Abstract
Bacteria of the SAR202 clade, within the phylum Chloroflexota, are ubiquitously distributed in the ocean but have not yet been cultivated in the lab. It has been proposed that ancient expansions of catabolic enzyme paralogs broadened the spectrum of organic compounds that SAR202 bacteria could oxidize, leading to transformations of the Earth’s carbon cycle. Here, we report the successful cultivation of SAR202 bacteria from surface seawater using dilution-to-extinction culturing. The growth of these strains is very slow (0.18–0.24 day−1) and is inhibited by exposure to light. The genomes, of ca. 3.08 Mbp, encode archaella (archaeal motility structures) and multiple sets of enzyme paralogs, including 80 genes coding for enolase superfamily enzymes and 44 genes encoding NAD(P)-dependent dehydrogenases. We propose that these enzyme paralogs participate in multiple parallel pathways for non-phosphorylative catabolism of sugars and sugar acids. Indeed, we demonstrate that SAR202 strains can utilize several substrates that are metabolized through the predicted pathways, such as sugars ʟ-fucose and ʟ-rhamnose, as well as their lactone and acid forms.
Similar content being viewed by others
Introduction
The SAR202 clade in the phylum Chloroflexota is ubiquitously distributed in the ocean, accounting for 10–30% of planktonic prokaryotes in the deep sea1,2,3,4,5,6,7. Various properties associated with organoheterotrophy and sulfur and nitrogen metabolism have been interpreted from SAR202 metagenome assemblies and single-cell genome sequences6,8,9,10,11,12. The seven groups (subclades) of SAR202 are individually distinct in the numbers and types of paralogs they contain8,12, suggesting a relationship between paralog evolution and niche specialization of the subclades.
Paralogous flavin-dependent monooxygenase genes in group III SAR202, in some cases exceeding 100 per genome, are proposed to have evolved to harvest carbon and energy from diverse organic molecules that accumulated in the oceans during the expansion of the Earth’s carbon cycle, following the rise of oxygenic phototrophy8,12,13. Similarly, in group I SAR202, large expansions of paralogs in the enolase protein superfamily are proposed to have evolved to enable these cells to metabolize compounds that resist biological oxidation because of their chiral complexity. The early branching of these paralogs in phylogenetic trees indicates that SAR202 cell evolution was a crucible for their diversification8,12,13.
SAR202 cells are found throughout ocean water columns, reaching highest numbers near the ocean surface, but they contribute a higher percentage of all plankton cells in the meso-, bathy-, hadal-, and abyssopelagic5,10,12. Group I and II SAR202 have rhodopsin genes in their genomes and are the most abundant SAR202 in epipelagic environments, whereas group III, lacking rhodopsin genes, is largely responsible for the high relative abundance of SAR202 in the dark ocean.
The cultivation of unrepresented cell types is a priority for microbiologists because cells frequently exhibit properties that cannot be easily predicted from their genomes14. A recent study has shown that genome-based inference fails to reliably predict catabolic pathways for more than 50% of carbon sources utilized by diverse prokaryotes that have well-curated phenotypic data15. Despite the recent surge in interest and technological advances in cultivation, a high proportion of prokaryotic groups remain uncultured16. The SAR202 clade has no cultured isolates yet, making it one of the “most wanted” in culture and a key “target for cultivation”14,17.
Here we report the successful cultivation of SAR202 bacteria. Twenty-four isolates of subclade I were retrieved from surface seawater samples by dilution-to-extinction in sterile seawater media. Metabolic reconstruction supported by experimental data with cells implicated enolase and dehydrogenase paralogs in non-phosphorylative sugar oxidation. We propose that multiple parallel metabolic pathways of this type enable these cells to harvest complex mixtures of sugar-related compounds from dissolved organic carbon pools. SAR202, which are found throughout the water column of modern oceans, evolved concurrently with the rise of oxygenic phototrophy13. We propose they expanded into the niche of harvesting dilute and diverse carbohydrate-related molecules as the oceans are oxidized.
