Abstract
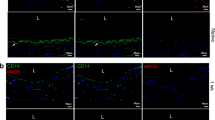
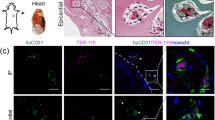
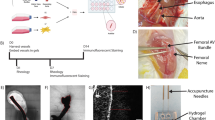
Notwithstanding the remarkable progress in vascular network engineering, implanted bioengineered microvessels mostly fail to form anastomoses with the host vasculature. Here we demonstrate that implants containing assembled human vascular networks (A-grafts) fail to engraft owing to their inability to engage non-inflammatory host neutrophils upon implantation into mice. By contrast, unassembled vascular cells (U-grafts) readily engage alternatively polarized neutrophils, which in turn serve as indispensable mediators of vascular assembly and anastomosis. The depletion of host neutrophils abrogated vascularization in U-grafts, whereas an adoptive transfer of neutrophils fully restored vascularization in myeloid-depleted mice. Neutrophil engagement was regulated by secreted factors and was progressively silenced as the vasculature matured. Exogenous addition of factors from U-grafts re-engaged neutrophils and enhanced revascularization in A-grafts, a process that was recapitulated by blocking Notch signalling. Our data suggest that the pro-vascularization potential of neutrophils can be harnessed to improve the engraftment of bioengineered tissues.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Griffith, L. G. & Naughton, G. Tissue engineering—current challenges and expanding opportunities. Science 295, 1009–1014 (2002).
Rouwkema, J., Rivron, N. C. & Van Blitterswijk, C. A. Vascularization in tissue engineering. Trends Biotechnol. 26, 434–441 (2008).
Novosel, E. C., Kleinhans, C. & Kluger, P. J. Vascularization is the key challenge in tissue engineering. Adv. Drug Deliv. Rev. 63, 300–311 (2011).
Schechner, J. S. et al. In vivo formation of complex microvessels lined by human endothelial cells in an immunodeficient mouse. Proc. Natl Acad. Sci. USA 97, 9191–9196 (2000).
Chen, Y.-C. et al. Functional human vascular network generated in photocrosslinkable gelatin methacrylate hydrogels. Adv. Funct. Mater. 22, 2027–2039 (2012).
Jain, R. K., Au, P., Tam, J., Duda, D. G. & Fukumura, D. Engineering vascularized tissue. Nat. Biotechnol. 23, 821–823 (2005).
Melero-Martin, J. M. et al. Engineering robust and functional vascular networks in vivo with human adult and cord blood-derived progenitor cells. Circ. Res. 103, 194–202 (2008).
Traktuev, D. O. et al. Robust functional vascular network formation in vivo by cooperation of adipose progenitor and endothelial cells. Circ. Res. 104, 1410–1420 (2009).
Chen, X. et al. Rapid anastomosis of endothelial progenitor cell-derived vessels with host vasculature is promoted by a high density of cotransplanted fibroblasts. Tissue Eng. Part A 16, 585–594 (2010).
Moisidis, E., Heath, T., Boorer, C., Ho, K. & Deva, A. K. A prospective, blinded, randomized, controlled clinical trial of topical negative pressure use in skin grafting. Plast. Reconstr. Surg. 114, 917–922 (2004).
Larry, M. W. & Eber, L. L. S. Considerations in nerve repair. Proc. Bayl. Univ. Med. Cent. 16, 152–156 (2003).
Giannoudis, P. V., Dinopoulos, H. & Tsiridis, E. Bone substitutes: an update. Injury 36, S20–S27 (2005).
Kølle, S., Fischer-Nielsen, A., Mathiasen, A. B. & Elberg, J. J. Enrichment of autologous fat grafts with ex-vivo expanded adipose tissue-derived stem cells for graft survival: a randomised placebo-controlled trial. Lancet 13, 1113–1120 (2013).
Mohr, F. W. et al. Coronary artery bypass graft surgery versus percutaneous coronary intervention in patients with three-vessel disease and left main coronary disease: 5-year follow-up of the randomised, clinical SYNTAX trial. Lancet 381, 629–638 (2013).
Serruys, P. W. et al. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N. Engl. J. Med. 360, 961–972 (2009).
Rogers, G. F. & Greene, A. K. Autogenous bone graft: basic science and clinical implications. J. Craniofac. Surg. 23, 323–327 (2012).
Nahrendorf, M. et al. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J. Exp. Med. 204, 3037–3047 (2007).
Grunewald, M. et al. VEGF-induced adult neovascularization: recruitment, retention, and role of accessory cells. Cell 124, 175–189 (2006).
