Abstract
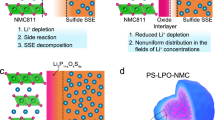
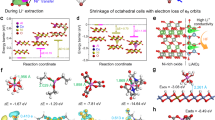
Engineered polycrystalline electrodes are critical to the cycling stability and safety of lithium-ion batteries, yet it is challenging to construct high-quality coatings at both the primary- and secondary-particle levels. Here we present a room-temperature synthesis route to achieve a full surface coverage of secondary particles and facile infusion into grain boundaries, and thus offer a complete ‘coating-plus-infusion’ strategy. Cobalt boride metallic glass was successfully applied to a Ni-rich layered cathode LiNi0.8Co0.1Mn0.1O2. It dramatically improved the rate capability and cycling stability, including under high-discharge-rate and elevated-temperature conditions and in pouch full-cells. The superior performance originates from a simultaneous suppression of the microstructural degradation of the intergranular cracking and of side reactions with the electrolyte. Atomistic simulations identified the critical role of strong selective interfacial bonding, which offers not only a large chemical driving force to ensure uniform reactive wetting and facile infusion, but also lowers the surface/interface oxygen activity, which contributes to the exceptional mechanical and electrochemical stabilities of the infused electrode.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data generated and analysed in this study are included in the manuscript and its Supplementary Information.
Change history
11 May 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41560-021-00845-2
References
Nitta, N., Wu, F., Lee, J. T. & Yushin, G. Li-ion battery materials: present and future. Mater. Today 18, 252–264 (2015).
Kim, J. et al. Prospect and reality of Ni‐rich cathode for commercialization. Adv. Energy Mater. 8, 1702028 (2018).
Li, W., Erickson, E. M. & Manthiram, A. High-nickel layered oxide cathodes for lithium-based automotive batteries. Nat. Energy 5, 26–34 (2020).
Edström, K., Gustafsson, T. & Thomas, J. O. The cathode–electrolyte interface in the Li-ion battery. Electrochim. Acta 50, 397–403 (2004).
Sharifi-Asl, S., Lu, J., Amine, K. & Shahbazian-Yassar, R. Oxygen release degradation in Li-ion battery cathode materials: mechanisms and mitigating approaches. Adv. Energy Mater. 9, 1900551 (2019).
Xie, Q., Li, W. & Manthiram, A. A Mg-doped high-nickel layered oxide cathode enabling safer, high-energy-density Li-ion batteries. Chem. Mater. 31, 938–946 (2019).
Schipper, F. et al. From surface ZrO2 coating to bulk Zr doping by high temperature annealing of nickel-rich lithiated oxides and their enhanced electrochemical performance in lithium ion batteries. Adv. Energy Mater. 8, 1701682 (2018).
Xu, X. et al. Radially oriented single-crystal primary nanosheets enable ultrahigh rate and cycling properties of LiNi0.8Co0.1Mn0.1O2 cathode material for lithium-ion batteries. Adv. Energy Mater. 9, 1803963 (2019).
Ofer, D., Pullen, A. W. & Sriramulu, S. Polycrystalline metal oxide, methods of manufacture thereof, and articles comprising the same. US patent 9,391,317 (2016).
Kim, J. et al. A highly stabilized nickel-rich cathode material by nanoscale epitaxy control for high-energy lithium-ion batteries. Energy Environ. Sci. 11, 1449–1459 (2018).
Yan, P. et al. Tailoring grain boundary structures and chemistry of Ni-rich layered cathodes for enhanced cycle stability of lithium-ion batteries. Nat. Energy 3, 600–605 (2018).
Xu, G.-L. et al. Building ultraconformal protective layers on both secondary and primary particles of layered lithium transition metal oxide cathodes. Nat. Energy 4, 484–494 (2019).
Li, L. et al. Hidden subsurface reconstruction and its atomic origins in layered oxide cathodes. Nano Lett. 20, 2756–2762 (2020).
Liu, H. et al. Intergranular cracking as a major cause of long-term capacity fading of layered cathodes. Nano Lett. 17, 3452–3457 (2017).
Yan, P. et al. Coupling of electrochemically triggered thermal and mechanical effects to aggravate failure in a layered cathode. Nat. Commun. 9, 2437 (2018).
Watanabe, S., Kinoshita, M., Hosokawa, T., Morigaki, K. & Nakura, K. Capacity fade of LiAlyNi1−x−yCoxO2 cathode for lithium-ion batteries during accelerated calendar and cycle life tests (surface analysis of LiAlyNi1−x−yCoxO2 cathode after cycle tests in restricted depth of discharge ranges). J. Power Sources 258, 210–217 (2014).
Jung, R., Metzger, M., Maglia, F., Stinner, C. & Gasteiger, H. A. Chemical versus electrochemical electrolyte oxidation on NMC111, NMC622, NMC811, LNMO, and conductive carbon. J. Phys. Chem. Lett. 8, 4820–4825 (2017).
