Abstract
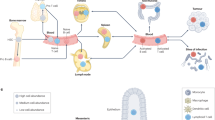
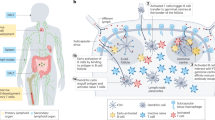
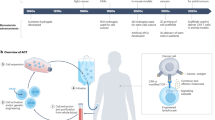
Vaccines and immunotherapies have provided enormous improvements for public health, but there are fundamental disconnects between where most studies are performed—in cell culture and animal models—and the ultimate application in humans. Engineering immune tissues and organs, such as bone marrow, thymus, lymph nodes and spleen, could be instrumental in overcoming these hurdles. Fundamentally, designed immune tissues could serve as in vitro tools to more accurately study human immune function and disease, while immune tissues engineered for implantation as next-generation vaccines or immunotherapies could enable direct, on-demand control over generation and regulation of immune function. In this Review, we discuss recent interdisciplinary strategies that are merging materials science and immunology to create engineered immune tissues in vitro and in vivo. We also highlight the hurdles facing these approaches and the need for comparison to existing clinical options, relevant animal models, and other emerging technologies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Lesokhin, A. M., Callahan, M. K., Postow, M. A. & Wolchok, J. D. On being less tolerant: enhanced cancer immunosurveillance enabled by targeting checkpoints and agonists of T cell activation. Sci. Transl. Med. 7, 280sr281 (2015).
Chen, D. S. & Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 541, 321–330 (2017).
DeFrancesco, L. CAR-T’s forge ahead, despite Juno deaths. Nat. Biotechnol. 35, 6–7 (2017).
Cadavid, D. et al. Safety and efficacy of opicinumab in acute optic neuritis (RENEW): a randomised, placebo-controlled, phase 2 trial. Lancet Neurol. 16, 189–199 (2017).
Antonelli, A. et al. Establishing human leukemia xenograft mouse models by implanting human bone marrow-like scaffold-based niches. Blood 128, 2949–2959 (2016).
Kapsenberg, M. L. Dendritic-cell control of pathogen-driven T-cell polarization. Nat. Rev. Immunol. 3, 984–993 (2003).
Loder, F. et al. B cell development in the spleen takes place in discrete steps and is determined by the quality of B cell receptor-derived signals. J. Exp. Med. 190, 75–89 (1999).
Nemazee, D. Mechanisms of central tolerance for B cells. Nat. Rev. Immunol. 17, 281–294 (2017).
Hogquist, K. A., Baldwin, T. A. & Jameson, S. C. Central tolerance: Learning self-control in the thymus. Nat. Rev. Immunol. 5, 772–782 (2005).
Takahama, Y. Journey through the thymus: stromal guides for T-cell development and selection. Nat. Rev. Immunol. 6, 127–135 (2006).
Mebius, R. E. & Kraal, G. Structure and function of the spleen. Nat. Rev. Immunol. 5, 606–616 (2005).
Butcher, E. C. & Picker, L. J. Lymphocyte homing and homeostasis. Science 272, 60–66 (1996).
Nutt, S. L., Hodgkin, P. D., Tarlinton, D. M. & Corcoran, L. M. The generation of antibody-secreting plasma cells. Nat. Rev. Immunol. 15, 160–171 (2015).
Crotty, S. Follicular helper CD4 T cells (T-FH). Annu. Rev. Immunol. 29, 621–663 (2011).
Bhatia, S. N. & Ingber, D. E. Microfluidic organs-on-chips. Nat. Biotechnol. 32, 760–772 (2014).
Takebe, T., Zhang, B. Y. & Radisic, M. Synergistic engineering: Organoids meet organs-on-a-chip. Cell Stem Cell 21, 297–300 (2017).
Sardi, M., Lubitz, A. & Giese, C. Modeling human immunity in vitro: improving artificial lymph node physiology by stromal cells. Appl. In Vitro Toxicol. 2, 143–150 (2016).
