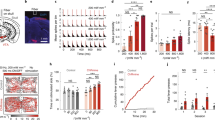
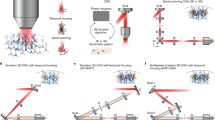
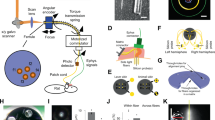
Abstract
Electrical neurostimulation is effective in the treatment of neurological disorders, but associated recording artefacts generally limit its applications to open-loop stimuli. Real-time and continuous closed-loop control of brain activity can, however, be achieved by pairing concurrent electrical recordings and optogenetics. Here we show that closed-loop optogenetic stimulation with excitatory opsins enables the precise manipulation of neural dynamics in brain slices from transgenic mice and in anaesthetized non-human primates. The approach generates oscillations in quiescent tissue, enhances or suppresses endogenous patterns in active tissue and modulates seizure-like bursts elicited by the convulsant 4-aminopyridine. A nonlinear model of the phase-dependent effects of optical stimulation reproduced the modulation of cycles of local-field potentials associated with seizure oscillations, as evidenced by the systematic changes in the variability and entropy of the phase-space trajectories of seizures, which correlated with changes in their duration and intensity. We also show that closed-loop optogenetic neurostimulation could be delivered using intracortical optrodes incorporating light-emitting diodes. Closed-loop optogenetic approaches may be translatable to therapeutic applications in humans.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The source data supporting the results in this study are available from the Newcastle University research repository at https://doi.org/10.25405/data.ncl.19519630. All data generated during the study are available from the corresponding author on reasonable request.
Code availability
Analysis and modelling code are available from the Newcastle University research repository at https://doi.org/10.25405/data.ncl.19519630.
References
Uhlhaas, P. J. & Singer, W. Neural synchrony in brain disorders: relevance for cognitive dysfunctions and pathophysiology. Neuron 52, 155–168 (2006).
Shanechi, M. M. Brain-machine interfaces from motor to mood. Nat. Neurosci. 22, 1554–1564 (2019).
Bouthour, W. et al. Biomarkers for closed-loop deep brain stimulation in Parkinson disease and beyond. Nat. Rev. Neurol. 15, 343–352 (2019).
Cagnan, H., Denison, T., McIntyre, C. & Brown, P. Emerging technologies for improved deep brain stimulation. Nat. Biotechnol. 37, 1024–1033 (2019).
Little, S. et al. Adaptive deep brain stimulation in advanced Parkinson disease. Ann. Neurol. 74, 449–457 (2013).
Skarpaas, T. L., Jarosiewicz, B. & Morrell, M. J. Brain-responsive neurostimulation for epilepsy (RNS® System). Epilepsy Res. 153, 68–70 (2019).
Grosenick, L., Marshel, J. H. & Deisseroth, K. Closed-loop and activity-guided optogenetic control. Neuron 86, 106–139 (2015).
Nicholson, E., Kuzmin, D. A., Leite, M., Akam, T. E. & Kullmann, D. M. Analogue closed-loop optogenetic modulation of hippocampal pyramidal cells dissociates gamma frequency and amplitude. eLife 7, e38346 (2018).
Sohal, V. S., Zhang, F., Yizhar, O. & Deisseroth, K. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 459, 698–702 (2009).
Siegle, J. H. & Wilson, M. A. Enhancement of encoding and retrieval functions through theta phase-specific manipulation of hippocampus. eLife 3, e03061 (2014).
Stark, E. et al. Pyramidal cell-interneuron interactions underlie hippocampal ripple oscillations. Neuron 83, 467–480 (2014).
Kanta, V., Pare, D. & Headley, D. B. Closed-loop control of gamma oscillations in the amygdala demonstrates their role in spatial memory consolidation. Nat. Commun. 10, 3970 (2019).
Hall, T. M., Nazarpour, K. & Jackson, A. Real-time estimation and biofeedback of single-neuron firing rates using local field potentials. Nat. Commun. 5, 5462 (2014).
Borisyuk, R. M. & Kirillov, A. B. Bifurcation analysis of a neural network model. Biol. Cybern. 66, 319–325 (1992).
Wang, Y., Goodfellow, M., Taylor, P. N. & Baier, G. Dynamic mechanisms of neocortical focal seizure onset. PLoS Comput. Biol. 10, e1003787 (2014).
