Abstract
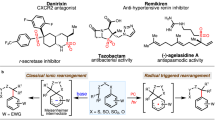
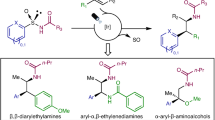
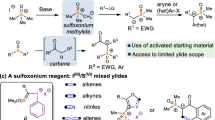
The asymmetric construction of all-carbon quaternary centres within acyclic settings represents a long-standing challenge for synthetic chemists. Alongside polar and radical methods, rearrangement reactions represent an attractive platform, but still broadly applicable methods are in high demand. Here we report an asymmetric, radical sulfinyl-Smiles rearrangement to access acyclic amides that bear an α-all-carbon quaternary centre. Our strategy uses enantioenriched N-arylsulfinyl acrylamides as acceptors for a variety of radicals produced in situ under mild photoredox conditions. The sulfinamido group not only directs the 1,4-migration of the aryl moiety onto the α-carbon of the amide, which thus governs its absolute configuration, but also functions as a traceless chiral auxiliary. The amides obtained in this multicomponent process are prevalent in pharmaceuticals, agrochemicals and bioactive natural products, and can be transformed into valuable chiral α,α-disubstituted acids, oxindoles as well as into β,β-disubstituted amines, highlighting the synthetic potential of this transformation.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Crystallographic data for structure 2.9 reported in this article has been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition number CCDC 1854836 (Supplementary Fig. 7). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All the other data that support the findings of this study are available within the article and its Supplementary Information.
References
Lovering, F., Bikker, J. & Humblet, C. Escape from flatland: increasing saturation as an approach to improving clinical success. J. Med. Chem. 52, 6752–6756 (2009).
Lovering, F. Escape from flatland 2: complexity and promiscuity. Med. Chem. Commun. 4, 515–519 (2013).
Ling, T. & Rivas, F. All-carbon quaternary centers in natural products and medicinal chemistry: recent advances. Tetrahedron 43, 6729–6777 (2016).
Carreira, E. M. & Kvaerno, L. Classics in Stereoselective Synthesis (Wiley-VCH, 2009).
Quasdorf, K. W. & Overman, L. E. Catalytic enantioselective synthesis of quaternary carbon stereocentres. Nature 516, 181–191 (2014).
Liu, Y., Han, S. J., Liu, W. B. & Stoltz, B. M. Catalytic enantioselective construction of quaternary stereocenters: assembly of key building blocks for the synthesis of biologically active molecules. Acc. Chem. Res. 48, 740–751 (2015).
Marek, I. et al. All-carbon quaternary stereogenic centers in acyclic systems through the creation of several C–C bonds per chemical step. J. Am. Chem. Soc. 136, 2682–2694 (2014).
Feng, J., Holmes, M. & Krische, M. J. Acyclic quaternary carbon stereocenters via enantioselective transition metal catalysis. Chem. Rev. 117, 12564–12580 (2017).
Kummer, D. A., Chain, W. J., Morales, M. R., Quiroga, O. & Myer, A. G. Stereocontrolled alkylative construction of quaternary carbon centers. J. Am. Chem. Soc. 130, 13231–13233 (2008).
Minko, Y., Pasco, M., Lercher, L., Botoshansky, M. & Marek, I. Forming all-carbon quaternary stereogenic centres in acyclic systems from alkynes. Nature 490, 522–526 (2012).
Masarwa, A. et al. Merging allylic carbon–hydrogen and selective carbon–carbon bond activation. Nature 505, 199–203 (2014).
Alam, R., Vollgraff, T., Eriksson, L. & Szabó, K. J. Synthesis of adjacent quaternary stereocenters by catalytic asymmetric allylboration. J. Am. Chem. Soc. 137, 11262–11265 (2015).
Mo, X. & Hall, D. G. Dual catalysis using boronic acid and chiral amine: acyclic quaternary carbons via enantioselective alkylation of branched aldehydes with allylic alcohols. J. Am. Chem. Soc. 138, 10762–10765 (2016).
Holmes, M., Nguyen, K. D., Schwartz, L. A., Luong, T. & Krische, M. J. Enantioselective formation of CF3-bearing all-carbon quaternary stereocenters via C–H functionalization of methanol: iridium catalyzed allene hydrohydroxymethylation. J. Am. Chem. Soc. 139, 8114–8117 (2017).
Starkov, P., Moore, J. T., Duquette, D. C., Stoltz, B. M. & Marek, I. Enantioselective construction of acyclic quaternary carbon stereocenters: palladium-catalyzed decarboxylative allylic alkylation of fully substituted amide enolates. J. Am. Chem. Soc. 139, 9615–9620 (2017).
Alexy, E. J., Zhang, H. & Stoltz, B. M. Catalytic enantioselective synthesis of acyclic quaternary centers: palladium-catalyzed decarboxylative allylic alkylation of fully substituted acyclic enol carbonates. J. Am. Chem. Soc. 140, 10109–10112 (2018).
Zhu, Y., Zhang, L. & Luo, S. Asymmetric α-photoalkylation of β-ketocarbonyls by primary amine catalysis: facile access to acyclic all-carbon quaternary stereocenters. J. Am. Chem. Soc. 136, 14642–14645 (2014).