Nojima, T. et al. In-vitro derived germinal centre B cells differentially generate memory B or plasma cells in vivo. Nat. Commun. 2, 465 (2011).
Kuzin, I. et al. Long-term immunologically competent human peripheral lymphoid tissue cultures in a 3D bioreactor. Biotechnol. Bioeng. 108, 1430–1440 (2011).
Tomei, A. A., Siegert, S., Britschgi, M. R., Luther, S. A. & Swartz, M. A. Fluid flow regulates stromal cell organization and CCL21 expression in a tissue-engineered lymph node microenvironment. J. Immunol. 183, 4273–4283 (2009).
Singh, A. Biomaterials innovation for next generation ex vivo immune tissue engineering. Biomaterials 130, 104–110 (2017).
Purwada, A. et al. Ex vivo engineered immune organoids for controlled germinal center reactions. Biomaterials 63, 24–34 (2015).
Purwada, A. & Singh, A. Immuno-engineered organoids for regulating the kinetics of B-cell development and antibody production. Nat. Protoc. 12, 168–182 (2017).
Beguelin, W. et al. EZH2 enables germinal centre formation through epigenetic silencing of CDKN1A and an Rb-E2F1 feedback loop. Nat. Commun. 8, 877 (2017).
Poznansky, M. C. et al. Efficient generation of human T cells from a tissue-engineered thymic organoid. Nat. Biotechnol. 18, 729–734 (2000).
Lee, J. & Kotov, N. A. Notch ligand presenting acellular 3D microenvironments for ex vivo human hematopoietic stem-cell culture made by layer-by-layer assembly. Small 5, 1008–1013 (2009).
Pinto, S. et al. An organotypic coculture model supporting proliferation and differentiation of medullary thymic epithelial cells and promiscuous gene expression. J. Immunol. 190, 1085–1093 (2013).
Nichols, J. E. et al. In vitro analog of human bone marrow from 3D scaffolds with biomimetic inverted colloidal crystal geometry. Biomaterials 30, 1071–1079 (2009).
Cook, M. M. et al. Micromarrows-three-dimensional coculture of hematopoietic stem cells and mesenchymal stromal cells. Tissue Eng. Part C 18, 319–328 (2012).
Torisawa, Y. S. et al. Bone marrow-on-a-chip replicates hematopoietic niche physiology in vitro. Nat. Methods 11, 663–669 (2014).
Tao, Z. M. & Ghoroghchian, P. P. Microparticle, nanoparticle, and stem cell-based oxygen carriers as advanced blood substitutes. Trends Biotechnol. 32, 466–473 (2014).
Giarratana, M. C. et al. Proof of principle for transfusion of in vitro-generated red blood cells. Blood 118, 5071–5079 (2011).
Thon, J. N. et al. Platelet bioreactor-on-a-chip. Blood 124, 1857–1867 (2014).
Trakarnsanga, K. et al. An immortalized adult human erythroid line facilitates sustainable and scalable generation of functional red cells. Nat. Commun. 8, 14750 (2017).
Kim, S. K., Moon, W. K., Park, J. Y. & Jung, H. Inflammatory mimetic microfluidic chip by immobilization of cell adhesion molecules for T cell adhesion. Analyst 137, 4062–4068 (2012).
Stachowiak, A. N. & Irvine, D. J. Inverse opal hydrogel-collagen composite scaffolds as a supportive microenvironment for immune cell migration. J. Biomed. Mater. Res. 85A, 815–828 (2008).
Haessler, U., Pisano, M., Wu, M. M. & Swartz, M. A. Dendritic cell chemotaxis in 3D under defined chemokine gradients reveals differential response to ligands CCL21 and CCL19. Proc. Natl Acad. Sci. USA 108, 5614–5619 (2011).
Gopalakrishnan, N. et al. Infection and immunity on a chip: a compartmentalised microfluidic platform to monitor immune cell behaviour in real time. Lab Chip 15, 1481–1487 (2015).
