Abstract
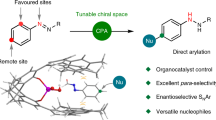
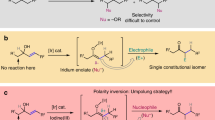
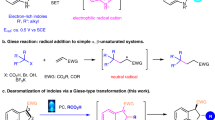
The umpolung functionalization of imines bears vast synthetic potential, but polarity inversion is less efficient compared with the carbonyl counterparts. Strong nucleophiles are often required to react with the N-electrophiles without catalytic and stereochemical control. Here we show an effective strategy to realize umpolung of imines promoted by organocatalytic aromatization. The attachment of strongly electron-withdrawing groups to imines could enhance the umpolung reactivity by both electronegativity and aromatic character, enabling the direct amination of (hetero)arenes with good efficiencies and stereoselectivities. Additionally, the application of chiral Brønsted acid catalyst furnishes (hetero)aryl C–N atropisomers or enantioenriched aliphatic amines via dearomative amination from N-electrophilic aromatic precursors. Control experiments and density functional theory calculations suggest an ionic mechanism for the umpolung reaction of imines. This disconnection expands the options to forge C–N bonds stereoselectively on (hetero)arenes, which represents an important synthetic pursuit, especially in medicinal chemistry.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under numbers CCDC 2181457 (E-1a), 2092420 (1), 2181572 (27), 2252804 (45), 2261521 (49), 2153493 (73), 1937417 (84) and 2252823 (102). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. The data supporting the findings of this work are provided in the Supplementary Information including experimental procedures and characterization of new compounds.
References
Seebach, D. Methods of reactivity umpolung. Angew. Chem. Int. Ed. 18, 239–258 (1979).
Smith, A. B. & Adams, C. M. Evolution of dithiane-based strategies for the construction of architecturally complex natural products. Acc. Chem. Res. 37, 365–377 (2004).
Shen, B., Makley, D. M. & Johnston, J. N. Umpolung reactivity in amide and peptide synthesis. Nature 465, 1027–1032 (2010).
Eymur, S., Göllü, M. & Tanyeli, C. Umpolung strategy: advances in catalytic C–C bond formations. Turk. J. Chem. 37, 586–609 (2013).
Zheng, X. et al. Umpolung of hemiaminals: titanocene-catalyzed dehydroxylative radical coupling reactions with activated alkenes. Angew. Chem. Int. Ed. 52, 3494–3498 (2013).
Scattolin, T., Deckers, K. & Schoenebeck, F. Efficient synthesis of trifluoromethyl amines through a formal umpolung strategy from the bench-stable precursor (Me4N)SCF3. Angew. Chem. Int. Ed. 56, 221–224 (2017).
Enders, D., Niemeier, O. & Henseler, A. Organocatalysis by N-heterocyclic carbenes. Chem. Rev. 107, 5606–5655 (2007).
Bugaut, X. & Glorius, F. Organocatalytic umpolung: N-heterocyclic carbenes and beyond. Chem. Soc. Rev. 41, 3511–3522 (2012).
Mahatthananchai, J. & Bode, J. W. On the mechanism of N-heterocyclic carbene-catalyzed reactions involving acyl azoliums. Acc. Chem. Res. 47, 696–707 (2014).
Flanigan, D. M., Romanov-Michailidis, F., White, N. A. & Rovis, T. Organocatalytic reactions enabled by N-heterocyclic carbenes. Chem. Rev. 115, 9307–9387 (2015).
Nakano, Y. & Lupton, D. W. Enantioselective N-heterocyclic carbene catalysis by the umpolung of α,β-unsaturated ketones. Angew. Chem. Int. Ed. 55, 3135–3139 (2016).
Wang, M. H. & Scheidt, K. A. Cooperative catalysis and activation with N-heterocyclic carbenes. Angew. Chem. Int. Ed. 55, 14912–14922 (2016).
Guo, C., Fleige, M., Janssen-Müller, D., Daniliuc, C. G. & Glorius, F. Cooperative N-heterocyclic carbene/palladium-catalyzed enantioselective umpolung annulations. J. Am. Chem. Soc. 138, 7840–7843 (2016).
