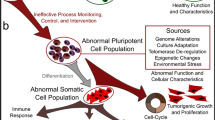
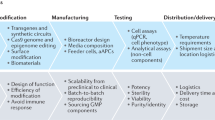
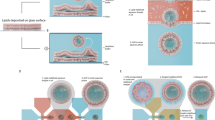
Abstract
The achievements of cell-based therapeutics have galvanized efforts to bring cell therapies to the market. To address the demands of the clinical and eventual commercial-scale production of cells, and with the increasing generation of large clinical datasets from chimeric antigen receptor T-cell immunotherapy, from transplants of engineered haematopoietic stem cells and from other promising cell therapies, an emphasis on biomanufacturing requirements becomes necessary. Robust infrastructure should address current limitations in cell harvesting, expansion, manipulation, purification, preservation and formulation, ultimately leading to successful therapy administration to patients at an acceptable cost. In this Review, we highlight case examples of cutting-edge bioprocessing technologies that improve biomanufacturing efficiency for cell therapies approaching clinical use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bianchi, M. et al. Restoration of NET formation by gene therapy in CGD controls aspergillosis. Blood 114, 2619 (2009).
Grossman, M. et al. Successful ex vivo gene therapy directed to liver in a patient with familial hypercholesterolaemia. Nat. Genet. 6, 335–341 (1994).
Bainbridge, J. W. B. et al. Long-term effect of gene therapy on Leber’s congenital amaurosis. N. Engl. J. Med. 372, 1887–1897 (2015).
Bennett, J. et al. Safety and durability of effect of contralateral-eye administration of AAV2 gene therapy in patients with childhood-onset blindness caused by RPE65 mutations: a follow-on phase 1 trial. Lancet 388, 661–672 (2016).
Clark, M. A., Jepson, M. A. & Hirst, B. H. Exploiting M cells for drug and vaccine delivery. Adv. Drug Deliv. Rev. 50, 81–106 (2001).
Culme-Seymour, E. J., Davie, N. L., Brindley, D. A., Edwards-Parton, S. & Mason, C. A decade of cell therapy clinical trials (2000–2010). Regen. Med. 7, 455–462 (2012).
National Cell Manufacturing Consortium Achieving Large-Scale, Cost-Effective, Reproducible Manufacturing of High-Quality Cells: A Technology Road Map to 2025 (Office of Science, Technology, and Policy, 2016); http://www.cellmanufacturingusa.org/sites/default/files/NCMC_Roadmap_021816_high_res-2.pdf
National Science and Technology Council Advanced Manufacturing: A Snapshot of Priority Technology Areas Across the Federal Government (Office of Science and Technology Policy, 2016).
Human Cells, Tissues, and Cellular and Tissue-based Products, FDA 21 CFR § 1271 (US Government Publishing Office, 2006).
Lipsitz, Y. Y., Timmins, N. E. & Zandstra, P. W. Quality cell therapy manufacturing by design. Nat. Biotechnol. 34, 393–400 (2016).
Pritchett, T. & Little, L. Hard cell: potency testing for cellular therapy products. BioProcess Int. 10, 36–48 (2012).
U.S. Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research Guidance for Industry: Potency Tests for Cellular and Gene Therapy Products (January, 2011).
Castrillo, A., Cardoso, M. & Rouse, L. Treatment of buffy coat platelets in platelet additive solution with the mirasol® pathogen reduction technology system. Transfus. Med. Hemother. 40, 44–48 (2013).
Marschner, S. & Goodrich, R. Pathogen reduction technology treatment of platelets, plasma and whole blood using riboflavin and UV light. Transfus. Med. Hemother. 38, 8–18 (2011).
Wang, J. F., Wu, Y. F., Harrintong, J. & McNiece, I. K. Ex vivo expansions and transplantations of mouse bone marrow-derived hematopoietic stem/progenitor cells. J. Zhejiang Univ. Sci. 5, 157–163 (2004).
Angelopoulou, M. et al. Cotransplantation of human mesenchymal stem cells enhances human myelopoiesis and megakaryocytopoiesis in NOD/SCID mice. Exp. Hematol. 31, 413–420 (2003).
