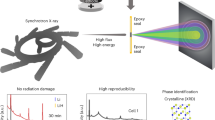
Abstract
Mn dissolution has been a long-standing, ubiquitous issue that negatively impacts the performance of Mn-based battery materials. Mn dissolution involves complex chemical and structural transformations at the electrode–electrolyte interface. The continuously evolving electrode–electrolyte interface has posed great challenges for characterizing the dynamic interfacial process and quantitatively establishing the correlation with battery performance. In this study, we visualize and quantify the temporally and spatially resolved Mn dissolution/redeposition (D/R) dynamics of electrochemically operating Mn-containing cathodes. The particle-level and electrode-level analyses reveal that the D/R dynamics is associated with distinct interfacial degradation mechanisms at different states of charge. Our results statistically differentiate the contributions of surface reconstruction and Jahn–Teller distortion to the Mn dissolution at different operating voltages. Introducing sulfonated polymers (Nafion) into composite electrodes can modulate the D/R dynamics by trapping the dissolved Mn species and rapidly establishing local Mn D/R equilibrium. This work represents an inaugural effort to pinpoint the chemical and structural transformations responsible for Mn dissolution via an operando synchrotron study and develops an effective method to regulate Mn interfacial dynamics for improving battery performance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within this Article and its Supplementary Information. Any other data are available from the corresponding authors on request.
References
Thackeray, M. M. & Amine, K. LiMn2O4 spinel and substituted cathodes. Nat. Energy 6, 566 (2021).
Kim, D. K. et al. Spinel LiMn2O4 nanorods as lithium ion battery cathodes. Nano Lett. 8, 3948–3952 (2008).
Xia, H., Luo, Z. & Xie, J. Nanostructured LiMn2O4 and their composites as high-performance cathodes for lithium-ion batteries. Prog. Nat. Sci.: Mater. Int. 22, 572–584 (2012).
Lun, Z. et al. Design principles for high-capacity Mn-based cation-disordered rocksalt cathodes. Chem 6, 153–168 (2020).
Li, H. et al. Toward high-energy Mn-based disordered-rocksalt Li-ion cathodes. Joule 6, 53–91 (2022).
Zhang, Y. et al. Investigating particle size‐dependent redox kinetics and charge distribution in disordered rocksalt cathodes. Adv. Funct. Mater. 32, 2110502 (2022).
Sun, X., Xiao, R., Yu, X. & Li, H. First-principles simulations for the surface evolution and Mn dissolution in the fully delithiated spinel LiMn2O4. Langmuir 37, 5252–5259 (2021).
Zhan, C., Wu, T., Lu, J. & Amine, K. Dissolution, migration, and deposition of transition metal ions in Li-ion batteries exemplified by Mn-based cathodes—a critical review. Energy Environ. Sci. 11, 243–257 (2018).
Tang, D. et al. Surface structure evolution of LiMn2O4 cathode material upon charge/discharge. Chem. Mater. 26, 3535–3543 (2014).
Zhou, G. et al. Mn ion dissolution mechanism for lithium-ion battery with LiMn2O4 cathode: in situ ultraviolet–visible spectroscopy and ab initio molecular dynamics simulations. J. Phys. Chem. Lett. 11, 3051–3057 (2020).
Zhu, X. et al. LiMnO2 cathode stabilized by interfacial orbital ordering for sustainable lithium-ion batteries. Nat. Sustain. 4, 392–401 (2021).
Lin, R. et al. Characterization of the structure and chemistry of the solid–electrolyte interface by cryo-EM leads to high-performance solid-state Li-metal batteries. Nat. Nanotechnol. 17, 768–776 (2022).
Cao, L. et al. Fluorinated interphase enables reversible aqueous zinc battery chemistries. Nat. Nanotechnol. 16, 902–910 (2021).
Liu, T. et al. In situ quantification of interphasial chemistry in Li-ion battery. Nat. Nanotechnol. 14, 50–56 (2019).
Xiang, Y. et al. Quantitatively analyzing the failure processes of rechargeable Li metal batteries. Sci. Adv. 7, eabj3423 (2021).
Liu, T. et al. Correlation between manganese dissolution and dynamic phase stability in spinel-based lithium-ion battery. Nat. Commun. 10, 4721 (2019).
Xu, C. et al. Bulk fatigue induced by surface reconstruction in layered Ni-rich cathodes for Li-ion batteries. Nat. Mater. 20, 84–92 (2021).