Murphy, J. J., Bastida, D., Paria, S., Fagnoni, M. & Melchiorre, P. Asymmetric catalytic formation of quaternary carbons by iminium ion trapping of radicals. Nature 532, 218–222 (2016).
Lenhart, D., Bauer, A., Pöthig, A. & Bach, T. Enantioselective visible-light-induced radical-addition reactions to 3-alkylidene indolin-2-ones. Chem. Eur. J. 22, 6519–6523 (2016).
Wang, Z., Yin, H. & Fu, G. C. Catalytic enantioconvergent coupling of secondary and tertiary electrophiles with olefins. Nature 563, 379–383 (2018).
Wu, L., Wang, F., Chen, P. & Liu, G. Enantioselective construction of quaternary all-carbon centers via copper-catalyzed arylation of tertiary carbon-centered radicals. J. Am. Chem. Soc. 141, 1887–1892 (2019).
Lin, J. S. et al. Cu/chiral phosphoric acid-catalyzed asymmetric three-component radical-initiated 1,2-dicarbofunctionalization of alkenes. J. Am. Chem. Soc. 141, 1074–1083 (2019).
Prakash Dasa, J. & Marek, I. Enantioselective synthesis of all-carbon quaternary stereogenic centers in acyclic systems. Chem. Commun. 47, 4593–4623 (2011).
Gu, Z., Herrmann, A. T., Stivala, C. E. & Zakarian, A. Stereoselective construction of adjacent quaternary chiral centers by the Ireland–Claisen rearrangement: stereoselection with esters of cyclic alcohols. Synlett. 2010, 1717–1722 (2010).
Ma, D., Miao, C.-B. & Sun, J. Catalytic enantioselective House–Meinwald rearrangement: efficient construction of all-carbon quaternary stereocenters. J. Am. Chem. Soc. 141, 13783–13787 (2019).
Holden, C. M. & Greaney, M. F. Modern aspects of the Smiles rearrangement. Chem. Eur. J. 23, 8992–9008 (2017).
Snape, T. J. A truce on the Smiles rearrangement: revisiting an old reaction—the Truce–Smiles rearrangement. Chem. Soc. Rev. 37, 2452–2458 (2008).
Clayden, J., Dufour, J., Grainger, D. M. & Helliwell, M. Substituted diarylmethylamines by stereospecific intramolecular electrophilic arylation of lithiated ureas. J. Am. Chem. Soc. 129, 7488–7489 (2007).
Tetlow, D. J. et al. Sequential double α-arylation of N-allylureas by asymmetric deprotonation and N→C aryl migration. Org. Lett. 12, 5442–5445 (2010).
Tait, M. et al. Amines bearing tertiary substituents by tandem enantioselective carbolithiation–rearrangement of vinylureas. Org. Lett. 15, 34–37 (2013).
Maury, J., Zawodny, W. & Clayden, J. Stereospecific intramolecular arylation of 2- and 3-pyridyl substituted alkylamines via configurationally stable α-pyridyl organolithiums. Org. Lett. 19, 472–475 (2017).
Zawodny, W. et al. Chemoenzymatic synthesis of substituted azepanes by sequential biocatalytic reduction and organolithium-mediated rearrangement. J. Am. Chem. Soc. 140, 17872–17877 (2018).
Loven, R. & Speckamp, W. N. Heterocyclic steroids XXVII. A novel 1,4 arylradical rearrangement. Tetrahedron Lett. 16, 1567–1570 (1972).
Motherwell, W. B. & Pennell, A. M. K. A novel route to biaryls via intramolecular free radical ipso substitution reactions. J. Chem. Soc. Chem. Commun. 1991, 877–879 (1991).
Allart-Simon, I., Gérard, S. & Sapi, J. Radical Smiles rearrangement: an update. Molecules 21, 878–889 (2016).
Henderson, A. R. P., Kosowan, J. R. & Wood, T. E. The Truce–Smiles rearrangement and related reactions: a review. Can. J. Chem. 95, 483–504 (2017).
Kong, W. et al. Stereoselective synthesis of highly functionalized indanes and dibenzocycloheptadienes through complex radical cascade reactions. Angew. Chem. Int. Ed. 54, 2487–2491 (2015).
Whalley, D. M., Duong, H. A. & Greaney, M. F. Alkene carboarylation through catalyst-free, visible-light-mediated Smiles rearrangement. Chem. Eur. J. 25, 1927–1930 (2019).
Monos, T. M., McAtee, R. C. & Stephenson, C. R. J. Arylsulfonylacetamides as bifunctional reagents for alkene aminoarylation. Science 361, 1369–1373 (2018).
Rabet, P. T. G., Boyd, S. & Greaney, M. F. Metal-free intermolecular aminoarylation of alkynes. Angew. Chem. Int. Ed. 56, 4183–4186 (2017).
Abrams, R. & Clayden, J. Photocatalytic difunctionalization of vinyl ureas by radical addition polar Truce–Smiles rearrangement cascades. Angew. Chem. Int. Ed. 59, 11600–11606 (2020).
