Abstract
Activation of metastatic reprogramming is critical for tumour metastasis. However, more detailed knowledge of the underlying mechanism is needed to enable targeted intervention. Here, we show that paraspeckle component 1 (PSPC1), identified in an aberrant 13q12.11 locus, is upregulated and associated with poor survival in patients with cancer. PSPC1 promotes tumorigenesis, epithelial-to-mesenchymal transition (EMT), stemness and metastasis in multiple cell types and in spontaneous mouse cancer models. PSPC1 is the master activator for transcription factors of EMT and stemness and accompanies c-Myc activation to facilitate tumour growth. PSPC1 increases transforming growth factor-β1 (TGF-β1) secretion through an interaction with phosphorylated and nuclear Smad2/3 to potentiate TGF-β1 autocrine signalling. Moreover, PSPC1 acts as a contextual determinant of the TGF-β1 pro-metastatic switch to alter Smad2/3 binding preference from tumour-suppressor to pro-metastatic genes. Having validated the PSPC1–Smads–TGF-β1 axis in various cancers, we conclude that PSPC1 is a master activator of pro-metastatic switches and a potential target for anti-metastasis drugs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Fidler, I. J. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat. Rev. Cancer 3, 453–458 (2003).
Massague, J. & Obenauf, A. C. Metastatic colonization by circulating tumour cells. Nature 529, 298–306 (2016).
Mani, S. A. et al. The epithelial–mesenchymal transition generates cells with properties of stem cells. Cell 133, 704–715 (2008).
Thiery, J. P., Acloque, H., Huang, R. Y. & Nieto, M. A. Epithelial–mesenchymal transitions in development and disease. Cell 139, 871–890 (2009).
Lamouille, S., Xu, J. & Derynck, R. Molecular mechanisms of epithelial–mesenchymal transition. Nat. Rev.Mol. Cell Biol. 15, 178–196 (2014).
Nieto, M. A., Huang, R. Y., Jackson, R. A. & Thiery, J. P. EMT: 2016. Cell 166, 21–45 (2016).
Ocana, O. H. et al. Metastatic colonization requires the repression of the epithelial–mesenchymal transition inducer Prrx1. Cancer Cell. 22, 709–724 (2012).
Tsai, J. H., Donaher, J. L., Murphy, D. A., Chau, S. & Yang, J. Spatiotemporal regulation of epithelial–mesenchymal transition is essential for squamous cell carcinoma metastasis. Cancer Cell. 22, 725–736 (2012).
Aceto, N. et al. Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell 158, 1110–1122 (2014).
Hou, J. M. et al. Clinical significance and molecular characteristics of circulating tumor cells and circulating tumor microemboli in patients with small-cell lung cancer. J. Clin. Oncol. 30, 525–532 (2012).
Cheung, K. J. & Ewald, A. J. A collective route to metastasis: seeding by tumor cell clusters. Science 352, 167–169 (2016).
Yu, M. et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 339, 580–584 (2013).
Hendrix, M.J. et al. Coexpression of vimentin and keratins by human melanoma tumor cells: correlation with invasive and metastatic potential.J. Natl Cancer Inst. 84, 165–174 (1992).
Ruscetti, M., Quach, B., Dadashian, E. L., Mulholland, D. J. & Wu, H. Tracking and functional characterization of epithelial–mesenchymal transition and mesenchymal tumor cells during prostate cancer metastasis. Cancer Res. 75, 2749–2759 (2015).
Pickup, M., Novitskiy, S. & Moses, H. L. The roles of TGFβ in the tumour microenvironment. Nat. Rev. Cancer 13, 788–799 (2013).
Moses, H. L., Roberts, A. B. & Derynck, R. The Discovery and early days of TGF-β: a historical perspective. Cold Spring Harb. Perspect. Biol. 8, a021865 (2016).
Penuelas, S. et al. TGF-β increases glioma-initiating cell self-renewal through the induction of LIF in human glioblastoma. Cancer Cell. 15, 315–327 (2009).
Ikushima, H. et al. Autocrine TGF-β signaling maintains tumorigenicity of glioma-initiating cells through Sry-related HMG-box factors. Cell Stem Cell 5, 504–514 (2009).
Massague, J. TGFβ signalling in context. Nat. Rev. Mol. Cell Biol. 13, 616–630 (2012).
