Abstract
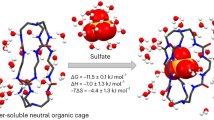
The convergent positioning of functional groups in biomacromolecules leads to good binding, catalytic and transport capabilities. Synthetic frameworks capable of convergently locking functional groups with minimized conformational uncertainty—leading to similar properties—are highly desirable but rare. Here we report C5-symmetric aromatic pentaamide macrocycles synthesized in one pot from the corresponding monomers. Their crystal structures reveal a star-shaped, fully constrained backbone that causes ten alternating NH/CH hydrogen-bond donors and five large amide dipoles to orient towards the centre of the macrocycle. With a highly electropositive cavity in a high-energy unbound state, the macrocycles bind anions in a 1:1 stoichiometry in solution, with high affinity for halides and very high affinity for oxoanions. We demonstrate that such macrocycles are able to transport anions across lipid bilayers with a high chloride selectivity and restore the depleted airway surface liquid of cystic fibrosis airway cell cultures.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Single-crystal X-ray structure data for macrocycle c5c (CCDC reference number: 2124841) can be obtained free of charge from the Cambridge Crystallographic Data Centre at www.ccdc.cam.ac.uk/data_request/cif. The source data for Supplementary Figs. 1, 2, 5, 6a,b, 10–14, 17 and 18 can be obtained free of charge from Figshare at https://doi.org/10.6084/m9.figshare.23302427 (ref. 60). Source data are provided with this paper.
Code availability
The codes used in the calculations of data from vesicle-based halide transport HPTS assays can be obtained free of charge from Figshare at https://doi.org/10.6084/m9.figshare.23302427 (ref. 60).
References
Whitty, A. Cooperativity and biological complexity. Nat. Chem. Biol. 4, 435–439 (2008).
Zhou, Y., Morais-Cabral, J. H., Kaufman, A. & MacKinnon, R. Chemistry of ion coordination and hydration revealed by a K+ channel–Fab complex at 2.0 Å resolution. Nature 414, 43–48 (2001).
Sui, H. X., Han, B. G., Lee, J. K., Walian, P. & Jap, B. K. Structural basis of water-specific transport through the AQP1 water channel. Nature 414, 872–878 (2001).
Dutzler, R., Campbell, E. B., Cadene, M., Chait, B. T. & MacKinnon, R. X-ray structure of a ClC chloride channel at 3.0 Å reveals the molecular basis of anion selectivity. Nature 415, 287–294 (2002).
Pflugrath, J. W. & Quiocho, F. A. Sulphate sequestered in the sulphate-binding protein of Salmonella typhimurium is bound solely by hydrogen bonds. Nature 314, 257–260 (1985).
Wu, X., Gilchrist, A. M. & Gale, P. A. Prospects and challenges in anion recognition and transport. Chem 6, 1296–1309 (2020).
Sessler, J. L., Gale, P. A. & Cho, W.-S. Anion Receptor Chemistry (RSC Publishing, 2006).
John, E. A., Massena, C. J. & Berryman, O. B. Helical anion foldamers in solution. Chem. Rev. 120, 2759–2782 (2020).
Borissov, A. et al. Anion recognition in water by charge-neutral halogen and chalcogen bonding foldamer receptors. J. Am. Chem. Soc. 141, 4119–4129 (2019).
Rebek, J. Jr. Model studies in molecular recognition. Science 235, 1478–1484 (1987).
Gong, B. et al. Creating nanocavities of tunable sizes: Hollow helices. Proc. Natl Acad. Sci. USA 99, 11583–11588 (2002).
Jiang, H., Léger, J. M. & Huc, I. Aromatic δ-peptides. J. Am. Chem. Soc. 125, 3448–3449 (2003).
Höger, S. Shape-persistent macrocycles: from molecules to materials. Chem. Eur. J. 10, 1320–1329 (2004).
Guieu, S., Crane, A. K. & MacLachlan, M. J. Campestarenes: novel shape-persistent Schiff base macrocycles. Chem. Commun. 47, 1169–1171 (2011).