Freiberg, A. T. S., Roos, M. K., Wandt, J., de Vivie-Riedle, R. & Gasteiger, H. A. Singlet oxygen reactivity with carbonate solvents used for Li-ion battery electrolytes. J. Phys. Chem. A 122, 8828–8839 (2018).
Zhan, C., Wu, T., Lu, J. & Amine, K. Dissolution, migration, and deposition of transition metal ions in Li-ion batteries exemplified by Mn-based cathodes—a critical review. Energy Environ. Sci. 11, 243–257 (2018).
Huang, Y. et al. Lithium manganese spinel cathodes for lithium-ion batteries. Adv. Energy Mater. 11, 2000997 (2021).
Zhu, Z. et al. Gradient Li-rich oxide cathode particles immunized against oxygen release by a molten salt treatment. Nat. Energy 4, 1049–1058 (2019).
Yan, P. et al. Injection of oxygen vacancies in the bulk lattice of layered cathodes. Nat. Nanotechnol. 14, 602–608 (2019).
Armstrong, A. R. et al. Demonstrating oxygen loss and associated structural reorganization in the lithium battery cathode Li[Ni0.2Li0.2Mn0.6]O2. J. Am. Chem. Soc. 128, 8694–8698 (2006).
House, R. A. et al. What triggers oxygen loss in oxygen redox cathode materials? Chem. Mater. 31, 3293–3300 (2019).
Yoon, M. et al. Unveiling nickel chemistry in stabilizing high-voltage cobalt-rich cathodes for lithium-ion batteries. Adv. Funct. Mater. 30, 1907903 (2020).
Jain, A. et al. Commentary: The Materials Project: a materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Mu, D. & Yang, C. Shen, B.-l. & Jiang, H. Oxidation resistance of borided pure cobalt. J. Alloy. Compd 479, 629–633 (2009).
Yang, Y., Kushima, A., Han, W., Xin, H. & Li, J. Liquid-like, self-healing aluminum oxide during deformation at room temperature. Nano Lett. 18, 2492–2497 (2018).
Li, J., Lenosky, T. J., Först, C. J. & Yip, S. Thermochemical and mechanical stabilities of the oxide scale of ZrB2+SiC and oxygen transport mechanisms. J. Am. Ceram. Soc. 91, 1475–1480 (2008).
Hasegawa, R. & Ray, R. Iron–boron metallic glasses. J. Appl. Phys. 49, 4174–4179 (1978).
Gaskell, P. H. A new structural model for amorphous transition metal silicides, borides, phosphides and carbides. J. Non-Cryst. Solids 32, 207–224 (1979).
Masa, J. et al. Amorphous cobalt boride (Co2B) as a highly efficient nonprecious catalyst for electrochemical water splitting: oxygen and hydrogen evolution. Adv. Energy Mater. 6, 1502313 (2016).
Deng, J. et al. Co–B nanoflakes as multifunctional bridges in ZnCo2O4 micro-/nanospheres for superior lithium storage with boosted kinetics and stability. Adv. Energy Mater. 9, 1803612 (2019).
Jiang, B. et al. A mesoporous non-precious metal boride system: synthesis of mesoporous cobalt boride by strictly controlled chemical reduction. Chem. Sci. 11, 791–796 (2020).
Chen, Z. et al. Study of cobalt boride-derived electrocatalysts for overall water splitting. Int. J. Hydrog. Energy 43, 6076–6087 (2018).
Zhang, C. et al. Revealing the role of NH4VO3 treatment in Ni-rich cathode materials with improved electrochemical performance for rechargeable lithium-ion batteries. Nanoscale 10, 8820–8831 (2018).
Yu, Y. et al. Optimal annealing of Al foil anode for prelithiation and full-cell cycling in Li-ion battery: the role of grain boundaries in lithiation/delithiation ductility. Nano Energy 67, 104274 (2020).
Miller, D. J., Proff, C., Wen, J. G., Abraham, D. P. & Bareño, J. Observation of microstructural evolution in Li battery cathode oxide particles by in situ electron microscopy. Adv. Energy Mater. 3, 1098–1103 (2013).
Kondrakov, A. O. et al. Anisotropic lattice strain and mechanical degradation of high- and low-nickel NCM cathode materials for Li-ion batteries. J. Phys. Chem. C. 121, 3286–3294 (2017).
Kim, J. et al. Controllable solid electrolyte interphase in nickel‐rich cathodes by an electrochemical rearrangement for stable lithium‐ion batteries. Adv. Mater. 30, 1704309 (2018).
Zhao, W. et al. High voltage operation of Ni‐rich NMC cathodes enabled by stable electrode/electrolyte interphases. Adv. Energy Mater. 8, 1800297 (2018).
