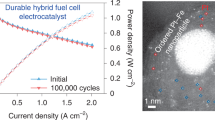
Abstract
The past 30 years have seen progress in the development of Pt-based nanocatalysts for the oxygen reduction reaction, and some are now in production on a commercial basis and used for polymer electrolyte fuel cells (PEFCs) for automotives and other applications. Further improvements in catalytic activity are required for wider uptake of PEFCs, however. In laboratories, researchers have developed various catalysts that have much higher activities than commercial ones, and these state-of-the-art catalysts have potential to improve energy conversion efficiencies and reduce the usage of platinum in PEFCs. There are several technical issues that must be solved before they can be applied in fuel cell vehicles, which require a high power density and practical durability, as well as high efficiency. In this Review, the development history of Pt-based nanocatalysts and recent analytical studies are summarized to identify the origin of these technical issues. Promising strategies for overcoming those issues are also discussed.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Yoshida, T. & Kojima, K. Toyota MIRAI fuel cell vehicle and progress toward a future hydrogen society. Interface 24, 45–49 (2015).
Lohse-Busch, H. et al. Automotive fuel cell stack and system efficiency and fuel consumption based on vehicle testing on a chassis dynamometer at minus 18 °C to positive 35 °C temperatures. Int. J. Hydrogen Energy 45, 861–872 (2020).
Kongkanand, A. & Mathias, M. F. The priority and challenge of high-power performance of low-platinum proton-exchange membrane fuel cells. J. Phys. Chem. Lett. 7, 1127–1137 (2016).
Thompson, S. T. et al. Direct hydrogen fuel cell electric vehicle cost analysis: system and high-volume manufacturing description, validation, and outlook. J. Power Sources 399, 304–313 (2018).
Wilson, M. S. & Gottesfeld, S. Thin-film catalyst layers for polymer electrolyte fuel-cell electrodes. J. Appl. Electrochem. 22, 1–7 (1992).
Mukerjee, S., Srinivasan, S., Soriaga, M. P. & McBreen, J. Role of structural and electronic properties of Pt and Pt alloys on electrocatalysis of oxygen reduction: an in situ XANES and EXAFS investigation. J. Electrochem. Soc. 142, 1409–1422 (1995).
Banham, D. et al. Critical advancements in achieving high power and stable nonprecious metal catalyst-based MEAs for real-world proton exchange membrane fuel cell applications. Sci. Adv. 4, eaar7180 (2018).
Chung, H. T. et al. Direct atomic-level insight into the active sites of a high-performance PGM-free ORR catalyst. Science 357, 479–483 (2017).
van der Vliet, D. F. et al. Unique electrochemical adsorption properties of Pt-skin surfaces. Angew. Chem. Int. Ed. 51, 3139–3142 (2012).
Stamenkovic, V. R. et al. Improved oxygen reduction activity on Pt3Ni(111) via increased surface site availability. Science 315, 493–497 (2007).
Stephens, I. E. L. et al. Tuning the activity of Pt(111) for oxygen electroreduction by subsurface alloying. J. Am. Chem. Soc. 133, 5485–5491 (2011).
Asano, M., Kawamura, R., Sasakawa, R., Todoroki, N. & Wadayama, T. Oxygen reduction reaction activity for strain-controlled Pt-based model alloy catalysts: surface strains and direct electronic effects induced by alloying elements. ACS Catal. 6, 5285–5289 (2016).
Wakisaka, M. et al. Unprecedented dependence of the oxygen reduction activity on Co content at Pt skin/Pt-Co(111) single crystal electrodes. Electrochem. Commun. 67, 47–50 (2016).
Norskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Viswanathan, V., Hansen, H. A., Rossmeisl, J. & Norskov, J. K. Universality in oxygen reduction electrocatalysis on metal surfaces. ACS Catal. 2, 1654–1660 (2012).
Jinnouchi, R., Kodama, K., Hatanaka, T. & Morimoto, Y. First principles based mean field model for oxygen reduction reaction. Phys. Chem. Chem. Phys. 13, 21070–21083 (2011).