Shojaei, F., Zhong, C., Wu, X., Yu, L. & Ferrara, N. Role of myeloid cells in tumor angiogenesis and growth. Trends Cell Biol. 18, 372–378 (2008).
De Palma, M. et al. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8, 211–226 (2005).
De Palma, M., Venneri, M. A., Roca, C. & Naldini, L. Targeting exogenous genes to tumor angiogenesis by transplantation of genetically modified hematopoietic stem cells. Nat. Med. 9, 789–795 (2003).
Nozawa, H., Chiu, C. & Hanahan, D. Infiltrating neutrophils mediate the initial angiogenic switch in a mouse model of multistage carcinogenesis. Proc. Natl Acad. Sci. USA 103, 12493–12498 (2006).
Bekes, E. M. et al. Tumor-recruited neutrophils and neutrophil TIMP-free MMP-9 regulate coordinately the levels of tumor angiogenesis and efficiency of malignant cell intravasation. Am. J. Pathol. 179, 1455–1470 (2011).
Piccard, H., Muschel, R. J. & Opdenakker, G. On the dual roles and polarized phenotypes of neutrophils in tumor development and progression. Crit. Rev. Oncol. Hematol. 82, 296–309 (2012).
Christoffersson, G. et al. VEGF-A recruits a proangiogenic MMP-9-delivering neutrophil subset that induces angiogenesis in transplanted hypoxic tissue. Blood 120, 4653–4662 (2012).
Massena, S. et al. Identification and characterization of VEGF-A-responsive neutrophils expressing CD49d, VEGFR1, and CXCR4 in mice and humans. Blood 126, 2016–2026 (2015).
Melero-Martin, J. M. et al. In vivo vasculogenic potential of human blood-derived endothelial progenitor cells. Blood 109, 4761–4768 (2007).
Lin, R.-Z., Chen, Y.-C., Moreno-Luna, R., Khademhosseini, A. & Melero-Martin, J. M. Transdermal regulation of vascular network bioengineering using a photopolymerizable methacrylated gelatin hydrogel. Biomaterials 34, 6785–6796 (2013).
Melero-Martin, J. M. et al. Host myeloid cells are necessary for creating bioengineered human vascular networks in vivo . Tissue Eng. Part A 16, 2457–2466 (2010).
Lin, R.-Z. et al. Human endothelial colony-forming cells serve as trophic mediators for mesenchymal stem cell engraftment via paracrine signaling. Proc. Natl Acad. Sci. USA 111, 10137–10142 (2014).
Fridlender, Z. G. et al. Polarization of tumor-associated neutrophil phenotype by TGF-β: “N1” versus “N2” TAN. Cancer Cell 16, 183–194 (2009).
Suchting, S. et al. The Notch ligand Delta-like 4 negatively regulates endothelial tip cell formation and vessel branching. Proc. Natl Acad. Sci. USA 104, 3225–3230 (2007).
Roca, C. & Adams, R. H. Regulation of vascular morphogenesis by Notch signaling. Genes Dev. 21, 2511–2524 (2007).
White, J. R. et al. Identification of a potent, selective non-peptide CXCR2 antagonist that inhibits interleukin-8-induced neutrophil migration. J. Biol. Chem. 273, 10095–10098 (1998).
Mantovani, A. The yin-yang of tumor-associated neutrophils. Cancer Cell 16, 173–174 (2009).
Fridlender, Z. G. & Albelda, S. M. Tumor-associated neutrophils: friend or foe? Carcinogenesis 33, 949–955 (2012).
Sagiv, J. Y. et al. Phenotypic diversity and plasticity in circulating neutrophil subpopulations in cancer. Cell Rep. 10, 562–573 (2015).
Fridlender, Z. G. et al. Transcriptomic analysis comparing tumor-associated neutrophils with granulocytic myeloid-derived suppressor cells and normal neutrophils. PLoS ONE 7, e31524 (2012).
Cuartero, M. I. et al. N2 neutrophils, novel players in brain inflammation after stroke: modulation by the PPARγ agonist rosiglitazone. Stroke 44, 3498–3508 (2013).
Ma, Y. et al. Temporal neutrophil polarization following myocardial infarction. Cardiovasc. Res. 110, 51–61 (2016).
Fantin, A. et al. Tissue macrophages act as cellular chaperones for vascular anastomosis downstream of VEGF-mediated endothelial tip cell induction. Blood 116, 829–840 (2010).