Jones, C. N. et al. Microfluidic chambers for monitoring leukocyte trafficking and humanized nano-proresolving medicines interactions. Proc. Natl Acad. Sci. USA 109, 20560–20565 (2012).
Dura, B. et al. Profiling lymphocyte interactions at the single-cell level by microfluidic cell pairing. Nat. Commun. 6, 5940 (2015).
Rosa, P. M., Gopalakrishnan, N., Ibrahim, H., Haug, M. & Halaas, O. The intercell dynamics of T cells and dendritic cells in a lymph node-on-a-chip flow device. Lab Chip 16, 3728–3740 (2016).
Ross, A. E., Belanger, M. C., Woodroof, J. F. & Pompano, R. R. Spatially resolved microfluidic stimulation of lymphoid tissue ex vivo. Analyst 142, 649–659 (2017).
Hori, Y., Winans, A. M., Huang, C. C., Horrigan, E. M. & Irvine, D. J. Injectable dendritic cell-carrying alginate gels for immunization and immunotherapy. Biomaterials 29, 3671–3682 (2008).
Ali, O. A., Huebsch, N., Cao, L., Dranoff, G. & Mooney, D. J. Infection-mimicking materials to program dendritic cells in situ. Nat. Mater. 8, 151–158 (2009).
Ali, O. A., Emerich, D., Dranoff, G. & Mooney, D. J. In situ regulation of DC subsets and T cells mediates tumour regression in mice. Sci. Transl. Med. 1, 8ra19 (2009).
Kim, J. et al. Injectable, spontaneously assembling, inorganic scaffolds modulate immune cells in vivo and increase vaccine efficacy. Nat. Biotechnol. 33, 64–72 (2015).
Verbeke, C. S. et al. Multicomponent injectable hydrogels for antigen-specific tolerogenic immune modulation. Adv. Healthc. Mater 6, 1600773 (2017).
Irvine, D. J., Swartz, M. A. & Szeto, G. L. Engineering synthetic vaccines using cues from natural immunity. Nat. Mater. 12, 978–990 (2013).
Cheung, A. S. & Mooney, D. J. Engineered materials for cancer immunotherapy. Nano Today 10, 511–531 (2015).
Northrup, L., Christopher, M. A., Sullivan, B. P. & Berkland, C. Combining antigen and immunomodulators: Emerging trends in antigen-specific immunotherapy for autoimmunity. Adv. Drug Deliv. Rev. 98, 86–98 (2016).
Tostanoski, L. H. & Jewell, C. M. Engineering self -assembled materials to study and direct immune function. Adv. Drug Deliv. Rev. 114, 60–78 (2017).
Pearson, R. M., Casey, L. M., Hughes, K. R., Miller, S. D. & Shea, L. D. In vivo reprogramming of immune cells: Technologies for induction of antigen-specific tolerance. Adv. Drug Deliv. Rev. 114, 240–255 (2017).
Kuai, R., Ochyl, L. J., Bahjat, K. S., Schwendeman, A. & Moon, J. J. Designer vaccine nanodiscs for personalized cancer immunotherapy. Nat. Mater. 16, 489–496 (2017).
Ishihara, J. et al. Matrix-binding checkpoint immunotherapies enhance antitumour efficacy and reduce adverse events. Sci. Transl. Med. 9, eaan0401 (2017).
Jewell, C. M., Lopez, S. C. B. & Irvine, D. J. In situ engineering of the lymph node microenvironment via intranodal injection of adjuvant-releasing polymer particles. Proc. Natl Acad. Sci. USA 108, 15745–15750 (2011).
Gammon, J. M. et al. Low-dose controlled release of mTOR inhibitors maintains T cell plasticity and promotes central memory T cells. J. Control Release 263, 151–161 (2017).
