Abstract
Efficient CO2 separation technologies are essential for mitigating climate change. Compared to traditional thermochemical methods, electrochemically mediated carbon capture using redox-tunable sorbents emerges as a promising alternative due to its versatility and energy efficiency. However, the undesirable linear free-energy relationship between redox potential and CO2 binding affinity in existing chemistry makes it fundamentally challenging to optimise key sorbent properties independently via chemical modifications. Here, we demonstrate a design paradigm for electrochemically mediated carbon capture sorbents, which breaks the undesirable scaling relationship by leveraging intramolecular hydrogen bonding in isoindigo derivatives. The redox potentials of isoindigos can be anodically shifted by >350 mV to impart sorbents with high oxygen stability without compromising CO2 binding, culminating in a system with minimised parasitic reactions. With the synthetic space presented, our effort provides a generalisable strategy to finetune interactions between redox-active organic molecules and CO2, addressing a longstanding challenge in developing effective carbon capture methods driven by non-conventional stimuli.
Similar content being viewed by others
Introduction
Carbon capture from stationary emitters or directly from the ambient environment, followed by sequestration or utilisation, is critical to mitigating climate change1,2,3. However, the incumbent wet chemical scrubbing methods for carbon dioxide (CO2) separation are technically and economically challenged by various inherent limitations, including high energy consumption for sorbent regeneration, thermal degradation, complexity in heat integration when retrofitting existing infrastructures, process equipment corrosion, and fugitive emission of volatile toxic sorbents to the environment4,5. Alternatively, electrochemically mediated carbon capture (EMCC) has emerged as a promising technology6,7,8,9,10,11. In EMCC, reversible CO2 capture and release is modulated by switching electrochemical potentials. Therefore, they can be operated isothermally at ambient pressure, powered by renewable energy sources, and modularly designed to accommodate the multiscale nature of carbon capture needs. Among the EMCC mechanisms explored to date, one popular strategy is to use redox-active organic compounds as CO2 carriers (redox-tunable Lewis bases), with quinones being the most studied class of molecules12,13,14. Electro-reduction of these molecules generates nucleophiles that form adducts with electrophilic CO2, which can be later oxidised to liberate pure CO2 while regenerating the sorbents (Fig. 1a). The past two decades have witnessed steady research progress in developing EMCC processes using redox-tunable sorbents8,10,15,16,17,18,19,20. Several bench-scale prototypes have been demonstrated for fixed-bed and flow-based CO2 separations attributed to materials and device-level engineering efforts17,18,19. In contrast, molecular-level design principles for precise sorbent property tuning remain largely unestablished beyond the simplistic method of structural substitution with electron-donating and withdrawing groups, despite their central role in EMCC.
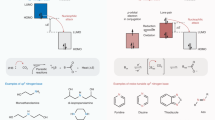
a A universal designing pattern for redox-tunable CO2 carriers containing α,β-unsaturated 1,4-diketone functionality. In previous designs, CO2 is only bonded to the reduced O centre via carbonate formation, where the binding affinity is sensitive to structural modification. b Bifunctional redox-tunable CO2 carrier design based on isoindigo. The secondary functionality of amide allows intramolecular hydrogen bonding, providing an extra handle to stablise the CO2 adduct, thereby enhancing the CO2 binding strength. c A summary of E1/2 of typical quinone-based sorbents and isoindigos under CO2, and their \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) in DMF (filled and empty dots represent the first and second E1/2 under CO2, respectively circle, isoindigos; diamond, benzoquinone; triangle, naphthoquinone; square, anthraquinone).
The lack of reliable sorbent chemistry has intrinsically hindered existing EMCC processes. A practical EMCC system usually requires chemical modification of redox-tunable sorbents to improve key properties such as solubility, processibility, and stability against impurities. For example, anodically shifting the redox potential will enhance the robustness of activated sorbent against molecular oxygen (O2), a common gas stream impurity, for improved carbon capture efficiency. Nevertheless, the CO2 binding affinity of existing redox-tunable Lewis base sorbents, such as quinones, is highly susceptible to chemical modifications, which decreases dramatically as the redox potential shifts anodically21,22,23. The seminal work from W. L. Bell et al. introduced a method to calculate the CO2 binding constant (\({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\)) of activated sorbent, which is widely adopted later to evaluate the CO2 binding strength12. As an example, anthraquinone exhibits a two-electron-transfer half-wave potential (E1/2) of −1.4 V vs. ferrocenium/ferrocene (Fc+/Fc) in N,N-dimethylformamide (DMF) under CO2 and a \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) of ~13.4 (Fig. 1c). The installation of electron-withdrawing groups (EWGs), such as one chloro group at 2-position, can anodically shift E1/2 to −1.25 V vs. Fc+/Fc, yet the \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) decreased substantially to 2.73 (Fig. 1c and Supplementary Table 1)21. A minimum \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) of ~3.0 in DMF is required to attain a practical efficiency for point source carbon capture (10% CO2), and the \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) must be >~5.5 for atmospheric CO2 concentration (400 ppm)12. Importantly, it is challenging to overcome the coupling between E1/2 and \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) as it is dictated by the fundamental principle in chemistry that electron deficiency facilitates reduction but in return weakens nucleophilicity24. Therefore, to fulfil the practical requirements of EMCC, it is pivotal to develop new classes of redox-tunable sorbents that can break the linear free-energy relationship between redox potential and CO2 binding strength to enable aerobic stability and high CO2 capacity (Supplementary Fig. 1).
Here, through analysing the structures of existing EMCC sorbents, we observe that the most representative quinoid species share a common pattern of α,β-unsaturated 1,4-diketone (Fig. 1a). Inspired by this structural pattern, we present a class of redox-tunable CO2 carriers based on isoindigo compounds, which can successfully overcome the undesirable coupling between redox potential and CO2 binding strength (Fig. 1b, c). With a molecular library of 21 examples and a combined experimental and computational effort, we show that the α,β-unsaturated 1,4-diketone in isoindigos plays the role of redox backbone for CO2 binding and the amide groups act as extra docking sites for CO2 complexation via intramolecular hydrogen bonding. This unique bifunctional structural design allows a wide range of chemical modifications to independently optimise key sorbent properties without sacrificing their abilities for CO2 binding. The isoindigo family culminates in an EMCC system that can operate at mild potentials (around −1 V vs. Fc+/Fc) with \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) maintained at ~9. The value is five orders of magnitude higher than tetrachloroquinone with alcohol additives22, which is the state-of-the-art sorbent chemistry with an attempt to break the linear free-energy relationship. Flow-based separation prototypes have also been demonstrated to evaluate the EMCC performance of the isoindigo sorbents, which can achieve CO2 capacity utilisation efficiencies up to ~80% and energy consumptions as low as 127.3 kJ mol−1 per CO2 capture/release cycle. With intrinsic O2 stability, high structural tunability, and synthetic feasibility, isoindigos are promising to serve as the next-generation EMCC sorbents. Moreover, this work demonstrates a generalisable strategy to overcome the intrinsic linear free-energy limits in redox-active organic species that can be broadly applied to EMCC and beyond.