Murdoch, C., Giannoudis, A. & Lewis, C. E. Mechanisms regulating the recruitment of macrophages into hypoxic areas of tumors and other ischemic tissues. Blood 104, 2224–2234 (2004).
Murdoch, C., Muthana, M., Coffelt, S. B. & Lewis, C. E. The role of myeloid cells in the promotion of tumour angiogenesis. Nat. Rev. Cancer 8, 618–631 (2008).
Benelli, R. et al. Neutrophils as a key cellular target for angiostatin: implications for regulation of angiogenesis and inflammation. FASEB J. 16, 267–269 (2002).
Baranski, J. D. et al. Geometric control of vascular networks to enhance engineered tissue integration and function. Proc. Natl Acad. Sci. USA 110, 7586–7591 (2013).
Riemenschneider, S. B. et al. Inosculation and perfusion of pre-vascularized tissue patches containing aligned human microvessels after myocardial infarction. Biomaterials 97, 51–61 (2016).
Ausprunk, D. H., Knighton, D. R. & Folkman, M. J. Vascularization of normal and neoplastic tissues grafted to the chick chorioallantois. Role of host and preexisting graft blood vessels. Am. J. Pathol. 79, 597–618 (1975).
Nolan, D. J. et al. Molecular signatures of tissue-specific microvascular endothelial cell heterogeneity in organ maintenance and regeneration. Dev. Cell 26, 204–219 (2013).
Lee, J.-H. et al. Lung stem cell differentiation in mice directed by endothelial cells via a BMP4–NFATc1–thrombospondin-1 axis. Cell 156, 440–455 (2014).
Hu, J. et al. Endothelial cell-derived angiopoietin-2 controls liver regeneration as a spatiotemporal rheostat. Science 343, 416–419 (2014).
Kusumbe, A. P., Ramasamy, S. K. & Adams, R. H. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature 507, 323–328 (2014).
Shen, Q. et al. Endothelial cells stimulate self-renewal and expand neurogenesis of neural stem cells. Science 304, 1338–1340 (2004).
Zhou, P. et al. Interrogating translational efficiency and lineage-specific transcriptomes using ribosome affinity purification. Proc. Natl Acad. Sci. USA 110, 15395–15400 (2013).
Wang, Y. et al. Ephrin-B2 controls VEGF-induced angiogenesis and lymphangiogenesis. Nature 465, 483–486 (2010).
Lin, R.-Z. et al. Induction of erythropoiesis using human vascular networks genetically engineered for controlled erythropoietin release. Blood 118, 5420–5428 (2011).
Acknowledgements
We thank S.-C. S. Jaminet and D. Li (Center for Vascular Biology, Beth Israel Deaconess Medical Center, Boston, USA) for quantitative reverse-transcription polymerase chain reaction analyses. Histology was supported by Core Facility of the Dana-Farber/Harvard Cancer Center (P30 CA06516). This work was supported by National Institutes of Health grants R00EB009096, R01AR069038, R01HL128452 and R21AI123883 to J.M.M.-M.
Author information
Authors and Affiliations
Contributions
R.-Z.L. and J.M.M.-M. conceived and designed the project. R.-Z.L., C.N.L., R.M.-L., J.N., P.Z., M.S., M.A.M. and J.M.M.-M. performed the experimental work. All authors discussed and analysed the data and edited the results. W.T.P., B.P. and S.E. provided crucial materials. R.-Z.L. and J.M.M.-M. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary notes. (PDF 4906 kb)
Rights and permissions
About this article
Cite this article
Lin, RZ., Lee, C., Moreno-Luna, R. et al. Host non-inflammatory neutrophils mediate the engraftment of bioengineered vascular networks. Nat Biomed Eng 1, 0081 (2017). https://doi.org/10.1038/s41551-017-0081
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-017-0081
This article is cited by
-
Engineering primitive multiscale chimeric vasculature by combining human microvessels with explanted murine vessels
Scientific Reports (2024)
-
Nanosilicate-functionalized nanofibrous membrane facilitated periodontal regeneration potential by harnessing periodontal ligament cell-mediated osteogenesis and immunomodulation
Journal of Nanobiotechnology (2023)
-
Pathological angiogenesis: mechanisms and therapeutic strategies
Angiogenesis (2023)
-
Autologous bone marrow-derived nucleated cell (aBMNC) transplantation improves endometrial function in patients with refractory Asherman’s syndrome or with thin and dysfunctional endometrium
Journal of Assisted Reproduction and Genetics (2023)
-
Liver ischaemia–reperfusion injury: a new understanding of the role of innate immunity
Nature Reviews Gastroenterology & Hepatology (2022)