Abstract
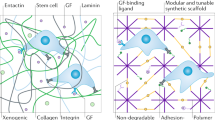
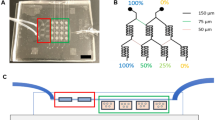
The physiological relevance of Matrigel as a cell-culture substrate and in angiogenesis assays is often called into question. Here, we describe an array-based method for the identification of synthetic hydrogels that promote the formation of robust in vitro vascular networks for the detection of putative vascular disruptors and that support human embryonic stem cell expansion and pluripotency. We identified hydrogel substrates that promote endothelial-network formation by primary human umbilical vein endothelial cells and by endothelial cells derived from human-induced pluripotent stem cells, and used the hydrogels with endothelial networks to identify angiogenesis inhibitors. The synthetic hydrogels showed superior sensitivity and reproducibility over Matrigel when known inhibitors were evaluated, as well as in a blinded screen of a subset of 38 chemicals, selected according to predicted vascular disruption potential, from the Toxicity ForeCaster library of the United States Environmental Protection Agency. We propose that the identified synthetic hydrogels are suitable alternatives to Matrigel for common cell-culture applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Knudsen, T. B. & Kleinstreuer, N. C. Disruption of embryonic vascular development in predictive toxicology. Birth Defects Res. C Embryo Today 93, 312–323 (2011).
De Falco, S. Antiangiogenesis therapy: an update after the first decade. Korean J. Intern. Med. 29, 1–11 (2014).
Folkman, J. & Haudenschild, C. Angiogenesis in vitro. Nature 288, 551–556 (1980).
Kubota, Y., Kleinman, H. K., Martin, G. R. & Lawley, T. J. Role of laminin and basement membrane in the morphological differentiation of human endothelial cells into capillary-like structures. J. Cell Biol. 107, 1589–1598 (1988).
Faulkner, A. et al. A thin layer angiogenesis assay: a modified basement matrix assay for assessment of endothelial cell differentiation. BMC Cell. Biol. 15, 41 (2014).
Arnaoutova, I. & Kleinman, H. K. In vitro angiogenesis: endothelial cell tube formation on gelled basement membrane extract. Nat. Protoc. 5, 628–635 (2010).
Crawford, Y. & Ferrara, N. VEGF inhibition: insights from preclinical and clinical studies. Cell Tissue Res. 335, 261–269 (2009).
Kerbel, R. & Folkman, J. Clinical translation of angiogenesis inhibitors. Nat. Rev. Cancer 2, 727–739 (2002).
Sennino, B. & McDonald, D. M. Controlling escape from angiogenesis inhibitors. Nat. Rev. Cancer 12, 699–709 (2012).
Kleinman, H. K. & Martin, G. R. Matrigel: basement membrane matrix with biological activity. Semin. Cancer Biol. 15, 378–386 (2005).
Xu, C. et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat. Biotechnol. 19, 971–974 (2001).
Villa-Diaz, L. G., Ross, A. M., Lahann, J. & Krebsbach, P. H. Concise review: the evolution of human pluripotent stem cell culture: from feeder cells to synthetic coatings. Stem Cells 31, 1–7 (2013).
Hughes, C., Postovit, L. & Lajoie, G. Matrigel: a complex protein mixture required for optimal growth of cell culture. Proteomics 10, 1886–1890 (2010).
Wood, J. A., Liliensiek, S. J., Russell, P., Nealey, P. F. & Murphy, C. J. Biophysical cueing and vascular endothelial cell behavior. Materials 3, 1620–1639 (2010).
Vukicevic, S. et al. Identification of multiple active growth factors in basement membrane Matrigel suggests caution in interpretation of cellular activity related to extracellular matrix components. Exp. Cell Res. 202, 1–8 (1992).
Stein, C. A., Larocca, R. V., Thomas, R., Mcatee, N. & Myers, C. E. Suramin—an anticancer drug with a unique mechanism of action. J. Clin. Oncol. 7, 499–508 (1989).
Prigozhina, N. L., Heisel, A. J., Seldeen, J. R., Cosford, N. D. & Price, J. H. Amphiphilic suramin dissolves Matrigel, causing an ‘inhibition’ artefact within in vitro angiogenesis assays. Int. J. Exp. Pathol. 94, 412–417 (2013).
Murphy, W. L., McDevitt, T. C. & Engler, A. J. Materials as stem cell regulators. Nat. Mater. 13, 547–557 (2014).
Murrow, L. M., Weber, R. J. & Gartner, Z. J. Dissecting the stem cell niche with organoid models: an engineering-based approach. Development 144, 998–1007 (2017).