Kong, W., Casimiro, M., Merino, E. & Nevado, C. Copper-catalyzed one-pot trifluoromethylation/aryl migration/desulfonylation and C(sp2)–N bond formation of conjugated osyl amides. J. Am. Chem. Soc. 135, 14480–14483 (2013).
Kong, W., Casimiro, Fuentes, N., Merino, E. & Nevado, C. Metal-free aryltrifluoromethylation of activated alkenes. Angew. Chem. Int. Ed. 52, 13086–13090 (2013).
Kong, W., Merino, E. & Nevado, C. Arylphosphonylation and arylazidation of activated alkenes. Angew. Chem. Int. Ed. 53, 5078–5082 (2014).
Kagan, H. B. in Organosulfur Chemistry in Asymmetric Synthesis (eds Toru, T. & Bolm, C) 1–29 (Wiley-VCH, 2008).
Fischer, H. & Radom, L. Factors controlling the addition of carbon‐centered radicals to alkenes—an experimental and theoretical perspective. Angew. Chem. Int. Ed. 40, 1340–1371 (2001).
Macmillan, D. W. C., Stephenson, C. R. J. & Yoon, T. P. Visible Light Photocatalysis in Organic Chemistry (Wiley-VCH, 2018).
Kaldre, D., Klose, I. & Maulide, N. Stereodivergent synthesis of 1,4-dicarbonyls by traceless charge–accelerated sulfonium rearrangement. Science 361, 664–667 (2018).
Roth, H. G., Romero, N. A. & Nicewicz, D. A. Experimental and calculated electrochemical potentials of common organic molecules for applications to single-electron redox chemistry. Synlett 27, 714–723 (2016).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Gillis, E. P., Eastman, K. J., Hill, M. D., Donnelly, D. J. & Meanwell, N. A. Applications of fluorine in medicinal chemistry. J. Med. Chem. 58, 8315–8359 (2015).
Tang, S. et al. Visible-light-induced perfluoroalkylation/arylmigration/desulfonylation cascades of conjugated tosyl amides. Tetrahedron Lett. 58, 329–332 (2017).
Sheng, R.-L., Okada, K. & Sekiguchi, S. Aromatic nucleophilic substitution. 9. Kinetics of the formation and decomposition of anionic σ complexes in the Smiles rearrangements of N-acetyl-β-aminoethyl 2-X-4-nitro-1-phenyl or N-acetyl-β-aminoethyl 5-nitro-2-pyridyl ethers in aqueous dimethyl sulfoxide. J. Org. Chem. 43, 441–447 (1978).
Knipe, A. C. & Sridhar, N. Role of intramolecular catalysis in the kinetics of Smiles rearrangement of N-[2-(p-nitrophenoxy)ethylene]diamine. J. Chem. Soc. Chem. Commun. 1979, 791–792 (1979).
Lennox, A. J. J. Meisenheimer complexes in SNAr reactions: intermediates or transition states? Angew. Chem. Int. Ed. 57, 14686–14688 (2018).
Acknowledgements
We thank P. Fiechter for the synthesis of the racemic substrate 2.32 and A. Linden for the X-ray diffraction analysis of 2.9. This work was supported by the European Research Council (ERC starting grant agreement no. 307948), the Swiss National Science Foundation (SNF 200020_146853) and the Comunidad de Madrid Research Talent Attraction Program (2018-T1/IND-10054 to E.M.).
Author information
Authors and Affiliations
Contributions
C.H., M.S.K., T.S. and M.M. performed the experiments. E.M. performed DFT calculations. E.M. and C.N. conceptualized the project, supervised, analysed the data and co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Yannick Landais and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
General information, reaction optimization data, experimental procedures, additional experiments, compound characterization data including spectroscopic and analytical data for all new compounds, X-ray crystallographic data, NMR and HPLC spectral data, computational details containing the Cartesian coordinates of computational structures, Supplementary Figs. 1–10, Tables 1–5 and references.
Supplementary Data
Crystallographic data for compound 2.9. CCDC 1854836.
Rights and permissions
About this article
Cite this article
Hervieu, C., Kirillova, M.S., Suárez, T. et al. Asymmetric, visible light-mediated radical sulfinyl-Smiles rearrangement to access all-carbon quaternary stereocentres. Nat. Chem. 13, 327–334 (2021). https://doi.org/10.1038/s41557-021-00668-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00668-4
This article is cited by
-
A general alkene aminoarylation enabled by N-centred radical reactivity of sulfinamides
Nature Chemistry (2024)
-
Chiral arylsulfinylamides as reagents for visible light-mediated asymmetric alkene aminoarylations
Nature Chemistry (2024)
-
Photoredox catalytic alkylarylation of alkynes with arylsulfonylacetate as bifunctional reagent
Science China Chemistry (2024)
-
SO2-Insertion induced enantioselective oxysulfonylation to access β-chiral sulfones with quaternary carbon stereocenters
Science China Chemistry (2024)
-
Nickel-catalysed regio- and stereoselective acylzincation of unsaturated hydrocarbons with organozincs and CO
Nature Synthesis (2023)