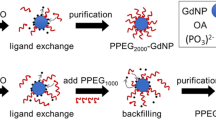
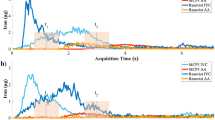
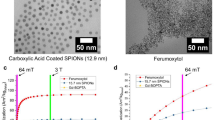
Abstract
Contrast agents for magnetic resonance imaging (MRI) improve anatomical visualizations. However, owing to poor image resolution in whole-body MRI, resolving fine structures is challenging. Here, we report that a nanoparticle with a polysaccharide supramolecular core and a shell of amorphous-like hydrous ferric oxide generating strong T1 MRI contrast (with a relaxivity coefficient ratio of ~1.2) facilitates the imaging, at resolutions of the order of a few hundred micrometres, of cerebral, coronary and peripheral microvessels in rodents and of lower-extremity vessels in rabbits. The nanoparticle can be synthesized at room temperature in aqueous solution and in the absence of surfactants, has blood circulation and renal clearance profiles that prevent opsonization, and leads to better imaging performance than Dotarem (gadoterate meglumine), a clinically approved gadolinium-based MRI contrast agent. The nanoparticle’s biocompatibility and imaging performance may prove advantageous in a broad range of preclinical and clinical applications of MRI.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The main data supporting the results of this study are available within the paper and its Supplementary Information. The raw and analysed datasets generated during the study are too large to be shared publicly, but they are available for research purposes from the corresponding authors upon reasonable request.
References
Hess, S. T., Girirajan, T. P. & Mason, M. D. Ultra-high resolution imaging by fluorescence photoactivation localization microscopy. Biophys. J. 91, 4258–4272 (2006).
Rust, M. J., Bates, M. & Zhuang, X. Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nat. Methods 3, 793–796 (2006).
Renaud, J.-P. et al. Cryo-EM in drug discovery: achievements, limitations and prospects. Nat. Rev. Drug Discov. 17, 471–492 (2018).
Dournes, G. et al. Lung morphology assessment of cystic fibrosis using MRI with ultra-short echo time at submillimeter spatial resolution. Eur. Radiol. 26, 3811–3820 (2016).
Feinberg, D. A. & Setsompop, K. Ultra-fast MRI of the human brain with simultaneous multi-slice imaging. J. Magn. Reson. 229, 90–100 (2013).
Kim, S.-G. & Ogawa, S. Biophysical and physiological origins of blood oxygenation level-dependent fMRI signals. J. Cereb. Blood Flow Metab. 32, 1188–1206 (2012).
Nowogrodzki, A. The world’s strongest MRI machines are pushing human imaging to new limits. Nature 563, 24–26 (2018).
Wahsner, J., Gale, E. M., Rodríguez-Rodríguez, A. & Caravan, P. Chemistry of MRI contrast agents: current challenges and new frontiers. Chem. Rev. 119, 957–1057 (2018).
Arami, H., Khandhar, A., Liggitt, D. & Krishnan, K. M. In vivo delivery, pharmacokinetics, biodistribution and toxicity of iron oxide nanoparticles. Chem. Soc. Rev. 44, 8576–8607 (2015).
Na, H. B., Song, I. C. & Hyeon, T. Inorganic nanoparticles for MRI contrast agents. Adv. Mater. 21, 2133–2148 (2009).
Ruehm, S. G., Christina, H., Violas, X., Corot, C. & Debatin, J. F. MR angiography with a new rapid‐clearance blood pool agent: initial experience in rabbits. Magn. Reson. Med. 48, 844–851 (2002).
Longmire, M., Choyke, P. L. & Kobayashi, H. Clearance properties of nano-sized particles and molecules as imaging agents: considerations and caveats. Nanomedicine (Lond.) 3, 703–717 (2008).
Choi, H. S. et al. Renal clearance of quantum dots. Nat. Biotechnol. 25, 1165–1170 (2007).
Zhang, X., Chen, Y., Zhao, N., Liu, H. & Wei, Y. Citrate modified ferrihydrite microstructures: facile synthesis, strong adsorption and excellent Fenton-like catalytic properties. RSC Adv. 4, 21575–21583 (2014).