Yoon, W.-S. et al. Investigation of the charge compensation mechanism on the electrochemically Li-ion deintercalated Li1–xCo1/3Ni1/3Mn1/3O2 electrode system by combination of soft and hard X-ray absorption spectroscopy. J. Am. Chem. Soc. 127, 17479–17487 (2005).
Lin, F. et al. Profiling the nanoscale gradient in stoichiometric layered cathode particles for lithium-ion batteries. Energy Environ. Sci. 7, 3077–3085 (2014).
Yang, L., Ravdel, B. & Lucht, B. L. Electrolyte reactions with the surface of high voltage LiNi0.5Mn1.5O4 cathodes for lithium-ion batteries. Electrochem. Solid-State Lett. 13, A95–A97 (2010).
Zheng, J. et al. Highly stable operation of lithium metal batteries enabled by the formation of a transient high-concentration electrolyte layer. Adv. Energy Mater. 6, 1502151 (2016).
Li, W. et al. Dynamic behaviour of interphases and its implication on high-energy-density cathode materials in lithium-ion batteries. Nat. Commun. 8, 14589 (2017).
Nguyen, T. T. D. et al. Understanding the thermal runaway of Ni-rich lithium-ion batteries. World Electr. Veh. J. 10, 79 (2019).
Lin, D., Liu, Y. & Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 12, 194–206 (2017).
Seo, D.-H. et al. The structural and chemical origin of the oxygen redox activity in layered and cation-disordered Li-excess cathode materials. Nat. Chem. 8, 692–697 (2016).
Luo, K. et al. Charge-compensation in 3d-transition-metal-oxide intercalation cathodes through the generation of localized electron holes on oxygen. Nat. Chem. 8, 684–691 (2016).
Hashigami, S. et al. Improvement of cycleability and rate-capability of LiNi0.5Co0.2Mn0.3O2 cathode materials coated with lithium boron oxide by an antisolvent precipitation method. Chem. Sel. 4, 8676–8681 (2019).
Park, J.-H. et al. Effect of residual lithium rearrangement on Ni-rich layered oxide cathodes for lithium-ion batteries. Energy Technol. 6, 1361–1369 (2018).
Kang, S. J., Mori, T., Narizuka, S., Wilcke, W. & Kim, H.-C. Deactivation of carbon electrode for elimination of carbon dioxide evolution from rechargeable lithium–oxygen cells. Nat. Commun. 5, 3937 (2014).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B: Condens. Matter Mater. Phys. 59, 1758 (1999).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Dudarev, S. L., Botton, G. A., Savrasov, S. Y., Humphreys, C. J. & Sutton, A. P. Electron-energy-loss spectra and the structural stability of nickel oxide: an LSDA + U study. Phys. Rev. B 57, 1505–1509 (1998).
Jain, A. et al. Formation enthalpies by mixing GGA and GGA + U calculations. Phys. Rev. B 84, 045115 (2011).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Acknowledgements
This work was supported by the Korea Institute of Energy Technology Evaluation and Planning (KETEP) and the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (no. 20172410100140). 2020 Research Funds (1.200029.1) of the Ulsan National Institute of Science and Technology (UNIST) is also acknowledged. Y.D. and J.L. acknowledge support from the Department of Energy, Basic Energy Sciences, under award no. DE-SC0002633 (Chemomechanics of Far-From-Equilibrium Interfaces).
Author information
Authors and Affiliations
Contributions
M.Y., Y.D., J.L. and J.C. conceived the project. M.Y. synthesized the materials and conducted the electrochemical testing. Y.D. conducted the simulations and theoretical analysis. M.Y. and J.H. conducted ex situ and in situ XRD measurements and analysis. H.C. and J.S. conducted the focused ion beam, TEM, SEM and XPS measurements. S.J.K. provided equipment for the DEMS measurements. M.Y. and K.A. assembled and tested the pouch-type full-cells. M.Y. and Y.D. analysed the data. M.Y., Y.D., J.L. and J.C. wrote the paper. All the authors discussed and contributed to the writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Energy thanks Payam Kaghazchi, David Wood III and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–33, Tables 1–9 and references.
Rights and permissions
About this article
Cite this article
Yoon, M., Dong, Y., Hwang, J. et al. Reactive boride infusion stabilizes Ni-rich cathodes for lithium-ion batteries. Nat Energy 6, 362–371 (2021). https://doi.org/10.1038/s41560-021-00782-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-021-00782-0
This article is cited by
-
Mn-based cathode materials for rechargeable batteries
Science China Chemistry (2024)
-
Defective oxygen inert phase stabilized high-voltage nickel-rich cathode for high-energy lithium-ion batteries
Nature Communications (2023)
-
Stalling oxygen evolution in high-voltage cathodes by lanthurization
Nature Energy (2023)
-
Anion-enrichment interface enables high-voltage anode-free lithium metal batteries
Nature Communications (2023)
-
Surface engineering to prevent oxygen evolution of high-voltage cathodes
Nature Energy (2023)