Abstract
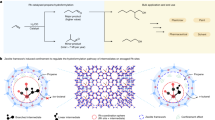
Catalytic hydrodeoxygenation is an essential step for bio-oil upgrading. However, hydrodeoxygenation usually requires a high hydrogen pressure and high temperature due to the good stability of the C–O bonds. Here we report an effective multiphase hydrodeoxygenation of lignin-based bio-oil at temperatures <100 °C and hydrogen pressures <1 atm using a synergetic catalyst system that consists of a low redox potential H4SiW12O40 (SiW12) and suspended Pt-on-carbon (Pt/C) particles. We propose that SiW12 plays three critical roles in bio-oil hydrodeoxygenation. First, it quickly oxidizes the H2 gas to form reduced SiW12 in the presence of Pt/C. Second, it transfers both electrons and H+ ions to the bulk phase to form active H* or H2 on the Pt/C surface. Third, the formation of the oxonium ion in a SiW12 superacid solution reduces the deoxygenation energy. The SiW12-enhanced proton-transfer hydrodeoxygenation mechanism is supported by density functional theory computations. As a result of the hydrogen buffer and acidic effect, up to a 90% yield of hydrocarbons (cyclohexane, benzene and their derivatives) was achieved from the hydrodeoxygenation of phenol and its derivatives.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available in the Article and Supplementary Information. Source data are provided with this paper.
References
Armstrong, R. C. et al. The frontiers of energy. Nat. Energy 1, 15020 (2016).
Liu, B. & Rajagopal, D. Life-cycle energy and climate benefits of energy recovery from wastes and biomass residues in the United States. Nat. Energy 4, 700–708 (2019).
Savage, N. Fuel options: the ideal biofuel. Nature 474, S9–S11 (2011).
Kunkes, E. L. et al. Catalytic conversion of biomass to monofunctional hydrocarbons and targeted liquid-fuel classes. Science 322, 417–421 (2008).
Ragauskas, A. J. et al. Lignin valorization: improving lignin processing in the biorefinery. Science 344, 1246843 (2014).
Duan, H. et al. Hydrodeoxygenation of water-insoluble bio-oil to alkanes using a highly dispersed Pd–Mo catalyst. Nat. Commun. 8, 591 (2017).
Xia, Q. et al. Direct hydrodeoxygenation of raw woody biomass into liquid alkanes. Nat. Commun. 7, 11162 (2016).
Saidi, M. et al. Upgrading of lignin-derived bio-oils by catalytic hydrodeoxygenation. Energ. Environ. Sci. 7, 103–129 (2014).
Lin, Z., Chen, R., Qu, Z. & Chen, J. G. Hydrodeoxygenation of biomass-derived oxygenates over metal carbides: from model surfaces to powder catalysts. Green Chem. 20, 2679–2696 (2018).
Mu, W., Ben, H., Ragauskas, A. & Deng, Y. Lignin pyrolysis components and upgrading—technology review. BioEnergy Res. 6, 1183–1204 (2013).
Jing, Y., Dong, L., Guo, Y., Liu, X. & Wang, Y. Chemicals from lignin: a review of the catalytic conversion involving hydrogen. ChemSusChem 13, 1–19 (2020).
Shafaghat, H., Rezaei, P. S. & Daud, W. M. A. W. Effective parameters on selective catalytic hydrodeoxygenation of phenolic compounds of pyrolysis bio-oil to high-value hydrocarbons. RSC Adv. 5, 103999–104042 (2015).
Wang, H., Male, J. & Wang, Y. Recent advances in hydrotreating of pyrolysis bio-oil and its oxygen-containing model compounds. ACS Catal. 3, 1047–1070 (2013).
Pritchard, J., Filonenko, G. A., van Putten, R., Hensen, E. J. M. & Pidko, E. A. Heterogeneous and homogeneous catalysis for the hydrogenation of carboxylic acid derivatives: history, advances and future directions. Chem. Soc. Rev. 44, 3808–3833 (2015).
Kozhevnikov, I. V. Advances in catalysis by heteropolyacids. Russian Chem. Rev. 56, 811–825 (1987).
Rausch, B., Symes, M. D., Chisholm, G. & Cronin, L. Decoupled catalytic hydrogen evolution from a molecular metal oxide redox mediator in water splitting. Science 345, 1326–1330 (2014).
Yang, G. et al. The nature of hydrogen adsorption on platinum in the aqueous phase. Angew. Chem. Int. Ed. 58, 3527–3532 (2019).
Singh, N. et al. Impact of pH on aqueous-phase phenol hydrogenation catalyzed by carbon-supported Pt and Rh. ACS Catal. 9, 1120–1128 (2019).
Zhao, C., He, J., Lemonidou, A. A., Li, X. & Lercher, J. A. Aqueous-phase hydrodeoxygenation of bio-derived phenols to cycloalkanes. J. Catal. 280, 8–16 (2011).
Güvenatam, B., Kurşun, O., Heeres, E. H. J., Pidko, E. A. & Hensen, E. J. M. Hydrodeoxygenation of mono- and dimeric lignin model compounds on noble metal catalysts. Catal. Today 233, 83–91 (2014).
Ohta, H. et al. Low temperature hydrodeoxygenation of phenols under ambient hydrogen pressure to form cyclohexanes catalysed by Pt nanoparticles supported on H-ZSM-5. Chem. Commun. 51, 17000–17003 (2015).
Alshehri, F., Feral, C., Kirkwood, K. & Jackson, S. D. Low temperature hydrogenation and hydrodeoxygenation of oxygen-substituted aromatics over Rh/silica: part 1: phenol, anisole and 4-methoxyphenol. React. Kinet. Mech. Catal. 128, 23–40 (2019).