Kulkarni, A., Siahrostami, S., Patel, A. & Norskov, J. K. Understanding catalytic activity trends in the oxygen reduction reaction. Chem. Rev. 118, 2302–2312 (2018).
Zhang, J. et al. Platinum monolayer electrocatalysts for O2 reduction: Pt monolayer on Pd(111) and on carbon-supported Pd nanoparticles. J. Phys. Chem. B 108, 10955–10964 (2004).
Aoki, N. et al. Electrochemical properties and single cell performance of Pd core-Pt shell structured catalyst synthesized by a simple direct displacement reaction. J. Electrochem. Soc. 167, 044513 (2020).
Kuttiyiel, K. A. et al. Pt monolayer on Au-stabilized PdNi core-shell nanoparticles for oxygen reduction reaction. Electrochim. Acta 110, 267–272 (2013).
Zhang, J., Yang, H. Z., Fang, J. Y. & Zou, S. Z. Synthesis and oxygen reduction activity of shape-controlled Pt3Ni nanopolyhedra. Nano Lett. 10, 638–644 (2010).
Choi, S. I. et al. Synthesis and characterization of 9 nm Pt-Ni octahedra with a record high activity of 3.3 A/mg(Pt) for the oxygen reduction reaction. Nano Lett. 13, 3420–3425 (2013).
Huang, X. Q. et al. High-performance transition metal-doped Pt3Ni octahedra for oxygen reduction reaction. Science 348, 1230–1234 (2015).
Niu, G. D. et al. Synthesis of Pt-Ni octahedra in continuous-flow droplet reactors for the scalable production of highly active catalysts toward oxygen reduction. Nano Lett. 16, 3850–3857 (2016).
Chen, C. et al. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces. Science 343, 1339–1343 (2014).
Li, M. F. et al. Ultrafine jagged platinum nanowires enable ultrahigh mass activity for the oxygen reduction reaction. Science 354, 1414–1419 (2016).
Bu, L. Z. et al. Biaxially strained PtPb/Pt core/shell nanoplate boosts oxygen reduction catalysis. Science 354, 1410–1414 (2016).
Tian, X. L. et al. Engineering bunched Pt-Ni alloy nanocages for efficient oxygen reduction in practical fuel cells. Science 366, 850–856 (2019).
Zhang, C. L., Hwang, S. Y., Trout, A. & Peng, Z. M. Solid-state chemistry-enabled scalable production of octahedral Pt-Ni alloy electrocatalyst for oxygen reduction reaction. J. Am. Chem. Soc. 136, 7805–7808 (2014).
Kuzume, A., Herrero, E. & Feliu, J. M. Oxygen reduction on stepped platinum surfaces in acidic media. J. Electroanal. Chem. 599, 333–343 (2007).
Hoshi, N., Nakamura, M. & Hitotsuyanagi, A. Active sites for the oxygen reduction reaction on the high index planes of Pt. Electrochim. Acta 112, 899–904 (2013).
Bandarenka, A. S., Hansen, H. A., Rossmeisl, J. & Stephens, I. E. L. Elucidating the activity of stepped Pt single crystals for oxygen reduction. Phys. Chem. Chem. Phys. 16, 13625–13629 (2014).
Jinnouchi, R., Kodama, K., Nagoya, A. & Morimoto, Y. Simulated volcano plot of oxygen reduction reaction on stepped Pt surfaces. Electrochim. Acta 230, 470–478 (2017).
Calle-Vallejo, F. et al. Why conclusions from platinum model surfaces do not necessarily lead to enhanced nanoparticle catalysts for the oxygen reduction reaction. Chem. Sci. 8, 2283–2289 (2017).
Garlyyev, B. et al. Optimizing the size of platinum nanoparticles for enhanced mass activity in the electrochemical oxygen reduction reaction. Angew. Chem. Int. Ed. 58, 9596–9600 (2019).
Strmcnik, D. et al. Enhanced electrocatalysis of the oxygen reduction reaction based on patterning of platinum surfaces with cyanide. Nat. Chem. 2, 880–885 (2010).
