Abstract
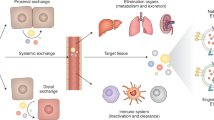
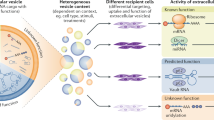
Extracellular vesicles (EVs) are biological nanoparticles that promote intercellular communication by delivering bioactive cargo over short and long distances. Short-distance communication takes place in the interstitium, whereas long-distance communication is thought to require transport through the blood circulation to reach distal sites. Extracellular vesicle therapeutics are frequently injected systemically, and diagnostic approaches often rely on the detection of organ-derived EVs in the blood. However, the mechanisms by which EVs enter and exit the circulation are poorly understood. Here, the lymphatic system and transport across the endothelial barrier through paracellular and transcellular routes are discussed as potential pathways for EV entry to and exit from the blood circulatory system.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Alberro, A., Iparraguirre, L., Fernandes, A. & Otaegui, D. Extracellular vesicles in blood: sources, effects, and applications. Int. J. Mol. Sci. 22, 8163 (2021).
Witwer, K. W. & Wolfram, J. Extracellular vesicles versus synthetic nanoparticles for drug delivery. Nat. Rev. Mater. 6, 103–106 (2021).
Busatto, S., Pham, A., Suh, A., Shapiro, S. & Wolfram, J. Organotropic drug delivery: synthetic nanoparticles and extracellular vesicles. Biomed. Microdevices 21, 46 (2019).
Beetler, D. J. et al. Extracellular vesicles as personalized medicine. Mol. Aspects Med. 91, 101155 (2022).
Walker, S. et al. Extracellular vesicle-based drug delivery systems for cancer treatment. Theranostics 9, 8001–8017 (2019).
Hu, T., Wolfram, J. & Srivastava, S. Extracellular vesicles in cancer detection: hopes and hypes. Trends Cancer 7, 122–133 (2020).
Iannotta, D., Yang, M., Celia, C., Di Marzio, L. & Wolfram, J. Extracellular vesicle therapeutics from plasma and adipose tissue. Nano Today 39, 101159 (2021).
Ghodasara, A., Raza, A., Wolfram, J., Salomon, C. & Popat, A. Clinical translation of extracellular vesicles. Adv. Healthc. Mater. https://doi.org/10.1002/adhm.202301010 (2023).
Dumas, S. J. et al. Phenotypic diversity and metabolic specialization of renal endothelial cells. Nat. Rev. Nephrol. 17, 441–464 (2021).
Jourde-Chiche, N. et al. Endothelium structure and function in kidney health and disease. Nat. Rev. Nephrol. 15, 87–108 (2019).
Wolfram, J. & Ferrari, M. Clinical cancer nanomedicine. Nano Today 25, 85–89 (2019).
Prabhakar, U. et al. Challenges and key considerations of the enhanced permeability and retention effect for nanomedicine drug delivery in oncology. Cancer Res. 73, 2412–2417 (2013).
Sindhwani, S. et al. The entry of nanoparticles into solid tumours. Nat. Mater. 19, 566–575 (2020).
Lessey-Morillon, E. C. et al. The RhoA guanine nucleotide exchange factor, LARG, mediates ICAM-1-dependent mechanotransduction in endothelial cells to stimulate transendothelial migration. J. Immunol. 192, 3390–3398 (2014).
Zeng, Z. et al. Cancer-derived exosomal miR-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat. Commun. 9, 5395 (2018).
Treps, L., Perret, R., Edmond, S., Ricard, D. & Gavard, J. Glioblastoma stem-like cells secrete the pro-angiogenic VEGF-A factor in extracellular vesicles. J. Extracell. Vesicles 6, 1359479 (2017).
Tominaga, N. et al. Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood–brain barrier. Nat. Commun. 6, 6716 (2015).
De La Cruz, E. M. How cofilin severs an actin filament. Biophys. Rev. 1, 51–59 (2009).
Chatterjee, V. et al. Endothelial microvesicles carrying Src-rich cargo impair adherens junction integrity and cytoskeleton homeostasis. Cardiovasc. Res. 116, 1525–1538 (2020).
Sperandio, M., Gleissner, C. A. & Ley, K. Glycosylation in immune cell trafficking. Immunol. Rev. 230, 97–113 (2009).
Goncalves, J. P., Deliwala, V. J., Kolarich, D., Souza-Fonseca-Guimaraes, F. & Wolfram, J. The cancer cell-derived extracellular vesicle glycocode in immunoevasion. Trends Immunol. 43, 864–867 (2022).
