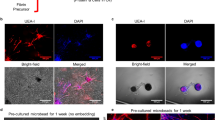
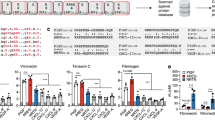
Abstract
Stem-cell-based therapies hold considerable promise for regenerative medicine. However, acute donor-cell death within several weeks after cell delivery remains a critical hurdle for clinical translation. Co-transplantation of stem cells with pro-survival factors can improve cell engraftment, but this strategy has been hampered by the typically short half-lives of the factors and by the use of Matrigel and other scaffolds that are not chemically defined. Here, we report a collagen–dendrimer biomaterial crosslinked with pro-survival peptide analogues that adheres to the extracellular matrix and slowly releases the peptides, significantly prolonging stem cell survival in mouse models of ischaemic injury. The biomaterial can serve as a generic delivery system to improve functional outcomes in cell-replacement therapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Nguyen, P. K., Neofytou, E., Rhee, J. W. & Wu, J. C. Potential strategies to address the major clinical barriers facing stem cell regenerative therapy for cardiovascular disease: a review. JAMA Cardiol. 1, 953–962 (2016).
Zwetsloot, P. P. et al. Cardiac stem cell treatment in myocardial infarction: a systematic review and meta-analysis of preclinical studies. Circ. Res. 118, 1223–1232 (2016).
Discher, D. E., Mooney, D. J. & Zandstra, P. W. Growth factors, matrices, and forces combine and control stem cells. Science 324, 1673–1677 (2009).
Pompe, T., Salchert, K., Alberti, K., Zandstra, P. & Werner, C. Immobilization of growth factors on solid supports for the modulation of stem cell fate. Nat. Protoc. 5, 1042–1050 (2010).
Hu, S. et al. Novel microRNA prosurvival cocktail for improving engraftment and function of cardiac progenitor cell transplantation. Circulation 124, S27–S34 (2011).
Laflamme, M. A. et al. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat. Biotechnol. 25, 1015–1024 (2007).
Seif-Naraghi, S. B. et al. Safety and efficacy of an injectable extracellular matrix hydrogel for treating myocardial infarction. Sci. Transl. Med. 5, 173 (2013).
Cha, C., Liechty, W. B., Khademhosseini, A. & Peppas, N. A. Designing biomaterials to direct stem cell fate. ACS Nano 6, 9353–9358 (2012).
Hahn, J. Y. et al. Pre-treatment of mesenchymal stem cells with a combination of growth factors enhances gap junction formation, cytoprotective effect on cardiomyocytes, and therapeutic efficacy for myocardial infarction. J. Am. Coll. Cardiol. 51, 933–943 (2008).
Zhang, Y., Alexander, P. B. & Wang, X. F. TGF-beta family signaling in the control of cell proliferation and survival. Cold Spring Harb. Perspect. Biol. 9, 1–22 (2017).
Hu, X. et al. A large-scale investigation of hypoxia-preconditioned allogeneic mesenchymal stem cells for myocardial repair in nonhuman primates: paracrine activity without remuscularization. Circ. Res. 118, 970–983 (2016).
Nguyen, P. K., Rhee, J. W. & Wu, J. C. Adult stem cell therapy and heart failure, 2000 to 2016: a systematic review. JAMA Cardiol. 1, 831–841 (2016).
Sheikh, A. Y. et al. In vivo functional and transcriptional profiling of bone marrow stem cells after transplantation into ischemic myocardium. Arterioscler. Thromb. Vasc. Biol. 32, 92–102 (2012).
Muraski, J. A. et al. Pim-1 regulates cardiomyocyte survival downstream of Akt. Nat. Med. 13, 1467–1475 (2007).
Penchala, S. C. et al. A biomimetic approach for enhancing the in vivo half-life of peptides. Nat. Chem. Biol. 11, 793–798 (2015).
Vo, T. N., Kasper, F. K. & Mikos, A. G. Strategies for controlled delivery of growth factors and cells for bone regeneration. Adv. Drug Deliv. Rev. 64, 1292–1309 (2012).
Dingal, P. C. & Discher, D. E. Combining insoluble and soluble factors to steer stem cell fate. Nat. Mater. 13, 532–537 (2014).
Burdick, J. A., Mauck, R. L. & Gerecht, S. To serve and protect: hydrogels to improve stem cell-based therapies. Cell Stem Cell 18, 13–15 (2016).
Davis, M. E. et al. Local myocardial insulin-like growth factor 1 (IGF-1) delivery with biotinylated peptide nanofibers improves cell therapy for myocardial infarction. Proc. Natl Acad. Sci. USA 103, 8155–8160 (2006).
Meghani, N. M., Amin, H. H. & Lee, B. J. Mechanistic applications of click chemistry for pharmaceutical drug discovery and drug delivery. Drug Discov. Today 22, 1604–1619 (2017).
Drzewiecki, K. E., Grisham, D. R., Parmar, A. S., Nanda, V. & Shreiber, D. I. Circular dichroism spectroscopy of collagen fibrillogenesis: a new use for an old technique. Biophys. J. 111, 2377–2386 (2016).
Hubbell, J. A. Cellular matrices: physiology in microfluidics. Nat. Mater. 7, 609–610 (2008).