Cruz-Acuna, R. & Garcia, A. J. Synthetic hydrogels mimicking basement membrane matrices to promote cell-matrix interactions. Matrix Biol. 57–58, 324–333 (2017).
Ranga, A. et al. Neural tube morphogenesis in synthetic 3D microenvironments. Proc. Natl Acad. Sci. USA 113, E6831–E6839 (2016).
Caiazzo, M. et al. Defined three-dimensional microenvironments boost induction of pluripotency. Nat. Mater. 15, 344–352 (2016).
Gjorevski, N. et al. Designer matrices for intestinal stem cell and organoid culture. Nature 539, 560–564 (2016).
Enemchukwu, N. O. et al. Synthetic matrices reveal contributions of ECM biophysical and biochemical properties to epithelial morphogenesis. J. Cell. Biol. 212, 113–124 (2016).
Huebsch, N. et al. Harnessing traction-mediated manipulation of the cell/matrix interface to control stem-cell fate. Nat. Mater. 9, 518–526 (2010).
Kloxin, A. M., Kasko, A. M., Salinas, C. N. & Anseth, K. S. Photodegradable hydrogels for dynamic tuning of physical and chemical properties. Science 324, 59–63 (2009).
Khetan, S. et al. Degradation-mediated cellular traction directs stem cell fate in covalently crosslinked three-dimensional hydrogels. Nat. Mater. 12, 458–465 (2013).
Fisher, O. Z., Khademhosseini, A., Langer, R. & Peppas, N. A. Bioinspired materials for controlling stem cell fate. Acc. Chem. Res. 43, 419–428 (2010).
Ranga, A. et al. 3D niche microarrays for systems-level analyses of cell fate. Nat. Commun. 5, 4324 (2014).
Singh, S. P. et al. A synthetic modular approach for modeling the role of the 3D microenvironment in tumor progression. Sci. Rep. 5, 17814 (2015).
Gill, B. J. et al. A synthetic matrix with independently tunable biochemistry and mechanical properties to study epithelial morphogenesis and EMT in a lung adenocarcinoma model. Cancer Res. 72, 6013–6023 (2012).
Beck, J. N., Singh, A., Rothenberg, A. R., Elisseeff, J. H. & Ewald, A. J. The independent roles of mechanical, structural and adhesion characteristics of 3D hydrogels on the regulation of cancer invasion and dissemination. Biomaterials 34, 9486–9495 (2013).
Raza, A., Ki, C. S. & Lin, C. C. The influence of matrix properties on growth and morphogenesis of human pancreatic ductal epithelial cells in 3D. Biomaterials 34, 5117–5127 (2013).
Ruoslahti, E. RGD and other recognition sequences for integrins. Annu. Rev. Cell. Dev. Biol. 12, 697–715 (1996).
Nagase, H. & Fields, G. B. Human matrix metalloproteinase specificity studies using collagen sequence-based synthetic peptides. Biopolymers 40, 399–416 (1996).
West, J. L. & Hubbell, J. A. Polymeric biomaterials with degradation sites for proteases involved in cell migration. Macromolecules 32, 241–244 (1999).
Morgan, C. R., Magnotta, F. & Ketley, A. D. Thiol/ene photocurable polymers. J. Polym. Sci. 15, 627–645 (1977).
Belair, D. G. & Murphy, W. L. Specific VEGF sequestering to biomaterials: influence of serum stability. Acta Biomater. 9, 8823–8831 (2013).
Belair, D. G. et al. Human vascular tissue models formed from human induced pluripotent stem cell derived endothelial cells. Stem Cell Rev. 11, 511–525 (2015).
Zhang, J. H., Chung, T. D. & Oldenburg, K. R. A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J. Biomol. Screen. 4, 67–73 (1999).
Kleinstreuer, N. C. et al. Environmental impact on vascular development predicted by high-throughput screening. Environ. Health Perspect. 119, 1596–1603 (2011).
El-Masri, H. et al. Integration of life-stage physiologically based pharmacokinetic models with adverse outcome pathways and environmental exposure models to screen for environmental hazards. Toxicol. Sci. 152, 230–243 (2016).
Knudsen, T. B. et al. Activity profiles of 309 ToxCast™ chemicals evaluated across 292 biochemical targets. Toxicology 282, 1–15 (2011).
Sipes, N. S. et al. Profiling 976 ToxCast chemicals across 331 enzymatic and receptor signaling assays. Chem. Res. Toxicol. 26, 878–895 (2013).