Kobayashi, S. & Ishitani, H. Catalytic enantioselective addition to imines. Chem. Rev. 99, 1069–1094 (1999).
Kobayashi, S., Mori, Y., Fossey, J. S. & Salter, M. M. Catalytic enantioselective formation of C–C bonds by addition to imines and hydrazones: a ten-year update. Chem. Rev. 111, 2626–2704 (2011).
Hummel, J. R., Boerth, J. A. & Ellman, J. A. Transition-metal-catalyzed C–H bond addition to carbonyls, imines, and related polarized π bonds. Chem. Rev. 117, 9163–9227 (2017).
Waser, M. & Novacek, J. An organocatalytic biomimetic strategy paves the way for the asymmetric umpolung of imines. Angew. Chem. Int. Ed. 54, 14228–14231 (2015).
Wu, Y., Hu, L., Li, Z. & Deng, L. Catalytic asymmetric umpolung reactions of imines. Nature 523, 445–450 (2015).
Liu, J., Cao, C.-G., Sun, H.-B., Zhang, X. & Niu, D. Catalytic asymmetric umpolung allylation of imines. J. Am. Chem. Soc. 138, 13103–13106 (2016).
Chen, P. et al. Phosphine-catalyzed asymmetric umpolung addition of trifluoromethyl ketimines to Morita–Baylis–Hillman carbonates. Angew. Chem. Int. Ed. 55, 13316–13320 (2016).
Hu, L., Wu, Y., Li, Z. & Deng, L. Catalytic asymmetric synthesis of chiral γ-amino ketones via umpolung reactions of imines. J. Am. Chem. Soc. 138, 15817–15820 (2016).
Patra, A. et al. N-Heterocyclic-carbene-catalyzed umpolung of imines. Angew. Chem. Int. Ed. 56, 2730–2734 (2017).
Hu, B. & Deng, L. Catalytic asymmetric synthesis of trifluoromethylated γ-amino acids through the umpolung addition of trifluoromethyl imines to carboxylic acid derivatives. Angew. Chem. Int. Ed. 57, 2233–2237 (2018).
Fiaud, J.-C. & Kagan, H. B. Une nouvelle synthese D’α amino-acides. Synthese asymetrique de l’alanine. Tetrahedron Lett. 11, 1813–1816 (1970).
Niwa, Y., Takayama, K. & Shimizu, M. Electrophilic amination with iminomalonate. Tetrahedron Lett. 42, 5473–5476 (2001).
Kattamuri, P. V. et al. Practical singly and doubly electrophilic aminating agents: a new, more sustainable platform for carbon–nitrogen bond formation. J. Am. Chem. Soc. 139, 11184–11196 (2017).
Mizota, I. & Shimizu, M. Umpolung reactions of α-imino esters: useful methods for the preparation of α-amino acid frameworks. Chem. Rec. 16, 688–702 (2016).
King, R. B. & Efraty, A. Pentamethylcyclopentadienyl derivatives of transition metals. II. Synthesis of pentamethylcyclopentadienyl metal carbonyls from 5-acetyl-1,2,3,4,5-pentamethylcyclopentadiene. J. Am. Chem. Soc. 94, 3773–3779 (1972).
Schleyer, Pv. R. & Pühlhofer, F. Recommendations for the evaluation of aromatic stabilization energies. Org. Lett. 4, 2873–2876 (2002).
Babinski, D. J. et al. Synchronized aromaticity as an enthalpic driving force for the aromatic Cope rearrangement. J. Am. Chem. Soc. 134, 16139–16142 (2012).
Xu, Y. et al. Deacylative transformations of ketones via aromatization-promoted C–C bond activation. Nature 567, 373–378 (2019).
Cho, S.-H., Kim, J.-Y., Kwak, J. & Chang, S. Recent advances in the transition metal-catalyzed twofold oxidative C–H bond activation strategy for C–C and C–N bond formation. Chem. Soc. Rev. 40, 5068–5083 (2011).
Louillat, M.-L. & Patureau, F. W. Oxidative C–H amination reactions. Chem. Soc. Rev. 43, 901–910 (2014).