Zhou, Y. et al. Application of approximate entropy on dynamic characteristics of epileptic absence seizure. Neural Regen. Res. 7, 572–577 (2012).
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Taylor, P. N. et al. Optimal control based seizure abatement using patient derived connectivity. Front. Neurosci. 9, 202 (2015).
Tang, E. & Bassett, D. S. Colloquium: control of dynamics in brain networks. Rev. Mod. Phys. 90, 031003 (2018).
Tognoli, E. & Kelso, J. A. The metastable brain. Neuron 81, 35–48 (2014).
Deco, G. et al. Awakening: predicting external stimulation to force transitions between different brain states. Proc. Natl Acad. Sci. USA 116, 18088–18097 (2019).
Jackson, A., Mavoori, J. & Fetz, E. E. Long-term motor cortex plasticity induced by an electronic neural implant. Nature 444, 56–60 (2006).
Zanos, S., Rembado, I., Chen, D. & Fetz, E. E. Phase-locked stimulation during cortical beta oscillations produces bidirectional synaptic plasticity in awake monkeys. Curr. Biol. 28, 2515–2526 (2018).
Suffczynski, P., Kalitzin, S. & Lopes Da Silva, F. H. Dynamics of non-convulsive epileptic phenomena modeled by a bistable neuronal network. Neuroscience 126, 467–484 (2004).
Taylor, P. N. et al. A computational study of stimulus driven epileptic seizure abatement. PLoS ONE 9, e114316 (2014).
Motamedi, G. K. et al. Optimizing parameters for terminating cortical afterdischarges with pulse stimulation. Epilepsia 43, 836–846 (2002).
Osorio, I. & Frei, M. G. Seizure abatement with single dc pulses: is phase resetting at play? Int. J. Neural Syst. 19, 149–156 (2009).
Berényi, A., Belluscio, M., Mao, D. & Buzsáki, G. Closed-loop control of epilepsy by transcranial electrical stimulation. Science 337, 735–737 (2012).
Arlotti, M. et al. Eight-hours adaptive deep brain stimulation in patients with Parkinson disease. Neurology 90, e971–e976 (2018).
Holt, A. B. et al. Phase-dependent suppression of beta oscillations in Parkinson’s disease patients. J. Neurosci. 39, 1119–1134 (2019).
Cagnan, H. et al. Stimulating at the right time: phase-specific deep brain stimulation. Brain 140, 132–145 (2017).
Brittain, J. S., Probert-Smith, P., Aziz, T. Z. & Brown, P. Tremor suppression by rhythmic transcranial current stimulation. Curr. Biol. 23, 436–440 (2013).
McNamara, C. G., Rothwell, M. & Sharott, A. Phase-dependent closed-loop modulation of neural oscillations in vivo. Preprint at bioRxiv https://doi.org/10.1101/2020.05.21.102335 (2020).
Paz, J. T. et al. Closed-loop optogenetic control of thalamus as a tool for interrupting seizures after cortical injury. Nat. Neurosci. 16, 64–70 (2013).
Krook-Magnuson, E., Armstrong, C., Oijala, M. & Soltesz, I. On-demand optogenetic control of spontaneous seizures in temporal lobe epilepsy. Nat. Commun. 4, 1376 (2013).
Lu, Y. et al. Optogenetic dissection of ictal propagation in the hippocampal-entorhinal cortex structures. Nat. Commun. 7, 10962 (2016).
Vormstein-Schneider, D. et al. Viral manipulation of functionally distinct interneurons in mice, non-human primates and humans. Nat. Neurosci. 23, 1629–1636 (2020).
de Lanerolle, N. C., Kim, J. H., Robbins, R. J. & Spencer, D. D. Hippocampal interneuron loss and plasticity in human temporal lobe epilepsy. Brain Res. 495, 387–395 (1989).
Andrioli, A., Alonso-Nanclares, L., Arellano, J. I. & DeFelipe, J. Quantitative analysis of parvalbumin-immunoreactive cells in the human epileptic hippocampus. Neuroscience 149, 131–143 (2007).
Raimondo, J. V., Burman, R. J., Katz, A. A. & Akerman, C. J. Ion dynamics during seizures. Front. Cell. Neurosci. 9, 419 (2015).