Subbaraman, R., Strmcnik, D., Stamenkovic, V. & Markovic, N. M. Three phase interfaces at electrified metal-solid electrolyte systems 1. Study of the Pt(hkl)-Nafion interface. J. Phys. Chem. C 114, 8414–8422 (2010).
Shinozaki, K., Morimoto, Y., Pivovar, B. S. & Kocha, S. S. Suppression of oxygen reduction reaction activity on Pt-based electrocatalysts from ionomer incorporation. J. Power Sources 325, 745–751 (2016).
Kodama, K. et al. Effect of the side-chain structure of perfluoro-sulfonic acid ionomers on the oxygen reduction reaction on the surface of Pt. ACS Catal. 8, 694–700 (2018).
Takeshita, T., Kamitaka, Y., Shinozaki, K., Kodama, K. & Morimoto, Y. Evaluation of ionomer coverage on Pt catalysts in polymer electrolyte membrane fuel cells by CO stripping voltammetry and its effect on oxygen reduction reaction activity. J. Electroanal. Chem. 871, 114250 (2020).
Gasteiger, H. A., Kocha, S. S., Sompalli, B. & Wagner, F. T. Activity benchmarks and requirements for Pt, Pt-alloy, and non-Pt oxygen reduction catalysts for PEMFCs. Appl. Catal. B 56, 9–35 (2005).
Greszler, T. A., Caulk, D. & Sinha, P. The impact of platinum loading on oxygen transport resistance. J. Electrochem. Soc. 159, F831–F840 (2012).
Ohma, A. et al. Analysis of proton exchange membrane fuel cell catalyst layers for reduction of platinum loading at Nissan. Electrochim. Acta 56, 10832–10841 (2011).
Nonoyama, N., Okazaki, S., Weber, A. Z., Ikogi, Y. & Yoshida, T. Analysis of oxygen-transport diffusion resistance in proton-exchange-membrane fuel cells. J. Electrochem. Soc. 158, B416–B423 (2011).
Owejan, J. P., Owejan, J. E. & Gu, W. B. Impact of platinum loading and catalyst layer structure on PEMFC performance. J. Electrochem. Soc. 160, F824–F833 (2013).
Schuler, T. et al. Fuel-cell catalyst-layer resistance via hydrogen limiting-current measurements. J. Electrochem. Soc. 166, F3020–F3031 (2019).
Suzuki, T., Kudo, K. & Morimoto, Y. Model for investigation of oxygen transport limitation in a polymer electrolyte fuel cell. J. Power Sources 222, 379–389 (2013).
Kudo, K., Jinnouchi, R. & Morimoto, Y. Humidity and temperature dependences of oxygen transport resistance of Nafion thin film on platinum electrode. Electrochim. Acta 209, 682–690 (2016).
Jinnouchi, R., Kudo, K., Kitano, N. & Morimoto, Y. Molecular dynamics simulations on O2 permeation through Nafion ionomer on platinum surface. Electrochim. Acta 188, 767–776 (2016).
Karan, K. Interesting facets of surface, interfacial, and bulk characteristics of perfluorinated ionomer films. Langmuir 35, 13489–13520 (2019).
Rolfi, A., Oldani, C., Merlo, L., Facchi, D. & Ruffo, R. New perfluorinated ionomer with improved oxygen permeability for application in cathode polymeric electrolyte membrane fuel cell. J. Power Sources 396, 95–101 (2018).
Katzenberg, A. et al. Highly permeable perfluorinated sulfonic acid ionomers for improved electrochemical devices: insights into structure-property relationships. J. Am. Chem. Soc. 142, 3742–3752 (2020).
Kamitaka, Y., Takeshita, T. & Morimoto, Y. MgO-templated mesoporous carbon as a catalyst support for polymer electrolyte fuel cells. Catalysts 8, 230 (2018).
Ott, S. et al. Ionomer distribution control in porous carbon-supported catalyst layers for high-power and low Pt-loaded proton exchange membrane fuel cells. Nat. Mater. 19, 77–85 (2020).
Yarlagadda, V. et al. Boosting fuel cell performance with accessible carbon mesopores. ACS Energy Lett. 3, 618–621 (2018).
