Abstract
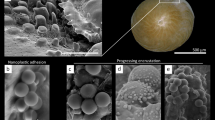
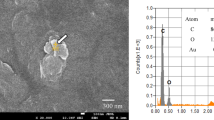
The role of aquatic organisms in the biological fragmentation of microplastics and their contribution to global nanoplastic pollution are poorly understood. Here we present a biological fragmentation pathway that generates nanoplastics during the ingestion of microplastics by rotifers, a commonly found and globally distributed surface water zooplankton relevant for nutrient recycling. Both marine and freshwater rotifers could rapidly grind polystyrene, polyethylene and photo-aged microplastics, thus releasing smaller particulates during ingestion. Nanoindentation studies of the trophi of the rotifer chitinous mastax revealed a Young’s modulus of 1.46 GPa, which was higher than the 0.79 GPa for polystyrene microparticles, suggesting a fragmentation mechanism through grinding the edges of microplastics. Marine and freshwater rotifers generated over 3.48 × 105 and 3.66 × 105 submicrometre particles per rotifer in a day, respectively, from photo-aged microplastics. Our data suggest the ubiquitous occurrence of microplastic fragmentation by different rotifer species in natural aquatic environments of both primary and secondary microplastics of various polymer compositions and provide previously unidentified insights into the fate of microplastics and the source of nanoplastics in global surface waters.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The raw data that support the findings of this study are available at the publicly accessible online repository Figshare with the identifier https://doi.org/10.6084/m9.figshare.24125016. Source data are provided with this paper.
References
Stubbins, A., Law, K. L., Muñoz, S. E., Bianchi, T. S. & Zhu, L. Plastics in the earth system. Science 373, 51–55 (2021).
Ross, P. S. et al. Pervasive distribution of polyester fibres in the Arctic Ocean is driven by Atlantic inputs. Nat. Commun. 12, 106 (2021).
Aves, A. R. et al. First evidence of microplastics in Antarctic snow. Cryosphere 16, 2127–2145 (2022).
Woodward, J., Li, J., Rothwell, J. & Hurley, R. Acute riverine microplastic contamination due to avoidable releases of untreated wastewater. Nat. Sustain. 4, 793–802 (2021).
Peng, X. et al. Microplastics contaminate the deepest part of the world’s ocean. Geochem. Perspect. Lett. 9, 1–5 (2018).
Santos, R. G., Machovsky-Capuska, G. E. & Andrades, R. Plastic ingestion as an evolutionary trap: toward a holistic understanding. Science 373, 56–60 (2021).
MacLeod, M., Arp, H. P. H., Tekman, M. B. & Jahnke, A. The global threat from plastic pollution. Science 373, 61–65 (2021).
Gigault, J. et al. Nanoplastics are neither microplastics nor engineered nanoparticles. Nat. Nanotechnol. 16, 501–507 (2021).
Vethaak, A. D. & Legler, J. Microplastics and human health. Science 371, 672–674 (2021).
Wagner, S. & Reemtsma, T. Things we know and don’t know about nanoplastic in the environment. Nat. Nanotechnol. 14, 300–301 (2019).
Gerritse, J., Leslie, H. A., Caroline, A., Devriese, L. I. & Vethaak, A. D. Fragmentation of plastic objects in a laboratory seawater microcosm. Sci. Rep. 10, 10945 (2020).
Dawson, A. L. et al. Turning microplastics into nanoplastics through digestive fragmentation by Antarctic krill. Nat. Commun. 9, 1001 (2018).
Wang, C., Zhao, J. & Xing, B. Environmental source, fate, and toxicity of microplastics. J. Hazard. Mater. 407, 124357 (2021).
Hewitt, D. P. & George, D. G. The population dynamics of Keratella cochlearis in a hypereutrophic tarn and the possible impact of predation by young roach. Hydrobiologia 147, 221–227 (1987).
Jeong, C. B. et al. Microplastic size-dependent toxicity, oxidative stress induction, and p-JNK and p-p38 activation in the monogonont rotifer (Brachionus koreanus). Environ. Sci. Technol. 50, 8849–8857 (2016).
Baer, A., Langdon, C., Mills, S., Schulz, C. & Hamre, K. Particle size preference, gut filling and evacuation rates of the rotifer Brachionus “Cayman” using polystyrene latex beads. Aquaculture 282, 75–82 (2008).
Stelzer, C. P., Riss, S. & Stadler, P. Genome size evolution at the speciation level: the cryptic species complex Brachionus plicatilis (Rotifera). BMC Evol. Biol. 11, 90 (2011).
Papakostas, S. et al. Integrative taxonomy recognizes evolutionary units despite widespread mitonuclear discordance: evidence from a rotifer cryptic species complex. Syst. Biol. 65, 508–524 (2016).
Gilbert, J. J. & Walsh, E. J. Brachionus calyciflorus is a species complex: mating behavior and genetic differentiation among four geographically isolated strains. Hydrobiologia 546, 257–265 (2005).
Drago, C. & Weithoff, G. Variable fitness response of two rotifer species exposed to microplastics particles: the role of food quantity and quality. Toxics 9, 305 (2021).
Fournier, S. B. et al. Nanopolystyrene translocation and fetal deposition after acute lung exposure during late-stage pregnancy. Part. Fibre Toxicol. 17, 55 (2020).
Kleinow, W. & Wratil, H. On the structure and function of the mastax of Brachionus plicatilis (Rotifera), a scanning electron microscope analysis. Zoomorphology 116, 169–177 (1996).
