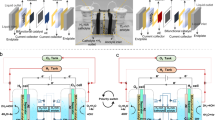
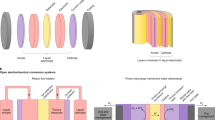
Abstract
Powered by renewable energy sources such as solar, marine, geothermal and wind, generation of storable hydrogen fuel through water electrolysis provides a promising path towards energy sustainability. However, state-of-the-art electrolysis requires support from associated processes such as desalination of water sources, further purification of desalinated water, and transportation of water, which often contribute financial and energy costs. One strategy to avoid these operations is to develop electrolysers that are capable of operating with impure water feeds directly. Here we review recent developments in electrode materials/catalysts for water electrolysis using low-grade and saline water, a significantly more abundant resource worldwide compared to potable water. We address the associated challenges in design of electrolysers, and discuss future potential approaches that may yield highly active and selective materials for water electrolysis in the presence of common impurities such as metal ions, chloride and bio-organisms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
11 May 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41560-021-00851-4
References
Vörösmarty, C. J. et al. Global threats to human water security and river biodiversity. Nature 467, 555–561 (2010).
Spröte, W. in A Concise Encyclopedia of the United Nations Vol. 07404 (ed Volger, H.) 147–152 (Brill, 2010).
The Future of Hydrogen (International Energy Agency, 2019).
Bezdek, R. H. The hydrogen economy and jobs of the future. Renew. Energy Environ. Sustain. 4, 1 (2019).
Moliner, R., Lázaro, M. J. & Suelves, I. Analysis of the strategies for bridging the gap towards the Hydrogen Economy. Int. J. Hydrog. Energy 41, 19500–19508 (2016).
Deutsch, T. G. & Turner, J. A. Semiconductor Materials for Photoelectrolysis: 2014 Annual Progress Report U. S. DOE Hydrogen & Fuel Cells Program (Department of Energy, 2014).
Ramsden, T., Ruth, M., Diakov, V., Laffen, M. & Timbario, T. A. Hydrogen Pathways: Updated Cost, Well-to-Wheels Energy Use, and Emissions for the Current Technology Status of Ten Hydrogen Production, Delivery, and Distribution Scenarios (National Renewable Energy Laboratory, 2013).
Ursúa, A., Gandía, L. M. & Sanchis, P. Hydrogen production from water electrolysis: current status and future trends. Proc. IEEE 100, 410–426 (2012).
Xiang, C., Papadantonakis, K. M. & Lewis, N. S. Principles and implementations of electrolysis systems for water splitting. Mater. Horiz. 3, 169–173 (2016).
Matute, G., Yusta, J. M. & Correas, L. C. Techno-economic modelling of water electrolysers in the range of several MW to provide grid services while generating hydrogen for different applications: a case study in Spain applied to mobility with FCEVs. Int. J. Hydrog. Energy 44, 17431–17442 (2019).
Chardonnet, C. et al. Study on Early Business Cases for H2 in Energy Storage and More Broadly Power To H2 Applications (EU Commission, 2017).
Karagiannis, I. C. & Soldatos, P. G. Water desalination cost literature: review and assessment. Desalination 223, 448–456 (2008).
Fourmond, V., Jacques, P. A., Fontecave, M. & Artero, V. H2 evolution and molecular electrocatalysts: Determination of Overpotentials and effect of homoconjugation. Inorg. Chem. 49, 10338–10347 (2010).
Bennett, J. E. Electrodes for generation of hydrogen and oxygen from seawater. Int. J. Hydrog. Energy 5, 401–408 (1980).
Katsounaros, I. et al. The effective surface pH during reactions at the solid-liquid interface. Electrochem. commun. 13, 634–637 (2011).
Auinger, M. et al. Near-surface ion distribution and buffer effects during electrochemical reactions. Phys. Chem. Chem. Phys. 13, 16384–16394 (2011).
Dionigi, F., Reier, T., Pawolek, Z., Gliech, M. & Strasser, P. Design criteria, operating conditions, and nickel-iron hydroxide catalyst materials for selective seawater electrolysis. ChemSusChem 9, 962–972 (2016). In this study, a general design criterion for oxygen-selective seawater splitting is derived from thermodynamic and kinetic considerations. Noble-metal-free NiFe double hydroxide electrocatalysts are demonstrated for selective oxygen evolution in seawater.