Ludvigsson, J., Wahlberg, J. & Casas, R. Intralymphatic injection of autoantigen in type 1 diabetes. New Engl. J. Med. 376, 697–699 (2017).
Tostanoski, L. H. et al. Reprogramming the local lymph node microenvironment promotes tolerance that is systemic and antigen specific. Cell Reports 16, 2940–2952 (2016).
Ugel, S. et al. In vivo administration of artificial antigen-presenting cells activates low-avidity t cells for treatment of cancer. Cancer Res. 69, 9376–9384 (2009).
Perica, K. et al. Enrichment and expansion with nanoscale artificial antigen presenting cells for adoptive immunotherapy. ACS Nano 9, 6861–6871 (2015).
Sunshine, J. C., Perica, K., Schneck, J. P. & Green, J. J. Particle shape dependence of CD8+T cell activation by artificial antigen presenting cells. Biomaterials 35, 269–277 (2014).
Meyer, R. A. et al. Biodegradable nanoellipsoidal artificial antigen presenting cells for antigen specific T-Cell activation. Small 11, 1519–1525 (2015).
Bruns., H. et al. CD47 enhances in vivo functionality of artificial antigen-presenting cells. Clin. Cancer Res. 21, 2075–2083 (2015).
Kosmides, A. K. et al. Biomimetic biodegradable artificial antigen presenting with PD-1 blockade to treat melanoma cells synergize. Biomaterials 118, 16–26 (2017).
Cuzzone, D. A., Albano, N. J., Aschen, S. Z., Ghanta, S. & Mehrara, B. J. Decellularized lymph nodes as scaffolds for tissue engineered lymph nodes. Lymphat. Res. Biol 13, 186–194 (2015).
Suematsu, S. & Watanabe, T. Generation of a synthetic lymphoid tissue-like organoid in mice. Nat. Biotechnol. 22, 1539–1545 (2004).
Okamoto, N., Chihara, R., Shimizu, C., Nishimoto, S. & Watanabe, T. Artificial lymph nodes induce potent secondary immune responses in naive and immunodeficient mice. J. Clin. Invest. 117, 997–1007 (2007).
Kobayashi, Y. & Watanabe, T. Gel-trapped lymphorganogenic chemokines trigger artificial tertiary lymphoid organs and mount adaptive immune responses in vivo. Front. Immunol 7, 316 (2016).
Stephan, S. B. et al. Biopolymer implants enhance the efficacy of adoptive T-cell therapy. Nat. Biotechnol. 33, 97–101 (2015).
Smith, T. T. et al. Biopolymers codelivering engineered T cells and STING agonists can eliminate heterogeneous tumours. J. Clin. Invest 127, 2176–2191 (2017).
Smith, T. T. et al. In situ programming of leukaemia-specific T cells using synthetic DNA nanocarriers. Nat. Nanotech 12, 813–820 (2017).
Hun, M. et al. Native thymic extracellular matrix improves in vivo thymic organoid T cell output, and drives in vitro thymic epithelial cell differentiation. Biomaterials 118, 1–15 (2017).
Fan, Y. et al. Bioengineering thymus organoids to restore thymic function and induce donor-specific immune tolerance to allografts. Mol. Ther. 23, 1262–1277 (2015).
Markert, M. L., Devlin, B. H. & McCarthy, E. A. Thymus transplantation. Clin. Immunol. 135, 236–246 (2010).
Mao, A. S. et al. Deterministic encapsulation of single cells in thin tunable microgels for niche modelling and therapeutic delivery. Nat. Mater. 16, 236–243 (2017).
Lee, J. et al. Implantable microenvironments to attract hematopoietic stem/cancer cells. Proc. Natl Acad. Sci. USA 109, 19638–19643 (2012).
Shih, Y.-R. et al. In vivo engineering of bone tissues with hematopoietic functions and mixed chimerism. Proc. Natl Acad. Sci. USA 114, 5419–5424 (2017).