Houck, K. A. et al. Profiling bioactivity of the ToxCast chemical library using BioMAP primary human cell systems. J. Biomol. Screen. 14, 1054–1066 (2009).
Belair, D. G., Schwartz, M. P., Knudsen, T. & Murphy, W. L. Human iPSC-derived endothelial cell sprouting assay in synthetic hydrogel arrays. Acta Biomater. 39, 12–24 (2016).
Tal, T. et al. Screening for chemical vascular disruptors in zebrafish to evaluate a predictive model for developmental vascular toxicity. Reprod. Toxicol. 70, 70–81 (2016).
Fawcett, T. An introduction to ROC analysis. Pattern Recogn. Lett. 27, 861–874 (2006).
Brodersen, K. H., Cheng Soon, O., Stephan, K. E. & Buhmann, J. M. The balanced accuracy and its posterior distribution. In 20th International Conference on Pattern Recognition 3121–3124 (IEEE, 2010).
Latham, A. M. et al. Indolinones and anilinophthalazines differentially target VEGF-A- and basic fibroblast growth factor-mediated responses in primary human endothelial cells. Br. J. Pharmacol. 165, 245–259 (2012).
Brossa, A. et al. Sunitinib but not VEGF blockade inhibits cancer stem cell endothelial differentiation. Oncotarget 6, 11295–11309 (2015).
Friis, T., Engel, A. M., Bendiksen, C. D., Larsen, L. S. & Houen, G. Influence of levamisole and other angiogenesis inhibitors on angiogenesis and endothelial cell morphology in vitro. Cancers (Basel) 5, 762–785 (2013).
Wood, J. M. et al. PTK787/ZK 222584, a novel and potent inhibitor of vascular endothelial growth factor receptor tyrosine kinases, impairs vascular endothelial growth factor-induced responses and tumor growth after oral administration. Cancer Res. 60, 2178–2189 (2000).
Donovan, D., Brown, N. J., Bishop, E. T. & Lewis, C. E. Comparison of three in vitro human ‘angiogenesis’ assays with capillaries formed in vivo. Angiogenesis 4, 113–121 (2001).
Francescone, R. A. III, Faibish, M. & Shao, R. A Matrigel-based tube formation assay to assess the vasculogenic activity of tumor cells. J. Vis. Exp. 7, 3040 (2011).
Song, J., Rolfe, B. E., Hayward, I. P., Campbell, G. R. & Campbell, J. H. Reorganization of structural proteins in vascular smooth muscle cells grown in collagen gel and basement membrane matrices (Matrigel): a comparison with their in situ counterparts. J. Struct. Biol. 133, 43–54 (2001).
Vernon, R., Angello, J., Iruelaarispe, M., Lane, T. & Sage, E. Reorganization of basement-membrane matrices by cellular traction promotes the formation of cellular networks in vitro. Lab. Invest. 66, 536–547 (1992).
Vernon, R. B. & Sage, E. H. Between molecules and morphology. Extracellular matrix and creation of vascular form. Am. J. Pathol. 147, 873–883 (1995).
De Smet, F., Segura, I., De Bock, K., Hohensinner, P. & Carmeliet, P. Mechanisms of vessel branching filopodia on endothelial tip cells lead the way. Arterioscler. Thromb. Vasc. Biol. 29, 639–649 (2009).
Maniotis, A. J. et al. Vascular channel formation by human melanoma cells in vivo and in vitro: vasculogenic mimicry. Am. J. Pathol. 155, 739–752 (1999).
Saunders, R. & Hammer, D. Assembly of human umbilical vein endothelial cells on compliant hydrogels. Cell. Mol. Bioeng. 3, 60–67 (2010).
Rundhaug, J. E. Matrix metalloproteinases and angiogenesis. J. Cell. Mol. Med. 9, 267–285 (2005).
Rashid, A., Kuppa, A., Kunwar, A. & Panda, D. Thalidomide (5HPP-33) suppresses microtubule dynamics and depolymerizes the microtubule network by binding at the vinblastine binding site on tubulin. Biochemistry 54, 2149–2159 (2015).
Satchi-Fainaro, R. et al. Inhibition of vessel permeability by TNP-470 and its polymer conjugate, caplostatin. Cancer Cell 7, 251–261 (2005).
Niwano, M. et al. Inhibition of tumor growth and microvascular angiogenesis by the potent angiogenesis inhibitor, TNP-470, in rats. Surg. Today 28, 915–922 (1998).