Chen, L., Fink, C., Fei, Z., Dyson, P. J. & Laurenczy, G. An efficient Pt nanoparticle–ionic liquid system for the hydrodeoxygenation of bio-derived phenols under mild conditions. Green Chem. 19, 5435–5441 (2017).
Ohta, H. et al. Surface modification of a supported Pt catalyst using ionic liquids for selective hydrodeoxygenation of phenols into arenes under mild conditions. Chem. Eur. J. 25, 14762–14766 (2019).
He, Z. & Wang, X. Hydrodeoxygenation of model compounds and catalytic systems for pyrolysis bio-oils upgrading. Catal. Sustain. Energy 1, 28–52 (2012).
Long, W. et al. Different crystal form titania supported ruthenium nanoparticles for liquid phase hydrodeoxygenation of guaiacol. J. Nanosci. Nanotechnol. 18, 8426–8436 (2018).
Si, Z., Zhang, X., Wang, C., Ma, L. & Dong, R. An overview on catalytic hydrodeoxygenation of pyrolysis oil and its model compounds. Catalysts 7, 169 (2017).
Mäki-Arvela, P. & Murzin, Y. D. Hydrodeoxygenation of lignin-derived phenols: from fundamental studies towards industrial applications. Catalysts 7, 265 (2017).
Garcia-Pintos, D., Voss, J., Jensen, A. D. & Studt, F. Hydrodeoxygenation of phenol to benzene and cyclohexane on Rh(111) and Rh(211) surfaces: insights from density functional theory. J. Phys. Chem. C 120, 18529–18537 (2016).
Wang, M., Shi, H., Camaioni, D. M. & Lercher, J. A. Palladium-catalyzed hydrolytic cleavage of aromatic C−O bonds. Angew. Chem. Int. Ed. 56, 2110–2114 (2017).
Yoon, Y., Rousseau, R., Weber, R. S., Mei, D. & Lercher, J. A. First-principles study of phenol hydrogenation on Pt and Ni catalysts in aqueous phase. J. Am. Chem. Soc. 136, 10287–10298 (2014).
Liu, D. et al. Competition and cooperation of hydrogenation and deoxygenation reactions during hydrodeoxygenation of phenol on Pt(111). J. Phys. Chem. C 121, 12249–12260 (2017).
Hensley, A. J. R., Wang, Y. & McEwen, J.-S. Phenol deoxygenation mechanisms on Fe(110) and Pd(111). ACS Catal. 5, 523–536 (2015).
Jordan, K. D., Michejda, J. A. & Burrow, P. D. Electron transmission studies of the negative ion states of substituted benzenes in the gas phase. J. Am. Chem. Soc. 98, 7189–7191 (1976).
Jiang, G., Nowakowski, D. J. & Bridgwater, A. V. Effect of the temperature on the composition of lignin pyrolysis products. Energy Fuel. 24, 4470–4475 (2010).
Staš, M., Chudoba, J., Kubička, D., Blažek, J. & Pospíšil, M. Petroleomic characterization of pyrolysis bio-oils: a review. Energy Fuel 31, 10283–10299 (2017).
Pourzolfaghar, H., Abnisa, F., Wan Daud, W. M. A. & Aroua, M. K. Atmospheric hydrodeoxygenation of bio-oil oxygenated model compounds: ae review. J. Anal. Appl. Pyrol. 133, 117–127 (2018).
Zhang, C. et al. Promoting lignin depolymerization and restraining the condensation via an oxidation−hydrogenation strategy. ACS Catal. 7, 3419–3429 (2017).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Mathew, K., Sundararaman, R., Letchworth-Weaver, K., Arias, T. A. & Hennig, R. G. Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J. Chem. Phys. 140, 084106 (2014).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Acknowledgements
W.L. thanks the RBI at Georgia Tech for scholarship support.
Author information
Authors and Affiliations
Contributions
Y.D. and W.L. conceived the project and designed the experiments. W.L. performed the electrochemical measurement and HDO experiments. W.S. and W. Yang helped with the HDO experiments and product analysis by GC. W. You and W.L. conducted the DFT calculations of proton-induced mechanisms. A.K. performed GC–mass spectrometry analysis of the HDO products. Y.G. performed the catalyst characterizations.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–5, Figs. 1–40 and refs. 1–55.
Supplementary Data
Source data for the TOFs of hydrocarbon generation during the phenol hydrodeoxygenation with different Pt-to-phenol ratios.
Source data
Source Data Fig. 3b
Source data for hydrodeoxygenations of phenol (at 75 °C) and guaiacol (at 95 °C) using N2 and H2 mixing gas (total pressure 1 atm).
Rights and permissions
About this article
Cite this article
Liu, W., You, W., Sun, W. et al. Ambient-pressure and low-temperature upgrading of lignin bio-oil to hydrocarbons using a hydrogen buffer catalytic system. Nat Energy 5, 759–767 (2020). https://doi.org/10.1038/s41560-020-00680-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-020-00680-x
This article is cited by
-
Selective hydrodeoxygenation of guaiacol to cyclohexanol using activated hydrochar-supported Ru catalysts
Frontiers of Chemical Science and Engineering (2024)
-
Electrocatalytic valorization of lignocellulose-derived aromatics at industrial-scale current densities
Nature Communications (2023)
-
Metal–support cooperation in Al(PO3)3-supported platinum nanoparticles for the selective hydrogenolysis of phenols to arenes
Nature Catalysis (2021)