Kapp, E. M. The precipitation of calcium and magnesium from sea water by sodium hydroxide. Biol. Bull. 55, 453–458 (1928).
Kirk, D. W. & Ledas, A. E. Precipitate formation during sea water electrolysis. Int. J. Hydrog. Energy 7, 925–932 (1982).
Dresp, S. & Strasser, P. Non-noble metal oxides and their application as bifunctional catalyst in reversible fuel cells and rechargeable air batteries. ChemCatChem 10, 4162–4171 (2018).
Dresp, S. et al. Direct electrolytic splitting of seawater: activity, selectivity, degradation, and recovery studied from the molecular catalyst structure to the electrolyzer cell level. Adv. Energy Mater. 8, 1800338 (2018). This work reports a nanostructured NiFe-layered double hydroxide and Pt nanoparticles for electrolysis of artificial alkaline seawater. Membrane-induced stability losses are investigated and a recovery effect is identified using an on/off cycle.
Adbel-Aal, H. K. & Hussein, I. A. Parametric study for saline water electrolysis: part 1-hydrogen production. Int. J. Hydrog. Energy 18, 485–489 (1993).
Oh, B. S. et al. Formation of hazardous inorganic by-products during electrolysis of seawater as a disinfection process for desalination. Sci. Total Environ. 408, 5958–5965 (2010).
Dresp, S., Dionigi, F., Klingenhof, M. & Strasser, P. Direct electrolytic splitting of seawater: opportunities and challenges. ACS Energy Lett. 4, 933–942 (2019).
Carmo, M., Fritz, D. L., Mergel, J. & Stolten, D. A comprehensive review on PEM water electrolysis. Int. J. Hydrog. Energy 38, 4901–4934 (2013).
Vincent, I. & Bessarabov, D. Low cost hydrogen production by anion exchange membrane electrolysis: A review. Renew. Sustain. Energy Rev. 81, 1690–1704 (2018).
Meng, Y. et al. Review: recent progress in low-temperature proton-conducting ceramics. J. Mater. Sci. 54, 9291–9312 (2019).
Laguna-Bercero, M. A. Recent advances in high temperature electrolysis using solid oxide fuel cells: a review. J. Power Sources 203, 4–16 (2012).
Mauritz, K. A. & Moore, R. B. State of understanding of Nafion. Chem. Rev. 104, 4535–4586 (2004).
Chae, K. J. et al. Mass transport through a proton exchange membrane (Nafion) in microbial fuel cells. Energy Fuels 22, 169–176 (2008).
Müller, M. et al. Water management in membrane electrolysis and options for advanced plants. Int. J. Hydrog. Energy 44, 10147–10155 (2019).
Schalenbach, M., Lueke, W. & Stolten, D. Hydrogen diffusivity and electrolyte permeability of the Zirfon PERL separator for alkaline water electrolysis. J. Electrochem. Soc. 163, 1480–1488 (2016).
Lim, C. K., Liu, Q., Zhou, J., Sun, Q. & Chan, S. H. High-temperature electrolysis of synthetic seawater using solid oxide electrolyzer cells. J. Power Sources 342, 79–87 (2017).
Hine, F., O’Brien, T. F. & Bommaraju, T. V. Handbook of Chlor-alkali Technology, Volume I: Fundamentals (Springer, 2005).
Karlsson, R. K. B. & Cornell, A. Selectivity between oxygen and chlorine evolution in the chlor-alkali and chlorate processes. Chem. Rev. 116, 2982–3028 (2016).
Kumari, S., Turner White, R., Kumar, B. & Spurgeon, J. M. Solar hydrogen production from seawater vapor electrolysis. Energy Environ. Sci. 9, 1725–1733 (2016).
Heremans, G. et al. Vapor-fed solar hydrogen production exceeding 15% efficiency using earth abundant catalysts and anion exchange membrane. Sustain. Energy Fuels 1, 2061–2065 (2017).
Kida, T. et al. Water vapor electrolysis with proton-conducting graphene oxide nanosheets. ACS Sustain. Chem. Eng. 6, 11753–11758 (2018).
McCrory, C. C. L. et al. Benchmarking hydrogen evolving reaction and oxygen evolving reaction electrocatalysts for solar water splitting devices. J. Am. Chem. Soc. 137, 4347–4357 (2015).
