Abstract
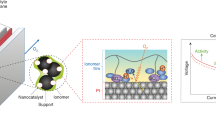
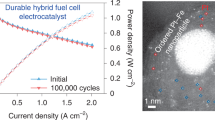
The proton exchange membrane fuel cell (PEMFC) as an attractive clean power source can promise a carbon-neutral future, but the widespread adoption of PEMFCs requires a substantial reduction in the usage of the costly platinum group metal (PGM) catalysts. Ultrafine nanocatalysts are essential to provide sufficient catalytic sites at a reduced PGM loading, but are fundamentally less stable and prone to substantial size growth in long-term operations. Here we report the design of a graphene-nanopocket-encaged platinum cobalt (PtCo@Gnp) nanocatalyst with good electrochemical accessibility and exceptional durability under a demanding ultralow PGM loading (0.070 mgPGM cm–2) due to the non-contacting enclosure of graphene nanopockets. The PtCo@Gnp delivers a state-of-the-art mass activity of 1.21 A mgPGM–1, a rated power of 13.2 W mgPGM–1 and a mass activity retention of 73% after an accelerated durability test. With the greatly improved rated power and durability, we project a 6.8 gPGM loading for a 90 kW PEMFC vehicle, which approaches that used in a typical catalytic converter.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data that support the findings of this study are available in the main text, figures and Supplementary Information files. Further data enquiries can be addressed to the corresponding authors upon reasonable request.
References
Debe, M. K. Electrocatalyst approaches and challenges for automotive fuel cells. Nature 486, 43–51 (2012).
Yarlagadda, V. et al. Boosting fuel cell performance with accessible carbon mesopores. ACS Energy Lett. 3, 618–621 (2018).
Tollefson, J. Worth its weight in platinum. Nature 450, 334–335 (2007).
Bossi, T. & Gediga, J. The environmental profile of platinum group metals. Johnson Matthey Technol. Rev. 61, 111–121 (2017).
James, B. D., Huya-Kouadio, J. M., Houchins, C. & DeSantis, D. A. Mass Production Cost Estimation of Direct H2 PEM Fuel Cell Systems for Transportation Applications: 2018 Update (US DOE, 2018).
Pollet, B. G., Kocha, S. S. & Staffell, I. Current status of automotive fuel cells for sustainable transport. Curr. Opin. Electrochem. 16, 90–95 (2019).
Gröger, O., Gasteiger, H. A. & Suchsland, J.-P. Electromobility: batteries or fuel cells? J. Electrochem. Soc. 162, A2605–A2622 (2015).
Hao, H. et al. Securing platinum-group metals for transport low-carbon transition. One Earth 1, 117–125 (2019).
Kongkanand, A. & Mathias, M. F. The priority and challenge of high-power performance of low-platinum proton-exchange membrane fuel cells. J. Phys. Chem. Lett. 7, 1127–1137 (2016).
Li, M. et al. Ultrafine jagged platinum nanowires enable ultrahigh mass activity for the oxygen reduction reaction. Science 354, 1414–1419 (2016).
Escudero-Escribano, M. et al. Tuning the activity of Pt alloy electrocatalysts by means of the lanthanide contraction. Science 352, 73–76 (2016).
Chen, C. et al. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces. Science 343, 1339–1343 (2014).
Greeley, J. et al. Alloys of platinum and early transition metals as oxygen reduction electrocatalysts. Nat. Chem. 1, 552–556 (2009).
Zhang, L. et al. Platinum-based nanocages with subnanometer-thick walls and well-defined, controllable facets. Science 349, 412–416 (2015).
Cui, C., Gan, L., Heggen, M., Rudi, S. & Strasser, P. Compositional segregation in shaped Pt alloy nanoparticles and their structural behaviour during electrocatalysis. Nat. Mater. 12, 765–771 (2013).
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: insights into materials design. Science 355, eaad4998 (2017).
Li, W., Chen, Z., Xu, L. & Yan, Y. A solution-phase synthesis method to highly active Pt-Co/C electrocatalysts for proton exchange membrane fuel cell. J. Power Sources 195, 2534–2540 (2010).