Results
Redox-tunable CO2 absorption of isoindigo
Isoindigo and its derivatives have been extensively utilised as core building blocks in organic semiconductors25,26,27,28, but have rarely been explored as redox molecules for organic electrodes. We envisage isoindigos bearing α,β-unsaturated 1,4-diketone functionalities to be redox-active and can complex with CO2 at the oxygen centres in the reduced state. A series of electrochemical experiments was conducted to validate the redox-driven interaction between isoindigo (IId) and CO2. First, we performed bulk electrolysis of IId using 0.25 M lithium perchlorate (LiClO4) in dimethyl sulfoxide (DMSO) as the supporting electrolyte under N2 or CO2 atmosphere (Fig. 2a). Under N2, the dark dispersion of IId turned greenish at the beginning of the reduction and became a clear yellow solution when the reaction was completed. This suggests the formation of the bisindolidenolate intermediate, which is expected to absorb CO2 as a Lewis base. Interestingly, the yellow solution quickly turned red when purged with CO2, implying the formation of the IId-CO2 adduct. To our delight, 1H and 13C nuclear magnetic resonance (NMR) spectra of the crude solution confirm the full conversion of IId into IId-CO2 with negligible side products (Fig. 2b). Characteristic peaks for the carbonate carbon were observed at 176 ~ 177 pm in 13C NMR. Besides, the proton on the lactam N shifts upfield to 10.33 ppm in IId-CO2 compared to that of 10.89 ppm in IId, strongly evidencing the formation of intramolecular hydrogen bonding with complexed CO2 (highlighted in red in Fig. 2a). The NMR spectra exhibit two types of amide hydrogens and carbonate carbons, which is probably due to the rotational isomerisation of IId-CO2 (Supplementary Fig. 2). The presence of intramolecular hydrogen bonding in IId-CO2 was further verified by variable-temperature (VT) 1H NMR, 2D 1H-13C Heteronuclear Single Quantum Coherence (HSQC) NMR, and Fourier transform infra-red (FT-IR) experiments (Supplementary Figs. 3–5, see Supplementary Note 1 for detailed analysis). The full NMR peak assignment of IId and IId-CO2 is given in Supplementary Fig. 2.
a Bulk electrolysis of isoindigo (IId) under N2 or CO2 atmosphere. The CO2 adduct can be obtained either by directly reducing IId (40 mM) in DMSO with 0.25 M LiClO4 under CO2 or by reducing IId under N2 followed by purging with CO2. b 1H NMR and 13C NMR spectra of the crude solution after bulk electrolysis under CO2. The minor isomer peaks are marked with an asterisk. c CV curves of IId (2.5 mM) in DMF using 0.1 M Nbu4PF6 as the supporting salt under N2 (grey) and CO2 (red) at a scan rate of −50 mV s−1 (5 mM ferrocene as the internal reference). ∆Epeak(2) is labelled for the calculation of \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\).
Since the discovery of the EMCC mechanism using quinones in 198812,13, to our best knowledge, the widely accepted electrochemically generated quinone-CO2 carbonate adduct is still a proposed structure and has not been confirmed in an EMCC process by non-ambiguous characterisations. This is probably due to the poor stability of the adducts and the transient bonding nature between the reduced sorbent and CO2. On the contrary, crude NMR spectra suggest bulk electro-reduction of IId under CO2 can yield the proposed adduct with full conversion (IId was fully consumed), indicating the high selectivity of this chemistry and the sufficient stability of the adduct, which we postulate to be the result of intramolecular hydrogen bonding. Noticeably, the IId-CO2 solution obtained from bulk electrolysis can be stably stored in ambient air with high water content. After 53 days, <9 mol% of IId-CO2 oxidised back to the neutral IId form (Supplementary Fig. 6).
To confirm the redox activity of IId, we measured its cyclic voltammetry (CV) using 0.1 M tetrabutylammonium hexafluorophosphate (NBu4PF6) in DMF as the supporting electrolyte (Fig. 2c). Under an inert N2 atmosphere, IId exhibits two major redox waves typical to stepwise two-electron transfer. Like quinoid species13, the two electron transfer steps correspond to the formation of anionic radicals (IId⋅−) and dianions (IId2−), respectively. The two reduction peaks under N2 emerge into one in the presence of CO2 with a nearly doubled peak current, indicating chemical interactions between reduced IId and CO2. This behaviour is analogous to the other redox-tunable CO2 sorbents reported previously based on oxygen or nitrogen binding centres, whose reaction with CO2 proceeds via an ECEC mechanism (E, electron transfer; C, chemical reaction)12,13,20. As a Lewis acid, CO2 can withdraw the electron density from IId⋅− to promote the second electron transfer, giving rise to an anodically shifted second reduction wave. Besides, the oxidation peak also shifts anodically and becomes quasi-reversible, corroborating the formation of the IId-CO2 adduct, which requires more energy for CO2 desorption.
This preliminary finding encouraged us to expand the chemical scope of isoindigos and search for more qualified EMCC compounds. Isoindigos can be synthesised through the condensation reaction between 2-oxindoles and isatins. This allows the modular design of redox sorbents, where the two building units can be modified separately and integrated into isoindigos in the last step, thereby addressing the synthetic barriers when engineering EMCC sorbents (Supplementary Note 2). Here, we demonstrate the structural modification of isoindigo at 5, 6, and N-positions with 21 examples (Fig. 3) and examine in detail the interplay between substituent groups and hydrogen bonding on the redox and CO2 binding behaviours of isoindigos to establish the underlying structure-property relationships. The main conclusions are summarised in Fig. 4 and will be discussed in detail in the following sections.
a CV of various isoindigos using 2.5 mM compound in DMF with 0.1 M NBu4PF6 under N2 (grey) or CO2 (coloured). The CV curves were recorded at a scan rate of −50 mV s−1 at 298 K. b DFT-optimised structure of IId; positions for introducing substituent groups are labelled, which are at 5, 6, and lactam N-position; intramolecular hydrogen bonding (a indicates bond length) in the neutral state is shown as blue dashed lines. c DFT-optimised structure of 5BIId suggests EWG can strengthen the hydrogen bonding (a). d DFT-optimised structures of 55BIId (left) and 66DBIId (right) showing that EWG substituent at 5-position is more effective in strengthening the hydrogen bonding (a). e DFT simulation showing that intramolecular hydrogen bonding (b) indicates bond length, red dashed lines) occurs in the CO2 adduct, stabilising the CO2 binding. Colour of atoms: grey: C; red: O; blue: N; brown: Br; white: H.