Results and discussion
The successful cultivation of the SAR202 clade
Dilution-to-extinction experiments with low-nutrient heterotrophic media (LNHM; Supplementary Table 1) in microtiter dishes retrieved twenty-four SAR202 group I isolates from surface samples (depth, 10 m) from two stations (GR1 and GR3) located nearby Garorim Bay of the Yellow Sea (Supplementary Fig. 1a). Four conditions, differing by catalase addition and light exposure (continuous dark vs. 14:10 h light-dark cycle) yielded 610 strains, of which 24 belonged to the SAR202 clade (Supplementary Table 2). All 24 SAR202 strains were obtained from cultures incubated continuously in dark (Supplementary Table 2).
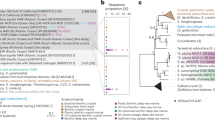
Phylogenetic comparisons showed that the isolates had nearly identical 16S rRNA gene sequences, were affiliated with the SAR202 group I (Fig. 1a)3,8,12, and corresponded to major (greater than 68%) amplicon sequence variant (ASV) of the SAR202 clade (0.7–1.0% in total prokaryotes) in the water samples (Supplementary Fig. 2).
a Phylogenetic tree based on 16S rRNA gene sequences showing the relationship among the 24 SAR202 strains isolated in this study and closely related sequences retrieved from various databases, including GTDB (marked in purple), IMG (yellow), GenBank, and EzBioCloud. The four SAR202 strains selected for genome sequencing are marked in red. RAxML (v8.2.12) was used for tree building with GTRGAMMA model. Bootstrap supporting values (≥70%; from 100 resamplings) are indicated. Dehalococcoides mccarty was set as an outgroup. Bar, 0.1 substitutions per nucleotide position. b Phylogenomic tree of the SAR202 clade. The four genomes of SAR202 strains from this study (marked in red) and other MAGs and SAGs from previous studies were classified using GTDB-Tk. Species cluster-representative genomes of the GTDB taxa, into which the analyzed SAR202 genomes were classified, were included for tree building. Designation of the SAR202 subgroups following the classification scheme of a previous study12 is indicated on the left side of the tree with color shadings. On the right side of the tree, taxonomic assignment of the genomes at the family and order levels according to the GTDB (release 202) is shown. The tree building was performed using RAxML (v8.2.12), with PROTGAMMAAUTO option, based on a concatenated alignment of core genes obtained by UBCG pipeline. Bootstrap supporting values (100 iterations) are indicated on the nodes. Bar, 0.1 substitution per amino acid position.
The growth of SAR202 isolates was very slow and inhibited by light
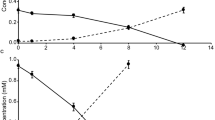
Four strains selected for further experiments, JH545, JH702, JH639, and JH1073, behaved similarly, growing slowly (0.18–0.24 day−1) and displaying sensitivity to light (Fig. 2a–c). Strain JH545 grew optimally at 15–20 °C (Supplementary Fig. 3), and therefore all subsequent experiments were performed at 20 °C. Approximately 50 days were required to reach stationary phase at maximum cell densities of ~2 × 108 cells mL−1 (Fig. 2a). Very little growth followed by gradual decline at 4 °C (Supplementary Fig. 3) suggests that strain JH545 belonging to the SAR202 group I may not be well-adapted to deep sea, where other members of the SAR202 clade (e.g., group III) are prevalent12.
a Initial growth curves of four representative SAR202 strains in low-nutrient heterotrophic medium (LNHM) under dark condition. b Growth curves of the four strains under continuous dark and light-dark conditions in LNHM. Light-dark cycle (14:10 h) was applied using LED lamps with a light intensity of ~155 μmol photons m–2 s–1. For the dark condition, tubes were wrapped in aluminum foil. c Growth curves of strain JH545 under dark condition and various light intensities in LNHM5x. All cultures were initially incubated in dark (i.e., the tubes were wrapped in aluminum foil). At the day indicated by a black arrow, some cultures were shifted to light condition (i.e., the aluminum foil was removed) and exposed to light with the following intensities: WL, ~45; ML, ~89; SL, ~134 μmol photons m–2 s–1. The experiment in c was performed in triplicates. Data are presented as mean values ± SEM. Note that the error bars are hidden when they are shorter than the size of the symbols. a–c Source data are provided as a Source data file. d–f Electron micrographs of strain JH545 cells observed by TEM (d), thin-section TEM (e), and SEM (f). Scale bars, 100 nm.