Feng, X. H. & Derynck, R. Specificity and versatility in TGF-β signaling through Smads. Annu. Rev. Cell. Dev. Biol. 21, 659–693 (2005).
Fox, A. H. et al. Paraspeckles: a novel nuclear domain. Curr. Biol. 12, 13–25 (2002).
Sasaki, Y. T., Ideue, T., Sano, M., Mituyama, T. & Hirose, T. MENε/β noncoding RNAs are essential for structural integrity of nuclear paraspeckles. Proc. Natl Acad. Sci. USA 106, 2525–2530 (2009).
Knott, G. J., Bond, C. S. & Fox, A. H. The DBHS proteins SFPQ, NONO and PSPC1: a multipurpose molecular scaffold. Nucleic Acids Res. 44, 3989–4004 (2016).
Kuwahara, S. et al. PSPC1, NONO, and SFPQ are expressed in mouse Sertoli cells and may function as coregulators of androgen receptor-mediated transcription. Biol. Reprod. 75, 352–359 (2006).
Chen, L. L. & Carmichael, G. G. Altered nuclear retention of mRNAs containing inverted repeats in human embryonic stem cells: functional role of a nuclear noncoding RNA. Mol. Cell. 35, 467–478 (2009).
Clemson, C. M. et al. An architectural role for a nuclear noncoding RNA: NEAT1 RNA is essential for the structure of paraspeckles. Mol. Cell. 33, 717–726 (2009).
Gao, X. et al. Paraspeckle protein 1 (PSPC1) is involved in the cisplatin induced DNA damage response-role in G1/S checkpoint. PLoS ONE 9, e97174 (2014).
Chen, Y. et al. Identification of druggable cancer driver genes amplified across TCGA datasets. PLoS ONE 9, e98293 (2014).
Santarius, T., Shipley, J., Brewer, D., Stratton, M. R. & Cooper, C. S. A census of amplified and overexpressed human cancer genes. Nat. Rev. Cancer 10, 59–64 (2010).
Chen, C. F., Yeh, S. H., Chen, D. S., Chen, P. J. & Jou, Y. S. Molecular genetic evidence supporting a novel human hepatocellular carcinoma tumor suppressor locus at 13q12.11. Genes. Chromosomes Cancer 44, 320–328 (2005).
Shiau, C. K., Gu, D. L., Chen, C. F., Lin, C. H. & Jou, Y. S. IGRhCellID: integrated genomic resources of human cell lines for identification. Nucleic Acids Res. 39, D520–D524 (2011).
Ginestier, C. et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 1, 555–567 (2007).
Guy, C. T., Cardiff, R. D. & Muller, W. J. Induction of mammary tumors by expression of polyomavirus middle T oncogene: a transgenic mouse model for metastatic disease. Mol. Cell Biol. 12, 954–961 (1992).
DuPage, M., Dooley, A. L. & Jacks, T. Conditional mouse lung cancer models using adenoviral or lentiviral delivery of Cre recombinase. Nat. Protoc. 4, 1064–1072 (2009).
Malanchi, I. et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 481, 85–89 (2011).
Brown, K. A. et al. Identification of novel Smad2 and Smad3 associated proteins in response to TGF-β1. J. Cell. Biochem. 105, 596–611 (2008).
Abdollah, S. et al. TβRI phosphorylation of Smad2 on Ser465 and Ser467 is required for Smad2–Smad4 complex formation and signaling. J. Biol. Chem. 272, 27678–27685 (1997).
Koinuma, D. et al. Chromatin immunoprecipitation on microarray analysis of Smad2/3 binding sites reveals roles of ETS1 and TFAP2A in transforming growth factor β signaling. Mol. Cell Biol. 29, 172–186 (2009).
Chen, C. R., Kang, Y. & Massague, J. Defective repression of c-myc in breast cancer cells: a loss at thecore of the transforming growth factor β growth arrest program. Proc. Natl Acad. Sci.USA 98, 992–999 (2001).
Dennler, S. et al. Direct binding of Smad3 and Smad4 to critical TGF β-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J. 17, 3091–3100 (1998).
Jang, C. W. et al. TGF-β induces apoptosis through Smad-mediated expression of DAP-kinase. Nat. Cell Biol. 4, 51–58 (2002).
Hahn, S. A. et al. DPC4, a candidate tumor suppressor gene at human chromosome 18q21.1. Science 271, 350–353 (1996).