Tuning the redox potential of isoindigos
We first examine the redox behaviours of derivatised isoindigos under N2 via CV. Delightfully, all the isoindigos tested display redox couples typical to stepwise two-electron transfer, underscoring the good electrochemical reversibility of these molecules (Fig. 4a, Supplementary Figs. 7–9).
Among the 21 examples of isoindigos, only IId, 6BIId, and 66DBIId exhibit less well-defined shapes for the second redox wave. This is probably due to the rotational isomerisation of the one-electron reduced isoindigo radical anions. As shown in Supplementary Fig. 10, the C = C bond connecting the two oxindole rings becomes a single bond in the radical anion, allowing free rotation of the two rings. This creates rotational isomers and gives rise to shoulder peaks in the second redox process. Adding strong EWGs or substitution groups at the 5-position of isoindigo can create dipole-dipole repulsion or steric hindrance, inhibiting rotational isomerisation and resulting in a more reversible second redox process. Similarly, adding CO2 onto isoindigos also creates dipole and steric hindrance, impeding the rotational isomerisation and leading to more defined CV curves.
Ideal EMCC sorbents shall have redox potentials more positive than the oxygen reduction potential (−1.35 V vs. Fc+/Fc in DMF, Supplementary Fig. 11) to minimise sorbent sensitivity towards O2. With stronger or increasing numbers of EWG introduced to the isoindigo rings, the redox potential exhibits an increasing anodic shift in the sequence of -F, -Br, -COOMe, -CONHR, and -NO2 substituent groups from mono- to tetra-substitution (Fig. 5a and Supplementary Table 2). Through a close examination of the structure-property relationship, we hypothesise that the anodic shift is attributed to not only the commonly expected electronic state tuning from the EWG substituents but also the intramolecular hydrogen bonding effect (a shown in Fig. 4b and c). This is because hydrogen bonding can decrease the electron density of the redox-active oxygen centre to facilitate reduction. The downfield shift of proton at 4-position (Ha) in isoindigos with EWG substituents suggests the formation of stronger hydrogen bonding (a) (Table 1 and Supplementary Table 2).
a The effect of substituent group on the first electron-transfer half-wave potentials under N2. b The plot between the first electron transfer half-wave potential under N2 and the change in CO2 binding constant relative to unmodified isoindigo. There is no linear correlation (R2 < 0.1) between the relative CO2 binding constants and the half-wave potentials.
To verify the above hypothesis, we show that EWG substituent at 5-position is more effective in facilitating electro-reduction than that at 6-position. This is because the former is in the ortho-position of Ha and more effective in pulling away the electron density, thereby inducing a stronger hydrogen bonding (Fig. 4d). For instance, 55DBIId and 66DBIId exhibit very close 1H NMR peaks for Hb (11.11 and 11.10 ppm, Supplementary Fig. 12), suggesting similar degrees of electron deficiency in these two molecules. In contrast, the chemical shift of Ha is 9.32 and 8.99 ppm for 55DBIId and 66DBIId, respectively, clearly indicating a stronger hydrogen bonding (a) in the 5-substituted species. Therefore, E1/2(IId/IId⋅−) of 55DBIId (−1.09 V vs. Fc+/Fc) is more positive compared to 66DBIId (−1.12 V vs. Fc+/Fc). This trend is consistent for all examples in our isoindigo family (Supplementary Table 2, e.g., 5BIId vs. 6BIId and 6MCIId vs. 5NIId vs. 5N6MCIId) and is further confirmed with DFT calculations (details vide post).
Counterintuitively, adding electron-donating groups (EDGs) such as methoxy shows a negligible influence on E1/2(IId/IId⋅−), as evidenced by IId vs. 55DMIId (both show E1/2 of −1.29 V vs. Fc+/Fc, Table 1), and 5NIId vs. 5M5NIId (both show E1/2 of −1.08 V vs. Fc+/Fc, Supplementary Table 2). This is likely due to the charge-transfer effect that lowers the energy level of the molecule, offsetting the electronic effect from EDGs. Specifically, isoindigo species are electron acceptors (n-type organic semiconductors)28, where charge transfer can be induced between the electron-deficient isoindigo rings and the electron-rich methoxy group. UV-vis absorption spectra suggest an optical bandgap of 1.90 ~ 1.98 eV for most isoindigos with or without chemical modification (Supplementary Table 2 and Supplementary Figs. 13–15). However, the bandgaps drop to 1.78 and 1.71 eV for methoxy substituted 55DMIId and 5M5NIId, manifesting charge transfer in these two compounds.
Breaking the scaling relationship between redox potential and CO2 affinity
After understanding the effect of molecular structures on the redox potentials of isoindigos, we further study their CO2 binding behaviours. All isoindigos in this work exhibit anodically shifted potential for the second electron transfer process under CO2, confirming their ability to form CO2 adducts upon electro-reduction. Importantly, through the combined effect of EWGs substitution and hydrogen bonding (a) discussed above, all isoindigos with unsubstituted Hb display E1/2 values anodic to oxygen reduction under CO2, implying favourable stability of isoindigo sorbents against O2.
In all previous reports on redox-tunable CO2 sorbents, anodically shifted redox potential always comes with significantly diminished CO2 binding affinity (Supplementary Table 1). However, another key finding of this work is that hydrogen atom (Hb) on the lactam-N of isoindigos can induce intermolecular hydrogen bonding with the complexed CO2 molecule to thermodynamically stabilise the CO2 adduct (Fig. 4e). As a result, regardless of E1/2, the \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) values of isoindigos with unsubstituted Hb were found to be relatively constant (Table 1 and Supplementary Table 2, see Supplementary Note 3 for details on \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) calculation), suggesting the high tolerance of such redox carriers to a wide range of chemical modifications.
To further demonstrate the role of hydrogen bonding (b) on CO2 adduct stabilisation, we synthesised two examples with N-substitutions to eliminate this hydrogen bonding (NNDPr66DBIId and NNDEHIId). Compared with 6,6’-dibromo substituted 66DBIId (\(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) = 9.59), the CO2 binding constant of N-alkylated NNDPr66DBIId (\(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) = 4.48) shows a dramatic decrease of five orders of magnitude (Table 1). A similar phenomenon was observed between NNDEHIId (\(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) = 6.62, Supplementary Table 2) and IId (\(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) = 9.34). Besides, we designed an isoindigo bearing a carboxylic acid group (6CIId), which serves as a free proton donor to disrupt the intramolecular hydrogen bonding (b). As a result, the CO2 binding ability of reduced 6CIId almost diminished with a low \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) of 2.87 (Supplementary Table 2).
To visualise our success in breaking the scaling relationship between redox potential and CO2 binding affinity, we plot the change in \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) relative to unmodified IId against E1/2 of the first electron transfer under N2 (Fig. 5b). Upon installing EWGs, E1/2 can be effectively shifted from −1.29 V to −0.97 V vs. Fc+/Fc. Nevertheless, the \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) values exhibit minimal changes within only one order of magnitude, as long as the lactam-N is unsubstituted to facilitate intramolecular hydrogen bonding. Furthermore, linear fitting shows a negligible correlation (R2 < 0.1), underlining that our molecular design strategy can indeed break the scaling relationship between redox potential and CO2 binding affinity.