The growth of all four strains was inhibited by light-dark cycles with broad spectrum LED lights (Fig. 2b), and experiments with strain JH545 showed that continuous exposure to light caused growth inhibition followed by cell death at all light intensities tested (~45–134 μmol photons m−2 s−1), with variations in response levels (Fig. 2c).
Although the isolates were pure cultures, TEM and SEM microscopy showed short rods (~0.8 × 0.4 μm), cocci (diameter, ~0.5 μm), discs, and discs with biconcave centers that sometimes appeared to be toroidal, as reported for the strains of Dehalococcoides18,19, a genus belonging to the same class (Dehalococcoidia) as SAR202 (Fig. 2d–f and Supplementary Fig. 4). Thin-section TEM images indicated monoderm cell envelopes, similar to other Chloroflexota (Fig. 2e)20,21.
Genomic features
General genome features and phylogenomics
The genomes of the four SAR202 strains were 3083–3094 kb in length with 51.8% GC content, ~87.5% coding density, and at least 99.9% average nucleotide identity (ANI) among the strains. The two genomes sequenced on PacBio platform (JH545 and JH1073) were assembled into one circularly closed contig, whereas the other two genomes sequenced with Illumina technology were composed of more than 30 contigs (~846 kb of N50 for both genomes) (Supplementary Fig. 5c). The circular map of the JH545 genome showed several regions with anomalous GC content and GC skew (Supplementary Fig. 5a), many of which overlapped with the predicted genomic islands (Supplementary Fig. 5b).
Genome-inferred metabolic features were nearly identical among the four genomes (e.g., COG profiles; Supplementary Table 3), leading us to focus on one of them, JH545, for further analysis (Fig. 3). In accord with previous studies, the genome annotation indicated central carbon and energy metabolism typical of aerobic organoheterotrophs, with some genes indicating capacities for lithotrophy by sulfide oxidation (sulfide:quinone oxidoreductase) and anaerobic respiration by nitrate reduction (NapAB) and N2O reduction (NosZ) (Supplementary Notes). The presence of NapAB and NosZ has been reported in the SAR202 MAGs obtained from the northern Gulf of Mexico “dead zone”, where the expression of these genes was detected in the samples with lowest dissolved oxygen9. In agreement with previous genome analyses of Dehalococcoidia members19,22,23, peptidoglycan biosynthesis was not encoded in the SAR202 genomes. The TEM images showed a layer outside of the cell membrane of JH545 (Fig. 2d, e), reminiscent of the S-layer observed in peptidoglycan-lacking Dehalococcoidia strains24,25.
Some key metabolic pathways are indicated by blue boxes. Detailed information on the enzymes corresponding to the numbers next to arrows is available in Supplementary Data 1. The colors of the transporters at the bottom indicate functional categories: pink, ABC transporters; blue, others. Figure created with BioRender.com.
Whole genome phylogenies of the Genome Taxonomy Database (GTDB) indicate that the SAR202 group I cells we cultured represent isolates of a hitherto-uncultured order (UBA1151) within a monophyletic superorder comprised of all SAR202 (Fig. 1b and Supplementary Notes). We propose the provisional taxonomic name “Candidatus Lucifugimonas marina”, which includes strains JH639, JH702, and JH1073, in addition to strain JH545 as the type strain. To accommodate this novel genus and species, we also propose the family “Candidatus Lucifugimonadaceae” fam. nov. and the new order “Candidatus Lucifugimonadales” ord. nov. within the class Dehalococcoidia (see Methods section).