González, O. C. et al. Role of KCC2-dependent potassium efflux in 4-aminopyridine-induced epileptiform synchronization. Neurobiol. Dis. 109, 137–147 (2018).
Ledri, M., Madsen, M. G., Nikitidou, L., Kirik, D. & Kokaia, M. Global optogenetic activation of inhibitory interneurons during epileptiform activity. J. Neurosci. 34, 3364–3377 (2014).
Chang, M. et al. Brief activation of GABAergic interneurons initiates the transition to ictal events through post-inhibitory rebound excitation. Neurobiol. Dis. 109, 102–116 (2018).
Magloire, V., Mercier, M. S., Kullmann, D. M. & Pavlov, I. GABAergic interneurons in seizures: investigating causality with optogenetics. Neuroscientist 25, 344–358 (2019).
Jirsa, V. K., Stacey, W. C., Quilichini, P. P., Ivanov, A. I. & Bernard, C. On the nature of seizure dynamics. Brain 137, 2210–2230 (2014).
Govorunova, E. G. et al. Kalium rhodopsins are natural light-gated potassium channels that mediate optogenetic inhibition. Nat. Neurosci. 25, 967–974 (2022).
Lo, P.-C. & Lee, Y.-Y. Applicability of phase-locked loop to tracking the rhythmic activity in EEGS. Circ. Syst. Signal Process. 19, 171–186 (2000).
Ladas, T. P., Chiang, C. C., Gonzalez-Reyes, L. E., Nowak, T. & Durand, D. M. Seizure reduction through interneuron-mediated entrainment using low frequency optical stimulation. Exp. Neurol. 269, 120–132 (2015).
Graham, R. T. et al. Synergistic positive feedback underlying seizure initiation. Preprint at bioRxiv https://doi.org/10.1101/2021.02.28.433224 (2021).
Alfonsa, H., Lakey, J. H., Lightowlers, R. N. & Trevelyan, A. J. Cl-out is a novel cooperative optogenetic tool for extruding chloride from neurons. Nat. Commun. 7, 13495 (2016).
Sahel, J. A. et al. Partial recovery of visual function in a blind patient after optogenetic therapy. Nat. Med. 27, 1223–1229 (2021).
Diester, I. et al. An optogenetic toolbox designed for primates. Nat. Neurosci. 14, 387–397 (2011).
Yazdan-Shahmorad, A. et al. A large-scale interface for optogenetic stimulation and recording in nonhuman primates. Neuron 89, 927–939 (2016).
Andersson, M. et al. Optogenetic control of human neurons in organotypic brain cultures. Sci. Rep. 6, 24818 (2016).
Shen, Y., Campbell, R. E., Côté, D. C. & Paquet, M. E. Challenges for therapeutic applications of opsin-based optogenetic tools in humans. Front. Neural Circ. 14, 41 (2020).
Lamont, C. et al. Silicone encapsulation of thin-film SiOx, SiOxNyand SiC for modern electronic medical implants: a comparative long-term ageing study. J. Neural Eng. 18, 055003 (2021).
Ramezani, R. et al. On-probe neural interface ASIC for combined electrical recording and optogenetic stimulation. IEEE Trans. Biomed. Circ. Syst. 12, 576–588 (2018).
Dehkhoda, F. et al. Self-sensing of temperature rises on light emitting diode based optrodes. J. Neural Eng. 15, 026012 (2018).
Berens, P. CircStat: a MATLAB toolbox for circular statistics. J. Stat. Softw. 31, 10 (2009).
Acknowledgements
The project CANDO (Controlling Abnormal Network Dynamics with Optogenetics) is co-funded by the Wellcome Trust (grant no. 102037; to A.J., A.O., S.N.B., G.J.C., M.O.C., P.D., M.K., F.E.N.L. and A.J.T.) and the Engineering and Physical Sciences Research Council (grant no. A000026; to A.J., A.O., S.N.B., G.J.C., M.O.C., P.D., M.K., F.E.N.L. and A.J.T.). We thank all members of the CANDO consortium (www.cando.ac.uk). A.J. is a Wellcome Trust Senior Research Fellow (grant no. 106149; to A.J.).
