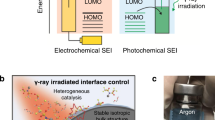
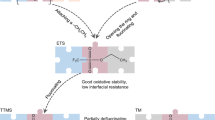
Abstract
Li+ desolvation in electrolytes and diffusion at the solid–electrolyte interphase (SEI) are two determining steps that restrict the fast charging of graphite-based lithium-ion batteries. Here we show that the low-solvent-coordination Li+ solvation structure could be induced near the inner Helmholtz plane on inorganic species. Specifically, Li3P could enable a lower Li+ desolvation barrier and faster Li+ diffusion capability through the SEI in comparison to the regular SEI components. We construct an ultrathin S-bridged phosphorus layer on a graphite surface, which in situ converts to crystalline Li3P-based SEI with high ionic conductivity. Our pouch cells with such a graphite anode show 10 min and 6 min (6C and 10C) charging for 91.2% and 80% of the capacity, respectively, as well as 82.9% capacity retention for over 2,000 cycles at a 6C charging rate. Our work reveals the importance of the SEI component and structure regulation for fast-charging LIBs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the article and its Supplementary Information files.
References
Liu, Y., Zhu, Y. & Cui, Y. Challenges and opportunities towards fast-charging battery materials. Nat. Energy 4, 540–550 (2019).
Cai, W. et al. A review on energy chemistry of fast-charging anodes. Chem. Soc. Rev. 49, 3806–3833 (2020).
Li, S. et al. Fast charging anode materials for lithium‐ion batteries: current status and perspectives. Adv. Funct. Mater. 32, 2200796 (2022).
Liu, H. et al. A disordered rock salt anode for fast-charging lithium-ion batteries. Nature 585, 63–67 (2020).
Griffith, K. J., Wiaderek, K. M., Cibin, G., Marbella, L. E. & Grey, C. P. Niobium tungsten oxides for high-rate lithium-ion energy storage. Nature 559, 556–563 (2018).
Meintz, A. et al. Enabling fast charging–vehicle considerations. J. Power Sources 367, 216–227 (2017).
Ni, Q. et al. An extremely fast charging Li3V2(PO4)3 cathode at a 4.8 V cutoff voltage for Li-ion batteries. ACS Energy Lett. 5, 1763–1770 (2020).
Tu, S. et al. Single-layer-particle electrode design for practical fast-charging lithium-ion batteries. Adv. Mater. 34, 2202892 (2022).
Wang, C.-Y. et al. Fast charging of energy-dense lithium-ion batteries. Nature 611, 485–490 (2022).
Lu, L. L. et al. Superior fast-charging lithium-ion batteries enabled by the high-speed solid-state lithium transport of an intermetallic Cu6Sn5 Network. Adv. Mater. 34, 2202688 (2022).
Ahmed, S. et al. Enabling fast charging–a battery technology gap assessment. J. Power Sources 367, 250–262 (2017).
Mao, C., Ruther, R. E., Li, J., Du, Z. & Belharouak, I. Identifying the limiting electrode in lithium ion batteries for extreme fast charging. Electrochem. Commun. 97, 37–41 (2018).
Tian, R. et al. Quantifying the factors limiting rate performance in battery electrodes. Nat. Commun. 10, 1933 (2019).
Jow, T. R., Delp, S. A., Allen, J. L., Jones, J.-P. & Smart, M. C. Factors limiting Li+ charge transfer kinetics in Li-ion batteries. J. Electrochem. Soc. 165, A361–A367 (2018).
Zhang, S. S., Ma, L., Allen, J. L. & Read, J. A. Stabilizing capacity retention of Li-ion battery in fast-charge by reducing particle size of graphite. J. Electrochem. Soc. 168, 040519 (2021).
Baek, M., Kim, J., Jin, J. & Choi, J. W. Photochemically driven solid electrolyte interphase for extremely fast-charging lithium-ion batteries. Nat. Commun. 12, 6807 (2021).
Chen, K. H. et al. Enabling 6C fast charging of Li‐ion batteries with graphite/hard carbon hybrid anodes. Adv. Energy Mater. 11, 2003336 (2020).
Sun, C. et al. 50C fast-charge Li-ion batteries using a graphite anode. Adv. Mater. 34, 2206020 (2022).
Kazyak, E., Chen, K. H., Chen, Y., Cho, T. H. & Dasgupta, N. P. Enabling 4C fast charging of lithium‐ion batteries by coating graphite with a solid‐state electrolyte. Adv. Energy Mater. 12, 2102618 (2021).
Lee, S. M. et al. A cooperative biphasic MoOx-MoPx promoter enables a fast-charging lithium-ion battery. Nat. Commun. 12, 39 (2021).
Heiskanen, S. K., Kim, J. & Lucht, B. L. Generation and evolution of the solid electrolyte interphase of lithium-ion batteries. Joule 3, 2322–2333 (2019).
Zhang, Z. et al. Capturing the swelling of solid-electrolyte interphase in lithium metal batteries. Science 375, 66–70 (2022).
Chen, J. et al. Electrolyte design for LiF-rich solid–electrolyte interfaces to enable high-performance microsized alloy anodes for batteries. Nat. Energy 5, 386–397 (2020).
Qin, N. et al. Over‐potential tailored thin and dense lithium carbonate growth in solid electrolyte interphase for advanced lithium ion batteries. Adv. Energy Mater. 12, 2103402 (2022).
Tan, J., Matz, J., Dong, P., Shen, J. & Ye, M. A growing appreciation for the role of LiF in the solid electrolyte interphase. Adv. Energy Mater. 11, 2100046 (2021).