Zhang, Z. et al. One-pot synthesis of highly anisotropic five-fold-twinned PtCu nanoframes used as a bifunctional electrocatalyst for oxygen reduction and methanol oxidation. Adv. Mater. 28, 8712–8717 (2016).
Wang, X. X. et al. Ordered Pt3Co intermetallic nanoparticles derived from metal–organic frameworks for oxygen reduction. Nano Lett. 18, 4163–4171 (2018).
Huang, L., Zheng, C. Y., Shen, B. & Mirkin, C. A. High-index-facet metal–alloy nanoparticles as fuel cell electrocatalysts. Adv. Mater. 32, 2002849 (2020).
Ott, S. et al. Ionomer distribution control in porous carbon-supported catalyst layers for high-power and low Pt-loaded proton exchange membrane fuel cells. Nat. Mater. 19, 77–85 (2019).
Qiao, Z. et al. 3D porous graphitic nanocarbon for enhancing the performance and durability of Pt catalysts: a balance between graphitization and hierarchical porosity. Energy Environ. Sci. 12, 2830–2841 (2019).
Wang, L. et al. Tunable intrinsic strain in two-dimensional transition metal electrocatalysts. Science 363, 870–874 (2019).
Wang, C. et al. Synthesis of homogeneous Pt-bimetallic nanoparticles as highly efficient electrocatalysts. ACS Catal. 1, 1355–1359 (2011).
He, D. S. et al. Ultrathin icosahedral Pt-enriched nanocage with excellent oxygen reduction reaction activity. J. Am. Chem. Soc. 138, 1494–1497 (2016).
Pizzutilo, E. et al. The space confinement approach using hollow graphitic spheres to unveil activity and stability of Pt–Co nanocatalysts for PEMFC. Adv. Energy Mater. 7, 1700835 (2017).
Mezzavilla, S. et al. Structure–activity–stability relationships for space-confined PtxNiy nanoparticles in the oxygen reduction reaction. ACS Catal. 6, 8058–8068 (2016).
DOE Technical Targets for Polymer Electrolyte Membrane Fuel Cell Components https://energy.gov/eere/fuelcells/doe-technical-targets-polymer-electrolyte-membrane-fuel-cell-components (US DOE, 2016).
Kodama, K., Nagai, T., Kuwaki, A., Jinnouchi, R. & Morimoto, Y. Challenges in applying highly active Pt-based nanostructured catalysts for oxygen reduction reactions to fuel cell vehicles. Nat. Nanotechnol. 16, 140–147 (2021).
Weber, A. Z. & Kusoglu, A. Unexplained transport resistances for low-loaded fuel-cell catalyst layers. J. Mater. Chem. A 2, 17207–17211 (2014).
Holby, E. F., Sheng, W., Shao-Horn, Y. & Morgan, D. Pt nanoparticle stability in PEM fuel cells: influence of particle size distribution and crossover hydrogen. Energy Environ. Sci. 2, 865–871 (2009).
Borup, R. L. et al. Recent developments in catalyst-related PEM fuel cell durability. Curr. Opin. Electrochem. 21, 192–200 (2020).
Tang, L., Li, X., Cammarata, R. C., Friesen, C. & Sieradzki, K. Electrochemical stability of elemental metal nanoparticles. J. Am. Chem. Soc. 132, 11722–11726 (2010).
Tang, L. et al. Electrochemical stability of nanometer-scale Pt particles in acidic environments. J. Am. Chem. Soc. 132, 596–600 (2010).
Du, L. et al. Low-PGM and PGM-free catalysts for proton exchange membrane fuel cells: stability challenges and material solutions. Adv. Mater. 33, 1908232 (2021).
Han, B. et al. Record activity and stability of dealloyed bimetallic catalysts for proton exchange membrane fuel cells. Energy Environ. Sci. 8, 258–266 (2015).
Braaten, J. P., Xu, X., Cai, Y., Kongkanand, A. & Litster, S. Contaminant cation effect on oxygen transport through the ionomers of polymer electrolyte membrane fuel cells. J. Electrochem. Soc. 166, F1337–F1343 (2019).