We further tested the bimolecular rate constant (kbimolecular) for the reaction between isoindigo radical anion and CO2 (Supplementary Figs. 16–19, Supplementary Table 3, see Supplementary Note 3 for details in measurement). With intramolecular hydrogen bonding (b) in the CO2 adduct, 6MCIId, 6AIIdSer, and 66DBIId display a similar rate constant of 22.5–18.6 M−1 s−1. However, the N,N-disubstituted NNDPr66DBIId exhibits a decreased rate constant of 3.3 M−1 s−1, manifesting that intramolecular hydrogen bonding (b) is also conducive to CO2 complexation kinetics.
At a CO2 concentration of 20% or 10%, the CV curves of isoindigos usually exhibit a positively shifted second reduction peak compared to that under N2; however, most do not completely emerge into the first (Supplementary Fig. 7 and 8). The phenomenon is attributed to the kinetic competition between the chemical transformation of IId⋅− into [IId-CO2]⋅− (r = kbimolecular[CO2][IId⋅−]29, where [CO2] and [IId⋅−] are the concentrations of CO2 and IId⋅−) and the electrochemical reduction of [IId-CO2]⋅−. Higher CO2 concentrations lead to higher r, which facilitates the formation of [IId-CO2]⋅− and ultimately leads to a single, merged reduction peak. Correspondingly, we tested the CV of 6MCIId at different scan rates and indeed observed the gradual merging of the two reduction peaks with slower scans (Supplementary Fig. 20). Therefore, the separation between the two cathodic peaks under low CO2 concentrations can serve as a qualitative indicator for CO2 complexation kinetics.
As a final note, CO2 complexation can be frustrated when introducing highly strong EWGs or strong hydrogen bonding acceptors to isoindigo (5N6MCIId and 6CIIdNa). Detailed analysis is included in Supplementary Note 4 and Supplementary Fig. 21.
Detailed investigation on the role of hydrogen bonding
Distinct from previously reported redox-tunable CO2 carriers, isoindigos possess intramolecular hydrogen bondings in both inactivated (neutral) and activated (reduced) forms, accounting for their unique EMCC properties. Therefore, we selected the seven most representative structures from our library and carefully compared various parameters such as NMR spectra, density functional theory (DFT)-optimised bond lengths, redox potentials, and CO2 binding constants to investigate the interplay between substituent groups and hydrogen bonding on the thermodynamic properties of isoindigos. Key data are summarised in Table 1 (also see Supplementary Figs. 22 and 23 for DFT-optimised structures). DFT calculation shows that the theoretical first electron transfer potential shifts anodically in the order of IId, 5BIId, 66DBIId, and 55DBIId, consistent with experimental observation (Supplementary Table 4). Besides, DFT-calculated CO2 binding constants (\(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\)) of the isoindigos agree well with the trend of our experimental results (Supplementary Table 5). DFT-optimised structures further confirm the formation of intramolecular hydrogen bonding (a) and (b).
The electron density of Ha at 4-position and Hb at N-position can be regarded as indicators of the bond strength of a and b, respectively. Specifically, 1H NMR spectra reveal a chemical shift of 9.06 ppm for Ha and 10.89 ppm for Hb in unmodified IId, corresponding to a DFT-optimised bond length of 1.963 Å for a and 1.954 Å for b. Introducing electron-withdrawing bromo groups at 5-position (55DBIId) downfield shifts Ha to 9.32 ppm and Hb to 11.11 ppm, resulting in an enhanced hydrogen bonding of 1.939 Å for a and 1.946 Å for b, respectively. Interestingly, the electronics and hydrogen bonding of each oxindole ring can be independently tuned in nonsymmetric isoindigos. Using the nonsymmetric 5BIId as an example, the chemical shifts of Ha and Hb are 9.07 and 10.96 ppm at the non-substituted side, corresponding to a DFT-optimised bond length of 1.962 Å for a and 1.957 Å for b, which is very close to IId. In contrast, Ha and Hb shift downfield to 9.31 and 11.05 ppm at the bromo-substituted side, corresponding to an enhanced hydrogen bonding of 1.936 Å for a and 1.943 Å for b.
Moreover, introducing the same EWG at different positions modulates the strength of hydrogen bonding (a) differently. For example, with the same dibromo-substitution, the chemical shift of Ha in 66DBIId (8.99 ppm) is upfield to that of 55DBIId (9.32 ppm). This results in a weakened hydrogen bonding (a) of 1.961 Å in 66DBIId than that of 1.939 Å in 55DBIId, explaining the more negative reduction potential of 66DBIId (−1.14 V vs. Fc+/Fc) than 55DBIId (−1.09 V vs. Fc+/Fc). The above results strongly suggest that, in addition to the electronic effect of substituent groups, intramolecular hydrogen bonding (a) is also vital in facilitating the reduction of isoindigo.
Although decreasing the electron density of isoindigos by introducing EWGs at 5,6-positions can effectively facilitate their reduction, counterintuitively, the reduced isoindigos exhibit negligible decay in CO2 affinities, underscoring the importance of intramolecular hydrogen bonding (b). Using DFT calculation, we found that the nucleophilicity of the oxygen centre indeed weakens when EWG is introduced, as suggested by the increased length of the carbonate C–O bond (c). For instance, the bond length of c on the oxindole ring with –COOMe group is increased by 1.2 pm compared to that on the non-substituted ring in 6MCIId. However, as mentioned above, hydrogen bonding (b) strengthens with stronger or increasing number of EWG substituents to keep the bond length of c nearly constant. Moreover, the DFT-optimised structure suggests that hydrogen bonding (b) is precluded in NNDPr66DBIId, and the carbonate bends out-of-plane to the reduced isoindigo rings due to steric repulsion. Thus, the bond length of c in NNDPr66DBIId is increased by 1.8 pm compared to 66DBIId, giving rise to a significant drop in \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) by almost five orders of magnitude.
The collective information above confirms our key conclusions. First, hydrogen bonding (a) can reduce the electron density at the redox centre and facilitate reduction. Second, hydrogen bonding (a) can be tuned by substituent groups, where EWG at 5-position is more effective than 6-position to shorten the bond length of a and hence facilitate reduction. Third, hydrogen bonding (b) stabilises the complexed CO2 when EWGs are introduced.
It is important to note that the effect of intramolecular hydrogen bonding on CO2 complexation has been briefly studied in prior works using quinones. However, it was observed that hydrogen bonding occupies the CO2 binding sites of quinones and diminishes the ability for CO2 capture14,30. Therefore, our work presents the first demonstration that intramolecular hydrogen bonding can facilitate CO2 adduct formation and break the intrinsic linear free-energy relationship of EMCC chemistries. This is attributed to the unique chemical structure of isoindigo that allows free rotation of the oxindole rings in the reduced state as supported by DFT simulation, breaking the intramolecular hydrogen bonding (a) to create space for CO2 complexation, which is further enhanced by the intramolecular hydrogen bonding (b) through the amide functionality.