Jiang, L. L. et al. Inhibiting solvent co-intercalation in a graphite anode by a localized high-concentration electrolyte in fast-charging batteries. Angew. Chem., Int. Ed. 60, 3402–3406 (2021).
Shi, J. et al. Improving the graphite/electrolyte interface in lithium-ion battery for fast charging and low temperature operation: fluorosulfonyl isocyanate as electrolyte additive. J. Power Sources 429, 67–74 (2019).
Yang, Y. et al. Rechargeable LiNi0.65Co0.15Mn0.2O2||graphite batteries operating at −60 °C. Angew. Chem., Int. Ed. 61, 202209619 (2022).
Yang, Y. et al. Synergy of weakly-solvated electrolyte and optimized interphase enables graphite anode charge at low temperature. Angew. Chem., Int. Ed. 61, 202208345 (2022).
Sun, Y. et al. Design of red phosphorus nanostructured electrode for fast-charging lithium-ion batteries with high energy density. Joule 3, 1080–1093 (2019).
Xu, K., von Cresce, A. & Lee, U. Differentiating contributions to ‘ion transfer’ barrier from interphasial resistance and Li+ desolvation at electrolyte/graphite interface. Langmuir 26, 11538–11543 (2010).
Li, Y. & Qi, Y. Energy landscape of the charge transfer reaction at the complex Li/SEI/electrolyte interface. Energy Environ. Sci. 12, 1286–1295 (2019).
Xin, S. et al. Smaller sulfur molecules promise better lithium-sulfur batteries. J. Am. Chem. Soc. 134, 18510–18513 (2012).
Zhou, J. et al. High-spin sulfur-mediated phosphorous activation enables safe and fast phosphorus anodes for sodium-ion batteries. Chem 6, 221–233 (2020).
Chuang, C.-C., Hsieh, Y.-Y., Chang, W.-C. & Tuan, H.-Y. Phosphorus-sulfur/graphene composites as flexible lithium-sulfur battery cathodes with super high volumetric capacity. Chem. Eng. J. 387, 123904 (2020).
Marino, C., El Kazzi, M., Berg, E. J., He, M. & Villevieille, C. Interface and safety properties of phosphorus-based negative electrodes in Li-ion batteries. Chem. Mater. 29, 7151–7158 (2017).
Shi, P. et al. The synergetic effect of lithium bisoxalatodifluorophosphate and fluoroethylene carbonate on dendrite suppression for fast charging lithium metal batteries. Small 16, 2001989 (2020).
Kim, N., Chae, S., Ma, J., Ko, M. & Cho, J. Fast-charging high-energy lithium-ion batteries via implantation of amorphous silicon nanolayer in edge-plane activated graphite anodes. Nat. Commun. 8, 812 (2017).
Huang, W. et al. Evolution of the solid-electrolyte interphase on carbonaceous anodes visualized by atomic-resolution cryogenic electron microscopy. Nano Lett. 19, 5140–5148 (2019).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Martínez, L., Andrade, R., Birgin, E. G. & Martínez, J. M. PACKMOL: a package for building initial configurations for molecular dynamics simulations. J. Comput. Chem. 30, 2157–2164 (2009).
Monti, S., Carravetta, V. & Ågren, H. Simulation of gold functionalization with cysteine by reactive molecular dynamics. J. Phys. Chem. Lett. 7, 272–276 (2016).
Jorgensen, W. L., Maxwell, D. S. & Tirado-Rives, J. Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236 (1996).
Bayly, C. I., Cieplak, P., Cornell, W. & Kollman, P. A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J. Phys. Chem. 97, 10269–10280 (1993).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Dauber-Osguthorpe, P. et al. Structure and energetics of ligand binding to proteins: Escherichia coli dihydrofolate reductase-trimethoprim, a drug-receptor system. Proteins: Struct. Funct. Bioinforma. 4, 31–47 (1988).
Campañá, C., Mussard, B. & Woo, T. K. Electrostatic potential derived atomic charges for periodic systems using a modified error functional. J. Chem. Theory Comput. 5, 2866–2878 (2009).
Hockney, R. W. & Eastwood, J. W. Computer Simulation Using Particles (CRC Press, 2021).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Fiorin, G., Klein, M. & Hénin, J. Using collective variables to drive molecular dynamics simulations. Mol. Phys. 111, 3345–3362 (2013).
Hohenberg, P. & Kohn, W. Inhomogeneous electron gas. Phys. Rev. 136, B864 (1964).
Kohn, W. & Sham, L. J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133 (1965).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558 (1993).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953 (1994).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188 (1976).
Acknowledgements
This work is financially supported by the Natural Science Foundation of China (Grant No. 52072137). We would like to thank the Analytical and Testing Center of Huazhong University of Science and Technology for providing the facilities to conduct the transmission electron microscopy characterizations.
Author information
Authors and Affiliations
Contributions
Y.S. and S.T. conceived the idea for this work. S.T. performed the material characterizations and electrochemical measurements with assistance from Y.Z., Z.C., X.W., R.Z., Y.O., W.W., X.L. and X.D. B.Z. performed the MD simulations and DFT calculations. L.W. discussed and edited the results. S.T. and B.Z. co-wrote the original paper. Y.S. reviewed and edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks James Tour and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–35 and Tables 1–3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tu, S., Zhang, B., Zhang, Y. et al. Fast-charging capability of graphite-based lithium-ion batteries enabled by Li3P-based crystalline solid–electrolyte interphase. Nat Energy 8, 1365–1374 (2023). https://doi.org/10.1038/s41560-023-01387-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-023-01387-5