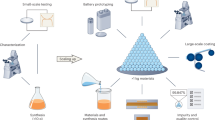
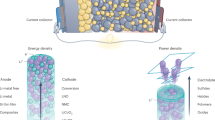
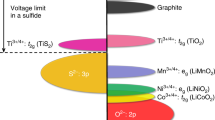
Abstract
Lithium-ion batteries are currently the most advanced electrochemical energy storage technology due to a favourable balance of performance and cost properties. Driven by forecasted growth of the electric vehicles market, the cell production capacity for this technology is continuously being scaled up. However, the demand for better performance, particularly higher energy densities and/or lower costs, has triggered research into post-lithium-ion technologies such as solid-state lithium metal, lithium–sulfur and lithium–air batteries as well as post-lithium technologies such as sodium-ion batteries. Currently, these technologies are being intensively studied with regard to material chemistry and cell design. In this Review, we expand on the current knowledge in this field. Starting with a market outlook and an analysis of technological differences, we discuss the manufacturing processes of these technologies. For each technology, we describe anode production, cathode production, cell assembly and conditioning. We then evaluate the manufacturing compatibility of each technology with the lithium-ion production infrastructure and discuss the implications for processing costs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Placke, T., Kloepsch, R., Dühnen, S. & Winter, M. Lithium ion, lithium metal, and alternative rechargeable battery technologies: the odyssey for high energy density. J. Solid State Electrochem. 21, 1939–1964 (2017). Comprehensively reviews the history of battery technologies and offers perspectives of lithium-ion and post-lithium-ion batteries.
Winter, M., Barnett, B. & Xu, K. Before Li ion batteries. Chem. Rev. 118, 11433–11456 (2018).
Duffner, F., Wentker, M., Greenwood, M. & Leker, J. Battery cost modeling: a review and directions for future research. Renew. Sustain. Energy Rev. 127, 109872 (2020). An in-depth analysis of the battery cost modelling literature based on various characteristics and identifying a trend towards state-of-the-art cell costs between US$100 kWh−1 and US$150 kWh−1.
Pillot, C. The Rechargeable Battery Market and Main Trends 2017–2030 (Avicenne Energy, 2019).
Vaalma, C., Buchholz, D., Weil, M. & Passerini, S. A cost and resource analysis of sodium-ion batteries. Nat. Rev. Mater. 3, 18013 (2018). An in-depth overview of SIBs, analysing the material and production properties and quantifying their implications for costs and cell performance.
Nanda, S., Gupta, A. & Manthiram, A. Anode-free full cells: a pathway to high-energy density lithium-metal batteries. Adv. Energy Mater. https://doi.org/10.1002/aenm.202000804 (2020).
Gallagher, K. G. et al. Quantifying the promise of lithium–air batteries for electric vehicles. Energy Environ. Sci. 7, 1555–1563 (2014).
Hagen, M. et al. Lithium–sulfur cells: the gap between the state-of-the-art and the requirements for high energy battery cells. Adv. Energy Mater. 5, 1401986 (2015).
Mineral Commodity Summaries (US Department of the Interior, US Geological Survey, 2018).
Schnell, J. et al. All-solid-state lithium-ion and lithium metal batteries—paving the way to large-scale production. J. Power Sources 382, 160–175 (2018).
Randau, S. et al. Benchmarking the performance of all-solid-state lithium batteries. Nat. Energy 5, 259–270 (2020). In-depth benchmarking analysis of all-solid-state battery performance data reported in the literature in various configurations.
Park, J. O. et al. A 1,000 Wh kg−1 Li–air battery: cell design and performance. J. Power Sources 419, 112–118 (2019).
Schmuch, R., Wagner, R., Hörpel, G., Placke, T. & Winter, M. Materials for automotive batteries: perspective on performance and cost of lithium-based rechargeable batteries. Nat. Energy 3, 267–278 (2018). Review of current and future materials for automotive batteries with regard to their production as well as performance and cost.