Cao, L. et al. Origin of magnetism in hydrothermally aged 2-line ferrihydrite suspensions. Environ. Sci. Technol. 51, 2643–2651 (2017).
Kim, B. H. et al. Large-scale synthesis of uniform and extremely small-sized iron oxide nanoparticles for high-resolution T1 magnetic resonance imaging contrast agents. J. Am. Chem. Soc. 133, 12624–12631 (2011).
Wei, H. et al. Exceedingly small iron oxide nanoparticles as positive MRI contrast agents. Proc. Natl Acad. Sci. USA 114, 2325–2330 (2017).
Jang, J. T. et al. Critical enhancements of MRI contrast and hyperthermic effects by dopant-controlled magnetic nanoparticles. Angew. Chem. Int. Ed. Engl. 48, 1234–1238 (2009).
Lee, J.-H. et al. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nat. Med. 13, 95–99 (2007).
Lu, Y. et al. Iron oxide nanoclusters for T1 magnetic resonance imaging of non-human primates. Nat. Biomed. Eng. 1, 637–643 (2017).
Livney, Y. D. et al. Swelling of dextran gel and osmotic pressure of soluble dextran in the presence of salts. J. Polym. Sci. Pol. Phys. 39, 2740–2750 (2001).
Penfield, J. G. & Reilly, R. F. Jr. What nephrologists need to know about gadolinium. Nat. Rev. Nephrol. 3, 654–668 (2007).
Todd, D. J., Kagan, A., Chibnik, L. B. & Kay, J. Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheumatol. 56, 3433–3441 (2007).
Gulani, V., Calamante, F., Shellock, F. G., Kanal, E. & Reeder, S. B. Gadolinium deposition in the brain: summary of evidence and recommendations. Lancet Neurol. 16, 564–570 (2017).
Huang, J. et al. Facile non-hydrothermal synthesis of oligosaccharide coated sub-5 nm magnetic iron oxide nanoparticles with dual MRI contrast enhancement effects. J. Mater. Chem. B 2, 5344–5351 (2014).
Wang, L. et al. Exerting enhanced permeability and retention effect driven delivery by ultrafine iron oxide nanoparticles with T1–T2 switchable magnetic resonance imaging contrast. ACS Nano 11, 4582–4592 (2017).
Bao, Y., Sherwood, J. & Sun, Z. Magnetic iron oxide nanoparticles as T1 contrast agents for magnetic resonance imaging. J. Mater. Chem. C 6, 1280–1290 (2018).
Lee, N. & Hyeon, T. Designed synthesis of uniformly sized iron oxide nanoparticles for efficient magnetic resonance imaging contrast agents. Chem. Soc. Rev. 41, 2575–2589 (2012).
Liu, J., Yu, M., Zhou, C. & Zheng, J. Renal clearable inorganic nanoparticles: a new frontier of bionanotechnology. Mater. Today 16, 477–486 (2013).
Ehlerding, E. B., Chen, F. & Cai, W. Biodegradable and renal clearable inorganic nanoparticles. Adv. Sci. 3, 1500223 (2016).
Zwanenburg, J. J., Hendrikse, J., Takahara, T., Visser, F. & Luijten, P. R. MR angiography of the cerebral perforating arteries with magnetization prepared anatomical reference at 7 T: comparison with time‐of‐flight. J. Magn. Reson. Imaging 28, 1519–1526 (2008).
Alistair Lammie, G. Pathology of small vessel stroke. Br. Med. Bull. 56, 296–306 (2000).
Faglia, E. et al. Angiographic evaluation of peripheral arterial occlusive disease and its role as a prognostic determinant for major amputation in diabetic subjects with foot ulcers. Diabetes Care 21, 625–630 (1998).
Stepansky, F. et al. Dynamic MR angiography of upper extremity vascular disease: pictorial review. Radiographics 28, e28 (2008).
Murray, C. J. & Lopez, A. D. Mortality by cause for eight regions of the world: Global Burden of Disease Study. Lancet 349, 1269–1276 (1997).
Mair, G. Lack of flow on time-of-flight MR angiography does not always indicate occlusion. BJR Case Rep. 2, 20150187 (2015).