Lin, F. et al. Surface reconstruction and chemical evolution of stoichiometric layered cathode materials for lithium-ion batteries. Nat. Commun. 5, 3529 (2014).
Liu, X. et al. Distinct charge dynamics in battery electrodes revealed by in situ and operando soft X-ray spectroscopy. Nat. Commun. 4, 2568 (2013).
Yuan, Y., Amine, K., Lu, J. & Shahbazian-Yassar, R. Understanding materials challenges for rechargeable ion batteries with in situ transmission electron microscopy. Nat. Commun. 8, 15806 (2017).
Jaumaux, P. et al. Localized water‐in‐salt electrolyte for aqueous lithium‐ion batteries. Angew. Chem. Int. Ed. 60, 19965–19973 (2021).
Suo, L. et al. ‘Water-in-salt’ electrolyte enables high-voltage aqueous lithium-ion chemistries. Science 350, 938–943 (2015).
Xu, J. et al. Aqueous electrolyte design for super-stable 2.5 V LiMn2O4 || Li4Ti5O12 pouch cells. Nat. Energy 7, 186–193 (2022).
Xie, J., Liang, Z. & Lu, Y.-C. Molecular crowding electrolytes for high-voltage aqueous batteries. Nat. Mater. 19, 1006–1011 (2020).
Wang, C. et al. Overlooked electrolyte destabilization by manganese (ii) in lithium-ion batteries. Nat. Commun. 10, 3423 (2019).
Leifer, N. et al. Studies of spinel-to-layered structural transformations in LiMn2O4 electrodes charged to high voltages. J. Phys. Chem. C 121, 9120–9130 (2017).
Vissers, D. R. et al. Role of manganese deposition on graphite in the capacity fading of lithium ion batteries. ACS Appl. Mater. Interfaces 8, 14244–14251 (2016).
Ren, Q., Yuan, Y. & Wang, S. Interfacial strategies for suppression of Mn dissolution in rechargeable battery cathode materials. ACS Appl. Mater. Interfaces 14, 23022–23032 (2021).
Xu, W. et al. Understanding the effect of Al doping on the electrochemical performance improvement of the LiMn2O4 cathode material. ACS Appl. Mater. Interfaces 13, 45446–45454 (2021).
Lee, S., Cho, Y., Song, H., Lee, K. T. & Cho, J. Carbon‐coated single‐crystal LiMn2O4 nanoparticle clusters as cathode material for high‐energy and high‐power lithium‐ion batteries. Angew. Chem. Int. Ed. 51, 8748–8752 (2012).
Wandt, J. et al. Transition metal dissolution and deposition in Li-ion batteries investigated by operando X-ray absorption spectroscopy. J. Mater. Chem. A 4, 18300–18305 (2016).
Gao, X. et al. Oxygen loss and surface degradation during electrochemical cycling of lithium-ion battery cathode material LiMn2O4. J. Mater. Chem. A 7, 8845–8854 (2019).
Santo, K. P. & Neimark, A. V. Effects of metal-polymer complexation on structure and transport properties of metal-substituted polyelectrolyte membranes. J. Colloid Interface Sci. 602, 654–668 (2021).
Kumar, R., Pasupathi, S., Pollet, B. G. & Scott, K. Nafion-stabilised platinum nanoparticles supported on titanium nitride: an efficient and durable electrocatalyst for phosphoric acid based polymer electrolyte fuel cells. Electrochim. Acta 109, 365–369 (2013).
Kuai, C. et al. Phase segregation reversibility in mixed-metal hydroxide water oxidation catalysts. Nat. Catal. 3, 743–753 (2020).
Yang, Y. et al. Quantification of heterogeneous degradation in Li‐ion batteries. Adv. Energy Mater. 9, 1900674 (2019).
Li, J. et al. Dynamics of particle network in composite battery cathodes. Science 376, 517–521 (2022).
Jang, D. H. & Oh, S. M. Electrolyte effects on spinel dissolution and cathodic capacity losses in 4 V Li/LixMn2O4 rechargeable cells. J. Electrochem. Soc. 144, 3342 (1997).
Sarapuu, A., Hussain, S., Kasikov, A., Pollet, B. G. & Tammeveski, K. Electroreduction of oxygen on Nafion®-coated thin platinum films in acid media. J. Electroanal. Chem. 848, 113292 (2019).
Yang, C. et al. A novel approach to fabricate membrane electrode assembly by directly coating the Nafion ionomer on catalyst layers for proton-exchange membrane fuel cells. ACS Sustain. Chem. Eng. 8, 9803–9812 (2020).