Qin, B. et al. Crystallographic evidence of an unusual, pentagon-shaped folding pattern in a circular aromatic pentamer. Org. Lett. 10, 5127–5130 (2008).
Lee, S. M., Chen, C. H. & Flood, A. H. A pentagonal cyanostar macrocycle with cyanostilbene CH donors binds anions and forms dialkylphosphate [3]rotaxanes. Nat. Chem. 5, 704–710 (2013).
Li, Y. & Flood, A. H. Pure C—H hydrogen bonding to chloride ions: a preorganized and rigid macrocyclic receptor. Angew. Chem. Int. Ed. 47, 2649–2652 (2008).
Bisson, A. P., Lynch, V. M., Monahan, M.-K. C. & Anslyn, E. V. Recognition of anions through NH—π hydrogen bonds in a bicyclic cyclophane—selectivity for nitrate. Angew. Chem. Int. Ed. Engl. 36, 2340–2342 (1997).
Sessler, J. L. & Davis, J. M. Sapphyrins: versatile anion binding agents. Acc. Chem. Res. 34, 989–997 (2001).
Brooks, S. J., Gale, P. A. & Light, M. E. Anion-binding modes in a macrocyclic amidourea. Chem. Commun. 41, 4344–4346 (2006).
Tresca, B. W. et al. Substituent effects in CH hydrogen bond interactions: linear free energy relationships and influence of anions. J. Am. Chem. Soc. 137, 14959–14967 (2015).
Bondy, C. R. & Loeb, S. J. Amide based receptors for anions. Coord. Chem. Rev. 240, 77–99 (2003).
Kang, S. O., Begum, R. A. & Bowman-James, K. Amide-based ligands for anion coordination. Angew. Chem. Int. Ed. 45, 7882–7894 (2006).
Choi, K. H. & Hamilton, A. D. Macrocyclic anion receptors based on directed hydrogen bonding interactions. Coord. Chem. Rev. 240, 101–110 (2003).
Choi, K. H. & Hamilton, A. D. Selective anion binding by a macrocycle with convergent hydrogen bonding functionality. J. Am. Chem. Soc. 123, 2456–2457 (2001).
Yuan, L. H. et al. Highly efficient, one-step macrocyclizations assisted by the folding and preorganization of precursor oligomers. J. Am. Chem. Soc. 126, 11120–11121 (2004).
Cao, R. K., Rossdeutcher, R. B., Wu, X. X. & Gong, B. Oligo(5-amino-N-acylanthranilic acids): amide bond formation without coupling reagent and folding upon binding anions. Org. Lett. 22, 7496–7501 (2020).
Juwarker, H. & Jeong, K. S. Anion-controlled foldamers. Chem. Soc. Rev. 39, 3664–3674 (2010).
Hua, Y., Liu, Y., Chen, C.-H. & Flood, A. H. Hydrophobic collapse of foldamer capsules drives picomolar-level chloride binding in aqueous acetonitrile solutions. J. Am. Chem. Soc. 135, 14401–14412 (2013).
Chang, K. J., Kang, B. N., Lee, M. H. & Jeong, K. S. Oligoindole-based foldamers with a helical conformation induced by chloride. J. Am. Chem. Soc. 127, 12214–12215 (2005).
Juwarker, H. et al. Anion binding of short, flexible aryl triazole oligomers. J. Org. Chem. 74, 8924–8934 (2009).
Helttunen, K. R. et al. Oligoamide foldamers as helical chloride receptors—the influence of electron-withdrawing substituents on anion-binding interactions. Chem. Asian J. 14, 647–654 (2019).
Liu, Y., Sengupta, A., Raghavachari, K. & Flood, A. H. Anion binding in solution: beyond the electrostatic regime. Chem 3, 411–427 (2017).
Qiao, B., Anderson, J. R., Pink, M. & Flood, A. H. Size-matched recognition of large anions by cyanostar macrocycles is saved when solvent-bias is avoided. Chem. Commun. 52, 8683–8686 (2016).
Pike, S. J., Hutchinson, J. J. & Hunter, C. A. H-bond acceptor parameters of anions. J. Am. Chem. Soc. 39, 6700–6706 (2017).