Finetuning the properties of isoindigos
Breaking the correlation between chemical modification and CO2 binding affinity greatly enhances the degree of freedom in finetuning sorbent properties. For instance, by installing EWGs such as methyl carboxylate, E1/2 of 66DMCIId under CO2 can be positively shifted by 300 mV compared to that of unmodified IId to impart O2 stability, while the \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) only drops slightly (from 9.34 to 8.07). Besides, 66DMCIId is almost insoluble in organic solvents such as DMF (solubility < 2.5 mM), suggesting its potential as absorbent electrodes in fixed-bed EMCC devices.
Unmodified IId has a moderate solubility in DMF ( ~ 230 mM), which needs to be improved for practical use in flow-based EMCC systems7. To facilitate chemical functionalisation, we introduced a carboxyl group to the 6-position of isoindigos, which can be easily connected with amino acids through amidation reactions. As a proof of concept, we utilised glycine methyl ester and serine methyl ester as solubility enhancers and three nonsymmetric isoindigos were prepared (6AIIdGly, 6AIIdSer, and 6B6AIIdSer). 6B6AIIdSer features halogen substitution on one oxindole ring to tune redox potentials and amino acid ester functionalisation on the other oxindole ring to enhance solubility. To our delight, the solubilities of 6AIIdGly, 6AIIdSer, and 6B6AIIdSer increase to 606, 830, and 568 mM in DMF, respectively (Supplementary Table 6), likely due to the enhanced molecular interaction between the polar functional groups (amide and carboxylate) and DMF solvent. This is supported by the fact that 6AIIdSer is more soluble than 6AIIdGly due to the additional hydroxyl group from the serine moiety.
Conventional amine scrubbing sorbents have raised environmental concerns due to their biotoxicity31,32. Here, we show that introducing amino ester functionalities into isoindigos substantially improves their biocompatibility with mammalian cells. Unmodified IId shows a LC50 (lethal concentration that causes 50% cell death) of 11.2, 50.4, and 15.8 μg ml−1 for NIH3T3/GFP mouse fibroblasts, U2OS.EGFP human osteosarcoma cells, and MCF10A human breast epithelial cells, respectively, after 48 h cell culture (Supplementary Fig. 24 and 25). In comparison, the serinate-modified counterpart does not display clear toxicity under concentrations up to 100 μg ml−1 for NIH3T3/GFP and U2OS.EGFP, and has a significantly improved LC50 of 89.4 μg ml−1 for MCF10A.
Effects of electrolytes, oxygen, and water
Before evaluating the CO2 capture performance of isoindigos in EMCC devices, we assessed the influence of electrolytes and common gas stream impurities such as water and O2 on their CO2 binding properties. Using 55DBIId as an example, the electrochemical behaviours remained almost unaffected up to a high O2 content (16% CO2 and 20% O2, Supplementary Fig. 26). Under N2, the CV curves of 55DBIId remain reversible even at a high water content of 10 vol% (Supplementary Fig. 27). The declining peak current is caused by the decreasing isoindigo solubility with increasing water content. However, the expected CO2 release at approximately −1 V vs. Fc+/Fc gets suppressed in the presence of 10 vol% water, possibly due to the involvement of pH-swing process under high water content that requires higher energy input for CO2 release.
We also studied the influence of supporting salt on the redox and CO2 binding behaviours of isoindigos (Supplementary Fig. 28). The most prominent effect comes from the choice of cation, where reducing the size of cation leads to anodically shifted reduction potential, as is explained by the electrostatic interaction between cation and reduced isoindigo. Smaller alkaline cations exhibit stronger Lewis acidity, allowing tighter binding with reduced isoindigo to facilitate electro-reduction. Therefore, a more acidic supporting salt cation can further enhance the robustness of isoindigo against O2. Nevertheless, it may also slow down the CO2 complexation kinetics due to the competition between cation and CO2 for binding with reduced isoindigo.
Evaluating the isoindigo sorbents in flow-based EMCC prototypes
Based on the CV peak potentials for CO2 capture and release, we estimated the theoretical minimum energy requirement for CO2 separation using isoindigo sorbents, which ranges from 9.8 to 27.6 kJ mol−1 CO2 (Supplementary Fig. 29). It is noteworthy that these molecules show very close onset potentials for CO2 capture and release such that the theoretical energetics calculated from CV peak potentials can be overestimated compared to previous calculations using onset values17. As a proof of concept, we evaluated their intrinsic capability for reversible CO2 capture and release in a flow-based EMCC prototype reported by us previously (Fig. 6a)17. Detailed testing conditions are provided in the Supplementary Information.
a Schematic of the flow-based EMCC prototype. b CO2 reading at the exit of the sorbent tank over 19 repeating capture/release cycles for ~100 h of operation. c, The CO2 reading of selected capture/release cycles overlaid, with the cumulative amount of CO2 captured/released in each cycle relative to the theoretical capacity. Lighter colours represent later cycles. The shaded regions indicate the capture, rest, release, and rest steps. For CO2 capture, 6MCIId was reduced at 10 mA for 60 min followed by a 75 min rest. For CO2 release, the adducts were oxidised at 10 mA to 0.05 mV followed by a ~ 120 min constant voltage hold, and finally rested for another 75 min. d Selected voltage-capacity curves for the 1st, 5th, 10th, and 15th capture/release cycle. e The CO2 capacity utilisation efficiency (blue squares), release/capture efficiency (red crosses), and electrochemical efficiency (empty grey squares) of the system. The liquid sorbent was composed of 10 ml 0.1 M 6MCIId in DMF with 0.25 M NaClO4 as the supporting salt. The sorbent tank was filled with plastic beads and purged with 10% CO2 at a flow rate of 2 standard cubic centimetres per minute (sccm). On the opposite side, a Fc tank was used to balance the charge, which was filled with 20 ml 0.1 M Fc in DMF with 0.25 M NaClO4 as the supporting salt, 4 mM ferrocenium tetrafluoroboronate (FcBF4) to facilitate Fc oxidation, and 10 mM 6MCIId to mitigate sorbent crossover.