Mullen, A. C. et al. Master transcription factors determine cell-type-specific responses to TGF-β signaling. Cell 147, 565–576 (2011).
Xu, R. H. et al. NANOG is a direct target of TGFβ/activin-mediated SMAD signaling in human ESCs. Cell Stem Cell 3, 196–206 (2008).
Zheng, H. & Kang, Y. Multilayer control of the EMT master regulators. Oncogene 33, 1755–1763 (2014).
Puisieux, A., Brabletz, T. & Caramel, J. Oncogenic roles of EMT-inducing transcription factors. Nat. Cell Biol. 16, 488–494 (2014).
Brabletz, T. et al. Variable β-catenin expression in colorectal cancers indicates tumor progression driven by the tumor environment. Proc. Natl Acad. Sci. USA 98, 10356–10361 (2001).
Vega, S. et al. Snail blocks the cell cycle and confers resistance to cell death. Genes Dev. 18, 1131–1143 (2004).
Gomis, R. R. et al. A FoxO-Smad synexpression group in human keratinocytes. Proc. Natl Acad. Sci. USA 103, 12747–12752 (2006).
Seoane, J., Le, H. V., Shen, L., Anderson, S. A. & Massague, J. Integration of Smad and forkhead pathways in the control of neuroepithelial and glioblastoma cell proliferation. Cell 117, 211–223 (2004).
Gomis, R. R., Alarcon, C., Nadal, C., Van Poznak, C. & Massague, J. C/EBPβ at the core of the TGFβ cytostatic response and its evasion in metastatic breast cancer cells. Cancer Cell. 10, 203–214 (2006).
David, C. J. et al. TGF-β tumor suppression through a lethal EMT. Cell 164, 1015–1030 (2016).
Xu, J. et al. 14-3-3ζ turns TGF-β’s function from tumor suppressor to metastasis promoter in breast cancer by contextual changes of Smad partners from p53 to Gli2. Cancer Cell. 27, 177–192 (2015).
Pattabiraman, D. R. & Weinberg, R. A. Tackling the cancer stem cells — what challenges do they pose? Nat. Rev. Drug. Discov. 13, 497–512 (2014).
ten Dijke, P. & van Dam, H. 14-3-3ζ turns TGF-β to the dark side. Cancer Cell. 27, 151–153 (2015).
Steeg, P. S. Targeting metastasis. Nat. Rev. Cancer 16, 201–218 (2016).
Wang, Y. W. et al. Identification of oncogenic point mutations and hyperphosphorylation of anaplastic lymphoma kinase in lung cancer. Neoplasia 13, 704–715 (2011).
Chen, C. F. et al. Overlapping high-resolution copy number alterations in cancer genomes identified putative cancer genes in hepatocellular carcinoma. Hepatology 52, 1690–1701 (2010).
Yang, M. H. et al. Direct regulation of TWIST by HIF-1α promotes metastasis. Nat. Cell Biol. 10, 295–305 (2008).
Yang, M. H. et al. Overexpression of NBS1 induces epithelial–mesenchymal transition and co-expression of NBS1 and Snail predicts metastasis of head and neck cancer. Oncogene 26, 1459–1467 (2007).
Chang, C. C., Lin, D. Y., Fang, H. I., Chen, R. H. & Shih, H. M. Daxx mediates the small ubiquitin-like modifier-dependent transcriptional repression of Smad4. J. Biol. Chem. 280, 10164–10173 (2005).
Cheng, G. Z. et al. Twist is transcriptionally induced by activation of STAT3 and mediates STAT3 oncogenic function. J. Biol. Chem. 283, 14665–14673 (2008).
Chang, C. Y., Lin, S. C., Su, W. H., Ho, C. M. & Jou, Y. S. Somatic LMCD1 mutations promoted cell migration and tumor metastasis in hepatocellular carcinoma. Oncogene 31, 2640–2652 (2012).
Acknowledgements
We thank the Common Equipment Core of IBMS and Academia Sinica for help with microscopy, DNA sequencing, the SPF animal facility and flow cytometry. We also acknowledge the National Center for Genome Medicine and Genomics for RNA-seq services, Y. T. Wang and P. C. Jin for their help with animal experiments, and R. Z. Lin and M. H. Yang for reading the manuscript. Our work was supported by grants from the Academia Sinica and Ministry of Science and Technology (MOST) (grant no. 106-0210-01-15-02) and from MOST (grant nos. 1062321B001051 and 1042320B001009MY3) of Taiwan.