Klusemann, J., Kleinow, W. & Peters, W. The hard parts (trophi) of the rotifer mastax do contain chitin: evidence from studies on Brachionus plicatilis. Histochemistry 94, 277–283 (1990).
Cornillac, A., Wurdak, E. & Clément, P. Biology of Rotifers (Springer, 1983).
Garvey, C. J. et al. Molecular-scale understanding of the embrittlement in polyethylene ocean debris. Environ. Sci. Technol. 54, 11173–11181 (2020).
Liu, Z. et al. Quantifying the dynamics of polystyrene microplastics UV-aging process. Environ. Sci. Technol. Lett. 9, 50–56 (2022).
Huang, Z. et al. Influence of protein configuration on aggregation kinetics of nanoplastics in aquatic environment. Water Res. 219, 118522 (2022).
Iyer, N. & Rao, T. Responses of the predatory rotifer Asplanchna intermedia to prey species differing in vulnerability: laboratory and field studies. Freshw. Biol. 36, 521–533 (1996).
Yuan, W., Liu, X., Wang, W., Di, M. & Wang, J. Microplastic abundance, distribution and composition in water, sediments, and wild fish from Poyang Lake, China. Ecotoxicol. Environ. Saf. 170, 180–187 (2019).
Wang, J., Wu, J., Yu, Y., Wang, T. & Gong, C. The specific list, quantitative distribution and change of zooplankton in the season of spring and autumn in Poyang Lake. J. Lake Sci. 15, 345–352 (2003).
Gilbert, J. J. Food niches of planktonic rotifers: diversification and implications. Limnol. Oceanogr. 67, 2218–2251 (2022).
Han, M. et al. Distribution of microplastics in surface water of the lower Yellow River near estuary. Sci. Total Environ. 707, 135601 (2020).
Fan, Y. et al. Spatiotemporal dynamics of microplastics in an urban river network area. Water Res. 212, 118116 (2022).
Janakiraman, A., Naveed, M. S. & Altaff, K. Impact of domestic sewage pollution on rotifer abundance in Adyar estuary. Int. J. Environ. Sci. 3, 689–696 (2012).
Cai, H., Chen, M., Du, F., Matthews, S. & Shi, H. Separation and enrichment of nanoplastics in environmental water samples via ultracentrifugation. Water Res. 203, 117509 (2021).
Nigamatzyanova, L. & Fakhrullin, R. Dark-field hyperspectral microscopy for label-free microplastics and nanoplastics detection and identification in vivo: a Caenorhabditis elegans study. Environ. Pollut. 271, 116337 (2021).
Stojicic, S., Zivkovic, S., Qian, W., Zhang, H. & Haapasalo, M. Tissue dissolution by sodium hypochlorite: effect of concentration, temperature, agitation, and surfactant. J. Endod. 36, 1558–1562 (2010).
Chopinet, L., Formosa, C., Rols, M. P., Duval, R. E. & Dague, E. Imaging living cells surface and quantifying its properties at high resolution using AFM in QI™ mode. Micron 48, 26–33 (2013).
de Vega, R. G. et al. Characterisation of microplastics and unicellular algae in seawater by targeting carbon via single particle and single cell ICP-MS. Anal. Chim. Acta 1174, 338737 (2021).
Podar, M. et al. Global prevalence and distribution of genes and microorganisms involved in mercury methylation. Sci. Adv. 1, e1500675 (2015).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (42192572, U2106213), Taishan Scholars Program of Shandong Province (tsqn201909051), Key Research and Development Program of Shandong Province (2020CXGC010703), Laoshan Laboratory (LSKJ202203901) and Fundamental Research Funds for the Central Universities (202172001, 202141003). We thank Z. Rao at the Chinese Academy of Sciences and J. Li at Zeiss for their assistance on HSI and RISE, respectively.
Author information
Authors and Affiliations
Contributions
J.Z. and B.X. conceptualized the idea and designed all the experiments, and J.Z. supervised the project. R.L., W.S., R.X. and J.Z. carried out the experiments. R.L., J.Z. and B.X wrote the manuscript. Z.W., D.S., X.L., T.Y., Z.L. and Y.D. supported the quantitative analysis of nanoplastics, and discussed and revised the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Chang-Bum Jeong, Guntram Weithoff, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 The quantitative results on the generation of nanoplastics from different microplastics by rotifers.
(A): The numbers of nanoplastics generated from PS fibers; (B): The numbers of nanoplastics generated from PS microplastics obtained from the food container. Data are presented as mean ± s.d. (n = 3 for all groups in Panel A and “PS debris in rotifers” group in Panel B; n = 4 for “original PS debris” group and “PS debris in medium” group in Panel B). Statistical analyses were performed using a one-sided ANOVA followed by LSD post hoc test.
Supplementary information
Supplementary Information
Supplementary Figs. 1–38, Videos 1–3 and Tables 1–3.
Supplementary Video 1
Uptake and grinding of PS microplastics by the rotifer.
Supplementary Video 2
Grinding of 10 μm PS microplastics by the rotifer 60 times before internalization.
Supplementary Video 3
Excretion of photo-aged PS microplastics and the fragments by the rotifer.
Supplementary Data 1
Source Data for Supplementary Figures.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, J., Lan, R., Wang, Z. et al. Microplastic fragmentation by rotifers in aquatic ecosystems contributes to global nanoplastic pollution. Nat. Nanotechnol. 19, 406–414 (2024). https://doi.org/10.1038/s41565-023-01534-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-023-01534-9