Figure 6b shows the cyclic capture-release performance of 6MCIId using 10% CO2 (balanced by N2) as the gas feed. The CO2 reading curves of each cycle are overlaid in Fig. 6c, where the cumulative CO2 captured/released is obtained by integrating these curves. For each cycle, 6MCIId was reduced at 10 mA for 60 min, and the decrease in CO2 concentration at the gas outlet confirmed carbon capture. The current was then stopped for 75 min, allowing the CO2 reading to gradually return to the baseline as the reduced isoindigo fully reacted with CO2. The CO2 adduct was subsequently oxidised following a constant current-constant voltage (CC-CV) protocol, and the continuous increase in CO2 concentration above 10% indicated CO2 desorption. Afterward, the current was set to zero again to ensure the complete release of the oversaturated CO2 from the sorbent electrolyte. The oxidation-reduction profiles of the flow system are shown in Fig. 6d. By integrating the voltage-capacity curves, the electrical energy consumption under 10% CO2 is estimated as 127.3 kJ mol−1 CO2 in the first cycle and 142.5 ± 8.2 kJ mol−1 CO2 over the first 16 cycles (Supplementary Fig. 30), which is comparable to other carbon capture technologies18,19,20,33,34,35,36,37,38.
Figure 6e summarises the three key metrics commonly used for evaluating EMCC performance. CO2 capacity utilisation, defined as the amount of CO2 captured relative to the theoretical value (one CO2 per electron), shows an average of 65% over 19 cycles, which is competitive against the state-of-the-art quinone-based sorbent reported recently19. The release/capture efficiency, defined as the ratio of the total amount of CO2 released and captured in each cycle, is averaged to be 97%, indicating the good reversibility of the sorbent. Considering a relatively constant electrochemical (Coulombic) efficiency of the EMCC prototype at ~84%, the major loss should be attributed to two factors: (1) our CC-CV protocol where the CO2 adduct was not fully oxidised; (2) the crossover of sorbents and counter electrolytes caused by membrane swelling, which limits all current nonaqueous redox-flow electrochemical systems. 6MCIId was also evaluated at a higher percentage of CO2 removal (Supplementary Fig. 31).
In addition to 6MCIId, we evaluated the performance of other isoindigo sorbents such as 55DBIId (Supplementary Fig. 32), 66DBIId (Supplementary Fig. 33), and 6B6AIIdSer (Supplementary Fig. 34). 55DBIId achieved an average CO2 utilisation efficiency of up to ~80% and an average release/capture efficiency of ~80%. Using 66DBIId, we studied 1H NMR of the crude sorbent electrolyte after 11 capture/release cycles over 50+ hours (Supplementary Fig. 35). The spectrum suggests the high stability of 66DBIId after cycling and also the severe crossover issue of the ferrocene counter electrolyte (three times the concentration of 66DBIId in the sorbent tank), which explains the decay in electrochemical capacity of current EMCC prototypes. Besides, we can recover 66DBIId with 87% yield from the sorbent electrolyte after cycling, corroborating the robustness of the sorbent. In addition, we found ~24% of 66DBIId isomerised into cis−66DBIId, supporting our hypothesis on the rotational isomerisation of reduced 66DBIId discussed earlier (Supplementary Fig. 10). Nevertheless, the reduction of both 66DBIId and its cis-isomer yield the same activated CO2 sorbent, which we believe does not affect the long-term stability of the EMCC prototype.
To our delight, 6B6AIIdSer exhibits a much lower oxidation potential for CO2 release, likely due to its higher solubility. Moreover, 6B6AIIdSer shows excellent cycling stability with negligible voltage decay over >40 cycles and ~200 h of operation with a capacity degradation rate of 2% (Supplementary Note 5).
We further tested the EMCC performance of 6MCIId using simulated flue gas (10% CO2 + 3% O2 balanced in N2) (Supplementary Fig. 36). The cell can run stably over ~90 h with reduction voltage maintained above −1.3 V, which minimised the parasitic oxygen reduction reaction. A CO2 capacity utilisation efficiency of ~50% was achieved with a near unity CO2 release/capture efficiency.
Finally, we evaluated the CO2 capture capability of 6MCIId under low CO2 concentration and its CO2 release capability under pure CO2. Using 1% CO2 with 0.3% O2 as the feed, we observe an early-stage energy consumption of 224.2 kJ mol−1 CO2 and a single pass CO2 removal of >90% (Supplementary Fig. 37). This suggests that our intramolecular hydrogen bonding strategy is effective in improving CO2 affinity for low-concentration CO2 capture. In another experiment, the CO2 capture-release behaviour under 100% CO2 headspace was quantified using mass flow metre (Supplementary Fig. 38). Under conditions similar to low-concentration CO2 capture, we show an early-stage energy consumption of 143.7 kJ mol−1 CO2 captured and 13.6 kJ mol−1 CO2 released, respectively.
In this study, we focus on exploring the fundamental chemistry of isoindigos as redox-active CO2 carriers and their potential to overcome the linear free-energy relationship that limits the structural modification of EMCC sorbents. The flow-based prototype in this work is, however, not an ultimate design for practical systems but a proof-of-concept demonstration to evaluate the performance of isoindigo at the lab scale. We believe future efforts can substantially improve the performance by optimising the electrolytes, electrodes, and membranes of EMCC devices.
Comparison of methods for estimating CO2 binding constants
As a final note, in this manuscript, we estimated the \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) of isoindigos using the prevalent method adopted for quinones, bipyridines, and benzyl thiolate12,21,22, providing an equitable comparison with previously reported redox-active CO2 carriers. Accordingly, we assume that the two-electron-reduced isoindigo binds to one molecule of CO2 and calculate the \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) based on ∆Epeak(2) under pure N2 and CO2 atmosphere, respectively (Supplementary Note 3). This method, therefore, eliminates the kinetic effects in CV under lower CO2 concentrations. Alternatively, we recorded the CV of IId and 6MCIId under various CO2 concentrations (Supplementary Fig. 39) and fitted the relationship between ∆Epeak(2) and CO2 concentration (Supplementary Fig. 40 and Supplementary Note 7). Similar \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) values were obtained for IId and 6MCIId on the order of 1012, further confirming the high tolerance of EWG in isoindigo structural motifs for strong CO2 binding. However, the fitting quality was unsatisfactory, with low R2 values and unreasonable number of binding sites. Therefore, we took the former method to calculate \({K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) for all isoindigos reported in this work.
Discussion
In summary, we demonstrate the rational design of a class of bifunctional redox-tunable CO2 carriers based on isoindigo and their derivatives. The unique intramolecular hydrogen bonding in isoindigo moieties enables a wide range of chemical modifications to facilitate electro-reduction, tune solubility, and preclude parasitic reactions without compromising their CO2 binding ability. With coupled experimental and computational studies, we provide an in-depth analysis of the structure-function relationships of isoindigos as EMCC sorbents. Compared to existing EMCC sorbents, isoindigo compounds have the following advantages: 1) a nearly constant \(\log {K}_{{{{{{{\rm{CO}}}}}}}_{2}}\) of ~9 with high tolerance to chemical modifications; 2) facile synthesis with the ease of encoding functionalities; 3) highly tunable redox potentials and solubilities; 4) improvable biocompatibility. In addition to flow-based EMCC, we envisage that isoindigos can also find applications as solid adsorbents in fixed-bed systems, due to their descent charge mobility and the abundant methods in synthesising and processing isoindigo-based polymers developed by the community of organic semiconductors. The work paves the way for engineering more reliable EMCC systems by breaking the fundamental barriers of the scaling relationship between redox potential and CO2 binding strength when designing redox-tunable CO2 sorbents.