Gong, M. et al. An advanced Ni–Fe layered double hydroxide electrocatalyst for water oxidation. J. Am. Chem. Soc. 135, 8452–8455 (2013).
Martindale, B. C. M. & Reisner, E. Bi-functional iron-only electrodes for efficient water splitting with enhanced stability through in situ electrochemical regeneration. Adv. Energy Mater. 6, 1502095 (2016). This report uses Fe-only materials as bi-functional electrocatalysts for both of the water splitting half reactions in alkaline media. The Fe materials show greater activity compared to previously reported bi-functional Co and Ni catalysts.
Huang, W.-H. & Lin, C.-Y. Iron phosphate modified calcium iron oxide as an efficient and robust catalyst in electrocatalyzing oxygen evolution from seawater. Faraday Discuss. 215, 205–215 (2019).
Juodkazytė, J. et al. Electrolytic splitting of saline water: durable nickel oxide anode for selective oxygen evolution. Int. J. Hydrog. Energy 44, 5929–5939 (2019). This study investigates a nickel oxide layer for electrocatalytic water oxidation and provides mechanistic insights into Ni(IV)-mediated electrocatalytic oxidation of water in alkaline chloride medium.
Trasatti, S. Electrocatalysis in the anodic evolution of oxygen and chlorine. Electrochim. Acta 29, 1503–1512 (1984).
Hansen, H. A. et al. Electrochemical chlorine evolution at rutile oxide (110) surfaces. Phys. Chem. Chem. Phys. 12, 283–290 (2010).
Exner, K. S., Anton, J., Jacob, T. & Over, H. Controlling selectivity in the chlorine evolution reaction over RuO2-based catalysts. Angew. Chem. Int. Ed. 53, 11032–11035 (2014).
Surendranath, Y. & Dinca, M. Electrolyte-dependent electrosynthesis and activity of cobalt-based water oxidation catalysts. J. Am. Chem. Soc. 131, 2615–2620 (2009).
Cheng, F. et al. Synergistic action of Co-Fe layered double hydroxide electrocatalyst and multiple ions of sea salt for efficient seawater oxidation at near-neutral pH. Electrochim. Acta 251, 336–343 (2017).
Zhao, Y. et al. Charge state manipulation of cobalt selenide catalyst for overall seawater electrolysis. Adv. Energy Mater. 8, 1801926 (2018).
Zeradjanin, A. R., Menzel, N., Schuhmann, W. & Strasser, P. On the faradaic selectivity and the role of surface inhomogeneity during the chlorine evolution reaction on ternary Ti-Ru-Ir mixed metal oxide electrocatalysts. Phys. Chem. Chem. Phys. 16, 13741–13747 (2014).
Macounová, K., Makarova, M., Jirkovský, J., Franc, J. & Krtil, P. Parallel oxygen and chlorine evolution on Ru1-xNixO2-y nanostructured electrodes. Electrochim. Acta 53, 6126–6134 (2008).
Kishor, K., Saha, S., Parashtekar, A. & Pala, R. G. S. Increasing chlorine selectivity through weakening of oxygen adsorbates at surface in Cu doped RuO2 during seawater electrolysis. J. Electrochem. Soc. 165, J3276–J3280 (2018).
Arikawa, T., Murakami, Y. & Takasu, Y. Simultaneous determination of chlorine and oxygen evolving at RuO2/Ti and RuO2-TiO2/Ti anodes by differential electrochemical mass spectroscopy. J. Appl. Electrochem. 28, 511–516 (1998).
Karlsson, R. K. B., Hansen, H. A., Bligaard, T., Cornell, A. & Pettersson, L. G. M. Ti atoms in Ru0.3Ti0.7O2 mixed oxides form active and selective sites for electrochemical chlorine evolution. Electrochim. Acta 146, 733–740 (2014).
Exner, K. S., Anton, J., Jacob, T. & Over, H. Chlorine evolution reaction on ruo2 (110): ab initio atomistic thermodynamics study - Pourbaix diagrams. Electrochim. Acta 120, 460–466 (2014).
Sohrabnejad-Eskan, I. et al. Temperature-dependent kinetic studies of the chlorine evolution reaction over RuO2(110) model electrodes. ACS Catal. 7, 2403–2411 (2017).