Yawer, M. A., Havel, V. & Sindelar, V. A bambusuril macrocycle that binds anions in water with high affinity and selectivity. Angew. Chem. Int. Ed. 54, 276–279 (2015).
Davis, A. P., Sheppard, D. N. & Smith, B. D. Development of synthetic membrane transporters for anions. Chem. Soc. Rev. 36, 348–357 (2007).
Gokel, G. W. & Negin, S. Synthetic ion channels: from pores to biological applications. Acc. Chem. Res. 46, 2824–2833 (2013).
Tomich, M., Bukovnik, U., Layman, J. & Schultz, B. D. Channel replacement therapy for cystic fibrosis. in Cystic Fibrosis - Renewed Hopes through Research (ed Sriramulu, D.) 291–332 (InTech, 2012).
Valkenier et al. Lipophilic balance – a new design principle for transmembrane anion carriers. Chem. Sci. 5, 1128–1134 (2014).
Sakai, N. & Matile, S. The determination of the ion selectivity of synthetic ion channels and pores in vesicles. J. Phys. Org. Chem. 19, 452–460 (2006).
Shen, Y. et al. Ultrasensitive liposome-based assay for the quantification of fundamental ion channel properties. Anal. Chim. Acta 1112, 8–15 (2020).
Matsui, H. et al. Evidence for periciliary liquid layer depletion, not abnormal ion composition, in the pathogenesis of cystic fibrosis airways disease. Cell 5, 1005–1015 (1998).
Jentzsch, A. V. et al. Transmembrane anion transport mediated by halogen-bond donors. Nat. Commun. 3, 905 (2012).
Rychkov, G. Y., Pusch, M., Roberts, M. L., Jentsch, T. J. & Bretag, A. H. Permeation and block of the skeletal muscle chloride channel, ClC-1, by foreign anions. J. Gen. Physiol. 111, 653–665 (1991).
Biwersi, J., Tulk, B. & Verkman, A. S. Long-wavelength chloride-sensitive fluorescent indicators. Anal. Biochem. 219, 139–143 (1994).
Seganish, J. L., Fettinger, J. C. & Davis, J. T. Facilitated chloride transport across phosphatidylcholine bilayers by an acyclic calixarene derivative: structure-function relationships. Supramol. Chem. 18, 257–264 (2006).
Kunz, W., Nostro, P. L. & Ninham, B. W. The present state of affairs with Hofmeister effects. Curr. Opin. Colloid Interface Sci. 9, 1–18 (2004).
Behera, H. & Madhavan, N. Anion-selective cholesterol decorated macrocyclic transmembrane ion carriers. J. Am. Chem. Soc. 139, 12919–12922 (2017).
Behr, J.-P., Kirch, M. & Lehn, J.-M. Carrier-mediated transport through bulk liquid membranes: dependence of transport rates and selectivity on carrier properties in a diffusion-limited process. J. Am. Chem. Soc. 107, 241–246 (1985).
Jentsch, T. J., Maritzen, T. & Zdebik, A. A. Chloride channel diseases resulting from impaired transepithelial transport or vesicular function. J. Clin. Invest. 115, 2039–2046 (2005).
Haq, I. J., Gray, M. A., Garnett, J. P., Ward, C. & Brodlie, M. Airway surface liquid homeostasis in cystic fibrosis: pathophysiology and therapeutic targets. Thorax 71, 284–287 (2016).
Alikadic, S. et al. Ciliary beat frequency in nasal and bronchial epithelial cells in patients with cystic fibrosis. Eur. Respir. J. 38, 4550 (2011).
Anderson, W. H. et al. The relationship of mucus concentration (hydration) to mucus osmotic pressure and transport in chronic bronchitis. Am. J. Respir. Crit. Care Med. 192, 182–190 (2015).
Goralski, J. L., Wu, D., Thelin, W. R., Boucher, R. C. & Button, B. The in vitro effect of nebulised hypertonic saline on human bronchial epithelium. Eur. Respir. J. 51, 1702652 (2018).