Niwa, H., Miyazaki, J. & Smith, A. G. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat. Genet. 24, 372–376 (2000).
Chambers, I. et al. Functional expression cloning of NANOG, a pluripotency sustaining factor in embryonic stem cells. Cell 113, 643–655 (2003).
Radzisheuskaya, A. & Silva, J. C. Do all roads lead to Oct4? The emerging concepts of induced pluripotency. Trends Cell. Biol. 24, 275–284 (2014).
Musah, S. et al. Glycosaminoglycan-binding hydrogels enable mechanical control of human pluripotent stem cell self-renewal. ACS Nano 6, 10168–10177 (2012).
Zhang, R. et al. A thermoresponsive and chemically defined hydrogel for long-term culture of human embryonic stem cells. Nat. Commun. 4, 1335 (2013).
Gharechahi, J. et al. The effect of Rho-associated kinase inhibition on the proteome pattern of dissociated human embryonic stem cells. Mol. Biosyst. 10, 640–652 (2014).
Nguyen, E. H., Zanotelli, M. R., Schwartz, M. P. & Murphy, W. L. Differential effects of cell adhesion, modulus and VEGFR-2 inhibition on capillary network formation in synthetic hydrogel arrays. Biomaterials 35, 2149–2161 (2014).
Schwartz, M. P. et al. Human pluripotent stem cell-derived neural constructs for predicting neural toxicity. Proc. Natl Acad. Sci. USA 112, 12516–12521 (2015).
Schukur, L., Zorlutuna, P., Cha, J. M., Bae, H. & Khademhosseini, A. Directed differentiation of size-controlled embryoid bodies towards endothelial and cardiac lineages in RGD-modified poly(ethylene glycol) hydrogels. Adv. Healthc. Mater. 2, 195–205 (2013).
Hansen, T. D. et al. Biomaterial arrays with defined adhesion ligand densities and matrix stiffness identify distinct phenotypes for tumorigenic and nontumorigenic human mesenchymal cell types. Biomater. Sci. 2, 745–756 (2014).
Fairbanks, B. D. et al. A versatile synthetic extracellular matrix mimic via thiol-norbornene photopolymerization. Adv. Mater. 21, 5005–5010 (2009).
Le, N. N. T., Zorn, S., Schmitt, S. K., Gopalan, P. & Murphy, W. L. Hydrogel arrays formed via differential wettability patterning enable combinatorial screening of stem cell behavior. Acta Biomater. 34, 93–103 (2015).
Hojjat-Farsangi, M. Small-molecule inhibitors of the receptor tyrosine kinases: promising tools for targeted cancer therapies. Int. J. Mol. Sci. 15, 13768–13801 (2014).
Fong, T. A. T. et al. SU5416 is a potent and selective inhibitor of the vascular endothelial growth factor receptor (Flk-1/KDR) that inhibits tyrosine kinase catalysis, tumor vascularization, and growth of multiple tumor types. Cancer Res. 59, 99–106 (1999).
Maynard, S. E. et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfimction, hypertension, and proteinuria in preeclampsia. J. Clin. Invest. 111, 649–658 (2003).
Clark, D. E. et al. A vascular endothelial growth factor antagonist is produced by the human placenta and released into the maternal circulation. Biol. Reprod. 59, 1540–1548 (1998).
Belgore, F. M., Blann, A. D. & Lip, G. Y. H. sFlt-1, a potential antagonist for exogenous VEGF. Circulation 102, E108–E108 (2000).
Hashizume, H. et al. Complementary actions of inhibitors of angiopoietin-2 and VEGF on tumor angiogenesis and growth. Cancer Res. 70, 2213–2223 (2010).
Rosenberg, S. A. Interleukin 2 for patients with renal cancer. Nat. Clin. Pract. Oncol. 4, 497 (2007).
Cao, Y. & Langer, R. Optimizing the delivery of cancer drugs that block angiogenesis. Sci. Transl. Med. 2, 15ps13 (2010).
Richard, A. M. et al. ToxCast chemical landscape: paving the road to 21st century toxicology. Chem. Res. Toxicol. 29, 1225–1251 (2016).
Nguyen, E. H. et al. Dataset for versatile synthetic alternatives to Matrigel for vascular toxicity screening and stem cell expansion. figsharehttp://dx.doi.org/10.6084/m9.figshare.c.3791386 (2017).