Petrykin, V., Macounova, K., Shlyakhtin, O. A. & Krtil, P. Tailoring the selectivity for electrocatalytic oxygen evolution on ruthenium oxides by zinc substitution. Angew. Chem. Int. Ed. 49, 4813–4815 (2010).
Nong, H. N. et al. A unique oxygen ligand environment facilitates water oxidation in hole-doped IrNiOx core–shell electrocatalysts. Nat. Catal. 1, 841–851 (2018).
Bergmann, A. et al. Unified structural motifs of the catalytically active state of Co(oxyhydr)oxides during the electrochemical oxygen evolution reaction. Nat. Catal. 1, 711–719 (2018).
Beermann, V. et al. Real-time imaging of activation and degradation of carbon supported octahedral Pt–Ni alloy fuel cell catalysts at the nanoscale using in situ electrochemical liquid cell STEM. Energy Environ. Sci. 12, 2476–2485 (2019).
Fabbri, E., Abbott, D. F., Nachtegaal, M. & Schmidt, T. J. Operando X-ray absorption spectroscopy: a powerful tool toward water splitting catalyst development. Curr. Opin. Electrochem. 5, 20–26 (2017).
Hsu, S.-H. et al. An earth-abundant catalyst-based seawater photoelectrolysis system with 17.9% solar-to-hydrogen efficiency. Adv. Mater. 30, 1707261 (2018). This study uses density functional theory and experimental analysis to design and test a transition metal hexacyanometallate–cobalt–carbonate/NiMoS system for selective O2 evolution. The integrated system achieves a solar-to-hydrogen efficiency of 17.9% in seawater at neutral pH.
Zeng, M. & Li, Y. Recent advances in heterogeneous electrocatalysts for the hydrogen evolution reaction. J. Mater. Chem. A 3, 14942–14962 (2015).
Vos, J. G., Wezendonk, T. A., Jeremiasse, A. W. & Koper, M. T. M. MnOx/IrOx as selective oxygen evolution electrocatalyst in acidic chloride solution. J. Am. Chem. Soc. 140, 10270–10281 (2018). This report demonstrates that deposition of MnOx onto IrOx decreases the ClER selectivity of the system in the presence of 30 mM Cl− from 86% to less than 7%, making it a highly OER-selective catalyst.
Izumiya, K. et al. Anodically deposited manganese oxide and manganese-tungsten oxide electrodes for oxygen evolution from seawater. Electrochim. Acta 43, 3303–3312 (1998).
Fujimura, K. et al. Anodically deposited manganese-molybdenum oxide anodes with high selectivity for evolving oxygen in electrolysis of seawater. J. Appl. Electrochem. 29, 765–771 (1999).
Fujimura, K. et al. Oxygen evolution on manganese-molybdenum oxide anodes in seawater electrolysis. Mater. Sci. Eng. A 267, 254–259 (1999).
Fujimura, K. et al. The durability of manganese–molybdenum oxide anodes for oxygen evolution in seawater electrolysis. Electrochim. Acta 45, 2297–2303 (2000).
El-Moneim, A. A., Kumagai, N., Asami, K. & Hashimoto, K. New nanocrystallinemanganese-molybdenum-tin oxdie anodes for oxygen evolution in seatwater electrolysis. ECS Trans. 1, 491–497 (2006).
Matsui, T. et al. Anodically deposited Mn-Mo-W oxide anodes for oxygen evolution in seawater electrolysis. J. Appl. Electrochem. 32, 993–1000 (2002).
El-Moneim, A. A. Mn-Mo-W-oxide anodes for oxygen evolution during seawater electrolysis for hydrogen production: effect of repeated anodic deposition. Int. J. Hydrog. Energy 36, 13398–13406 (2011).
Abdel Ghany, N. A., Kumagai, N., Meguro, S., Asami, K. & Hashimoto, K. Oxygen evolution anodes composed of anodically deposited Mn-Mo-Fe oxides for seawater electrolysis. Electrochim. Acta 48, 21–28 (2002).
Kato, Z., Bhattarai, J., Kumagai, N., Izumiya, K. & Hashimoto, K. Durability enhancement and degradation of oxygen evolution anodes in seawater electrolysis for hydrogen production. Appl. Surf. Sci. 257, 8230–8236 (2011).