Methods
Synthesis of 6MCIId
To a mixture of isatin (1.47 g, 10 mmol) and methyl 2-oxindole-6-carboxylate (1.91 g, 10 mmol) in acetic acid (50 ml) was added 37% HCl solution (0.5 ml). The mixture was heated at reflux for 1 day under Argon atmosphere. The mixture was cooled to room temperature, filtered, and washed with water, ethanol, and ethyl acetate. The solid was dried in the vacuum oven at 60 °C for 15 h to afford a dark red powder (2.93 g, 92%). 1H NMR (400 MHz, DMSO) δ 11.08 (s, 1H), 10.94 (s, 1H), 9.15 (d, J = 8.4 Hz, 1H), 9.07 (d, J = 8.0 Hz, 1H), 7.57 (dd, J = 8.4, 1.7 Hz, 1H), 7.38 (td, J = 7.7, 1.2 Hz, 1H), 7.34 (d, J = 1.6 Hz, 1H), 7.02 – 6.94 (m, 1H), 6.85 (d, J = 7.3 Hz, 1H), 3.87 (s, 3H). 13C NMR (101 MHz, DMSO) δ 168.68, 168.62, 165.57, 144.70, 143.91, 135.73, 133.47, 131.95, 131.79, 129.83, 129.06, 125.68, 121.98, 121.51, 121.28, 109.69, 109.29, 52.29.
Electrochemical measurements
Electrochemical measurements were performed with a BioLogic VSP potentiostat from BioLogic Science Instruments. Cyclic voltammetry (CV) utilised a glassy carbon electrode (3 mm diameter) as the working electrode, a platinum wire as the counter electrode, and a silver wire as the quasi-reference electrode, with ferrocene as the internal reference. In a standard CV test, isoindigo (2.5 mM) was dissolved in anhydrous DMF with 100 mM NBu4PF6 as the supporting electrolyte. CV were typically recorded at a scanning rate of −50 mV s–1 with a cut-off potential from −1.5 to 0.5 V (prior to ferrocene calibration). To examine the effects of ionic species on the redox behaviour of the sorbent molecules, supporting electrolyte salts including 100 mM LiClO4, NaClO4, KClO4, NBu4ClO4, sodium triflate (NaOTf), or sodium bis(trifluoromethanesulfonyl)imide (NaTFSI) was employed, respectively. The electrochemical data were gathered and analysed by EC-lab V11.50.
Flow-based EMCC prototype
In a standard setup, a scintillation vial (20 ml) with a septum cap, serving as the sorbent tank, was continuously purged with CO2 feed gas (balanced with N2) at a controlled flow rate using an Alicat mass flow controller. Simulated flue gas conditions were mimicked using a 10% CO2 and 3% O2 mix, balanced by N2. CO2 levels were continuously monitored at the gas outlet using an infra-red-based CO2 sensor (SprintIR-W 100%), with data recorded via Labview 2021. To minimise mixing time in the overhead space, the tank was filled with plastic beads from McMaster-Carr. The Fc tank was maintained without air exposure. Sorbent and Fc electrolytes were circulated at a flow rate of 10 ml min–1 through a commercial flow cell (Scribner) by a two-channel peristaltic pump (Masterflex). The flow cell incorporated two graphite plates with 5 cm2 interdigitated flow fields, pressing against two pieces of carbon paper electrodes (Sigracet 28 AA) on each side of the graphite electrodes. A Nafion 115 membrane, flanked by polypropylene sheets, was placed between the carbon electrodes. The cell was sealed by Kalrez fluoropolymer elastomer gaskets (0.02-inch thick). CO2 capture was conducted in constant current mode, while release followed a constant current/constant voltage protocol: the adduct was first oxidised at a constant current until reaching a cut-off voltage and then held at that voltage until the current value fell below 5% of the original constant current). For experiments using 5 cm2 flow fields, the CO2 capture-release was cycled at a current of 10 mA with a cut-off potential of 0.05 V in the release process. For experiments using 25 cm2 flow fields, the CO2 capture-release was conducted at a current of 50 mA with a cut-off potential of 0.3 V in the release process. The cycling protocols of each experiment are provided in the corresponding figure captions. For experiments using 6MCIId sorbent, we added 10 mM 6MCIId and 4 mM ferrocenium tetrafluoroboronate (FcBF4) into the CE tank to mitigate sorbent crossover and facilitate the reduction of the oxidised ferrocenium in the counter electrolyte, respectively. For less soluble 55DBIId and 66DBIId, the sorbent was dispersed in dimethylacetamide (DMAc) to form a slurry catholyte.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data generated or analysed during this study are included in the manuscript and its Supplementary Information. The main data generated in this study are provided in the Supplementary Information/Source Data file. Data are also available from the corresponding author upon request. Source data are provided with this paper.
References
Mac Dowell, N., Fennell, P. S., Shah, N. & Maitland, G. C. The role of CO2 capture and utilization in mitigating climate change. Nat. Clim. Change 7, 243–249 (2017).
Deutz, S. & Bardow, A. Life-cycle assessment of an industrial direct air capture process based on temperature–vacuum swing adsorption. Nat. Energy 6, 203–213 (2021).
Bui, M. et al. Carbon capture and storage (CCS): the way forward. Energy Environ. Sci. 11, 1062–1176 (2018).
Chu, S. Carbon capture and sequestration. Science 325, 1599–1599 (2009).
Sanz-Pérez, E. S., Murdock, C. R., Didas, S. A. & Jones, C. W. Direct capture of CO2 from ambient air. Chem. Rev. 116, 11840–11876 (2016).
Sharifian, R., Wagterveld, R. M., Digdaya, I. A., Xiang, C. & Vermaas, D. A. Electrochemical carbon dioxide capture to close the carbon cycle. Energy Environ. Sci. 14, 781–814 (2021).
Liu, Y., Lucas, É., Sullivan, I., Li, X. & Xiang, C. Challenges and opportunities in continuous flow processes for electrochemically mediated carbon capture. iScience 25, 105153 (2022).
Rheinhardt, J. H., Singh, P., Tarakeshwar, P. & Buttry, D. A. Electrochemical capture and release of carbon dioxide. ACS Energy Lett. 2, 454–461 (2017).
Renfrew, S. E., Starr, D. E. & Strasser, P. Electrochemical approaches toward CO2 capture and concentration. ACS Catal. 10, 13058–13074 (2020).
Barlow, J. M. et al. Molecular design of redox carriers for electrochemical CO2 capture and concentration. Chem. Soc. Rev. 51, 8415–8433 (2022).