Ball, L. M. et al. Cotransplantation of ex vivo expanded mesenchymal stem cells accelerates lymphocyte recovery and may reduce the risk of graft failure in haploidentical hematopoietic stem-cell transplantation. Blood 110, 2764–2767 (2007).
Le Blanc, K. et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 371, 1579–1586 (2008).
Le Blanc, K. et al. Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 363, 1439–1441 (2004).
Ringden, O. et al. Mesenchymal stem cells for treatment of therapy-resistant graft-versus-host disease. Transplantation 81, 1390–1397 (2006).
Parekkadan, B. et al. Aire controls mesenchymal stem cell-mediated suppression in chronic colitis. Mol. Ther. 20, 178–186 (2012).
Parekkadan, B., Tilles, A. W. & Yarmush, M. L. Bone marrow-derived mesenchymal stem cells ameliorate autoimmune enteropathy independently of regulatory T cells. Stem Cells 26, 1913–1919 (2008).
Parekkadan, B. et al. Bone marrow stromal cell transplants prevent experimental enterocolitis and require host CD11b+ splenocytes. Gastroenterology 140, 966–975 (2011).
Duijvestein, M. et al. Autologous bone marrow-derived mesenchymal stromal cell treatment for refractory luminal Crohn’s disease: results of a phase I study. Gut 59, 1662–1669 (2010).
Semont, A. et al. Mesenchymal stem cells increase self-renewal of small intestinal epithelium and accelerate structural recovery after radiation injury. Adv. Exp. Med. Biol. 585, 19–30 (2006).
Shumakov, V. I., Onishchenko, N. A., Rasulov, M. F., Krasheninnikov, M. E. & Zaidenov, V. A. Mesenchymal bone marrow stem cells more effectively stimulate regeneration of deep burn wounds than embryonic fibroblasts. Bull. Exp. Biol. Med. 136, 192–195 (2003).
Huang, J. et al. Genetic modification of mesenchymal stem cells overexpressing CCR1 increases cell viability, migration, engraftment, and capillary density in the injured myocardium. Circ. Res. 106, 1753–1762 (2010).
Noiseux, N. et al. Mesenchymal stem cells overexpressing Akt dramatically repair infarcted myocardium and improve cardiac function despite infrequent cellular fusion or differentiation. Mol. Ther. 14, 840–850 (2006).
Ortiz, L. A. et al. Mesenchymal stem cell engraftment in lung is enhanced in response to bleomycin exposure and ameliorates its fibrotic effects. Proc. Natl Acad. Sci. USA 100, 8407–8411 (2003).
Gupta, N. et al. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J. Immunol. 179, 1855–1863 (2007).
Lee, J. W. et al. Therapeutic effects of human mesenchymal stem cells in ex vivo human lungs injured with live bacteria. Am. J. Respir. Crit. Care Med. 187, 751–760 (2013).
Matthay, M. A. et al. Therapeutic potential of mesenchymal stem cells for severe acute lung injury. Chest 138, 965–972 (2010).
Parekkadan, B. et al. Mesenchymal stem cell-derived molecules reverse fulminant hepatic failure. PLoS ONE 2, e941 (2007).
van Poll, D. et al. Mesenchymal stem cell-derived molecules directly modulate hepatocellular death and regeneration in vitro and in vivo. Hepatology 47, 1634–1643 (2008).
Togel, F. et al. Administered mesenchymal stem cells protect against ischemic acute renal failure through differentiation-independent mechanisms. Am. J. Physiol. Ren. Physiol. 289, F31–42 (2005).
Elman, J. S. et al. Pharmacokinetics of natural and engineered secreted factors delivered by mesenchymal stromal cells. PLoS ONE 9, e89882 (2014).
Parekkadan, B. & Milwid, J. M. Mesenchymal stem cells as therapeutics. Annu. Rev. Biomed. Eng. 12, 87–117 (2010).
Rowley, J., Abraham, E., Campbell, A., Brandwein, H. & Oh, S. Meeting lot-size challenges of manufacturing adherent cells for therapy. BioProcess Int. 10, 7 (2012).
Lechanteur, C. et al. Clinical-scale expansion of mesenchymal stromal cells: a large banking experience. J. Transl. Med. 14, 145 (2016).