Author information
Authors and Affiliations
Contributions
The in vitro experiments were performed and analysed by M.T., A.H., D.W., F.E.N.L., M.O.C., A.J.T. and A.J. The in vivo experiments were performed and analysed by B.Z., M.T., S.N.B. and A.J. Virology and post-mortem histology were performed by C.G., F.M., E.E.M. and G.J.C. The simulations were performed by Y.W., F.H., M.K. and A.J. The closed-loop algorithm was developed by S.N.B. and A.J. The optrode was designed and manufactured by E.E.-C., A.S.I., R.G.B., S.T., A.P., N.P., J.G., N.D., T.G.C., P.D. and A.O. A.J. wrote the manuscript, with input from all authors.
Corresponding author
Ethics declarations
Competing interests
A.J., M.O.C., S.N.B. and P.D. are named as inventors in patent numbers EP3490669A1 (granted) and US20190168021 (pending), filed by Newcastle University, which cover aspects of the closed-loop stimulation method reported in this work. The other authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Henry H. Yin, Andrew Sharott and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Realigning phase shifts relative to high gamma envelope.
a LFP spectra from three different Emx1-ChR2 mouse brain slice sessions with different closed-loop filter frequencies. Bottom plot shows that the phase shifts and frequencies of driven oscillations varied across sessions. b Cross-correlation (over all phase conditions) reveals a varying phase relationship between the LFP and high gamma envelope for different sessions, while the stimulation to high gamma phase was relatively constant. c Schematic of the phase adjustment procedure. (1) During the experiment, the FIR filter generated optical stimulation that was phase shifted relative to the LFP (by an amount set by the experimenter). This stimulation drove neural activity, for which we used the high gamma envelope as a surrogate measure, which then produced LFP. (2) To assess the phase relationship between neural activity and resultant LFP, at the end of the experiment we measured the zero-lag phase of the overall LFP-gamma cross-correlation. (3) We added these two phase shifts to produce an adjusted phase shift of stimulation relative to high gamma envelope. We hypothesized that this overall phase-shift would determine whether the closed-loop dynamics was characterised by positive or negative feedback. d LFP spectra replotted after adjusting phase-shift relative to high gamma envelope. In addition, the frequency axis has been normalised by the filter frequency. As a result, the frequencies of oscillations driven by different phase-shifts are closely aligned. d Phase-shifts (relative to LFP) that drove maximal oscillation plotted against relative phase between LFP and high gamma envelope. Solid line indicates stimulation in phase with high gamma. Maximal modulation was typically obtained for phase-shifts advanced by 0–90° relative to this (circular mean 38°, dashed line).
Extended Data Fig. 2 Implantable LED array used in NHP experiments.
a Images of the silicon fork optrode comprising four shanks with 2 CREE DA2432 LEDs per shank encapsulated with silicone elastomer (NuSil MED-6015). Scale bar: 1 mm. b Measured light power output of each LED for different supply currents (blue). Also shown is the light power output for the commercial light source used for the other datasets. The horizontal axis encompasses the full range of stimulation intensities in both cases. Note that the datasets analysed here used a single LED to activate the tissue.
Extended Data Fig. 3 Replication of in vitro results using viral delivery of opsins.
a Example oscillations driven by CLOSe in quiescent brain slices taken from mice injected with AAV8-hSyn-Chronos-GFP, one of the viruses used in the NHP experiments. b Modulation of LFP power relative to no stimulation for different CLOSe phase-shifts (10 Hz filter). c Phase-dependent modulation of LFP for four different filter frequencies in slices taken from three animals. Colour indicates adjusted phase-shift associated with maximal oscillation. Dashed line shows log-linear fit to data. d 4-AP was then applied to induce seizure-like events. CLOSe modulated the duration of oscillatory bursts. Left: Max/min modulation of burst duration across 3 animals for raw data and sinusoidal fit. Colour shows phase-shift associated with max/min for individual animals. Right: Average modulation of burst duration averaged across three animals. e Same but for LFP power at seizure frequency. Error bars indicate s.e.m. over sessions. R and P values from circular-linear correlation over datasets.