Muraglia, K. A. et al. Small-molecule ion channels increase host defences in cystic fibrosis airway epithelia. Nature 567, 405–408 (2019).
te Velde, G. et al. Chemistry with ADF. J. Comput. Chem. 22, 931–967 (2001).
Liu, Z. H., Ma, Q. Q., Liu, Y. X. & Wang, Q. M. 4-(N,N-Dimethylamino)pyridine hydrochloride as a recyclable catalyst for acylation of inert alcohols: substrate scope and reaction mechanism. Org. Lett. 16, 236–239 (2014).
Zhang, Z. G. et al. Efficient synthesis of cyclic carnonates from atmospheric CO2 using a positive charge delocalized ionic liquid catalyst. ACS Sustain. Chem. Eng. 5, 2841–2846 (2017).
Cao, R. et al. Aromatic pentaamide macrocycles bind anions with high-affinity for transport across biomembranes. Figshare https://doi.org/10.6084/m9.figshare.23302427 (2023).
Gong, B. Linear and cyclic aromatic oligoamides, methods of making same and uses thereof. US patent PCT/US2021/050041 (2021).
Acknowledgements
We acknowledge support from the US National Science Foundation (CHE-2304878 to B.G. for the conceptualization, design, data collection, analysis, decision to publish or preparation of the manuscript; CHE-1905094 and 2108538 to B.G. for preparing some of the synthetic building blocks; and CHE-2108597 to D.P.M. for the computational studies) for financial support; the Cystic Fibrosis Foundation (BUTTON19G0) and the NIH (R01HL125280) to B.B. for assessing the effect of c5a on restoring the ASL of cultured CF cells; the National Key R&D Program of China (2020YFA0908100), the NSFC (81627801) and the K. C. Wong Education Foundation (Hong Kong) to Z.S. for the design and performance of experiments on anion transport, for the determination of transport selectivity and for the preparation of the manuscript; and the Rabinowitz Honors College Research Assistant Program and a Lister Endowed Fellowship from Hofstra University (to D.P.M.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. Computations were carried out at the Center for Computational Research at the University at Buffalo (http://hdl.handle.net/10477/79221).
Author information
Authors and Affiliations
Contributions
R.C. and R.B.R. designed and conducted the syntheses and also performed the binding studies. T.A.S. performed ITC experiments. Y.S., Y.Z., R.B.R. and Z.S. performed the vesicle-based assays on anion transport. X.W. processed the X-ray data. D.P.M., L.S.B., K.R. and E.Z. performed the computational studies. B.B designed and, along with M.I.G. and M.F.F., performed experiments on cystic fibrosis cell cultures. T.S. helped with the NMR experiments. B.G. conceived and supervised the project. Z.S. and B.G. co-wrote the paper. All authors participated in discussion and editing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
A provisional US patent application (PCT/US2021/050041) regarding the synthesis, anion binding, anion transport and potential therapeutic uses involving macrocycles c5 has been filed61. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Maija Nissinen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–19, Table 1, synthetic procedures, additional NMR and mass spectrometry spectra, descriptions of anion transport, cell culture and imaging and computational studies.
Supplementary Data 1
Crystallographic data for compound c5c; CCDC reference 2124841.
Supplementary Data 2
Crystallographic data for compound c5a, including cif file, checkcif file, structural factors and justification for A- and B-level alerts.
Supplementary Data 3
The xyz coordinates of computed structures.
Source data
Source Data Fig. 2
UV and NMR titration data of c5a with Cl− for Fig. 2a,b.
Source Data Fig. 3
Values of binding constants of c5a with different anions versus the diameter of the corresponding anions for Fig. 3c.
Source Data Fig. 4
The numerical values of data for halide ion transport by vesicle assay with and without c5a for Fig. 4b–d.
Source Data Fig. 5
The numerical values of the thickness of the ASL layers of control and treated (with c5a) CF cells for Fig. 5c.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cao, R., Rossdeutcher, R.B., Zhong, Y. et al. Aromatic pentaamide macrocycles bind anions with high affinity for transport across biomembranes. Nat. Chem. 15, 1559–1568 (2023). https://doi.org/10.1038/s41557-023-01315-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01315-w