Yang, M. et al. Extracellular vesicle glucose transporter-1 and glycan features in monocyte-endothelial inflammatory interactions. Nanomedicine 42, 102515 (2022).
Walker, S. A. et al. Glycan node analysis of plasma-derived extracellular vesicles. Cells 9, 1946 (2020).
Williams, C. et al. Glycosylation of extracellular vesicles: current knowledge, tools and clinical perspectives. J. Extracell. Vesicles 7, 1442985 (2018).
Pendiuk Goncalves, J. et al. Glycan node analysis detects varying glycosaminoglycan levels in melanoma-derived extracellular vesicles. Int. J. Mol. Sci. 9, 1946 (2023).
Li, Y. et al. EV-origin: enumerating the tissue-cellular origin of circulating extracellular vesicles using exLR profile. Comput Struct. Biotechnol. J. 18, 2851–2859 (2020).
Baluk, P. et al. Functionally specialized junctions between endothelial cells of lymphatic vessels. J. Exp. Med. 204, 2349–2362 (2007).
Trzewik, J., Mallipattu, S. K., Artmann, G. M., Delano, F. A. & Schmid-Schönbein, G. W. Evidence for a second valve system in lymphatics: endothelial microvalves. FASEB J. 15, 1711–1717 (2001).
Breslin, J. W. et al. Lymphatic vessel network structure and physiology. Compr. Physiol. 9, 207–299 (2018).
Liu, D. et al. CD97 promotion of gastric carcinoma lymphatic metastasis is exosome dependent. Gastric Cancer 19, 754–766 (2016).
Shimizu, A. et al. Exosomal CD47 plays an essential role in immune evasion in ovarian cancer. Mol. Cancer Res. 19, 1583–1595 (2021).
Tessandier, N. et al. Platelets disseminate extracellular vesicles in lymph in rheumatoid arthritis. Arterioscler. Thromb. Vasc. Biol. 40, 929–942 (2020).
Welsh, J. D., Kahn, M. L. & Sweet, D. T. Lymphovenous hemostasis and the role of platelets in regulating lymphatic flow and lymphatic vessel maturation. Blood 128, 1169–1173 (2016).
Mehta, D. & Malik, A. B. Signaling mechanisms regulating endothelial permeability. Physiol. Rev. 86, 279–367 (2006).
Fernández-Hernando, C. et al. Genetic evidence supporting a critical role of endothelial caveolin-1 during the progression of atherosclerosis. Cell Metab. 10, 48–54 (2009).
Morad, G. et al. Tumor-derived extracellular vesicles breach the intact blood–brain barrier via transcytosis. ACS Nano 13, 13853–13865 (2019).
Chen, C. C. et al. Elucidation of exosome migration across the blood–brain barrier model in vitro. Cell. Mol. Bioeng. 9, 509–529 (2016).
Gonda, A., Kabagwira, J., Senthil, G. N. & Wall, N. R. Internalization of exosomes through receptor-mediated endocytosis. Mol. Cancer Res. 17, 337–347 (2019).
Mulcahy, L. A., Pink, R. C. & Carter, D. R. F. Routes and mechanisms of extracellular vesicle uptake. J. Extracell. Vesicles 3, 24641 (2014).
Feng, Y. et al. The blocking of integrin-mediated interactions with maternal endothelial cells reversed the endothelial cell dysfunction induced by EVs, derived from preeclamptic placentae. Int. J. Mol. Sci. 23, 13115 (2022).
Fomina, A. F., Deerinck, T. J., Ellisman, M. H. & Cahalan, M. D. Regulation of membrane trafficking and subcellular organization of endocytic compartments revealed with FM1-43 in resting and activated human T cells. Exp. Cell. Res. 291, 150–166 (2003).
Morelli, A. E. et al. Endocytosis, intracellular sorting, and processing of exosomes by dendritic cells. Blood 104, 3257–3266 (2004).
Wei, X. et al. Surface phosphatidylserine is responsible for the internalization on microvesicles derived from hypoxia-induced human bone marrow mesenchymal stem cells into human endothelial cells. PLoS ONE 11, e0147360 (2016).
He, C., Hu, Y., Yin, L., Tang, C. & Yin, C. Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials 31, 3657–3666 (2010).
Lu, F., Wu, S. H., Hung, Y. & Mou, C. Y. Size effect on cell uptake in well‐suspended, uniform mesoporous silica nanoparticles. Small 5, 1408–1413 (2009).