Jones, M. et al. Genetic stability of bone marrow-derived human mesenchymal stromal cells in the quantum system. Cytotherapy 15, 1323–1339 (2013).
Hanley, P. J. et al. Efficient manufacturing of therapeutic mesenchymal stromal cells with the use of the quantum cell expansion system. Cytotherapy 16, 1048–1058 (2014).
Schnitzler, A. C. et al. Bioprocessing of human mesenchymal stem/stromal cells for therapeutic use: current technologies and challenges. Biochem. Eng. J. 108, 3–13 (2016).
Rafiq, Q. A., Coopman, K. & Hewitt, C. J. Scale-up of human mesenchymal stem cell culture: current technologies and future challenges. Curr. Opin. Chem. Eng. 2, 8–16 (2013).
Estrada, J. C. et al. Culture of human mesenchymal stem cells at low oxygen tension improves growth and genetic stability by activating glycolysis. Cell Death Differ. 19, 743–755 (2012).
Sieblist, C. et al. Insights into large-scale cell-culture reactors: II. Gas-phase mixing and CO2 stripping. Biotechnol. J. 6, 1547–1556 (2011).
Nienow, A. W. Reactor engineering in large scale animal cell culture. Cytotechnology 50, 9–33 (2006).
Tan, K. Y., Reuveny, S. & Oh, S. K. W. Recent advances in serum-free microcarrier expansion of mesenchymal stromal cells: parameters to be optimized. Biochem. Biophys. Res. Commun. 473, 769–773 (2016).
dos Santos, F. et al. Toward a clinical-grade expansion of mesenchymal stem cells from human sources: a microcarrier-based culture system under xeno-free conditions. Tissue Eng. Part C Methods 17, 1201–1210 (2011).
Cunha, B. et al. Exploring continuous and integrated strategies for the up- and downstream processing of human mesenchymal stem cells. J. Biotechnol. 213, 97–108 (2015).
Rafiq, Q. A., Brosnan, K. M., Coopman, K., Nienow, A. W. & Hewitt, C. J. Culture of human mesenchymal stem cells on microcarriers in a 5 l stirred-tank bioreactor. Biotechnol. Lett. 35, 1233–1245 (2013).
Yang, H. S., Jeon, O., Bhang, S. H., Lee, S. H. & Kim, B. S. Suspension culture of mammalian cells using thermosensitive microcarrier that allows cell detachment without proteolytic enzyme treatment. Cell Transplant. 19, 1123–1132 (2010).
Shekaran, A. et al. Biodegradable ECM-coated PCL microcarriers support scalable human early MSC expansion and in vivo bone formation. Cytotherapy 18, 1332–1344 (2016).
Hanga, M. P. et al. Expansion of bone marrow-derived human mesenchymal stem/stromal cells (hMSCs) using a two-phase liquid/liquid system. J. Chem. Technol. Biotechnol. 92, 1577–1589 (2017).
Sart, S., Tsai, A.-C., Li, Y. & Ma, T. Three-dimensional aggregates of mesenchymal stem cells: cellular mechanisms, biological properties, and applications. Tissue Eng. Part B Rev. 20, 365–380 (2014).
Dykes, J., Lenshof, A., Astrand-Grundstrom, I. B., Laurell, T. & Scheding, S. Efficient removal of platelets from peripheral blood progenitor cell products using a novel micro-chip based acoustophoretic platform. PLoS ONE 6, e23074 (2011).
Lupu, M., Gooley, T., Zellmer, E., Graves, S. S. & Storb, R. Principles of peripheral blood mononuclear cell apheresis in a preclinical canine model of hematopoietic cell transplantation. J. Vet. Intern. Med. 22, 74–82 (2008).
The dose makes the poison. Nat. Nanotech. 6, 329 (2011).
Perin, E. C. et al. A phase II dose-escalation study of allogeneic mesenchymal precursor cells in patients with ischemic or nonischemic heart failure. Circ. Res. 117, 576–584 (2015).
Hatch, A., Hansmann, G. & Murthy, S. K. Engineered alginate hydrogels for effective microfluidic capture and release of endothelial progenitor cells from whole blood. Langmuir 27, 4257–4264 (2011).