Diederichsen, K. M. et al. Electrochemical methods for carbon dioxide separations. Nat. Rev. Methods Prim. 2, 68 (2022).
Bell, W. L., Miedaner, A., Smart, J. C., DuBois, D. L. & Verostko, C. E. Synthesis and evaluation of electroactive CO2 carriers. SAE Trans. 97, 544–552 (1988).
Mizen, M. B. & Wrighton, M. S. Reductive addition of CO2 to 9,10‐phenanthrenequinone. J. Electrochem. Soc. 136, 941–946 (1989).
Nagaoka, T., Nishii, N., Fujii, K. & Ogura, K. Mechanisms of reductive addition of CO2 to quinones in acetonitrile. J. Electroanal. Chem. 322, 383–389 (1992).
Rahimi, M., Khurram, A., Hatton, T. A. & Gallant, B. Electrochemical carbon capture processes for mitigation of CO2 emissions. Chem. Soc. Rev. 51, 8676–8695 (2022).
Scovazzo, P., Poshusta, J., DuBois, D., Koval, C. & Noble, R. Electrochemical separation and concentration of <1% carbon dioxide from nitrogen. J. Electrochem. Soc. 150, D91 (2003).
Gurkan, B., Simeon, F. & Hatton, T. A. Quinone reduction in ionic liquids for electrochemical CO2 separation. ACS Sustain. Chem. Eng. 3, 1394–1405 (2015).
Voskian, S. & Hatton, T. A. Faradaic electro-swing reactive adsorption for CO2 capture. Energy Environ. Sci. 12, 3530–3547 (2019).
Diederichsen, K. M., Liu, Y., Ozbek, N., Seo, H. & Hatton, T. A. Toward solvent-free continuous-flow electrochemically mediated carbon capture with high-concentration liquid quinone chemistry. Joule 6, 221–239 (2021).
Li, X., Zhao, X., Liu, Y., Hatton, T. A. & Liu, Y. Redox-tunable Lewis bases for electrochemical carbon dioxide capture. Nat. Energy 7, 1065–1075 (2022).
Simeon, F. et al. Electrochemical and molecular assessment of quinones as CO2-binding redox molecules for carbon capture. J. Phys. Chem. C. 126, 1389–1399 (2022).
Barlow, J. M. & Yang, J. Y. Oxygen-stable electrochemical CO2 capture and concentration with quinones using alcohol additives. J. Am. Chem. Soc. 144, 14161–14169 (2022).
Bui, A. T., Hartley, N. A., Thom, A. J. W. & Forse, A. C. Trade-off between redox potential and the strength of electrochemical CO2 capture in quinones. J. Phys. Chem. C. 126, 14163–14172 (2022).
Hansch, C., Leo, A. & Taft, R. W. A survey of hammett substituent constants and resonance and field parameters. Chem. Rev. 91, 165–195 (1991).
Wang, E., Mammo, W. & Andersson, M. R. 25th anniversary article: isoindigo-based polymers and small molecules for bulk heterojunction solar cells and field effect transistors. Adv. Mater. 26, 1801–1826 (2014).
Bogdanov, A. V. & Mironov, V. F. Recent advances in the application of isoindigo derivatives in materials chemistry. Beilstein J. Org. Chem. 17, 1533–1564 (2021).
Kiss, F. L., Corbet, B. P., Simeth, N. A., Feringa, B. L. & Crespi, S. Predicting the substituent effects in the optical and electrochemical properties of N,N′-substituted isoindigos. Photochem. Photobiol. Sci. 20, 927–938 (2021).
Wei, X., Zhang, W. & Yu, G. Semiconducting polymers based on isoindigo and its derivatives: synthetic tactics, structural modifications, and applications. Adv. Funct. Mater. 31, 2010979 (2021).
Xu, Y. et al. Assessing the kinetics of quinone–CO2 adduct formation for electrochemically mediated carbon capture. ACS Sustain. Chem. Eng. 11, 11333–11341 (2023).
Schimanofsky, C. et al. Direct electrochemical CO2 capture using substituted anthraquinones in homogeneous solutions: a joint experimental and theoretical study. J. Phys. Chem. C. 126, 14138–14154 (2022).
del Rio, B. et al. The biogenic amines putrescine and cadaverine show in vitro cytotoxicity at concentrations that can be found in foods. Sci. Rep. 9, 120 (2019).
Rohr, A. C. et al. Potential toxicological effects of amines used for carbon capture and storage and their degradation products. Energy Procedia 37, 759–768 (2013).
Eisaman, M. D. et al. CO2 separation using bipolar membrane electrodialysis. Energy Environ. Sci. 4, 1319–1328 (2011).
Wang, M. et al. Flue gas CO2 capture via electrochemically mediated amine regeneration: System design and performance. Appl. Energy 255, 113879 (2019).
Wang, M., Herzog, H. J. & Hatton, T. A. CO2 capture using electrochemically mediated amine regeneration. Ind. Eng. Chem. Res. 59, 7087–7096 (2020).
Jin, S., Wu, M., Gordon, R. G., Aziz, M. J. & Kwabi, D. G. pH swing cycle for CO2 capture electrochemically driven through proton-coupled electron transfer. Energy Environ. Sci. 13, 3706–3722 (2020).
Jin, S., Wu, M., Jing, Y., Gordon, R. G. & Aziz, M. J. Low energy carbon capture via electrochemically induced pH swing with electrochemical rebalancing. Nat. Commun. 13, 2140 (2022).
Liu, Y., Ye, H.-Z., Diederichsen, K. M., Van Voorhis, T. & Hatton, T. A. Electrochemically mediated carbon dioxide separation with quinone chemistry in salt-concentrated aqueous media. Nat. Commun. 11, 2278 (2020).
Acknowledgements
We acknowledge support from the Johns Hopkins University, the Scialog program sponsored jointly by Research Corporation for Science Advancement and the Alfred P. Sloan Foundation, with additional support from Climate Pathfinders Foundation (grant #28438 X.L.), and the David and Lucile Packard Foundation. X.Z. acknowledges FDCT of Macau SAR (grant#0024/2022/ITP).
Author information
Authors and Affiliations
Contributions
X.L. and Ya.L. conceived of the project and designed the experiments. X.L. conducted the experiments and analysed the data. X.Z. performed the DFT simulations under the supervision of Yu.L. L.Z. helped with the CV experiments and solubility test. A.M. helped analyse the carbon capture results. Y.X. performed the reaction rate tests and analysed the data. Z.F. conducted the biocompatibility tests under the supervision of L.G. Ya.L. supervised the whole project. X.L. and Ya.L. co-wrote the manuscript. All authors discussed the results and revised or commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, X., Zhao, X., Zhang, L. et al. Redox-tunable isoindigos for electrochemically mediated carbon capture. Nat Commun 15, 1175 (2024). https://doi.org/10.1038/s41467-024-45410-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-45410-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.