Arinicheva, Y. et al. in Advanced Ceramics for Energy Conversion and Storage (ed. Guillon, O.) 549–709 (Elsevier, 2020).
Andre, D., Hain, H., Lamp, P., Maglia, F. & Stiaszny, B. Future high-energy density anode materials from an automotive application perspective. J. Mater. Chem. A 5, 17174–17198 (2017).
Myung, S. T. et al. Nickel-rich layered cathode materials for automotive lithium-ion batteries: achievements and perspectives. ACS Energy Lett. 2, 196–223 (2017).
Scrosati, B., Garche, J. & Tillmetz, W. Advances in Battery Technologies for Electric Vehicles (Elsevier, 2015).
Blomgren, G. E. The development and future of lithium ion batteries. J. Electrochem. Soc. 164, A5019–A5025 (2017).
Lee, H., Yanilmaz, M., Toprakci, O., Fu, K. & Zhang, X. A review of recent developments in membrane separators for rechargeable lithium-ion batteries. Energy Environ. Sci. 7, 3857–3886 (2014).
Nayak, P. K., Yang, L., Brehm, W. & Adelhelm, P. From lithium-ion to sodium-ion batteries: advantages, challenges, and surprises. Angew. Chem. Int. Ed. 57, 102–120 (2018).
Adelhelm, P. et al. From lithium to sodium: cell chemistry of room temperature sodium–air and sodium–sulfur batteries. Beilstein J. Nanotechnol. 6, 1016–1055 (2015).
Pang, Q., Liang, X., Kwok, C. Y. & Nazar, L. F. Advances in lithium–sulfur batteries based on multifunctional cathodes and electrolytes. Nat. Energy 1, 1–11 (2016).
Betz, J. et al. Theoretical versus practical energy: a plea for more transparency in the energy calculation of different rechargeable battery systems. Adv. Energy Mater. 9, 1803170 (2019).
Janek, J. & Zeier, W. G. A solid future for battery development. Nat. Energy 1, 1–4 (2016).
Bachman, J. C. et al. Inorganic solid-state electrolytes for lithium batteries: mechanisms and properties governing ion conduction. Chem. Rev. 116, 140–162 (2016).
Famprikis, T., Canepa, P., Dawson, J. A., Islam, M. S. & Masquelier, C. Fundamentals of inorganic solid-state electrolytes for batteries. Nat. Mater. 18, 1278–1291 (2019).
Takada, K. Progress in solid electrolytes toward realizing solid-state lithium batteries. J. Power Sources 394, 74–85 (2018).
Bruce, P. G., Freunberger, S. A., Hardwick, L. J. & Tarascon, J.-M. Li–O2 and Li–S batteries with high energy storage. Nat. Mater. 11, 19–29 (2012). Provides a comprehensive technological overview of LSBs and LABs and presents the main challenges to overcome to use these technologies in mass applications.
Choi, J. W. & Aurbach, D. Promise and reality of post-lithium-ion batteries with high energy densities. Nat. Rev. Mater. 1 16013 (2016).
Mizuno, F., Nakanishi, S., Kotani, Y., Yokoishi, S. & Iba, H. Rechargeable Li–air batteries with carbonate-based liquid electrolytes. Electrochemistry 78, 403–405 (2010).
Liu, Q.-C. et al. A flexible and wearable lithium–oxygen battery with record energy density achieved by the interlaced architecture inspired by bamboo slips. Adv. Mater. 28, 8413–8418 (2016).
Asadi, M. et al. A lithium–oxygen battery with a long cycle life in an air-like atmosphere. Nature 555, 502–506 (2018).
Kwade, A. et al. Current status and challenges for automotive battery production technologies. Nat. Energy 3, 290–300 (2018). Offers an in-depth description of lithium-ion production steps, focusing on technical properties, production costs, scale-up issues and future process innovations.
Turetskyy, A. et al. Toward data‐driven applications in lithium‐ion battery cell manufacturing. Energy Technol. 8, 1900136 (2020).