Author information
Authors and Affiliations
Contributions
H.-W.Y., E.-C.H., S.-S.L., Y.-D.L., S.-Y.L., C.-F. Chen and Y.-S.J. designed and performed the research. S.-S.L., D.-L.G., C.-M.H. and J.-H.S. performed bioinformatics analyses. Y.-C.L., C.-T.C. C.-F. Cheng, R.-H.C. and R.-B.Y. developed and maintained the rodent technologies. C.-Y.C. and P.-H.T. performed the pathological analyses, and H.-W.Y. and Y.-S.J. analysed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Altered PSPC1 genome and gene expression in cancers of TCGA.
. (a) Genomic DNA alterations of PSPC1 in multiple cancers of TCGA datasets retrieved from cBioportal database. PSPC1 is commonly amplified (red color), deleted (blue color) and mutated (green color) in multiple cancer types. (b) Distribution of PSPC1 expression in different subtypes of breast cancer patients by transcriptome analysis of datasets in TCGA-BRCA (RNA_Seq) and in GSE18229 and GSE45827 (microarrays). n values are provided in the graphs and denote the number of tumours Data are mean +/- sd. (c) Specificity test of PSPC1 antibody to PSPC1 wild type A549 and MDA-MB-231 (red rectangles) and PSPC1 deficient SK-Hep1 (null), A549-PSPC1KO (KO), MDA-MB-231-PSPC1KO (KO) and H1355 (null) (black rectangles) in 40X and 100X magnification. The intensity of PSPC1 staining in IHC score was calculated from 100 cells per sample. ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). The source data are provided in Supplementary Table 3.
Supplementary Figure 2 Expression of PSPC1 enhanced EMT, cell migration, cell invasion in SK-Hep1 and knockdown of PSPC1 in A549, MDA- MB-231 and PC3 cells.
. (a-c) EMT and stemness characteristics were performed by detecting Immunofluorescence expression of E-cadherin (green) and vimentin (red) (representative images are shown from 3 independent experiments) (a), and morphological changes of cell lines in spheroids cultures and in 2D cultures (b), and E-cadherin and vimentin expression by western blotting analysis (c). (d, e) PSPC1 expression increased cell migration (d) and invasion (e) in vitro but reversed by knocking down PSPC1 with shRNAs in cell lines. Error bars are mean +/- sd. n = 3 independent experiments. *P<0.05, **P<0.001, and ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). The source data are provided in Supplementary Table 3.
Supplementary Figure 3 Expression of PSPC1 enhanced stemness and drug resistance in various cancer cell lines including expression of PSPC1 in SK-Hep1 and knockdown of PSPC1 in A549, MDA- MB-231 and PC3 cells.
. (a, b) PSPC1 expression increased tumor metastasis to lung in nude mice models as shown in representative images and fluorescence intensity compared to day zero corresponding to tumor size (n=5 or n=10 tumours as indicated) (a) and relative number of tumor nodules in metastatic lung tissues in vivo (b). In contrast, knockdown of PSPC1 in A549, MDA-MB-231 and PC3 reversed these effects. Data in a and b are from 1 experiment. (c) In stem cell-like ALDH1+ population, relative expression of PSPC1, EMT-TFs and CSC-TFs was increased in compared with that in ALDH1- population in A549, MDA-MB-231 and PC3 cells. (d, e) Expression of PSPC1 increased the ALDH1+ population and the tumor spheroid formation in 3 serial passages but diminished these stemness features by knocking down PSPC1, respectively. (f) Expression of PSPC1 increased drug resistance to doxorubicin treatments in SK-Hep1. (g-j) Expression of PSPC1 reduced apoptotic cell (Annexin V+) population after treatments of dosages of chemotherapeutic drugs cisplatin, 5-FU and taxol (g). In contrast, knockdown PSPC1 in A549 (h), MDA-MB-231 (i) and PC3 (j) increased apoptotic cell populations. *P<0.05, **P<0.001, and ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). All error bars are mean +/- sd. n =3 independent experiments. The source data are provided in Supplementary Table 3.