Wang, B. L. et al. Microfluidic high-throughput culturing of single cells for selection based on extracellular metabolite production or consumption. Nat. Biotechnol. 32, 473–478 (2014).
Chabert, M. & Viovy, J.-L. Microfluidic high-throughput encapsulation and hydrodynamic self-sorting of single cells. Proc. Natl Acad. Sci. USA 105, 3191–3196 (2008).
A safety, tolerability, and efficacy study of VC-01™ combination product in subjects with type I diabetes mellitus. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/results/NCT02239354 (2015).
Hsu, C. H., Chen, C., Irimia, D. & Toner, M. Fast sorting of CD4+ T cells from whole blood using glass microbubbles. Technology (Singap. World Sci.) 3, 38–44 (2015).
Toner, M. & Irimia, D. Blood-on-a-chip. Annu. Rev. Biomed. Eng. 7, 77–103 (2005).
Sethu, P. et al. Microfluidic isolation of leukocytes from whole blood for phenotype and gene expression analysis. Anal. Chem. 78, 5453–5461 (2006).
Simon, M. G. et al. Increasing label-free stem cell sorting capacity to reach transplantation-scale throughput. Biomicrofluidics 8, 064106 (2014).
Nakamura, H. & Funahashi, J. Electroporation: past, present and future. Dev. Growth Differ. 55, 15–19 (2013).
Zhao, Y. et al. Multiple injections of electroporated autologous T cells expressing a chimeric antigen receptor mediate regression of human disseminated tumor. Cancer Res. 70, 9053–9061 (2010).
Jin, Z. et al. The hyperactive Sleeping Beauty transposase SB100X improves the genetic modification of T cells to express a chimeric antigen receptor. Gene Ther. 18, 849–856 (2011).
Raja Manuri, P. V. et al. piggyBac transposon/transposase system to generate CD19-specific T cells for the treatment of B-lineage malignancies. Hum. Gene Ther. 21, 427–437 (2010).
Saito, S. et al. Anti-leukemic potency of piggyBac-mediated CD19-specific T cells against refractory Philadelphia chromosome–positive acute lymphoblastic leukemia. Cytotherapy 16, 1257–1269 (2014).
Ramanayake, S. et al. Low-cost generation of Good Manufacturing Practice-grade CD19-specific chimeric antigen receptor–expressing T cells using piggyBac gene transfer and patient-derived materials. Cytotherapy 17, 1251–1267 (2015).
van der Loo, J. C. & Wright, J. F. Progress and challenges in viral vector manufacturing. Hum. Mol. Genet. 25, R42–R52 (2016).
Lim, W. A. & June, C. H. The principles of engineering immune cells to treat cancer. Cell 168, 724–740 (2017).
Sadelain, M. CD19 CAR T cells. Cell 171, 1471 (2017).
Neelapu, S. S. et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Lynn, R. C. et al. High-affinity FRβ-specific CAR T cells eradicate AML and normal myeloid lineage without HSC toxicity. Leukemia 30, 1355–1364 (2016).
Maus, M. V. & June, C. H. Making better chimeric antigen receptors for adoptive T-cell therapy. Clin. Cancer Res. 22, 1875–1884 (2016).
Zhang, B. et al. The significance of controlled conditions in lentiviral vector titration and in the use of multiplicity of infection (MOI) for predicting gene transfer events. Genet. Vaccin. Ther. 2, 6 (2004).
Eichler, F. et al. Hematopoietic stem-cell gene therapy for cerebral adrenoleukodystrophy. N. Engl. J. Med. 377, 1630–1638 (2017).
Thrasher, A. J. & Williams, D. A. Evolving gene therapy in primary immunodeficiency. Mol. Ther. 25, 1132–1141 (2017).
Hacein-Bey-Abina, S. et al. A modified gamma-retrovirus vector for X-linked severe combined immunodeficiency. N. Engl. J. Med. 371, 1407–1417 (2014).
Wu, C. & Dunbar, C. E. Stem cell gene therapy: the risks of insertional mutagenesis and approaches to minimize genotoxicity. Front. Med. 5, 356–371 (2011).