Zhu, J. L. & Kaufman, L. J. Collagen I self-assembly: revealing the developing structures that generate turbidity. Biophys. J. 106, 1822–1831 (2014).
Jin, E. et al. Acid-active cell-penetrating peptides for in vivo tumor-targeted drug delivery. J. Am. Chem. Soc. 135, 933–940 (2013).
Xu, P. et al. Targeted charge-reversal nanoparticles for nuclear drug delivery. Angew. Chem. Int. Ed. Engl. 46, 4999–5002 (2007).
Lee, S. S. et al. Sulfated glycopeptide nanostructures for multipotent protein activation. Nat. Nanotechnol. 12, 821–829 (2017).
Niiyama, H., Huang, N. F., Rollins, M. D. & Cooke, J. P. Murine model of hindlimb ischemia. J. Vis. Exp. 23, e1035 (2009).
Hu, S. et al. Effects of cellular origin on differentiation of human induced pluripotent stem cell-derived endothelial cells. JCI Insight 1, e85558 (2016).
Li, Z. et al. Imaging survival and function of transplanted cardiac resident stem cells. J. Am. Coll. Cardiol. 53, 1229–1240 (2009).
Smith, R. R. et al. Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens. Circulation 115, 896–908 (2007).
Lutolf, M. P., Gilbert, P. M. & Blau, H. M. Designing materials to direct stem-cell fate. Nature 462, 433–441 (2009).
Kutschka, I. et al. Collagen matrices enhance survival of transplanted cardiomyoblasts and contribute to functional improvement of ischemic rat hearts. Circulation 114, I167–I173 (2006).
Kraehenbuehl, T. P. et al. Human embryonic stem cell-derived microvascular grafts for cardiac tissue preservation after myocardial infarction. Biomaterials 32, 1102–1109 (2011).
Simpson, D., Liu, H., Fan, T. H., Nerem, R. & Dudley, S. C. Jr. A tissue engineering approach to progenitor cell delivery results in significant cell engraftment and improved myocardial remodeling. Stem Cells 25, 2350–2357 (2007).
Cao, F. et al. In vivo visualization of embryonic stem cell survival, proliferation, and migration after cardiac delivery. Circulation 113, 1005–1014 (2006).
Sun, N. et al. Feeder-free derivation of induced pluripotent stem cells from adult human adipose stem cells. Proc. Natl Acad. Sci. USA 106, 15720–15725 (2009).
Acknowledgements
We would like to thank J. Tao for her assistance with the performance of the bindingassay detailed in Supplementary Fig. 16. We would also like to thank Stanford Bio-X (A.S.L.), the National Institutes of Health (grants HL133272 (J.C.W.), HL132875 (J.C.W.),113006 (J.C.W.), EB009035 (J.C.W.) and HL134830-01 (P.K.N.)) and California Institute of Regenerative Medicine (CIRM; grants DR2-05394 and RT3-07798 (J.C.W.)) for funding support forthis study.
Author information
Authors and Affiliations
Contributions
A.S.L., M.I., M.A.L., J.R. and P.K.N. conceived, performed and interpreted the experiments and wrote the manuscript. W.S., M.I. and J.R. formulated and produced the col×D×pep cocktail and characterized it by biophysical and biochemical methods. X.Z., S.P., W.Y.Z. and M.B.P. injected col×D×pep with cells into the animals and performed BLI. A.V.M. performed atomic force microscopy, dynamic light scattering and Raman experiments. X.Z., X.Q., S.P., W.X.H., N.G.K. and W.Y.Z. performed BLI, MRI, echo, Doppler assays and data analysis. S.P., V.S.F., W.Y.Z. and A.D.E. performed the western blot and immunostaining experiments. E.L. performed RNA sequencing. C.K.F.C. performed dissection and fluorescence microscopy experiments and provided experimental advice. P.K.N. performed the imaging experiments and data analysis, provided experimental advice and contributed to manuscript writing. J.R. conceived col×D×pep cocktail formulation, provided experimental advice and contributed to manuscript writing. V.R.P. performed BMMNCs culture. J.C.W. conceived the idea, provided experimental advice and funding support, and contributed to manuscript writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, figures, tables and references.
Rights and permissions
About this article
Cite this article
Lee, A.S., Inayathullah, M., Lijkwan, M.A. et al. Prolonged survival of transplanted stem cells after ischaemic injury via the slow release of pro-survival peptides from a collagen matrix. Nat Biomed Eng 2, 104–113 (2018). https://doi.org/10.1038/s41551-018-0191-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-018-0191-4
This article is cited by
-
Enabling Non-invasive Tracking of Vascular Endothelial Cells Derived from Induced Pluripotent Stem Cells Using Nuclear Imaging
Cardiovascular Engineering and Technology (2024)
-
Generation of Autologous Vascular Endothelial Cells for Patients with Peripheral Artery Disease
Journal of Cardiovascular Translational Research (2023)
-
The sustained PGE2 release matrix improves neovascularization and skeletal muscle regeneration in a hindlimb ischemia model
Journal of Nanobiotechnology (2022)
-
Comparison of the treatment efficacy of umbilical mesenchymal stem cell transplantation via renal subcapsular and parenchymal routes in AKI-CKD mice
Stem Cell Research & Therapy (2022)
-
Constructing a cell microenvironment with biomaterial scaffolds for stem cell therapy
Stem Cell Research & Therapy (2021)