Acknowledgements
The authors acknowledge funding from the National Institutes of Health (NIH; R01HL093282-01A1, R21EB016381-01, 1UH2TR000506-01, T32HL007889, T32HL07936, R01EB10039, R24 EY022883, R01 EY026078, P30 EY016665, P30 CA 014520 and 5 P30 CA 014520-01), the Biotechnology Training Program (NIGMS5T32GM08349), the National Science Foundation (GE-0718123), the University of Wisconsin-Madison Graduate Engineering Research Scholars program, the Environmental Protection Agency (STAR grant no. 83573701), the Chemical Safety for Sustainability Research Program, the Office of Research and Development, the Virtual Tissue Models Project and the National Center for Computational Biology, the University of Wisconsin-Madison Molecular and Environmental Toxicity Center Training Program (NIH T32 ES007015), the Gates Millennium Scholars Program and the Retina Research Foundation, and an unrestricted departmental award from Research to Prevent Blindness. N.S. was a recipient of the Research to Prevent Blindness Stein Innovation Award. Mechanical testing data were obtained using the Ares LS2 rheometer at the University of Wisconsin-Madison Soft Materials Laboratory. This study made use of the National Magnetic Resonance Facility at Madison, which is supported by NIH grant P41GM103399 (NIGMS; old number: P41RR002301). Equipment was purchased with funds from the University of Wisconsin-Madison, the NIH (P41GM103399, S10RR02781, S10RR08438, S10RR023438, S10RR025062 and S10RR029220), the NSF (DMB-8415048, OIA-9977486 and BIR-9214394) and the United States Department of Agriculture. The authors acknowledge M. L. Dombroe for assistance with the HUVEC cultures and the laboratory of O. Mezu-Ndubuisi for providing mice for use in the aortic ring sprouting assays. The US Environmental Protection Agency (EPA), through its Office of Research and Development, funded and managed part of the research described here. The views expressed in this paper are those of the authors and do not necessarily reflect the views or policies of the EPA.
Author information
Authors and Affiliations
Contributions
E.H.N., W.T.D., C.S.L., M.F., D.G.B., T.B.K. and G.E.A. contributed to the conception and design of the endothelial-network experiments. E.H.N., W.T.D., M.F., M.P.S., C.S.L. and M.A.S. contributed to the execution of the endothelial-network experiments. E.H.N., W.T.D., M.F., M.P.S., C.S.L., D.G.B., M.A.S. and T.B.K. contributed to the analysis and figure preparation of the endothelial-network experiments. N.N.T.L. contributed to the conception, design and execution of the hESC experiments. E.H.N. and N.N.T.L. contributed to the analysis and figure preparation of the hESC experiments. E.H.N. and C.S.L. contributed to the conception, design and execution of the hydrogel characterization experiments. E.H.N., W.T.D., T.B.K. and W.L.M. drafted the manuscript. N.S. and W.L.M. supervised the work throughout data collection and manuscript preparation. W.L.M. approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
C.S.L. is an employee and stockholder of Stem Pharm. W.L.M. is a founder and stockholder of Stem Pharm.
Supplementary information
Supplementary Information
Supplementary discussion, results, methods, figures and references. (PDF 3663 kb)
Supplementary Video 1
Endothelial-network formation by HUVECs over the course of 24 hours after seeding onto PEG hydrogels. (WMV 9353 kb)
Supplementary Video 2
Endothelial-network formation by HUVECs over the course of 24 hours after seeding onto Matrigel. (WMV 6650 kb)
Supplementary Video 3
Endothelial-network formation by iPSC-ECs over the course of 24 hours after seeding onto PEG hydrogels. (WMV 12369 kb)
Supplementary Video 4
Formation process of the thin hydrogel array. (MP4 25323 kb)
Rights and permissions
About this article
Cite this article
Nguyen, E., Daly, W., Le, N. et al. Versatile synthetic alternatives to Matrigel for vascular toxicity screening and stem cell expansion. Nat Biomed Eng 1, 0096 (2017). https://doi.org/10.1038/s41551-017-0096
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-017-0096
This article is cited by
-
Biomaterial-based platforms for tumour tissue engineering
Nature Reviews Materials (2023)
-
Bead-jet printing enabled sparse mesenchymal stem cell patterning augments skeletal muscle and hair follicle regeneration
Nature Communications (2022)
-
Towards organoid culture without Matrigel
Communications Biology (2021)
-
The Essential Need for a Validated Potency Assay for Cell-Based Therapies in Cardiac Regenerative and Reparative Medicine. A Practical Approach to Test Development
Stem Cell Reviews and Reports (2021)
-
Factors to consider when interrogating 3D culture models with plate readers or automated microscopes
In Vitro Cellular & Developmental Biology - Animal (2021)