Liu, W. J., Wu, B. L. & Cha, C. S. Surface diffusion and the spillover of H-adatoms and oxygen-containing surface species on the surface of carbon black and Pt/C porous electrodes. J. Electroanal. Chem. 476, 101–108 (1999).
Chan, K. & Eikerling, M. A pore-scale model of oxygen reduction in ionomer-free catalyst layers of PEFCs. J. Electrochem. Soc. 158, B18–B28 (2011).
Liu, J. J. & Zenyuk, I. Proton transport in ionomer-free regions of polymer electrolyte fuel cells and implications for oxygen reduction reaction. Curr. Opin. Electrochem. 12, 202–208 (2018).
Hu, L. M., Zhang, M. X., Babu, S. K., Kongkanand, A. & Litster, S. Ionic conductivity over metal/water interfaces in ionomer-free fuel cell electrodes. ChemElectroChem 6, 2659–2666 (2019).
Darling, R. M. & Meyers, J. P. Kinetic model of platinum dissolution in PEMFCs. J. Electrochem. Soc. 150, A1523–A1527 (2003).
Topalov, A. A. et al. Dissolution of platinum: limits for the deployment of electrochemical energy conversion? Angew. Chem. Int. Ed. 51, 12613–12615 (2012).
Pizzutilo, E. et al. On the need of improved accelerated degradation protocols (ADPs): examination of platinum dissolution and carbon corrosion in half-cell tests. J. Electrochem. Soc. 163, F1510–F1514 (2016).
Lopes, P. P. et al. Eliminating dissolution of platinum-based electrocatalysts at the atomic scale. Nat. Mater. 19, 1207–1214 (2020).
Wilson, M. S., Garzon, F. H., Sickafus, K. E. & Gottesfeld, S. Surface-area loss of supported platinum in polymer electrolyte fuel-cells. J. Electrochem. Soc. 140, 2872–2877 (1993).
Ferreira, P. J. et al. Instability of Pt/C electrocatalysts in proton exchange membrane fuel cells: a mechanistic investigation. J. Electrochem. Soc. 152, A2256–A2271 (2005).
Holby, E. F., Sheng, W. C., Shao-Horn, Y. & Morgan, D. Pt nanoparticle stability in PEM fuel cells: influence of particle size distribution and crossover hydrogen. Energy Environ. Sci. 2, 865–871 (2009).
Jinnouchi, R., Toyoda, E., Hatanaka, T. & Morimoto, Y. First principles calculations on site-dependent dissolution potentials of supported and unsupported Pt particles. J. Phys. Chem. C 114, 17557–17568 (2010).
Chattot, R. et al. Surface distortion as a unifying concept and descriptor in oxygen reduction reaction electrocatalysis. Nat. Mater. 17, 827–833 (2018).
Braaten, J. P., Xu, X. M., Cai, Y., Kongkanand, A. & Litster, S. Contaminant cation effect on oxygen transport through the ionomers of polymer electrolyte membrane fuel cells. J. Electrochem. Soc. 166, F1337–F1343 (2019).
Borup, R. L. et al. Recent developments in catalyst-related PEM fuel cell durability. Curr. Opin. Electrochem. 21, 192–200 (2020).
Wang, D. L. et al. Structurally ordered intermetallic platinum-cobalt core-shell nanoparticles with enhanced activity and stability as oxygen reduction electrocatalysts. Nat. Mater. 12, 81–87 (2013).
Beermann, V. et al. Rh-Doped Pt-Ni octahedral nanoparticles: understanding the correlation between elemental distribution, oxygen reduction reaction, and shape stability. Nano Lett. 16, 1719–1725 (2016).
Zhao, X. et al. High-performance nitrogen-doped intermetallic PtNi catalyst for the oxygen reduction reaction. ACS Catal. 10, 10637–10645 (2020).
Zhao, X. et al. Simultaneous improvements in performance and durability of an octahedral PtNix/C electrocatalyst for next-generation fuel cells by continuous, compressive, and concave Pt skin layers. ACS Catal. 7, 4642–4654 (2017).