Dreger, H., Bockholt, H., Haselrieder, W. & Kwade, A. Discontinuous and continuous processing of low-solvent battery slurries for lithium nickel cobalt manganese oxide electrodes. J. Electron. Mater. 44, 4434–4443 (2015).
Efficient Battery Electrode Slurry Production (Bühler Group, 2017).
Zhang, Z., Zeng, T., Lai, Y., Jia, M. & Li, J. A comparative study of different binders and their effects on electrochemical properties of LiMn2O4 cathode in lithium ion batteries. J. Power Sources 247, 1–8 (2014).
Rollag, K., Juarez-Robles, D., Du, Z., Wood, D. L. & Mukherjee, P. P. Drying temperature and capillarity-driven crack formation in aqueous processing of Li-ion battery electrodes. ACS Appl. Energy Mater. 2, 4464–4476 (2019).
Hawley, W. B. & Li, J. Electrode manufacturing for lithium-ion batteries—analysis of current and next generation processing. J. Energy Storage 25, 100862 (2019).
Flynn, J. C. & Marsh, C. Development and experimental results of continuous coating technology for lithium-ion electrodes. In Proc. 13th Annual Battery Conference on Applications and Advances (eds Frank, H. A. & Seo, E. T.) 81–84 (IEEE, 1998).
Song, Z. et al. Origami lithium-ion batteries. Nat. Commun. 5, 3140 (2014).
Schmitt, M., Scharfer, P. & Schabel, W. Slot die coating of lithium-ion battery electrodes: investigations on edge effect issues for stripe and pattern coatings. J. Coat. Technol. Res. 11, 57–63 (2014).
Carvalho, M. S. & Kheshgi, H. S. Low-flow limit in slot coating: theory and experiments. AIChE J. 46, 1907–1917 (2000).
Romero, O. J., Suszynski, W. J., Scriven, L. E. & Carvalho, M. S. Low-flow limit in slot coating of dilute solutions of high molecular weight polymer. J. Nonnewton. Fluid Mech. 118, 137–156 (2004).
Jaiser, S., Friske, A., Baunach, M., Scharfer, P. & Schabel, W. Development of a three-stage drying profile based on characteristic drying stages for lithium-ion battery anodes. Dry. Technol. 35, 1266–1275 (2017).
Jiang, Z., Zhao, F., Guan, Y. & Qiu, Z. Research on vacuum drying process and internal heat conduction of Li-ion battery core. Theor. Appl. Mech. Lett. 9, 120–129 (2019).
Meyer, C., Bockholt, H., Haselrieder, W. & Kwade, A. Characterization of the calendering process for compaction of electrodes for lithium-ion batteries. J. Mater. Process. Technol. 249, 172–178 (2017).
Takeda, O. et al. Electrowinning of lithium from LiOH in molten chloride. J. Electrochem. Soc. 161, D820–D823 (2014).
Meyer, H. C. Jr in Handling and Uses of the Alkali Metals Vol. 19, Ch. 2 (ACS, 1957).
Wietelmann, U. Surface-passivated lithium metal and method for the production thereof. US patent 13,515,579 (2012).
Schnell, J. et al. Prospects of production technologies and manufacturing costs of oxide-based all-solid-state lithium batteries. Energy Environ. Sci. 12, 1818–1833 (2019). Evaluates and identifies production technologies for oxidic SSBs, focusing on the ceramic processing technologies required to produce SEs and composite cathodes.
Lee, H., Song, J., Kim, Y.-J., Park, J.-K. & Kim, H.-T. Structural modulation of lithium metal–electrolyte interface with three-dimensional metallic interlayer for high-performance lithium metal batteries. Sci. Rep. 6, 30830 (2016).
Fu, J. et al. In situ formation of a bifunctional interlayer enabled by a conversion reaction to initiatively prevent lithium dendrites in a garnet solid electrolyte. Energy Environ. Sci. 12, 1404–1412 (2019).