Singh, N., Shi, J., June, C. H. & Ruella, M. Genome-editing technologies in adoptive T cell immunotherapy for cancer. Curr. Hematol. Malig. Rep. 12, 522–529 (2017).
Moore, J. C. et al. Efficient, high-throughput transfection of human embryonic stem cells. Stem Cell Res. Ther. 1, 23 (2010).
Li, L. H. et al. Rapid and efficient nonviral gene delivery of CD154 to primary chronic lymphocytic leukemia cells. Cancer Gene. Ther. 13, 215–224 (2006).
Li, L. H. et al. Highly efficient, large volume flow electroporation. Technol. Cancer Res. Treat. 1, 341–350 (2002).
Fratantoni, J. C., Dzekunov, S., Singh, V. & Liu, L. N. A non-viral gene delivery system designed for clinical use. Cytotherapy 5, 208–210 (2003).
Beatty, G. L. et al. Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol. Res. 2, 112–120 (2014).
Kebriaei, P. et al. Phase I trials using Sleeping Beauty to generate CD19-specific CAR T cells. J. Clin. Invest. 126, 3363–3376 (2016).
Ren, J. et al. Multiplex genome editing to generate universal CAR T cells resistant to PD1 inhibition. Clin. Cancer Res. 23, 2255–2266 (2017).
Sharei, A. et al. Cell squeezing as a robust, microfluidic intracellular delivery platform. J. Vis. Exp. 81, e50980 (2013).
Kim, J. A. et al. A novel electroporation method using a capillary and wire-type electrode. Biosens. Bioelectron. 23, 1353–1360 (2008).
O’Dea, S. et al. Vector-free intracellular delivery by reversible permeabilization. PLoS ONE 12, e0174779 (2017).
Yang, F. et al. Genetic engineering of human stem cells for enhanced angiogenesis using biodegradable polymeric nanoparticles. Proc. Natl Acad. Sci. USA 107, 3317–3322 (2010).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Scudellari, M. How iPS cells changed the world. Nature 534, 310 (2016).
Di Foggia, V., Makwana, P., Ali, R. R. & Sowden, J. C. Induced pluripotent stem cell therapies for degenerative disease of the outer retina: disease modeling and cell replacement. J. Ocul. Pharmacol. Ther. 32, 240–252 (2016).
Focosi, D. et al. Induced pluripotent stem cells in hematology: current and future applications. Blood Cancer J. 4, e211 (2014).
Imaizumi, Y. & Okano, H. Modeling human neurological disorders with induced pluripotent stem cells. J. Neurochem. 129, 388–399 (2014).
Vaccarino, F. M. et al. Annual research review: the promise of stem cell research for neuropsychiatric disorders. J. Child Psychol. Psychiatry 52, 504–516 (2011).
Haggarty, S. J., Silva, M. C., Cross, A., Brandon, N. J. & Perlis, R. H. Advancing drug discovery for neuropsychiatric disorders using patient-specific stem cell models. Mol. Cell. Neurosci. 73, 104–115 (2016).
Bravery, C. A. Do human leukocyte antigen-typed cellular therapeutics based on induced pluripotent stem cells make commercial sense? Stem Cells Dev. 24, 1–10 (2015).
Jacquet, L. et al. Strategy for the creation of clinical grade hESC line banks that HLA-match a target population. EMBO Mol. Med. 5, 10–17 (2013).
Fusaki, N., Ban, H., Nishiyama, A., Saeki, K. & Hasegawa, M. Efficient induction of transgene-free human pluripotent stem cells using a vector based on Sendai virus, an RNA virus that does not integrate into the host genome. Proc. Jpn Acad. Ser. B Phys. Biol. Sci. 85, 348–362 (2009).
Yu, J. et al. Human induced pluripotent stem cells free of vector and transgene sequences. Science 324, 797–801 (2009).
Sridhar, A., Ohlemacher, S. K., Langer, K. B. & Meyer, J. S. Robust differentiation of mRNA-reprogrammed human induced pluripotent stem cells toward a retinal lineage. Stem Cells Transl. Med. 5, 417–426 (2016).
