral epithelial dysplasia as a predictor of malignant progression
BMC Oral Health (2023)
-
Innate lymphoid cells and innate-like T cells in cancer — at the crossroads of innate and adaptive immunity
Nature Reviews Cancer (2023)