Takenaka, S. et al. Improvement in the durability of pt electrocatalysts by coverage with silica layers. J. Phys. Chem. C 111, 15133–15136 (2007).
Wen, Z. H., Liu, J. & Li, J. H. Core/shell Pt/C nanoparticles embedded in mesoporous carbon as a methanol-tolerant cathode catalyst in direct methanol fuel cells. Adv. Mater. 20, 743–747 (2008).
Chung, D. Y. et al. Highly durable and active PtFe nanocatalyst for electrochemical oxygen reduction reaction. J. Am. Chem. Soc. 137, 15478–15485 (2015).
Yamada, H., Kato, H. & Kodama, K. Cell performance and durability of Pt/C cathode catalyst covered by dopamine derived carbon thin layer for polymer electrolyte fuel cells. J. Electrochem. Soc. 167, 084508 (2020).
Newton, J. E., Preece, J. A., Rees, N. V. & Horswell, S. L. Nanoparticle catalysts for proton exchange membrane fuel cells: can surfactant effects be beneficial for electrocatalysis? Phys. Chem. Chem. Phys. 16, 11435–11446 (2014).
Kodama, K., Jinnouchi, R., Takahashi, N., Murata, H. & Morimoto, Y. Activities and stabilities of Au-modified stepped-Pt single-crystal electrodes as model cathode catalysts in polymer electrolyte fuel cells. J. Am. Chem. Soc. 138, 4194–4200 (2016).
Shao, M. H., Peles, A. & Shoemaker, K. Electrocatalysis on platinum nanoparticles: particle size effect on oxygen reduction reaction activity. Nano Lett. 11, 3714–3719 (2011).
Perez-Alonso, F. J. et al. The effect of size on the oxygen electroreduction activity of mass-selected platinum nanoparticles. Angew. Chem. Int. Ed. 51, 4641–4643 (2012).
Tian, Z. Q., Jiang, S. P., Liu, Z. C. & Li, L. Polyelectrolyte-stabilized Pt nanoparticles as new electrocatalysts for low temperature fuel cells. Electrochem. Commun. 9, 1613–1618 (2007).
Zhou, Z. Y., Kang, X. W., Song, Y. & Chen, S. W. Ligand-mediated electrocatalytic activity of Pt nanoparticles for oxygen reduction reactions. J. Phys. Chem. C. 116, 10592–10598 (2012).
Chung, Y. H., Chung, D. Y., Jung, N. & Sung, Y. E. Tailoring the electronic structure of nanoelectrocatalysts induced by a surface-capping organic molecule for the oxygen reduction reaction. J. Phys. Chem. Lett. 4, 1304–1309 (2013).
Wada, N., Nakamura, M. & Hoshi, N. Structural effects on the oxygen reduction reaction on Pt single-crystal electrodes modified with melamine. Electrocatalysis 11, 275–281 (2020).
Miyabayashi, K., Nishihara, H. & Miyake, M. Platinum nanoparticles modified with alkylamine derivatives as an active and stable catalyst for oxygen reduction reaction. Langmuir 30, 2936–2942 (2014).
Yamazaki, S., Asahi, M. & Ioroi, T. Promotion of oxygen reduction on a porphyrazine-modified Pt catalyst surface. Electrochim. Acta 297, 725–734 (2019).
Ioroi, T., Siroma, Z., Fujiwara, N., Yamazaki, S. & Yasuda, K. Sub-stoichiometric titanium oxide-supported platinum electrocatalyst for polymer electrolyte fuel cells. Electrochem. Commun. 7, 183–188 (2005).
Takasaki, F. et al. Carbon-free Pt electrocatalysts supported on SnO2 for polymer electrolyte fuel cells: electrocatalytic activity and durability. J. Electrochem. Soc. 158, B1270–B1275 (2011).
Dou, M. L. et al. SnO2 nanocluster supported Pt catalyst with high stability for proton exchange membrane fuel cells. Electrochim. Acta 92, 468–473 (2013).
Fabbri, E., Rabis, A., Kotz, R. & Schmidt, T. J. Pt nanoparticles supported on Sb-doped SnO2 porous structures: developments and issues. Phys. Chem. Chem. Phys. 16, 13672–13681 (2014).