Jenkins, M. J. & Farid, S. S. Human pluripotent stem cell-derived products: advances towards robust, scalable and cost-effective manufacturing strategies. Biotechnol. J. 10, 83–95 (2015).
Lin, H., Li, Q. & Lei, Y. An integrated miniature bioprocessing for personalized human induced pluripotent stem cell expansion and differentiation into neural stem cells. Sci. Rep. 7, 40191 (2017).
Serra, M., Brito, C., Correia, C. & Alves, P. M. Process engineering of human pluripotent stem cells for clinical application. Trends Biotechnol. 30, 350–359 (2012).
Hong, S. G., Dunbar, C. E. & Winkler, T. Assessing the risks of genotoxicity in the therapeutic development of induced pluripotent stem cells. Mol. Ther. 21, 272–281 (2013).
Nguyen, H. T., Geens, M. & Spits, C. Genetic and epigenetic instability in human pluripotent stem cells. Hum. Reprod. Update 19, 187–205 (2013).
Lund, R. J., Narva, E. & Lahesmaa, R. Genetic and epigenetic stability of human pluripotent stem cells. Nat. Rev. Genet. 13, 732–744 (2012).
Trainor, N., Pietak, A. & Smith, T. Rethinking clinical delivery of adult stem cell therapies. Nat. Biotechnol. 32, 729 (2014).
Lipsitz, Y. Y., Timmins, N. E. & Zandstra, P. W. Quality cell therapy manufacturing by design. Nat. Biotechnol. 34, 393–400 (2016).
Choudhary, P. et al. Directing differentiation of pluripotent stem cells toward retinal pigment epithelium lineage. Stem Cells Transl. Med. 6, 490–501 (2017).
Ding, S. et al. Synthetic small molecules that control stem cell fate. Proc. Natl Acad. Sci. USA 100, 7632–7637 (2003).
Saxena, P. et al. A programmable synthetic lineage-control network that differentiates human IPSCs into glucose-sensitive insulin-secreting beta-like cells. Nat. Commun. 7, 11247 (2016).
Kamat, V. et al. MicroRNA screen of human embryonic stem cell differentiation reveals miR-105 as an enhancer of megakaryopoiesis from adult CD34+ cells. Stem Cells 32, 1337–1346 (2014).
Yu, Z., Li, Y., Fan, H., Liu, Z. & Pestell, R. miRNAs regulate stem cell self-renewal and differentiation. Front. Genet. 3, 191 (2012).
Jiang, C. et al. MicroRNA-184 promotes differentiation of the retinal pigment epithelium by targeting the AKT2/mTOR signaling pathway. Oncotarget 7, 52340–52353 (2016).
Loewer, S. et al. Large intergenic non-coding RNA-RoR modulates reprogramming of human induced pluripotent stem cells. Nat. Genet. 42, 1113–1117 (2010).
Li, M. et al. Phenotypic and functional characterization of human bone marrow stromal cells in hollow-fibre bioreactors. J. Tissue Eng. Regen. Med. 6, 369–377 (2012).
Yagi, H. et al. Long-term superior performance of a stem cell/hepatocyte device for the treatment of acute liver failure. Tissue Eng. Part A 15, 3377–3388 (2009).
de Vos, P. et al. Multiscale requirements for bioencapsulation in medicine and biotechnology. Biomaterials 30, 2559–2570 (2009).
Rokstad, A. M. A., Lacik, I., de Vos, P. & Strand, B. L. Advances in biocompatibility and physico-chemical characterization of microspheres for cell encapsulation. Adv. Drug Deliv. Rev. 67–68, 111–130 (2014).
National Diabetes Statistics Report, 2014: Estimates of Diabetes and its Burden in the United States (Center for Disease Control and Prevention, 2014).
Qi, M. Transplantation of encapsulated pancreatic islets as a treatment for patients with type 1 diabetes mellitus. Adv. Med. 2014, 429710 (2014).
Desai, T. & Shea, L. D. Advances in islet encapsulation technologies. Nat. Rev. Drug Discov. 16, 338–350 (2016).
Scharp, D. W. & Marchetti, P. Encapsulated islets for diabetes therapy: history, current progress, and critical issues requiring solution. Adv. Drug Deliv. Rev. 67–68, 35–73 (2014).