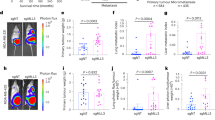
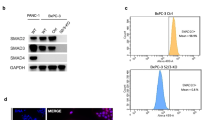
Supplementary Figure 4 Knockdown, knockout and functional characterization of mouse PSPC1 (mPSPC1) in ALDH1+ CSC populations in MMTV-PyMT breast and K-rasLSL-G12D/+;p53fl/fl (KP) lung spontaneous tumor models.
. (a, b) Knockdown efficiency of mPSPC1 by specific shRNAs with western blotting analysis (blots are representative of 3 independent experiments). (c-e, g) Knockdown mPSPC1 reduced ALDH1 stem cell-like populations with and without treatments of ALDH1 inhibitor DEAB. Error bars are mean +/- sd, n=3 independent experiments. (f, h) Knockdown mPSPC1 decreased cell migration capabilities in tumor cells isolated from tumors of two spontaneous mouse models. Error bars are mean +/- sd, n=3 independent experiments. (i-k) Knockout mPSPC1 by CRISPR/Cas9 in MMTV-PyMT tumor cells as shown by western blotting analysis. Data in 1-j are from one experiment. (k) reduced tumor xenograft rate (blots are representative of 3 independent experiments) (i) and tumor weight (j) in compared to control. Error bars are mean +/- sd, n=5. *P<0.05, **P<0.001, and ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). The source data are provided in Supplementary Table 3.
Supplementary Figure 5 PSPC1 NOPS/Coil domain interacts with Smad2/3 to enhance TGFβ1 signaling-mediated EMT and stemness.
. (a, b) PSPC1 interacted with Smad2/3 through NOPS-coil domain were determined by series deletion constructs and specific NOPS-coil domain deleted construct of PSPC1 in IP experiments and by SBE4 promoter-luciferase reporter assays with and without 5 ng/ml TGFβ1 treatment for 6 hours. (c-f) Expression of PSPC1 NOPS-/-mutation in SK-Hep1 abrogated PSPC1 enhanced expression of EMT-TFs and CSC-TFs (c), cell migration (e), and ALDH1 activity containing population in comparison with that of mock and wild type PSPC1-expressing SK-Hep1 (d, f). DEAB is the ALDH1 activity specific inhibitor and SSC is Side Scatter. (g) Phosphorylation of Smad2/3 is required for interaction with PSPC1 by using Smad2/3-3 serine to alanine mutants (n= 3 independent experiments). (h-k) PSPC1 knockdown resistance mutant (PSPC1shmut) avoids shPSPC1#2 knockdown effects on knockdown efficiency by western blotting) (h), TGFβ1 expression at RNA (i) and protein levels (j), and SBE4 promoter luciferase reporter assays (k) in A549 and MDA-MB-231. .All blots are representative of 3 independent experiments. All error bars are mean +/- sd, n = 3 independent experiments. *P<0.05, **P<0.001, and ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). The source data are provided in Supplementary Table 3.
Supplementary Figure 6 Expression of PSPC1 is required for TGFβ1–enhanced EMT and stemness.
. (a-d) PSPC1 increased cell migration (a), spheroid formation (b), and expression (c) and (d) binding to the promoters of EMT-TFs and CSC-TFs could be suppressed by treatment of 20 mM SB431542 (data representative of 3 independent experiments). (e-j) TGFβ1 (5 ng/ml, 24 hr) treatments induced cell migration (e, h), spheroids formation (f, i) and upregulation of EMT-TFs and CSCs-TFs expression (g, j) in A549 cells (e-g) and MDAMB231 (h-j) were abolished by knocking down PSPC1. All error bars are mean +/- sd, n = 3 independent experiments. *P<0.05, **P<0.001, and ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). The source data are provided in Supplementary Table 3.
Supplementary Figure 7 PSPC1 augmented TGFβ1-mediated growth inhibitory effects in non-cancerous cells and increase of cell proliferation capability in cancer cells.
