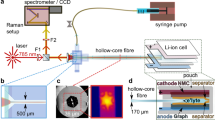
Abstract
Improvements to battery performance, reliability and lifetime are essential to meet the expansive demands for energy storage. As part of this, continuous monitoring of the dynamic chemistry inside cells offers an exciting path to minimizing parasitic reactions and maximizing sustainability. Building upon recent fibre-optic/battery innovations, we report the use of operando infrared fibre evanescent wave spectroscopy to monitor electrolyte evolution in 18650 Na-ion and Li-ion cells under real working conditions. This approach enables identification of chemical species and reveals electrolyte and additive decomposition mechanisms during cycling, thereby providing important insights into the growth and nature of the solid–electrolyte interphase, the dynamics of solvation and their complex interrelations. Moreover, by directly embedding fibres within the electrode material, we demonstrate simultaneous observations of both the material structural evolution and the Na(Li) inventory changes upon cycling. This illuminating sensing method has the power to reveal the otherwise opaque chemical phenomena occurring within each key battery component.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The datasets generated during the current study are available in the article and its Supplementary Information.
References
Larcher, D. & Tarascon, J.-M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 7, 19–29 (2015).
Wang, F. et al. Tracking lithium transport and electrochemical reactions in nanoparticles. Nat. Commun. 3, 1201 (2012).
Blanc, F., Leskes, M. & Grey, C. P. In situ solid-state NMR spectroscopy of electrochemical cells: batteries, supercapacitors, and fuel cells. Acc. Chem. Res. 46, 1952–1963 (2013).
Zhang, Y. et al. Revealing electrolyte oxidation via carbonate dehydrogenation on Ni-based oxides in Li-ion batteries by in situ Fourier transform infrared spectroscopy. Energy Environ. Sci. 13, 183–199 (2020).
Sathiya, M. et al. Electron paramagnetic resonance imaging for real-time monitoring of Li-ion batteries. Nat. Commun. 6, 6276 (2015).
Grey, C. P. & Tarascon, J. M. Sustainability and in situ monitoring in battery development. Nat. Mater. 16, 45–56 (2016).
Huang, J., Boles, S. T. & Tarascon, J.-M. Sensing as the key to battery lifetime and sustainability. Nat. Sustain. 5, 194–204 (2022).
Peng, J. et al. High precision strain monitoring for lithium ion batteries based on fiber Bragg grating sensors. J. Power Sources 433, 226692 (2019).
Sommer, L. W. et al. Embedded fiber optic sensing for accurate state estimation in advanced battery management systems. MRS Online Proc. Libr. 1681, 1–7 (2014).
Huang, J. et al. Operando decoding of chemical and thermal events in commercial Na(Li)-ion cells via optical sensors. Nat. Energy 5, 674–683 (2020).
Desai, P. et al. Deciphering interfacial reactions via optical sensing to tune the interphase chemistry for optimized Na-ion electrolyte formulation. Adv. Energy Mater. 11, 2101490 (2021).
Huang, J. et al. Monitoring battery electrolyte chemistry via in-operando tilted fiber Bragg grating sensors. Energy Environ. Sci. https://doi.org/10.1039/D1EE02186A (2021).
Manap, H., Dooly, G., O’Keeffe, S. & Lewis, E. Ammonia detection in the UV region using an optical fiber sensor. Sens. 2009 IEEE https://doi.org/10.1109/ICSENS.2009.5398215 (2009).
Michel, K. et al. Monitoring of pollutant in waste water by infrared spectroscopy using chalcogenide glass optical fibers. Sens. Actuators B Chem. 101, 252–259 (2004).
Yan, D., Popp, J., Pletz, M. W. & Frosch, T. Highly sensitive broadband Raman sensing of antibiotics in step-index hollow-core photonic crystal fibers. ACS Photonics 4, 138–145 (2017).
Cubillas, A. M. et al. Photonic crystal fibres for chemical sensing and photochemistry. Chem. Soc. Rev. 42, 8629–8648 (2013).