Mailyan, A. K. et al. Cutting-edge and time-honored strategies for stereoselective construction of C–N bonds in total synthesis. Chem. Rev. 116, 4441–4557 (2016).
Ruiz-Castillo, P. & Buchwald, S. L. Applications of palladium-catalyzed C–N cross-coupling reactions. Chem. Rev. 116, 12564–12649 (2016).
Kainz, Q. M. et al. Asymmetric copper-catalyzed C–N cross-couplings induced by visible light. Science 351, 681–684 (2016).
Paudyal, M. P. et al. Dirhodium-catalyzed C–H arene amination using hydroxylamines. Science 353, 1144–1147 (2016).
Takahashi, I., Suzuki, Y. & Kitagawa, O. Asymmetric synthesis of atropisomeric compounds with an N–C chiral axis. Org. Prep. Proced. Int. 46, 1–23 (2014).
Ricci, A. Amino Group Chemistry: from Synthesis to the Life Sciences (Wiley–VCH, 2008).
Tsuji, N. et al. Activation of olefins via asymmetric Brønsted acid catalysis. Science 359, 1501–1505 (2018).
Schreyer, L. et al. Confined acids catalyze asymmetric single aldolizations of acetaldehyde enolates. Science 362, 216–219 (2018).
Akiyama, T., Itoh, J., Yokota, K. & Fuchibe, K. Enantioselective Mannich-type reaction catalyzed by a chiral Brønsted acid. Angew. Chem. Int. Ed. 43, 1566–1568 (2004).
Uraguchi, D. & Terada, M. Chiral Brønsted acid-catalyzed direct Mannich reactions via electrophilic activation. J. Am. Chem. Soc. 126, 5356–5357 (2004).
Zhuo, C.-X., Zhang, W. & You, S.-L. Catalytic asymmetric dearomatization reactions. Angew. Chem. Int. Ed. 51, 12662–12686 (2012).
Gustafson, J., Lim, D. & Miller, S. J. Dynamic kinetic resolution of biaryl atropisomers via peptide-catalyzed asymmetric bromination. Science 328, 1251–1255 (2010).
Barrett, K. T., Metrano, A. J., Rablen, P. R. & Miller, S. J. Spontaneous transfer of chirality in an atropisomerically enriched two-axis system. Nature 509, 71–75 (2014).
Iorio, N. D. et al. Remote control of axial chirality: aminocatalytic desymmetrization of N-arylmaleimides via vinylogous Michael addition. J. Am. Chem. Soc. 136, 10250–10253 (2014).
Zhang, J.-W. et al. Discovery and enantiocontrol of axially chiral urazoles via organocatalytic tyrosine click reaction. Nat. Commun. 7, 10677 (2016).
Crawford, J. M., Stone, E. A., Metrano, A. J., Miller, S. J. & Sigman, M. S. Parameterization and analysis of peptide-based catalysts for the atroposelective bromination of 3-arylquinazolin-4(3H)-ones. J. Am. Chem. Soc. 140, 868–871 (2018).
Li, T.-Z., Liu, S.-J., Tan, W. & Shi, F. Catalytic asymmetric construction of axially chiral indole-based frameworks: an emerging area. Chem. Eur. J. 26, 15779–15792 (2020).
Frisch, M. J. et al. Gaussian 16, revision A.03 (Gaussian, 2016).
Chai, J. D. et al. Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Cossi, M. et al. Energies, structures, and electronic properties of molecules in solution with the C-PCM solvation model. J. Comput. Chem. 24, 669–681 (2003).
Luchini, G. et al. GoodVibes 3.0.1 10.5281/zenodo.595246 (2019).
Grimme, S. Supramolecular binding thermodynamics by dispersion-corrected density functional theory. Chem. Eur. J. 18, 9955–9964 (2012).
Li, Y. et al. Improved force-field parameters for QM/MM simulations of the energies of adsorption for molecules in zeolites and a free rotor correction to the rigid rotor harmonic oscillator model for adsorption enthalpies. J. Phys. Chem. C 119, 1840–1850 (2015).
Grimme, S. et al. Fully automated quantum-chemistry-based computation of spin-spincoupled nuclear magnetic resonance spectra. Angew. Chem. Int. Ed. 56, 14763–14769 (2017).