. (a, b) PSPC1 increased TGFβ1 mRNA expression (a) and transcription of SBE4 promoter luciferase reporter assays (b) in non-cancerous Beas2B and MCF-10A cells. (c) In non-cancerous cells (HaCaT, MCF-10A and Beas2B) or PSPC1 deficient (SK-Hep1) cells, expression of PSPC1 in these cells confers more resistance to TGFβ1-induced cell death resulted in lower growth inhibition than mock. (d) in contrast, in PSPC1 highly expressed cancer cells (MDA-MB-231 and A549), knockdown PSPC1 resulted in higher TGFβ1-mediated growth inhibition than mock control. TGFβ1 5 ng/ml was treated for 48 hours. (e) Expression of PSPC1 down-regulated p21, p57 and DAPK1 but increased c-Myc in SK-Hep1. In contrast, knockdown PSPC1 reverted expression of abovementioned genes in A549 and MDA-MB-231 by western blotting analysis. (f) Expression of PSPC1 down-regulated p21, p57 and DAPK1 but increased c-Myc in HaCaT and MCF-10A by western blotting analysis (blots in e and f are representative of 3 independent experiments). (g, h) PSPC1 knockdown up-regulated tumor suppressive genes p15, p21, p57 and DAPK but reduced expression of oncogenic genes C-Myc and PAI-1 in Smad4-deficient pancreatic cancer cells AsPC1 and BxPC3 could be further potentiated by re-expression of Smad4. (i) PSPC1 increased cell proliferation in PSPC1-expressing SK-Hep1. In contrast, knockdown PSPC1 reduced cell proliferation capability in A549 and MDA-MB-231. All error bars are mean +/- sd, n = 3 independent experiments. *P<0.05, **P<0.001, and ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). The source data are provided in Supplementary Table 3.
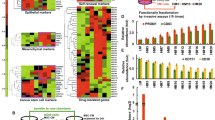
Supplementary Figure 8 PSPC1 enabled the TGFβ1 pro-metastatic switch conducted in the CRISPR/Cas9 knockout A549 by transcriptome (GSE103293), gene enrichment and functional analysis.
. (a, b) Knockout PSPC1 up-regulated cell death-related genes and down-regulated migration and viability genes could be further potentiated by TGFβ1 treatments as shown by IPA analysis (a) and heatmap clustering (b). Data in a and b are from 1 experiment. (c, d) Treatment of TGFβ1 is unable to rescue PSPC1 enhanced cell migration and ALDH1+ cell population in A549PSPC1KO in compared to A549PSPC1wildtype. (e) PSPC1 knockout (PSPC1KO) reduced tumor xenograft rate in compared to parental A549 in limited dilution assays in nude mice (n= 5 mice per genotype, 1 experiment). (f) PSPC1 Knockdown in A549 and MDA-MB-231 and expression in SK-Hep1 did not alter expression of 14-3-3ζ at protein levels by western blotting analysis (blot representative of 3 independent experiments). (g, h) Expression of 14-3-3ζ did not rescue cell migration and ALDH1+ population in PSPC1 knockdown A549 and MDA-MB-231. All error bars are mean +/- sd, n = 3 independent experiments. *P<0.05, **P<0.001, and ***P<0.0001, determined by two-tailed Student’s t-test (95% confidence interval). The source data are provided in Supplementary Table 3.
Supplementary Figure 9
. Unprocessed images of all blots.
Supplementary information
Supplementary Information
Supplementary Figures 1–9 and Supplementary Table legends.
Supplementary Table 1
Altered PSPC1 gene expression in cancers in the TCGA.
Supplementary Table 2
GSEA of transcriptome (GSE103293).
Supplementary Table 3
Source Data.
Supplementary Table 4
List of antibodies.
Supplementary Table 5
List of oligonucleotides for shRNA, sgRNA, real-time PCR and standard ChIP.
Rights and permissions
About this article
Cite this article
Yeh, HW., Hsu, EC., Lee, SS. et al. PSPC1 mediates TGF-β1 autocrine signalling and Smad2/3 target switching to promote EMT, stemness and metastasis. Nat Cell Biol 20, 479–491 (2018). https://doi.org/10.1038/s41556-018-0062-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-018-0062-y
This article is cited by
-
Kinesin Family Member-18A (KIF18A) Promotes Cell Proliferation and Metastasis in Hepatocellular Carcinoma
Digestive Diseases and Sciences (2024)
-
M2 tumor-associated macrophage mediates the maintenance of stemness to promote cisplatin resistance by secreting TGF-β1 in esophageal squamous cell carcinoma
Journal of Translational Medicine (2023)
-
Regulation and signaling pathways in cancer stem cells: implications for targeted therapy for cancer
Molecular Cancer (2023)
-
IL20RB signaling enhances stemness and chemotherapy resistance in pancreatic cancer
Journal of Translational Medicine (2023)
-
Suppression of TGFβR-Smad3 pathway alleviates the syrinx induced by syringomyelia
Cell & Bioscience (2023)