Colby, L. A. & Morenko, B. J. Clinical considerations in rodent bioimaging. Comp. Med. 54, 623–630 (2004).
Kang, H. et al. Renal clearable organic nanocarriers for bioimaging and drug delivery. Adv. Mater. 28, 8162–8168 (2016).
Albanese, A., Tang, P. S. & Chan, W. C. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu. Rev. Biomed. Eng. 14, 1–16 (2012).
Aggarwal, P., Hall, J. B., McLeland, C. B., Dobrovolskaia, M. A. & McNeil, S. E. Nanoparticle interaction with plasma proteins as it relates to particle biodistribution, biocompatibility and therapeutic efficacy. Adv. Drug Deliv. Rev. 61, 428–437 (2009).
Owens, D. E. III & Peppas, N. A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 307, 93–102 (2006).
Zhou, Z. et al. Engineered iron-oxide-based nanoparticles as enhanced T1 contrast agents for efficient tumor imaging. ACS Nano 7, 3287–3296 (2013).
Vinluan, R. D. III & Zheng, J. Serum protein adsorption and excretion pathways of metal nanoparticles. Nanomedicine 10, 2781–2794 (2015).
Saha, A. K. & Brewer, C. F. Determination of the concentrations of oligosaccharides, complex type carbohydrates, and glycoproteins using the phenol-sulfuric acid method. Carbohydr. Res. 254, 157–167 (1994).
Cornell, R. M. & Schwertmann, U. in The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses 533 (John Wiley & Sons, 2003).
Na, H. B. et al. Versatile PEG-derivatized phosphine oxide ligands for water-dispersible metal oxide nanocrystals. Chem. Commun. 5167–5169 (2007).
Shin, T.-H. et al. A magnetic resonance tuning sensor for the MRI detection of biological targets. Nat. Protoc. 13, 2664–2684 (2018).
Ohi, M., Li, Y., Cheng, Y. & Walz, T. Negative staining and image classification—powerful tools in modern electron microscopy. Biol. Proc. Online 6, 23–34 (2004).
Acknowledgements
This work was supported by the Korea Healthcare Technology R&D Project, Ministry for Health & Welfare, Republic of Korea (HI08C2149) and Institute for Basic Science (IBS-R026-D1). We thank H. Y. Kim for helping to synthesize SAIO, K. Kim and B.-K.Yu for helping to perform the TEM analyses, and J. Park for helping to perform the MRI analyses.
Author information
Authors and Affiliations
Contributions
T.-H.S., B.W.C. and J. Cheon conceived of and designed the project. T.-H.S., J. Cheong, S. Kang, S. Kim, Y.L., W.S. and J.D.L. synthesized SAIO, performed the material characterizations and conducted the animal experiments. P.K.K., J.-Y.J. and B.W.C. worked on the MRI experiments and analyses. T.-H.S. and J. Cheon wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
T.-H.S. is a founder of Inventera Pharmaceuticals, a startup company that develops nanoimaging agents.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–15, Tables 1–4, equation (1) and references.
Rights and permissions
About this article
Cite this article
Shin, TH., Kim, P.K., Kang, S. et al. High-resolution T1 MRI via renally clearable dextran nanoparticles with an iron oxide shell. Nat Biomed Eng 5, 252–263 (2021). https://doi.org/10.1038/s41551-021-00687-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-021-00687-z
This article is cited by
-
Physiological principles underlying the kidney targeting of renal nanomedicines
Nature Reviews Nephrology (2024)
-
Doping engineering of iron oxide nanoparticles towards high performance and biocompatible T1-weighted MRI contrast agents
Rare Metals (2024)
-
Hypersensitive MR angiography based on interlocking stratagem for diagnosis of cardiac-cerebral vascular diseases
Nature Communications (2023)
-
Advances in magnetic nanoparticle-based magnetic resonance imaging contrast agents
Nano Research (2023)
-
Biodegradable and biocompatible exceedingly small magnetic iron oxide nanoparticles for T1-weighted magnetic resonance imaging of tumors
Journal of Nanobiotechnology (2022)