Archaellum
A gene cluster for archaellum, an archaeal motility structure, was predicted in the four SAR202 genomes. This gene cluster is similar to conserved arrangements of archaellum genes observed in archaea26, 27 and includes six tandem copies of flaB (encoding archaellin), followed by the genes flaGFHIJ (Supplementary Fig. 6). Six additional copies of flaB genes were scattered throughout the genome. A candidate gene for FlaK, a family of prepilin peptidases that remove signal peptides from archaellin26, was also predicted based on the assignment to COG1989 and the presence of domain PF01478 (Peptidase_A24; Type IV leader peptidase family)28.
Archaella are related to type IV pili and have no evolutionary relationship to bacterial flagella. Although a Chloroflexota MAG from aquifer sediment was previously reported to harbor a gene cluster for archaellum29, bacterial isolates have never been reported to harbor archaella. Searches for FlaB homologs in the IMG database revealed that two SAR202 MAGs from the Gulf of Mexico had archaellum gene clusters very similar to that of JH545 (Supplementary Fig. 6)9. Exploration of FlaB (K07325) distribution using the AnnoTree database showed that most (45 of 52) bacterial genomes harboring flaB were affiliated to Chloroflexota. The remaining seven bacterial genomes having flaB were distributed among seven phyla, indicating that this gene is very rare among other bacteria. Of the 45 flaB-harboring Chloroflexota genomes, 40 were among the 212 Dehalococcoidia genomes in the database, and the remaining 5 were in Anaerolineae. This distribution suggests that archaella genes might have been transferred from Archaea to an ancestor of Chloroflexota and retained in the class Dehalococcoidia. A phylogenetic analysis of FlaB sequences found in strain JH545, several Chloroflexota genomes, and representative archaeal isolates, confirmed this inference. The FlaB sequences of Chloroflexota formed a monophyletic clade, which was located as a sister clade of an archaeal FlaB group (Supplementary Fig. 7). Investigation on the 45 Chloroflexota genomes having FlaB (found in AnnoTree) and BlastP searches at NCBI (FlaB of strain JH545 as a query) revealed that at least two Chloroflexota strains harboring archaella gene cluster (flaB and several other genes) have been cultivated: Litorilinea aerophila30,31 and Aggregatilinea lenta32 (Supplementary Fig. 6). No archaella were observed during microscopic examination of the SAR202 cells.
Heliorhodopsin
Two copies of heliorhodopsin (HeR) genes were detected in the JH545 genome. In a recent study of marine SAR202 MAG/SAGs, rhodopsin genes were found in 28 group I and II genomes, all retrieved from water depths of less than 150 m. An HeR gene was reported in a single group II genome12. Therefore, this finding represents the presence of HeR in SAR202 group I. The two HeR copies found in the JH545 genome exhibited ~83% amino acid identity and were located one gene downstream of DNA photolyase, which repairs DNA damage caused by UV exposure using visible light (Supplementary Fig. 8a). Multiple sequence alignment showed that the two copies differed in the amino acid residue that caused a spectral shift in an Ala scanning mutagenesis study33 (W163 in 48C12; Supplementary Fig. 8b), suggesting that the two HeR copies might have different absorption spectra. Although the function of HeR remains unresolved, it has recently been suggested that it might function as a light sensor that regulates responses to light-induced oxidative stress34,35. Given that no HeR has been found in SAR202 group III, which is abundant in the dark ocean12, the possession of HeR by JH545 isolated from coastal surface water may indicate adaptation to the euphotic habitat.
Transporters and sulfatases
In accord with a previous study9, a large number of major facilitator superfamily (MFS) transporters were found in the JH545 genome, including 42 proteins assigned to COG0477 (Supplementary Table 3). MFS is a very large family of membrane transporters that are known to transport a variety of compounds, including mono- and oligosaccharides, amino acids, and nucleosides36,37. It is noteworthy that some substrates that we report below enhance the growth of JH545 (see the next section on COG4948) are known to be transported by MFS proteins38,39,40.
Eighteen proteins in the JH545 genome were assigned to COG3119, annotated as arylsulfatase A or a related enzyme (Supplementary Table 3). Sulfatase paralogs have been reported previously in SAR202 groups I and II12. Arylsulfatases catalyze the desulfation of sulfated carbohydrates in some catabolic pathways, for example the degradation pathway of ulvan by a marine bacteria41.