Sulek, M., Adams, J., Kaberline, S., Ricketts, M. & Waldecker, J. R. In situ metal ion contamination and the effects on proton exchange membrane fuel cell performance. J. Power Sources 196, 8967–8972 (2011).
Hoene, J. V., Charles, R. G. & Hickam, W. M. Thermal decomposition of metal acetylacetonates: mass spectrometer studies. J. Phys. Chem. 62, 1098–1101 (1958).
Grimm, S. et al. Gas-phase aluminium acetylacetonate decomposition: revision of the current mechanism by VUV synchrotron radiation. Phys. Chem. Chem. Phys. 23, 15059–15075 (2021).
Fei, L.-f et al. Direct observation of carbon nanostructure growth at liquid–solid interfaces. Chem. Commun. 50, 826–828 (2014).
Picher, M., Lin, P. A., Gomez-Ballesteros, J. L., Balbuena, P. B. & Sharma, R. Nucleation of graphene and its conversion to single-walled carbon nanotubes. Nano Lett. 14, 6104–6108 (2014).
Fan, H. et al. Dynamic state and active structure of Ni–Co catalyst in carbon nanofiber growth revealed by in situ transmission electron microscopy. ACS Nano 15, 17895–17906 (2021).
Zhao, Z. et al. Tailoring a three-phase microenvironment for high-performance oxygen reduction reaction in proton exchange membrane fuel cells. Matter 3, 1774–1790 (2020).
Cullen, D. A. et al. New roads and challenges for fuel cells in heavy-duty transportation. Nat. Energy 6, 462–474 (2021).
Chong, L. et al. Ultralow-loading platinum–cobalt fuel cell catalysts derived from imidazolate frameworks. Science 362, 1276–1281 (2018).
Jia, Q. et al. Improved oxygen reduction activity and durability of dealloyed PtCox catalysts for proton exchange membrane fuel cells: strain, ligand, and particle size effects. ACS Catal. 5, 176–186 (2015).
Li, J. et al. Hard-magnet L10-CoPt nanoparticles advance fuel cell catalysis. Joule 3, 124–135 (2019).
Papadias, D. D. et al. Durability of Pt–Co alloy polymer electrolyte fuel cell cathode catalysts under accelerated stress tests. J. Electrochem. Soc. 165, F3166–F3177 (2018).
Slack, J. J. et al. Nanofiber fuel cell MEAs with a PtCo/C cathode. J. Electrochem. Soc. 166, F3202–F3209 (2019).
Fuel Cell Technologies Office Multi-year Research, Development, and Demonstration Plan https://www.energy.gov/eere/fuelcells/downloads/fuel-cell-technologies-office-multi-year-research-development-and-22 (US DOE, 2017).
Zhao, Z. et al. Pt-based nanocrystal for electrocatalytic oxygen reduction. Adv. Mater. 31, 1808115 (2019).
Kleen, G. & Padgett, E. Durability-Adjusted Fuel Cell System Cost (US DOE, 2021).
Baker, D. R., Caulk, D. A., Neyerlin, K. C. & Murphy, M. W. Measurement of oxygen transport resistance in PEM fuel cells by limiting current methods. J. Electrochem. Soc. 156, B991–B1003 (2009).
Garsany, Y., Atkinson, R. W., Gould, B. D. & Swider-Lyons, K. E. High power, low-Pt membrane electrode assemblies for proton exchange membrane fuel cells. J. Power Sources 408, 38–45 (2018).
Papageorgopoulos, D. Fuel Cell R&D Overview (US DOE, 2019).
Kongkanand, A. Highly Accessible Catalysts for Durable High Power Performance (US DOE, 2020).
Stariha, S. et al. Recent advances in catalyst accelerated stress tests for polymer electrolyte membrane fuel cells. J. Electrochem. Soc. 165, F492–F501 (2018).
Zhao, Z. et al. Tailoring a three-phase microenvironment for high-performance oxygen reduction reaction in proton exchange membrane fuel cells. Matter 3, 1774–1790 (2020).
Huang, P. Y. et al. Grains and grain boundaries in single-layer graphene atomic patchwork quilts. Nature 469, 389–392 (2011).