Shapiro, A. et al. Islet transplantation in type 1 diabetes: ongoing challenges, refined procedures, and long-term outcome. Rev. Diabet. Stud. 9, 385–406 (2012).
Hrvatin, S. et al. Differentiated human stem cells resemble fetal, not adult, β cells. Proc. Natl Acad. Sci. USA 111, 3038–3043 (2014).
Pagliuca, F. W. et al. Generation of functional human pancreatic β cells in vitro. Cell 159, 428–439 (2014).
O’Sullivan, E. S., Vegas, A., Anderson, D. G. & Weir, G. C. Islets transplanted in immunoisolation devices: a review of the progress and the challenges that remain. Endocr. Rev. 32, 827–844 (2011).
Open-label investigation of the safety and effectiveness of DIABECELL(R) in patients with type I diabetes mellitus. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00940173 (2014).
Open-label investigation of the safety and efficacy of DIABECELL in patients with type 1 diabetes mellitus. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01736228 (2015).
Barkai, U. et al. Enhanced oxygen supply improves islet viability in a new bioartificial pancreas. Cell Transplant. 22, 1463–1476 (2013).
Neufeld, T. et al. The efficacy of an immunoisolating membrane system for islet xenotransplantation in minipigs. PLoS ONE 8, e70150 (2013).
Ludwig, B. et al. Transplantation of human islets without immunosuppression. Proc. Natl Acad. Sci. USA 110, 19054–19058 (2013).
Ludwig, B. et al. A novel device for islet transplantation providing immune protection and oxygen supply. Horm. Metab. Res. 42, 918–922 (2010).
Ludwig, B. et al. Islet transplantation at the Dresden diabetes center: five years’ experience. Horm. Metab. Res. 47, 4–8 (2015).
Pepper, A. R. et al. Diabetes is reversed in a murine model by marginal mass syngeneic islet transplantation using a subcutaneous cell pouch device. Transplantation 99, 2294–2300 (2015).
A phase I/II study of the safety and efficacy of Sernova’s Cell PouchTM for therapeutic islet transplantation ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01652911 (2016).
Tuch, B. E. et al. Safety and viability of microencapsulated human islets transplanted into diabetic humans. Diabetes Care 32, 1887–1889 (2009).
de Groot, M., Schuurs, T. A. & van Schilfgaarde, R. Causes of limited survival of microencapsulated pancreatic islet grafts. J. Surg. Res. 121, 141–150 (2004).
Vegas, A. J. et al. Combinatorial hydrogel library enables identification of materials that mitigate the foreign body response in primates. Nat. Biotechnol. 34, 345–352 (2016).
Vegas, A. J. et al. Long-term glycemic control using polymer-encapsulated human stem cell-derived beta cells in immune-competent mice. Nat. Med. 22, 306–311 (2016).
Martín-Banderas, L. et al. Making drops in microencapsulation processes. Lett. Drug Des. Discov. 7, 300–309 (2010).
Hunt, C. J. Cryopreservation of human stem cells for clinical application: a review. Transfus. Med. Hemother. 38, 107–123 (2011).
Sauer-Heilborn, A., Kadidlo, D. & McCullough, J. Patient care during infusion of hematopoietic progenitor cells. Transfusion 44, 907–916 (2004).
Khera, N. et al. Limiting the daily total nucleated cell dose of cryopreserved peripheral blood stem cell products for autologous transplantation improves infusion-related safety with no adverse impact on hematopoietic engraftment. Biol. Blood Marrow Transplant. 18, 220–228 (2012).
Best, B. P. Cryoprotectant toxicity: facts, issues, and questions. Rejuvenation Res 18, 422–436 (2015).
Song, Y. S. et al. Microfluidics for cryopreservation. Lab Chip 9, 1874–1881 (2009).
Buchanan, S. S. et al. Cryopreservation of stem cells using trehalose: evaluation of the method using a human hematopoietic cell line. Stem Cells Dev. 13, 295–305 (2004).
Eroglu, A. et al. Intracellular trehalose improves the survival of cryopreserved mammalian cells. Nat. Biotechnol. 18, 163–167 (2000).