Kato, Z. et al. Electrochemical characterization of degradation of oxygen evolution anode for seawater electrolysis. Electrochim. Acta 116, 152–157 (2014).
Kato, Z. et al. The influence of coating solution and calcination condition on the durability of Ir1-xSnxO2/Ti anodes for oxygen evolution. Appl. Surf. Sci. 388, 640–644 (2016).
Obata, K. & Takanabe, K. A Permselective CeOx coating to improve the stability of oxygen evolution electrocatalysts. Angew. Chem. Int. Ed. 57, 1616–1620 (2018).
Balaji, R. et al. An alternative approach to selective sea water oxidation for hydrogen production. Electrochem. commun. 11, 1700–1702 (2009).
Lu, X. et al. A sea-change: manganese doped nickel/nickel oxide electrocatalysts for hydrogen generation from seawater. Energy Environ. Sci. 11, 1898–1910 (2018). This study demonstrates a highly active HER catalyst electrode which exhibits Pt-like performances in both neutral electrolytes and natural seawater. The Mn doped NiO/Ni heterostructured electrode was formed by pyrolysing a Mn-MOF/Ni-F precursor in an inert environment.
Bard, A. J., Parsons, R. & Jordan, J. Standard Potentials in Aqueous Solution (Routledge, 1985).
Nørskov, J. K. et al. Trends in the exchange current for hydrogen evolution. J. Electrochem. Soc. 152, J23 (2005).
Zheng, Y., Jiao, Y., Jaroniec, M. & Qiao, S. Z. Advancing the electrochemistry of the hydrogen-evolution reaction through combining experiment and theory. Angew. Chem. Int. Ed. 54, 52–65 (2015).
Dinh, C.-T. et al. Multi-site electrocatalysts for hydrogen evolution in neutral media by destabilization of water molecules. Nat. Energy 4, 107–114 (2019).
Song, L. J. & Meng, H. M. Effect of carbon content on Ni-Fe-C electrodes for hydrogen evolution reaction in seawater. Int. J. Hydrog. Energy 35, 10060–10066 (2010).
Miao, J. et al. Hierarchical Ni-Mo-S nanosheets on carbon fiber cloth: A flexible electrode for efficient hydrogen generation in neutral electrolyte. Sci. Adv. 1, e1500259 (2015).
Schalenbach, M. et al. Gas permeation through nafion. Part 1: measurements. J. Phys. Chem. C. 119, 25145–25155 (2015).
Li, H., Tang, Q., He, B. & Yang, P. Robust electrocatalysts from an alloyed Pt-Ru-M (M = Cr, Fe, Co, Ni, Mo)-decorated Ti mesh for hydrogen evolution by seawater splitting. J. Mater. Chem. A 4, 6513–6520 (2016).
Golgovici, F. et al. Ni–Mo alloy nanostructures as cathodic materials for hydrogen evolution reaction during seawater electrolysis. Chem. Pap. 72, 1889–1903 (2018).
Zhang, Y., Li, P., Yang, X., Fa, W. & Ge, S. High-efficiency and stable alloyed nickel based electrodes for hydrogen evolution by seawater splitting. J. Alloy. Compd. 732, 248–256 (2018).
Zheng, J., Zhao, Y., Xi, H. & Li, C. Seawater splitting for hydrogen evolution by robust electrocatalysts from secondary M (M = Cr, Fe, Co, Ni, Mo) incorporated Pt. RSC Adv. 8, 9423–9429 (2018).
Raj, I. A. & Vasu, K. I. Transition metal-based hydrogen electrodes in alkaline solution — electrocatalysis on nickel based binary alloy coatings. J. Appl. Electrochem. 20, 32–38 (1990).
Esposito, D. V. Membrane-coated electrocatalysts - an alternative approach to achieving stable and tunable electrocatalysis. ACS Catal. 8, 457–465 (2018).
Vos, J. G. & Koper, M. T. M. Measurement of competition between oxygen evolution and chlorine evolution using rotating ring-disk electrode voltammetry. J. Electroanal. Chem. 819, 260–268 (2018). This report investigates the selectivity between chlorine evolution and oxygen evolution in aqueous media. Using a new method to quickly study chlorine evolution rates the authors demonstrate that oxygen evolution and chlorine evolution proceed independently on a glassy carbon supported IrOx catalyst.