Tsai, C.-L. et al. High conductivity of mixed phase Al-substituted Li7La3Zr2O12. J. Electroceram. 35, 25–32 (2015).
Jin, S. et al. Solid–solution-based metal alloy phase for highly reversible lithium metal anode. J. Am. Chem. Soc. 142, 8818–8826 (2020).
Qian, J. et al. Anode-free rechargeable lithium metal batteries. Adv. Funct. Mater. 26, 7094–7102 (2016).
Teragawa, S., Aso, K., Tadanaga, K., Hayashi, A. & Tatsumisago, M. Liquid-phase synthesis of a Li3PS4 solid electrolyte using N-methylformamide for all-solid-state lithium batteries. J. Mater. Chem. A 2, 5095–5099 (2014).
Kotobuki, M., Munakata, H., Kanamura, K., Sato, Y. & Yoshida, T. Compatibility of Li7La3Zr2O12 solid electrolyte to all-solid-state battery using Li metal anode. J. Electrochem. Soc. 157, A1076 (2010).
Ahmed, S., Nelson, P. A. & Dees, D. W. Study of a dry room in a battery manufacturing plant using a process model. J. Power Sources 326, 490–497 (2016).
Jung, Y. S., Oh, D. Y., Nam, Y. J. & Park, K. H. Issues and challenges for bulk-type all-solid-state rechargeable lithium batteries using sulfide solid electrolytes. Isr. J. Chem. 55, 472–485 (2015).
Sahu, G. et al. Air-stable, high-conduction solid electrolytes of arsenic-substituted Li4 SnS4. Energy Environ. Sci. 7, 1053–1058 (2014).
Muramatsu, H., Hayashi, A., Ohtomo, T., Hama, S. & Tatsumisago, M. Structural change of Li2S–P2S5 sulfide solid electrolytes in the atmosphere. Solid State Ion. 182, 116–119 (2011).
Li, Y., Han, J.-T., Wang, C.-A., Xie, H. & Goodenough, J. B. Optimizing Li+ conductivity in a garnet framework. J. Mater. Chem. 22, 15357–15361 (2012).
Miara, L. et al. About the compatibility between high voltage spinel cathode materials and solid oxide electrolytes as a function of temperature. ACS Appl. Mater. Interfaces 8, 26842–26850 (2016).
Xia, W. et al. Reaction mechanisms of lithium garnet pellets in ambient air: the effect of humidity and CO2. J. Am. Ceram. Soc. 100, 2832–2839 (2017).
Hanft, D., Exner, J. & Moos, R. Thick-films of garnet-type lithium ion conductor prepared by the Aerosol Deposition Method: the role of morphology and annealing treatment on the ionic conductivity. J. Power Sources 361, 61–69 (2017).
Schnell, J., Knörzer, H., Imbsweiler, A. J. & Reinhart, G. Solid versus liquid—a bottom‐up calculation model to analyze the manufacturing cost of future high‐energy batteries. Energy Technol. 8, 1901237 (2020).
Ma, Z. et al. A review of cathode materials and structures for rechargeable lithium–air batteries. Energy Environ. Sci. 8, 2144–2198 (2015).
McCloskey, B. D. et al. Limitations in rechargeability of Li–O2 batteries and possible origins. J. Phys. Chem. Lett. 3, 3043–3047 (2012).
Padbury, R. & Zhang, X. Lithium–oxygen batteries—limiting factors that affect performance. J. Power Sources 196, 4436–4444 (2011).
Ludwig, B., Zheng, Z., Shou, W., Wang, Y. & Pan, H. Solvent-free manufacturing of electrodes for lithium-ion batteries. Sci. Rep. 6, 23150 (2016). Presents a method for solvent-free electrode production using electrostatic spraying and hot rolling as a basis for materials dispensing and binder activation.