Thirumala, S., Wu, X., Gimble, J. M. & Devireddy, R. V. Evaluation of polyvinylpyrrolidone as a cryoprotectant for adipose tissue-derived adult stem cells. Tissue Eng. Part C Methods 16, 783–792 (2010).
Shivakumar, S. B. et al. Cryopreservation of human Wharton’s jelly-derived mesenchymal stem cells following controlled rate freezing protocol using different cryoprotectants; a comparative study. Int. J. Stem Cells 8, 155–169 (2015).
Miyamoto, Y. et al. Cryopreservation of human adipose tissue-derived stem/progenitor cells using the silk protein sericin. Cell Transplant. 21, 617–622 (2012).
Moll, G. et al. Do cryopreserved mesenchymal stromal cells display impaired immunomodulatory and therapeutic properties? Stem Cells 32, 2430–2442 (2014).
Robinson, N. J., Picken, A. & Coopman, K. Low temperature cell pausing: an alternative short-term preservation method for use in cell therapies including stem cell applications. Biotechnol. Lett. 36, 201–209 (2014).
Lipsitz, Y. Y. et al. A roadmap for cost-of-goods planning to guide economic production of cell therapy products. Cytotherapy 19, 1383–1391 (2017).
Hassan, S. et al. Allogeneic cell therapy bioprocess economics and optimization: downstream processing decisions. Regen. Med. 10, 591–609 (2015).
Porter, D. L., Levine, B. L., Kalos, M., Bagg, A. & June, C. H. Chimeric antigen receptor–modified T cells in chronic lymphoid leukemia. N. Engl. J. Med. 365, 725–733 (2011).
Acknowledgements
We thank a number of colleagues for feedback on a draft of the manuscript, specifically B. Hampson and T. Heathman from Hitachi Chemical Advanced Therapeutics Solutions. This work was supported in part by the Shriners Hospitals for Children (B.P.) and by the National Institutes of Health Grant R01EB012521 (B.P.).
Author information
Authors and Affiliations
Contributions
All authors contributed to the writing of subsections of the manuscript. A.A., M.L., D.S., R.P. and B.P. contributed to all sections and to figure preparation. D.K., J.T. and R.D. contributed to the content relevant to iPSCs and ESCs. C.L. contributed to content on biopreservation. O.S.F., R.M.O. and D.G.A. contributed to the discussion of cell–material composites. S.L., M.V.M. and J.R. contributed to the cell-engineering discussion. R.N.B. contributed to the content on cell expansion.
Corresponding author
Ethics declarations
Competing interests
A.A., M.L., O.S.F., D.K., M.V.M., J.R., J.T., R.M.O. and S.L. declare no competing interests. D.S., C.L. and R.P. are employees of Hitachi Chemical Advanced Therapeutics Solutions. R.D. owns equity in BlueRock Therapeutics. D.G.A. is a founder and equity shareholder in Siglion Therapeutics. R.N.B. is an employee and equity shareholder of Sentien Biotechnologies, Inc. B.P. is a founder and equity shareholder of Sentien Biotechnologies, Inc.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary tables and references
Supplementary Data
Phase-II and Phase-III clinical trials that include the use of cell therapies
Rights and permissions
About this article
Cite this article
Aijaz, A., Li, M., Smith, D. et al. Biomanufacturing for clinically advanced cell therapies. Nat Biomed Eng 2, 362–376 (2018). https://doi.org/10.1038/s41551-018-0246-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-018-0246-6
This article is cited by
-
Induced pluripotent stem cells (iPSCs): molecular mechanisms of induction and applications
Signal Transduction and Targeted Therapy (2024)
-
Advanced cryopreservation engineering strategies: the critical step to utilize stem cell products
Cell Regeneration (2023)
-
Whole tumour cell-based vaccines: tuning the instruments to orchestrate an optimal antitumour immune response
British Journal of Cancer (2023)
-
Peptide-mediated delivery of CRISPR enzymes for the efficient editing of primary human lymphocytes
Nature Biomedical Engineering (2023)
-
A knockout run for CRISPRed cells
Nature Biomedical Engineering (2023)