Théry, C. et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 7, 1535750 (2018).
Gould, S. J. & Raposo, G. As we wait: coping with an imperfect nomenclature for extracellular vesicles. J. Extracell. Vesicles 2, 20389 (2013).
Sousa de Almeida, M. et al. Understanding nanoparticle endocytosis to improve targeting strategies in nanomedicine. Chem. Soc. Rev. 50, 5397–5434 (2021).
Nazarenko, I. et al. Cell surface tetraspanin Tspan8 contributes to molecular pathways of exosome-induced endothelial cell activation. Cancer Res. 70, 1668–1678 (2010).
Yuan, D. et al. Macrophage exosomes as natural nanocarriers for protein delivery to inflamed brain. Biomaterials 142, 1–12 (2017).
Joshi, B. S. & Zuhorn, I. S. Heparan sulfate proteoglycan-mediated dynamin-dependent transport of neural stem cell exosomes in an in vitro blood–brain barrier model. Eur. J. Neurosci. 53, 706–719 (2021).
Ihrcke, N. S., Wrenshall, L. E., Lindman, B. J. & Platt, J. L. Role of heparan sulfate in immune system-blood vessel interactions. Immunol. Today 14, 500–505 (1993).
Chanda, D. et al. Fibronectin on the surface of extracellular vesicles mediates fibroblast invasion. Am. J. Respir. Cell Mol. Biol. 60, 279–288 (2019).
Purushothaman, A. et al. Fibronectin on the surface of myeloma cell-derived exosomes mediates exosome-cell interactions. J. Biol. Chem. 291, 1652–1663 (2016).
Deng, Z. et al. Tumor cell cross talk with tumor-associated leukocytes leads to induction of tumor exosomal fibronectin and promotes tumor progression. Am. J. Pathol. 180, 390–398 (2012).
Mertens, G., Cassiman, J. J., Van den Berghe, H., Vermylen, J. & David, G. Cell surface heparan sulfate proteoglycans from human vascular endothelial cells. Core protein characterization and antithrombin III binding properties. J. Biol. Chem. 267, 20435–20443 (1992).
Matsumoto, J. et al. Transmission of α-synuclein-containing erythrocyte-derived extracellular vesicles across the blood–brain barrier via adsorptive mediated transcytosis: another mechanism for initiation and progression of Parkinson’s disease? Acta Neuropathol. Commun. 5, 71 (2017).
Banks, W. A. et al. Transport of extracellular vesicles across the blood–brain barrier: brain pharmacokinetics and effects of inflammation. Int. J. Mol. Sci. 21, 4407 (2020).
Hervé, F., Ghinea, N. & Scherrmann, J.-M. CNS delivery via adsorptive transcytosis. AAPS J. 10, 455–472 (2008).
Banks, W. A., Kastin, A. J., Brennan, J. M. & Vallance, K. L. Adsorptive endocytosis of HIV-1gp120 by blood–brain barrier is enhanced by lipopolysaccharide. Exp. Neurol. 156, 165–171 (1999).
Wurdinger, T. et al. Extracellular vesicles and their convergence with viral pathways. Adv. Virol. 2012, 767694 (2012).
Banks, W. A. et al. Transport of human immunodeficiency virus type 1 pseudoviruses across the blood–brain barrier: role of envelope proteins and adsorptive endocytosis. J. Virol. 75, 4681–4691 (2001).
Ben-Zvi, A. et al. Mfsd2a is critical for the formation and function of the blood–brain barrier. Nature 509, 507–511 (2014).
Andreone, B. J. et al. Blood–brain barrier permeability is regulated by lipid transport-dependent suppression of caveolae-mediated transcytosis. Neuron 94, 581–594.e5 (2017).
Nguyen, L. N. et al. Mfsd2a is a transporter for the essential omega-3 fatty acid docosahexaenoic acid. Nature 509, 503–506 (2014).
Busatto, S. et al. Lipoprotein-based drug delivery. Adv. Drug Deliv. Rev. 159, 377–390 (2020).
Simonsen, J. B. What are we looking at? Extracellular vesicles, lipoproteins, or both. Circ. Res. 121, 920–922 (2017).
Toth, E. A. et al. Formation of a protein corona on the surface of extracellular vesicles in blood plasma. J. Extracell. Vesicles 10, e12140 (2021).
Sodar, B. W. et al. Low-density lipoprotein mimics blood plasma-derived exosomes and microvesicles during isolation and detection. Sci. Rep. 6, 24316 (2016).