Holzapfel, B. M. et al. Tissue engineered humanized bone supports human hematopoiesis in vivo. Biomaterials 61, 103–114 (2015).
Reinisch, A. et al. A humanized bone marrow ossicle xenotransplantation model enables improved engraftment of healthy and leukemic human hematopoietic cells. Nat. Med 22, 812–821 (2016).
Abarrategi, A. et al. Versatile humanized niche model enables study of normal and malignant human hematopoiesis. J. Clin. Invest 127, 543–548 (2017).
Ben-David, U. et al. Patient-derived xenografts undergo mouse-specific tumour evolution. Nat. Genet. 49, 1567–1575 (2017).
Schaupper, M., Jeltsch, M., Rohringer, S., Redl, H. & Holnthoner, W. Lymphatic vessels in regenerative medicine and tissue engineering. Tissue Eng. Part B 22, 395–407 (2016).
Randolph, G. J., Ivanov, S., Zinselmeyer, B. H. & Scallan, J. P. The lymphatic system: integral roles in immunity. Annu. Rev. Immunol. 35, 31–52 (2017).
Gibot, L. et al. Tissue-engineered 3D human lymphatic microvascular network for in vitro studies of lymphangiogenesis. Nat. Protoc. 12, 1077–1088 (2017).
Groom, J. R. et al. CXCR3 chemokine receptor-ligand interactions in the lymph node optimize CD4(+) T helper 1 cell differentiation. Immunity 37, 1091–1103 (2012).
Malhotra, D. et al. Transcriptional profiling of steady-state and inflamed lymph node stroma reveals potential hematopoietic-stromal cross-talk pathways and suggests an active role for stroma during ongoing immune responses. J. Immunol. 188, 176.22 (2012).
Song, J. et al. Extracellular matrix of secondary lymphoid organs impacts on B-cell fate and survival. Proc. Natl Acad. Sci. USA 110, E2915–E2924 (2013).
Cohen, J. N. et al. Tolerogenic properties of lymphatic endothelial cells are controlled by the lymph node microenvironment. PLOS ONE 9, e87740 (2014).
Kastenmuller, W. et al. Peripheral prepositioning and local CXCL9 chemokine-mediated guidance orchestrate rapid memory CD8(+) T cell responses in the lymph node. Immunity 38, 502–513 (2013).
Burrell, B. E. & Bromberg, J. S. Fates of CD4+T cells in a tolerant environment depend on timing and place of antigen exposure. Am. J. Transplant 12, 576–589 (2012).
Dubrot, J. et al. Lymph node stromal cells acquire peptide-MHCII complexes from dendritic cells and induce antigen-specific CD4(+) T cell tolerance. J. Exp. Med. 211, 1153–1166 (2014).
Hirosue, S. et al. Steady-state antigen scavenging, cross-presentation, and CD8(+) T cell priming: a new role for lymphatic endothelial cells. J. Immunol. 192, 5002–5011 (2014).
Tamburini, B. A., Burchill, M. A. & Kedl, R. M. Antigen capture and archiving by lymphatic endothelial cells following vaccination or viral infection. Nat. Commun. 5, 3989 (2014).
Warren, K. J., Iwami, D., Harris, D. G., Bromberg, J. S. & Burrell, B. E. Laminins affect T cell trafficking and allograft fate. J. Clin. Invest. 124, 2204–2218 (2014).
Astarita, J. L. et al. The CLEC-2-podoplanin axis controls the contractility of fibroblastic reticular cells and lymph node microarchitecture. Nat. Immunol. 16, 75–84 (2015).
Nia, H. T. et al. Solid stress and elastic energy as measures of tumour mechanopathology. Nat. Biomed. Eng. 1, 0004 (2016).
Nolan, D. J. et al. Molecular signatures of tissue-specific microvascular endothelial cell heterogeneity in organ maintenance and regeneration. Dev. Cell 26, 204–219 (2013).