Kakinuma, K. et al. Electronic states and transport phenomena of Pt nanoparticle catalysts supported on Nb-doped SnO2 for polymer electrolyte fuel cells. ACS Appl. Mater. Interfaces 11, 34957–34963 (2019).
Beermann, V. et al. Real-time imaging of activation and degradation of carbon supported octahedral Pt-Ni alloy fuel cell catalysts at the nanoscale using in situ electrochemical liquid cell STEM. Energy Environ. Sci. 12, 2476–2485 (2019).
van der Vliet, D. F. et al. Mesostructured thin films as electrocatalysts with tunable composition and surface morphology. Nat. Mater. 11, 1051–1058 (2012).
Hernandez-Fernandez, P. et al. Mass-selected nanoparticles of PtxY as model catalysts for oxygen electroreduction. Nat. Chem. 6, 732–738 (2014).
Pan, L. J., Ott, S., Dionigi, F. & Strasser, P. Current challenges related to the deployment of shape-controlled Pt alloy oxygen reduction reaction nanocatalysts into low Pt-loaded cathode layers of proton exchange membrane fuel cells. Curr. Opin. Electrochem. 18, 61–71 (2019).
Zalitis, C., Kucernak, A., Lin, X. Q. & Sharman, J. Electrochemical measurement of intrinsic oxygen reduction reaction activity at high current densities as a function of particle size for Pt4-xCox/C (x = 0, 1, 3) catalysts. ACS Catal. 10, 4361–4376 (2020).
Inaba, M. et al. Benchmarking high surface area electrocatalysts in a gas diffusion electrode: measurement of oxygen reduction activities under realistic conditions. Energy Environ. Sci. 11, 988–994 (2018).
Rasmussen, K. D., Wenzel, H., Bangs, C., Petavratzi, E. & Liu, G. Platinum demand and potential bottlenecks in the global green transition: a dynamic material flow analysis. Environ. Sci. Technol. 53, 11541–11551 (2019).
Proietti, E. et al. Iron-based cathode catalyst with enhanced power density in polymer electrolyte membrane fuel cells. Nat. Commun. 2, 416 (2011).
Uddin, A. et al. High power density platinum group metal-free cathodes for polymer electrolyte fuel cells. ACS Appl. Mater. Interfaces 12, 2216–2224(2020).
Gittleman, C. S., Kongkanand, A., Masten, D. & Gu, W. B. Materials research and development focus areas for low cost automotive proton-exchange membrane fuel cells. Curr. Opin. Electrochem. 18, 81–89 (2019).
Mun, Y. et al. Versatile strategy for tuning ORR activity of a single Fe-N4 site by controlling electron-withdrawing/donating properties of a carbon plane. J. Am. Chem. Soc. 141, 6254–6262 (2019).
Chong, L. et al. Ultralow-loading platinum-cobalt fuel cell catalysts derived from imidazolate frameworks. Science 362, 1276–1281 (2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Nanotechnology thanks Zongping Shao and other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kodama, K., Nagai, T., Kuwaki, A. et al. Challenges in applying highly active Pt-based nanostructured catalysts for oxygen reduction reactions to fuel cell vehicles. Nat. Nanotechnol. 16, 140–147 (2021). https://doi.org/10.1038/s41565-020-00824-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-020-00824-w
This article is cited by
-
Unusual Sabatier principle on high entropy alloy catalysts for hydrogen evolution reactions
Nature Communications (2024)
-
Identifying the distinct roles of dual dopants in stabilizing the platinum-nickel nanowire catalyst for durable fuel cell
Nature Communications (2024)
-
In situ modulating coordination fields of single-atom cobalt catalyst for enhanced oxygen reduction reaction
Nature Communications (2024)
-
Machine-learning-accelerated design of high-performance platinum intermetallic nanoparticle fuel cell catalysts
Nature Communications (2024)
-
Constructing inter-diffusive PtCuNi/WO3 interface to enhance the catalytic activity and stability in oxygen reduction
Tungsten (2024)