Busatto, S. et al. Brain metastases-derived extracellular vesicles induce binding and aggregation of low-density lipoprotein. J. Nanobiotechnol. 18, 162 (2020).
Busatto, S. et al. Considerations for extracellular vesicle and lipoprotein interactions in cell culture assays. J. Extracell. Vesicles 11, e12202 (2022).
Lozano-Andrés, E. et al. Physical association of low density lipoprotein particles and extracellular vesicles unveiled by single particle analysis. Preprint at https://doi.org/10.1101/2022.08.31.506022 (2022).
Pham, M.-T. et al. Endosomal egress and intercellular transmission of hepatic ApoE-containing lipoproteins and its exploitation by the hepatitis C virus. PLoS Pathog. 19, e1011052 (2023).
Broad, K. et al. Unraveling multilayered extracellular vesicles: speculation on cause. J. Extracell. Vesicles 12, e12309 (2023).
Phinney, D. G. et al. Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nat. Commun. 6, 8472 (2015).
Dixson, A. C., Dawson, T. R., Di Vizio, D. & Weaver, A. M. Context-specific regulation of extracellular vesicle biogenesis and cargo selection. Nat. Rev. Mol. Cell Biol. 4, 454–476 (2023).
Dallas, S. L., Prideaux, M. & Bonewald, L. F. The osteocyte: an endocrine cell … and more. Endocr. Rev. 34, 658–690 (2013).
Abbott, N. J., Ronnback, L. & Hansson, E. Astrocyte–endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 7, 41–53 (2006).
Xie, Y., Bagby, T. R., Cohen, M. S. & Forrest, M. L. Drug delivery to the lymphatic system: importance in future cancer diagnosis and therapies. Expert Opin. Drug Deliv. 6, 785–792 (2009).
Parker, R. J., Hartman, K. D. & Sieber, S. M. Lymphatic absorption and tissue disposition of liposome-entrapped [14C]adriamycin following intraperitoneal administration to rats. Cancer Res. 41, 1311–1317 (1981).
Fujimoto, Y., Okuhata, Y., Tyngi, S., Namba, Y. & Oku, N. Magnetic resonance lymphography of profundus lymph nodes with liposomal gadolinium-diethylenetriamine pentaacetic acid. Biol. Pharm. Bull. 23, 97–100 (2000).
Kang, M., Jordan, V., Blenkiron, C. & Chamley, L. W. Biodistribution of extracellular vesicles following administration into animals: a systematic review. J. Extracell. Vesicles 10, e12085 (2021).
Amruta, A., Iannotta, D., Cheetham, S. W., Lammers, T. & Wolfram, J. Vasculature organotropism in drug delivery. Adv. Drug Deliv. Rev. 201, 115054 (2023).
Li, C. et al. The role of exosomal miRNAs in cancer. J. Transl. Med. 20, 6 (2022).
Crowl, J. T., Gray, E. E., Pestal, K., Volkman, H. E. & Stetson, D. B. Intracellular nucleic acid detection in autoimmunity. Annu. Rev. Immunol. 35, 313–336 (2017).
Snaebjornsson, M. T., Janaki-Raman, S. & Schulze, A. Greasing the wheels of the cancer machine: the role of lipid metabolism in cancer. Cell Metab. 31, 62–76 (2020).
Lei, K. et al. Cancer-cell stiffening via cholesterol depletion enhances adoptive T-cell immunotherapy. Nat. Biomed. Eng. 5, 1411–1425 (2021).
Acknowledgements
Partial funding for this work was provided by The University of Queensland, Australia (J.W.), the National Institute on Aging, National Institutes of Health, United States under award number R01AG076537 (J.W.) and The Medical Research Future Fund, Australia under award number MRF2019485 (J.W.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the organizations and funding agencies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests. J.W. is listed on an extracellular vesicle grant from Ionis Pharmaceuticals, but the article topic is not a primary focus of the grant and the grant funding did not contribute to this work.
Peer review
Peer review information
Nature Nanotechnology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Iannotta, D., A, A., Kijas, A.W. et al. Entry and exit of extracellular vesicles to and from the blood circulation. Nat. Nanotechnol. 19, 13–20 (2024). https://doi.org/10.1038/s41565-023-01522-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-023-01522-z
This article is cited by
-
The ‘communicatome’ of pregnancy: spotlight on cellular and extravesicular chimerism
EMBO Molecular Medicine (2024)