Iftakhar-E-Khuda, I. et al. Gene-expression profiling of different arms of lymphatic vasculature identifies candidates for manipulation of cell traffic. Proc. Natl Acad. Sci. USA 113, 10643–10648 (2016).
Lee, T. T. et al. Light-triggered in vivo activation of adhesive peptides regulates cell adhesion, inflammation and vascularization of biomaterials. Nat. Mater. 14, 352–360 (2015).
Sadtler, K. et al. Developing a pro-regenerative biomaterial scaffold microenvironment requires T helper 2 cells. Science 352, 366–370 (2016).
Doloff, J. C. et al. Colony stimulating factor-1 receptor is a central component of the foreign body response to biomaterial implants in rodents and non-human primates. Nat. Mater. 16, 671–680 (2017).
Guc, E. et al. Local induction of lymphangiogenesis with engineered fibrin-binding VEGF-C promotes wound healing by increasing immune cell trafficking and matrix remodeling. Biomaterials 131, 160–175 (2017).
Wang, H. M. et al. Enzyme-catalyzed formation of supramolecular hydrogels as promising vaccine adjuvants. Adv. Funct. Mater. 26, 1822–1829 (2016).
Sharp, F. A. et al. Uptake of particulate vaccine adjuvants by dendritic cells activates the NALP3 inflammasome. Proc. Natl Acad. Sci. USA 106, 870–875 (2009).
Andorko, J. I., Hess, K. L. & Jewell, C. M. Harnessing biomaterials to engineer the lymph node microenvironment for immunity or tolerance. Aaps J. 17, 323–338 (2015).
Hess, K. L. et al. Engineering immunological tolerance using quantum dots to tune the density of self-antigen display. Adv. Funct. Mater. 27, 1700290 (2017).
Singha, S. et al. Peptide-MHC-based nanomedicines for autoimmunity function as T-cell receptor microclustering devices. Nat. Nanotech. 12, 701–710 (2017).
Novkovic, M. et al. Topological small-world organization of the fibroblastic reticular cell network determines lymph node functionality. PLOS Biol. 14, e1002515 (2016).
Acknowledgements
This work was supported in part by the United States Department of Veterans Affairs grant no. 1I01BX003690, NIH grant no. R01EB026896, NSF CAREER grant no. 1351688, the National Multiple Sclerosis Society grant no. RG-1501-02968, NIH grant no. 1R01AI062765, NIH grant no. 1R01AI114496, the Damon Runyon Foundation grant no. DRR3415, and Juvenile Diabetes Research Foundation grant no. 2-SRA-2016-319-S-B. H.B.E. is a trainee of the NIH T32 Training Program in Cell and Molecular Biology (T32GM080201). C.M.J. is a young investigator of the Alliance for Cancer Gene Therapy (grant no. 15051543) and the Melanoma Research Alliance (grant no. 348963).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests. The content in this review do not reflect the views of the Department of Veterans Affairs or the United States Government.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gosselin, E.A., Eppler, H.B., Bromberg, J.S. et al. Designing natural and synthetic immune tissues. Nature Mater 17, 484–498 (2018). https://doi.org/10.1038/s41563-018-0077-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-018-0077-6
This article is cited by
-
Elasticity-controlled jamming criticality in soft composite solids
Nature Communications (2024)
-
Engineering the lymph node environment promotes antigen-specific efficacy in type 1 diabetes and islet transplantation
Nature Communications (2023)
-
Biomimetic nanoparticles deliver mRNAs encoding costimulatory receptors and enhance T cell mediated cancer immunotherapy
Nature Communications (2021)
-
Biomaterial-enabled induction of pancreatic-specific regulatory T cells through distinct signal transduction pathways
Drug Delivery and Translational Research (2021)
-
Recapitulating T cell infiltration in 3D psoriatic skin models for patient-specific drug testing
Scientific Reports (2020)