Al-Shroofy, M. et al. Solvent-free dry powder coating process for low-cost manufacturing of LiNi1/3Mn1/3Co1/3O2 cathodes in lithium-ion batteries. J. Power Sources 352, 187–193 (2017).
Park, D.-W., Cañas, N. A., Wagner, N. & Friedrich, K. A. Novel solvent-free direct coating process for battery electrodes and their electrochemical performance. J. Power Sources 306, 758–763 (2016).
Bailey, A. G. The science and technology of electrostatic powder spraying, transport and coating. J. Electrostat. 45, 85–120 (1998).
Yan, B., Liu, J., Song, B., Xiao, P. & Lu, L. Li-rich thin film cathode prepared by pulsed laser deposition. Sci. Rep. 3, 3332 (2013).
Baggetto, L., Unocic, R. R., Dudney, N. J. & Veith, G. M. Fabrication and characterization of Li–Mn–Ni–O sputtered thin film high voltage cathodes for Li-ion batteries. J. Power Sources 211, 108–118 (2012).
Koike, S. & Tatsumi, K. Preparation and performances of highly porous layered LiCoO2 films for lithium batteries. J. Power Sources 174, 976–980 (2007).
Reinhart, G. et al. in Future Trends in Production Engineering (eds Schuh, G. et al.) 3–12 (Springer, 2013).
Schilling, A., Schmitt, J., Dietrich, F. & Dröder, K. Analyzing bending stresses on lithium-ion battery cathodes induced by the assembly process. Energy Technol. 4, 1502–1508 (2016).
Zhang, S. S. A review on the separators of liquid electrolyte Li-ion batteries. J. Power Sources 164, 351–364 (2007).
Baumeister, M. & Fleischer, J. Integrated cut and place module for high productive manufacturing of lithium-ion cells. CIRP Ann. 63, 5–8 (2014).
Lee, S. S., Kim, T. H., Hu, S. J., Cai, W. W. & Abell, J. A. Joining technologies for automotive lithium-ion battery manufacturing: a review. In Proc. ASME International Manufacturing Science and Engineering Conference Vol. 1, 541–549 (ASMEDC, 2010).
Günter, F. J. et al. Influence of the cell format on the electrolyte filling process of lithium‐ion cells. Energy Technol. 8, 1801108 (2019).
Günter, F. J., Burgstaller, C., Konwitschny, F. & Reinhart, G. Influence of the electrolyte quantity on lithium-ion cells. J. Electrochem. Soc. 166, A1709–A1714 (2019).
Lamination and Stacking of Cells (Manz, 2019).
Sakti, A., Michalek, J. J., Fuchs, E. R. H. & Whitacre, J. F. A techno-economic analysis and optimization of Li-ion batteries for light-duty passenger vehicle electrification. J. Power Sources 273, 966–980 (2015).
Devine, J. in ASM Handbook: Welding, Brazing, and Soldering Vol. 6 (eds Olson, D. L. et al.) 324–327 (1993).
McGrogan, F. P. et al. Compliant yet brittle mechanical behavior of Li2S-P2S5 lithium-ion-conducting solid electrolyte. Adv. Energy Mater. 7, 1602011 (2017).
Hao, F., Han, F., Liang, Y., Wang, C. & Yao, Y. Architectural design and fabrication approaches for solid-state batteries. MRS Bull. 43, 775–781 (2018).
An, S. J. et al. The state of understanding of the lithium-ion-battery graphite solid electrolyte interphase (SEI) and its relationship to formation cycling. Carbon 105, 52–76 (2016).
Wood, D. L., Li, J. & Daniel, C. Prospects for reducing the processing cost of lithium ion batteries. J. Power Sources 275, 234–242 (2015).
Arora, P., White, R. E. & Doyle, M. Capacity fade mechanisms and side reactions in lithium-ion batteries. J. Electrochem. Soc. 145, 3647–3667 (1998).