Bertucci, A. et al. Detection of unamplified genomic DNA by a PNA-based microstructured optical fiber (MOF) Bragg-grating optofluidic system. Biosens. Bioelectron. 63, 248–254 (2015).
Miele, E. et al. Hollow-core optical fibre sensors for operando Raman spectroscopy investigation of Li-ion battery liquid electrolytes. Nat. Commun. 13, 1651 (2022).
Ellis, L. D. et al. A new method for determining the concentration of electrolyte components in lithium-ion cells, using Fourier transform infrared spectroscopy and machine learning. J. Electrochem. Soc. 165, A256–A262 (2018).
Buteau, S. & Dahn, J. R. Analysis of thousands of electrochemical impedance spectra of lithium-ion cells through a machine learning inverse model. J. Electrochem. Soc. 166, A1611–A1622 (2019).
Corvec, M. L. et al. Fast and non-invasive medical diagnostic using mid infrared sensor: the AMNIFIR project. Innov. Res. Biomed. Eng. 37, 116–123 (2016).
Calvez, L. Chalcogenide glasses and glass-ceramics: transparent materials in the infrared for dual applications. Comptes Rendus Phys. 18, 314–322 (2017).
Cresce, A. V. et al. Solvation behavior of carbonate-based electrolytes in sodium ion batteries. Phys. Chem. Chem. Phys. 19, 574–586 (2016).
Yan, G. et al. A new electrolyte formulation for securing high temperature cycling and storage performances of Na-ion batteries. Adv. Energy Mater. 9, 1901431 (2019).
Eshetu, G. G. et al. Comprehensive insights into the reactivity of electrolytes based on sodium ions. ChemSusChem 9, 462–471 (2016).
Yan, G. et al. Assessment of the electrochemical stability of carbonate-based electrolytes in Na-ion batteries. J. Electrochem. Soc. 165, A1222 (2018).
Angell, C. L. The infra-red spectra and structure of ethylene carbonate. Trans. Faraday Soc. 52, 1178 (1956).
Yoshida, H. et al. Degradation mechanism of alkyl carbonate solvents used in lithium-ion cells during initial charging. J. Power Sources 68, 311–315 (1997).
Nyquist, R. A. & Settineri, S. E. Infrared study of ethylene carbonate in various solvents and solvent systems. Appl. Spectrosc. 45, 1075–1084 (1991).
Pan, Y. et al. Investigation of the solid electrolyte interphase on hard carbon electrode for sodium ion batteries. J. Electroanal. Chem. 799, 181–186 (2017).
Aurbach, D. et al. On the use of vinylene carbonate (VC) as an additive to electrolyte solutions for Li-ion batteries. Electrochim. Acta 47, 1423–1439 (2002).
Sasaki, T., Abe, T., Iriyama, Y., Inaba, M. & Ogumi, Z. Suppression of an alkyl dicarbonate formation in Li-ion cells. J. Electrochem. Soc. 152, A2046 (2005).
Kosova, N. V. & Rezepova, D. O. Na1+yVPO4F1+y (0 ≤ y ≤ 0.5) as cathode materials for hybrid Na/Li batteries. Inorganics 5, 19 (2017).
Boivin, É. Crystal chemistry of vanadium phosphates as positive electrode materials for Li-ion and Na-ion batteries. https://tel.archives-ouvertes.fr/tel-03648931 (2017).
Ait Salah, A. et al. FTIR features of lithium-iron phosphates as electrode materials for rechargeable lithium batteries. Spectrochim. Acta A Mol. Biomol. Spectrosc. 65, 1007–1013 (2006).
Bianchini, M. et al. Comprehensive investigation of the Na3V2(PO4)2F3–NaV2(PO4)2F3 system by operando high resolution synchrotron X-ray diffraction. Chem. Mater. 27, 3009–3020 (2015).
Bianchini, M. et al. Na3V2(PO4)2F3 revisited: a high-resolution diffraction study. Chem. Mater. 26, 4238–4247 (2014).
Becke, A. D. A new mixing of Hartree–Fock and local density‐functional theories. J. Chem. Phys. 98, 1372–1377 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Erba, A., Baima, J., Bush, I., Orlando, R. & Dovesi, R. Large-scale condensed matter DFT simulations: performance and capabilities of the CRYSTAL code. J. Chem. Theory Comput. 13, 5019–5027 (2017).