Lindbergh, G. & Simonsson, D. The effect of chromate addition on cathodic reduction of hypochlorite in hydroxide and chlorate solutions. J. Electrochem. Soc. 137, 3094–3099 (2006).
Endrődi, B. et al. Towards sustainable chlorate production: The effect of permanganate addition on current efficiency. J. Clean. Prod. 182, 529–537 (2018).
Ma, Y. Y. et al. Highly efficient hydrogen evolution from seawater by a low-cost and stable CoMoP@C electrocatalyst superior to Pt/C. Energy Environ. Sci. 10, 788–798 (2017).
Kim, K., Kim, H., Lim, J. H. & Lee, S. J. Development of a desalination membrane bioinspired by mangrove roots for spontaneous filtration of sodium ions. ACS Nano 10, 11428–11433 (2016).
Kumar, A., Phillips, K. R., Thiel, G. P., Schröder, U. & Lienhard, J. H. Direct electrosynthesis of sodium hydroxide and hydrochloric acid from brine streams. Nat. Catal. 2, 106–113 (2019).
Schiermeier, Q., Tollefson, J., Scully, T., Witze, A. & Morton, O. Energy alternatives: electricity without carbon. Nature 454, 816–823 (2008).
Gao, S. et al. Electrocatalytic H2 production from seawater over Co, N-codoped nanocarbons. Nanoscale 7, 2306–2316 (2015).
Ma, Y. Y. et al. Highly efficient hydrogen evolution from seawater by a low-cost and stable CoMoP@C electrocatalyst superior to Pt/C. Energy Environ. Sci. 10, 788–798 (2017).
Zhao, Y., Tang, Q., He, B. & Yang, P. Carbide decorated carbon nanotube electrocatalyst for high-efficiency hydrogen evolution from seawater. RSC Adv. 6, 93267–93274 (2016).
Jin, H. et al. Single-crystal nitrogen-rich two-dimensional Mo5N6 nanosheets for efficient and stable seawater splitting. ACS Nano 12, 12761–12769 (2018).
Sun, Y. et al. Electrodeposited cobalt-sulfide catalyst for electrochemical and photoelectrochemical hydrogen generation from water. J. Am. Chem. Soc. 135, 17699–17702 (2013).
Zhao, Y., Jin, B., Vasileff, A., Jiao, Y. & Qiao, S. Interfacial nickel nitride/sulfide as a bifunctional electrode for highly efficient overall water/seawater electrolysis. J. Mater. Chem. A 7, 8117–8121 (2019). This study describes a bifunctional NiNS electrocatalyst for overall water splitting near neutral pH and in seawater.
Acknowledgements
W.T., M.F., R.S.E., A.J.C. and P.F. acknowledge financial support from INTERREG Atlantic Area programme (Grant reference EAPA_190_2016). P.F. acknowledges support from Royal Society Alumni programme. F.D., S.D. and P.S. gratefully acknowledge financial support by the German Research Foundation (DFG) through Grant reference number STR 596/8-1 and the federal ministry for economic affairs and energy (Bundesministerium für Wirtschaft und Energie, BMWi) under grant number 03EIV041F. P.S. acknowledges partial funding by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany´s Excellence Strategy – EXC 2008/1 – 390540038 (zum Teil gefördert durch die Deutsche Forschungsgemeinschaft (DFG) im Rahmen der Exzellenzstrategie des Bundes und der Länder – EXC 2008/1 – 390540038).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tong, W., Forster, M., Dionigi, F. et al. Electrolysis of low-grade and saline surface water. Nat Energy 5, 367–377 (2020). https://doi.org/10.1038/s41560-020-0550-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-020-0550-8
This article is cited by
-
Ultrastable electrocatalytic seawater splitting at ampere-level current density
Nature Sustainability (2024)
-
A silicon photoanode protected with TiO2/stainless steel bilayer stack for solar seawater splitting
Nature Communications (2024)
-
Durable electrocatalysts for seawater splitting at the ampere level
Nature Sustainability (2024)
-
High-entropy materials for energy and electronic applications
Nature Reviews Materials (2024)
-
Efficient bubble/precipitate traffic enables stable seawater reduction electrocatalysis at industrial-level current densities
Nature Communications (2024)