Bieker, G., Winter, M. & Bieker, P. Electrochemical in situ investigations of SEI and dendrite formation on the lithium metal anode. Phys. Chem. Chem. Phys. 17, 8670–8679 (2015).
Cheng, X.-B. et al. A review of solid electrolyte interphases on lithium metal anode. Adv. Sci. 3, 1500213 (2016).
Peled, E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems—the solid electrolyte interphase model. J. Electrochem. Soc. 126, 2047–2051 (1979).
Mao, C. et al. Balancing formation time and electrochemical performance of high energy lithium-ion batteries. J. Power Sources 402, 107–115 (2018).
An, S. J., Li, J., Du, Z., Daniel, C. & Wood, D. L. Fast formation cycling for lithium ion batteries. J. Power Sources 342, 846–852 (2017).
Kato, Y. et al. High-power all-solid-state batteries using sulfide superionic conductors. Nat. Energy 1, 16030 (2016).
Tagawa, K. & Brodd, R. J. in Lithium-Ion Batteries (eds Yoshio M. et al.) 181–194 (Springer, 2009).
Verma, P., Maire, P. & Novák, P. A review of the features and analyses of the solid electrolyte interphase in Li-ion batteries. Electrochim. Acta 55, 6332–6341 (2010).
London Metal Exchange (LME, accessed 30 January 2020); https://www.lme.com/
Duffner, F., Mauler, L., Wentker, M., Leker, J. & Winter, M. Large-scale automotive battery cell manufacturing: analyzing strategic and operational effects on manufacturing costs. Intern. J. Prod. Econ. 232, 107982 (2021). Forecast of large-scale lithium-ion battery manufacturing costs based on more than 250 parameters relating to technical parameters, product characteristics, operating conditions and factor prices.
Cano, Z. P. et al. Batteries and fuel cells for emerging electric vehicle markets. Nat. Energy 3, 279–289 (2018).
Manthiram, A., Yu, X. & Wang, S. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2, 16103 (2017).
Lee, Y.-G. et al. High-energy long-cycling all-solid-state lithium metal batteries enabled by silver–carbon composite anodes. Nat. Energy 5, 299–308 (2020).
Lee, H. C. et al. High-energy-density Li–O2 battery at cell scale with folded cell structure. Joule 3, 542–556 (2019).
Aurbach, D., McCloskey, B. D., Nazar, L. F. & Bruce, P. G. Advances in understanding mechanisms underpinning lithium–air batteries. Nat. Energy 1, 1–11 (2016).
Moores, S. Dawn of the Battery Megafactories and the Impact on Industrial Minerals (Benchmark Mineral Intelligence, 2019).
Schünemann, J. H. Modell zur Bewertung der Herstellkosten von Lithiumionenbatteriezellen. PhD thesis, Technische Univ. Braunschweig (2015).
Acknowledgements
This work was supported by the German Ministry of Education and Research (BMBF) through the project ProLiFest (03XP0253A). We acknowledge A. Bar for the assistance in preparing Figs. 1–5.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
F.D. and N.K. are employees at Porsche Consulting GmbH. All other authors have no competing interests.
Additional information
Peer review information Nature Energy thanks Jianlin Li, Yan Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Duffner, F., Kronemeyer, N., Tübke, J. et al. Post-lithium-ion battery cell production and its compatibility with lithium-ion cell production infrastructure. Nat Energy 6, 123–134 (2021). https://doi.org/10.1038/s41560-020-00748-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-020-00748-8
This article is cited by
-
Improving rechargeable magnesium batteries through dual cation co-intercalation strategy
Nature Communications (2024)
-
Cost and performance analysis as a valuable tool for battery material research
Nature Reviews Materials (2024)
-
Metal electrodes for next-generation rechargeable batteries
Nature Reviews Electrical Engineering (2024)
-
Recycling of solid-state batteries
Nature Energy (2024)
-
A hybrid four-stage detection model to pre-identify the sustainable manufacturing process of Li-ion battery pack
The Journal of Supercomputing (2024)