Grimme, S. Exploration of chemical compound, conformer, and reaction space with metadynamics simulations based on tight-binding quantum chemical calculations. J. Chem. Theory Comput. 15, 2847–2862 (2019).
Grimme, S. et al. A robust and accurate tight-binding quantum chemical method for structures, vibrational frequencies, and noncovalent interactions of large molecular systems parametrized for all spd-block elements (Z = 1–86). J. Chem. Theory Comput. 13, 1989–2009 (2017).
Bannwarth, C. et al. GFN2-xTB—an accurate and broadly parametrized self-consistent tight-binding quantum chemical method with multipole electrostatics and densitydependent dispersion contributions. J. Chem. Theory Comput. 15, 1652–1671 (2019).
Pracht, P. et al. A robust non-self-consistent tight binding quantum chemistry method for large molecules. Preprint at ChemRxiv 10.26434/chemrxiv.8326202.v1 (2019).
Acknowledgements
We are grateful for financial support from the National Natural Science Foundation of China (nos. 21825105 and 22231004 to B.T. and no. 22271135 to S.-H.X.), National Key R&D Program of China (nos. 2022YFA1503703 and 2021YFF0701604 to B.T.), National Science Foundation (CHE-1764328 to K.N.H.), Guangdong Innovative Program (no. 2019BT02Y335 to B.T.), Shenzhen Science and Technology Program (KQTD20210811090112004 and JCYJ20210324120205016 to B.T. and JCYJ20210324105005015 to S.-H.X.). Computational work was supported by the Center for Computational Science and Engineering at Southern University of Science and Technology, and the Extreme Science and Engineering Discovery Environment, which is supported by the National Science Foundation (OCI-1053575 to K.N.H.). The authors appreciate the assistance of SUSTech Core Research Facilities.
Author information
Authors and Affiliations
Contributions
B.T. conceived and directed the project. Y.-H.C. designed and performed experiments. S.-L.L, Y.-W.L., J.K.C. and S.-H.X. helped with the collection of some new compounds and data analysis. M.D. and P.Y. performed the DFT calculations and mechanism analysis. K.N.H. directed the DFT calculations and mechanism analysis. B.T., Y.-H.C., M.D., J.K.C., S.-H.X. and K.N.H. wrote the paper with input from all other authors. All authors discussed the results and commented on the manuscript. Y.-H.C. and M.D. contributed equally to this work
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–797, Tables 1–5, experimental procedures, synthetic procedures, characterization data and DFT calculations.
Supplementary Data 1
Crystallographic data for compound E-1a; CCDC reference 2181457.
Supplementary Data 2
Structure factors of compound E-1a; CCDC reference 2181457.
Supplementary Data 3
Crystallographic data for compound 1; CCDC reference 2092420.
Supplementary Data 4
Structure factors of compound 1; CCDC reference 2092420.
Supplementary Data 5
Crystallographic data for compound 27; CCDC reference 2181572.
Supplementary Data 6
Structure factors of compound 27; CCDC reference 2181572.
Supplementary Data 7
Crystallographic data for compound 45; CCDC reference 2252804.
Supplementary Data 8
Structure factors of compound 45; CCDC reference 2252804.
Supplementary Data 9
Crystallographic data for compound 49; CCDC reference 2261521.
Supplementary Data 10
Structure factors of compound 49; CCDC reference 2261521.
Supplementary Data 11
Crystallographic data for compound 73; CCDC reference 2153493.
Supplementary Data 12
Structure factors of compound 73; CCDC reference 2153493.
Supplementary Data 13
Crystallographic data for compound 84; CCDC reference 1937417.
Supplementary Data 14
Structure factors of compound 84; CCDC reference 1937417.
Supplementary Data 15
Crystallographic data for compound 102; CCDC reference 2252823.
Supplementary Data 16
Structure factors of compound 102; CCDC reference 2252823.
Supplementary Data 17
Calculations archive files in XYZ format.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, YH., Duan, M., Lin, SL. et al. Organocatalytic aromatization-promoted umpolung reaction of imines. Nat. Chem. 16, 408–416 (2024). https://doi.org/10.1038/s41557-023-01384-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01384-x