Stariha, S. et al. Recent advances in catalyst accelerated stress tests for polymer electrolyte membrane fuel cells. J. Electrochem. Soc. 165, F492–F501 (2018).
Garrick, T. R., Moylan, T. E., Carpenter, M. K. & Kongkanand, A. Electrochemically active surface area measurement of aged Pt alloy catalysts in PEM fuel cells by CO stripping. J. Electrochem. Soc. 164, F55–F59 (2016).
Yarlagadda, V. et al. Boosting fuel cell performance with accessible carbon mesopores. ACS Energy Lett. 3, 618–621 (2018).
Garsany, Y., Atkinson, R. W., Gould, B. D. & Swider-Lyons, K. E. High power, low-Pt membrane electrode assemblies for proton exchange membrane fuel cells. J. Power Sources 408, 38–45 (2018).
Baker, D. R., Caulk, D. A., Neyerlin, K. C. & Murphy, M. W. Measurement of oxygen transport resistance in PEM fuel cells by limiting current methods. J. Electrochem. Soc. 156, B991–B1003 (2009).
Acknowledgements
Y.H., X.D., Z.Z., Z.L., A.Z., W.X. and B.P. acknowledge the Office of Naval Research (ONR) grant N00014-18-1-2155. Y.H., Z.Z., Z.L. and A.Z. acknowledge the Defense University Research Instrumentation Program (DURIP) grant N00014-18-1-2271. X.Y. and X.P. acknowledge the DOE, Office of Basic Energy Sciences, Division of Materials Sciences and Engineering grant DE-SC0014430. We acknowledge the use of facilities and instrumentation at the UC Irvine Materials Research Institute (IMRI), which is supported in part by the National Science Foundation through the UC Irvine Materials Research Science and Engineering Center (DMR-2011967). We also acknowledge assistance in additional electron microscopy characterizations by C. Wang and L. Han (UC Irvine). C. Wang, L. Han and H.L.X. acknowledge National Science Foundation grant CHE-1900401 and the TEM resources of the Center for Functional Nanomaterials (CFN), which is a US DOE Office of Science User Facility at Brookhaven National Laboratory under contract no. DE-SC0012704.
Author information
Authors and Affiliations
Contributions
For the experimental design and execution, the synthesis of the electrocatalysts, structural characterization, MEA fabrication and electrochemical testing were carried out by Z.Z., Z.L., A.Z., W.X. and B.P., the S/TEM and EDS characterizations by X.Y. and Z.Z. Supervision was carried out by Y.H. (conceptualization, project design, syntheses and evaluation of the catalysts), X.P. (STEM and EELS) and H.L.X. (additional EM studies). The original draft was written by Z.Z., X.D. and Y.H., and edited by Z.Z., Z.L., X.D. and Y.H.
Corresponding author
Ethics declarations
Competing interests
Patent application filed on the catalyst development as US Provisional Application no. 63/222,293 by the UCLA inventors: Y.H., X.D., Z.Z. and Z.L.; the remaining authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Yadong Yin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–10 and Tables 1–4.
Rights and permissions
About this article
Cite this article
Zhao, Z., Liu, Z., Zhang, A. et al. Graphene-nanopocket-encaged PtCo nanocatalysts for highly durable fuel cell operation under demanding ultralow-Pt-loading conditions. Nat. Nanotechnol. 17, 968–975 (2022). https://doi.org/10.1038/s41565-022-01170-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-022-01170-9
This article is cited by
-
Unveiling the nature of Pt-induced anti-deactivation of Ru for alkaline hydrogen oxidation reaction
Nature Communications (2024)
-
Electrocatalytic hydrogenation of acetonitrile to ethylamine in acid
Nature Communications (2024)
-
Recent progress of Pt-based oxygen reduction reaction catalysts for proton exchange membrane fuel cells
Rare Metals (2024)
-
Constructing ultrafine Pt nanoparticles anchored on N-doped porous carbon nanofibers for efficient and stable oxygen reduction reaction
Science China Materials (2023)
-
PtNi-W/C with Atomically Dispersed Tungsten Sites Toward Boosted ORR in Proton Exchange Membrane Fuel Cell Devices
Nano-Micro Letters (2023)