Dovesi, R. et al. Quantum-mechanical condensed matter simulations with CRYSTAL. WIREs Comput. Mol. Sci. 8, e1360 (2018).
Maschio, L., Kirtman, B., Rérat, M., Orlando, R. & Dovesi, R. Ab initio analytical Raman intensities for periodic systems through a coupled perturbed Hartree-Fock/Kohn-Sham method in an atomic orbital basis. I. Theory. J. Chem. Phys. 139, 164101 (2013).
Maschio, L., Kirtman, B., Rérat, M., Orlando, R. & Dovesi, R. Ab initio analytical Raman intensities for periodic systems through a coupled perturbed Hartree-Fock/Kohn-Sham method in an atomic orbital basis. II. Validation and comparison with experiments. J. Chem. Phys. 139, 164102 (2013).
Acknowledgements
J.-M.T. acknowledges the International Balzan Prize Foundation and the LABEX STOREXII for funding. C.G.-M. and J.-M.T. acknowledge Brurker for the instrumental set-up. We thank Tiamat for providing the NVPF/HC 18650 cells and R. Dugas for his assistance in the cell fabrication. We thank R. Chometon for the scanning electron microscopy images. M.B.Y. acknowledges the support of the French Agence Nationale de la Recherche (ANR) under reference ANR-17-CE05-10 (project VASELinA). S.T.B. acknowledges the support from the ENERSENSE research initiative (68024013) at the Norwegian University of Science and Technology (NTNU), Norway. Finally, we gladly thank S. Mariyappan, P. Desai and D. Larcher for valuable discussions and comments.
Author information
Authors and Affiliations
Contributions
C.G.-M. and J.-M.T. conceived the idea and designed the experiments with the help of C.B.-P., J.L.A. and X.H.Z., who provided the adequate fibre. C.G.-M. performed the electrochemical tests, optical tests and data analysis. M.B.Y. and M.-L.D. conducted the theoretical analysis. C.B.-P., J.H., L.A.B., S.T.B., C.L., X.H.Z, J.L.A. and J.-M.T. conjointly discussed the data and their meaning. Finally, C.G.-M., M.-L.D., S.T.B. and J.-M.T. wrote the paper, with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
A patent related to the work has been submitted (application no. PCT/EP2022/071395) by the Centre National de la Rercherche Scientifique, Collège de France, Sorbonne Université, Université de Rennes 1, Ecole Nationale Supérieure de Chimie de Rennes and Institut National des Sciences Appliquées de Rennes. The inventors are Jean-Marie Tarasco, Charlotte Gervillié, Catherine Boussard, Xiang-Hua Zhang and Jean-Luc Adam. The patent covers a method for operando characterization of chemical species within a battery using infrared fibre evanescent wave spectroscopy.
Peer review
Peer review information
Nature Energy thanks Yifei Yu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–23.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gervillié-Mouravieff, C., Boussard-Plédel, C., Huang, J. et al. Unlocking cell chemistry evolution with operando fibre optic infrared spectroscopy in commercial Na(Li)-ion batteries. Nat Energy 7, 1157–1169 (2022). https://doi.org/10.1038/s41560-022-01141-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-022-01141-3
This article is cited by
-
Operando monitoring of dendrite formation in lithium metal batteries via ultrasensitive tilted fiber Bragg grating sensors
Light: Science & Applications (2024)
-
Work-function effect of Ti3C2/Fe-N-C inducing solid electrolyte interphase evolution for ultra-stable sodium storage
Nano Research (2024)
-
Nonintrusive thermal-wave sensor for operando quantification of degradation in commercial batteries
Nature Communications (2023)
-
Detangling electrolyte chemical dynamics in lithium sulfur batteries by operando monitoring with optical resonance combs
Nature Communications (2023)
-
Operando monitoring of thermal runaway in commercial lithium-ion cells via advanced lab-on-fiber technologies
Nature Communications (2023)