Sharma, P. P. & Kim, D. A facile and sustainable enhancement of anti-oxidation stability of Nafion membrane. Membranes 12, 521 (2022).
Acknowledgements
The work was supported by the National Science Foundation under CBET 1912885 (F.L.). This research used resources of the Advanced Photon Source, US Department of Energy (DOE), Office of Science User Facility, operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. The use of the Stanford Synchrotron Radiation Lightsource (SSRL), SLAC National Accelerator Laboratory, was supported by the US DOE, Office of Science, Office of Basic Energy Sciences, under contract no. DE-AC02-76SF00515. This research used the electron microscopy resources of the Center for Functional Nanomaterials (CFN), US DOE, Office of Science User Facility, at Brookhaven National Laboratory under contract no. DE-SC0012704. Y.Z. and F.L. thank the beamline scientist R. Davis at SSRL for help with the hard XAS measurements.
Author information
Authors and Affiliations
Contributions
F.L. conceived and led the project. F.L. and Y.Z. designed the experiments. Y.Z. performed the materials synthesis, electrochemical tests, synchrotron measurements and data analysis. A.H. and D.X. assisted with the XFM measurements. D.X. assisted with the hard XAS measurements. S.S. and D.N. performed the soft XAS measurements. S.H. performed the TEM measurements and data analysis. F.M.M. participated in the data discussion. R.B.M. participated in the experimental design and data discussion. L.L. assisted with the XFM measurements and participated in the discussion and data analysis. Y.Z. and F.L. wrote the manuscript with inputs from all the co-authors. All the authors approved the final draft of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Yong-Mook Kang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Cell configuration for the in-situ X-ray fluorescence microscopy (XFM) experiment.
The three-electrode cell used for the in situ and operando X-ray fluorescence microscopy (XFM) experiment. The X-rays hit the electrode from the side of the current collector (carbon paper).
Extended Data Fig. 2 XFM images of LMO particles at different states of charge.
The XFM images at the single-particle level of LMO electrodes at different states of charge during the first cycle, showing a heterogeneous Mn D/R behaviours.
Extended Data Fig. 3 Mn deposition on the counter electrode.
(a) The photo of a graphite paper used as the counter electrode during electrochemical cycling, where the arrows indicate the original and cycled regions; (b) XFM image of the region labelled by blue frame in (a), showing a much higher Mn concentration in the cycled region and confirming that the dissolved Mn species can deposit on the counter electrode surface. Note that XFM is extremely elementally sensitive, the minor Mn concentration in the pristine graphite originates from the impurity.
Extended Data Fig. 4 Mn D/R dynamics of Mn3O4 held at 1.2 V.
The Mn D/R dynamics of Mn3O4 held at 1.2 V. The CA protocol is applied for 21 minutes to keep the voltage at 1.2 V and monitor the Mn concentration variation. Each XFM image takes 7 minutes.
Extended Data Fig. 5 Mn D/R dynamics of MnO2 held at 1.2 V.
The Mn D/R dynamics of MnO2 held at 1.2 V. The CA protocol is applied for 21 minutes to keep the voltage at 1.2 V and monitor the Mn concentration variation. Each XFM image takes 7 minutes.
Extended Data Fig. 6 Mn D/R dynamics of LMO cycled in 2 M LiTFSI-TEGDME electrolyte.
(a) The XFM images of Mn concentration in the LMO electrode during electrochemical cycling in 2 M LiTFSI in tetraethylene glycol dimethyl ether (TEG-DME) solvents; (b) the corresponding Mn D/R behaviours during the first cycle.
Extended Data Fig. 7 Trapped Mn ions in the Nafion membrane.
(a) The photos of casted Nafion membrane; (b) the EDS mapping of Mn element for pristine and soaked Nafion membranes. The Mn signal in pristine membranes is caused by the uncertainty from the instrument, the actual Mn atomic ratio is zero.
Supplementary information
Supplementary Information
Supplementary Figs. 1–18 and Tables 1–5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Hu, A., Xia, D. et al. Operando characterization and regulation of metal dissolution and redeposition dynamics near battery electrode surface. Nat. Nanotechnol. 18, 790–797 (2023). https://doi.org/10.1038/s41565-023-01367-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-023-01367-6
This article is cited by
-
Progress on Transition Metal Ions Dissolution Suppression Strategies in Prussian Blue Analogs for Aqueous Sodium-/Potassium-Ion Batteries
Nano-Micro Letters (2024)
-
Getting to the bottom of transition metal dissolution
Nature Nanotechnology (2023)