Extended Data Fig. 4 CLOSe modulates the frequency and duration of high-amplitude seizure states in vivo in NHPs.
a Percentage of time spent in seizure state under no stimulation (black) and different CLOSe phase-shifts (coloured) for an example dataset. Seizure state was defined as smoothed, rectified LFP amplitude exceeding a particular threshold, ranging from zero up to the maximum amplitude observed in the no stimulation condition. b Change in time spent in seizure states under CLOSe+ (45° phase-shift) and CLOSe− (270° phase-shift) relative to no stimulation for the four datasets. c Average duration of seizure states exceeding each threshold level. d Relative change in seizure duration under CLOSe+ and CLOSe−. e Frequency of seizure states exceeding the threshold level. f Relative change in seizure frequency under CLOSe+ and CLOSe−. Thick bars in panels b,d,f indicate significant thresholds with significant phase-dependent modulation (P < 0.05, circular-linear correlation over datasets).
Extended Data Fig. 5 Seizure trajectories under different CLOSe phase-shifts in silico and in vitro.
a 2-D delay-embedded LFP trajectories during seizure bursts for the in silico model under all CLOSe phase-shifts. Colour indicates when in the cycle stimulation was delivered. b 3-D trajectories viewed from angle indicated in panel a. c,d Same but for example mouse in vitro session.
Extended Data Fig. 6 Comparison of closed-loop and open-loop stimulation strategies.
a Model simulations comparing CLOSe- versus open-loop delivery of the same optical pattern. Left: Example simulations of LFP under CLOSe- (red) and associated stimulation pattern (cyan). This light pattern was then delivered during a second run of the model (open-loop aligned, yellow). In this case, the only difference was the use of a new noise seed, leading to subtle differences in the seizure dynamics following the onset of stimulation (dashed line). We also delivered the same stimulation pattern during a third run of the model (open-loop random, blue). In this case, the model used the new noise seed and the onset of stimulation occurred at a different phase of the seizure cycle. Right: Summary of 100 model simulations. Despite using the same stimulation pattern, open-loop stimulation performed significantly worse than CLOSe-, and on average extended seizure durations relative to no stimulation. Boxes show median and interquartile range, whiskers show max/min. P values from two-tailed, paired t-test over simulations. b Model simulations comparing no stimulation versus open-loop stimulation at constant frequency. Left: Example simulations of LFP under no stimulation (black) and open-loop stimulation at 20 Hz (grey). Note that open-loop stimulation entrained the seizure cycles and extended the duration of seizures. Right: Relative duration of seizure bursts for increasing intensity of stimulation at different frequencies (average of 100 model stimulations per intensity). Burst duration increases monotonically for all frequencies. Note also that 20 Hz (the frequency of seizure cycles under no stimulation) is most effective at extending seizure duration. c Left: Example seizure bursts elicited by bath application of 4-AP to Emx1-ChR2 mouse brain slices with no stimulation (black) and with open-loop stimulation at constant frequency (grey) triggered by burst onset. Note that open-loop stimulation entrains the seizure cycles and extends the burst duration. Also shown are example seizure bursts under CLOSe with different phase-shifts (colours). Right: Burst duration and LFP power at seizure frequency relative to no stimulation under CLOSe+ (blue), CLOSe− (red) and open-loop stimulation (grey). Mean of 4 sessions from 3 animals. Error bars indicate s.e.m. over sessions. P values from two-tailed, paired t-test over sessions.
Extended Data Fig. 7 Model predictions for different closed-loop optogenetic seizure suppression strategies.
Optogenetic activation/suppression of excitatory/inhibitory neurons can be considered as weak perturbations of the seizure trajectory in different directions in the model state-space. Peripheral plots show predicted impact on seizure burst duration of closed-loop optogenetic strategies that deliver each types of stimulation in a phase-dependent manner. Strategies that suppress excitatory neurons and/or activate inhibitory neurons are generally more effective than activation of excitatory neurons and/or suppression of inhibitory neurons. Nevertheless, most strategies can result in either lengthening or shortening of seizures depending on the phase at which stimulation is delivered, and in all cases, there are particular phases which are more effective at suppressing seizures. Thus optogenetic seizure suppression strategies using a range of different opsins/cell-targets may be improved by delivering stimulation in a closed-loop manner.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2 and caption for Supplementary Video 1.
Supplementary video 1
Model trajectories with different CLOSe phase shifts.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zaaimi, B., Turnbull, M., Hazra, A. et al. Closed-loop optogenetic control of the dynamics of neural activity in non-human primates. Nat. Biomed. Eng 7, 559–575 (2023). https://doi.org/10.1038/s41551-022-00945-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-022-00945-8