Genomes of SAR202 group I have the highest proportion of COG4948 paralogs among all prokaryotes, and these paralogs are highly divergent
The genomes of cultivated SAR202 we report encoded 80 COG4948 proteins in the mandelate racemase family within the enolase superfamily (~2.8% of CDS; Supplementary Table 3). Many marine bacteria have minimal genomes with few paralogs, so the unusually large sets of paralogs present in diverse SAR202 groups, such as COG4948 and COG2141 in the groups I and III, respectively, attracted attention when they were first discovered12.
We sought to establish whether the expansion of COG4948 in SAR202 group I is unusual in the context of prokaryotic diversity. We analyzed the genomes in GTDB, one of the most phylogenetically comprehensive genome databases. Because COG annotation is not scalable, we counted the numbers of the two Pfam domains (PF02746 and PF13378) corresponding to COG4948 (see Materials and Methods for details) and calculated the proportion of COG4948 proteins among all CDSs in each genome. Among the 47,894 species cluster-representative genomes of GTDB (R202), all 48 SAR202 group I (o__UBA1151 in GTDB) genomes ranked in the top 66 except one that ranked the 134th, in proportions of COG4948 (Fig. 4a), demonstrating that the paralog expansion of COG4948 is a prominent feature of SAR202 group I across all prokaryotes.
a Proportion of COG4948 proteins among the species cluster-representative genomes of GTDB (release 202; 47,894 genomes). Only the top 200 genomes with the highest proportion are included. Genomes of o_UBA1151 (SAR202 group I) are indicated with a distinct color. All 48 o__UBA1151 genomes are included in this figure owing to the high proportion of COG4948. The black arrow on the y-axis indicates the proportion of the four SAR202 genomes sequenced in this study (~2.8%). Source data are provided as a Source data file. b SSN (sequence similarity network) analysis of diverse COG4948 proteins predicted in the JH545 genome. UniProt proteins with the two Pfam domains, PF02746 and PF13378, were included in the analysis together with the 80 COG4948 proteins of strain JH545. Only the protein clusters including the JH454 proteins were retained in this figure after applying a threshold cutoff value to the alignment score (≥150). Nodes (proteins) are colored by their phylum-level taxonomy, following the color codes at the bottom. The 80 COG4948 proteins of the JH545 genome are indicated in red triangles. The four proteins that have been studied experimentally are indicated with black rectangles (UniProt ID: D8ADB5, C9A1P5, C6CBG9, and A8RQK7).
We analyzed sequence divergence among the 80 COG4948 proteins of the JH545 genome by building a sequence similarity network (SSN) using the Enzyme Function Initiative’s Enzyme Similarity Tool (EFI-EST)42. Most of the ~70 clusters that contained JH545 proteins as their members included only one JH545 gene, indicating that the JH545 COG4948 proteins are highly divergent (Fig. 4b). While many of the largest COG4948 clusters included diverse bacterial phyla, many small clusters were comprised mainly of Chloroflexota, suggesting that the COG4948 protein family, which is distributed widely across prokaryotes, diversified in Chloroflexota. Only the largest cluster, which included two JH545 proteins, contained biochemically studied proteins43 (Fig. 4b).
Abundant COG paralogs of SAR202 participate in the degradation of sugars, lactones, and sugar acids
We propose that seven sets of paralogs (COG1028, COG0667, COG3618, COG4948, COG1063, COG3836, and COG0329), including the most abundant COG4948 paralogs, act concertedly in parallel pathways that harvest energy from diverse carbohydrates (Fig. 5b), and we provide experimental evidence that cultured SAR202 group I cells respond to predicted substrates of these pathways (Fig. 5a). The largest COG sets in the SAR202 genomes of this study, in addition to the mandelate racemases (COG4948), included NAD(P)-dependent dehydrogenases (COG1028), aldolases (COG3836), and oxidoreductases (COG0667; Supplementary Table 3). These enzymes are found in non-phosphorylative pathways of various sugars and their acid catabolism (e.g., fucose, rhamnose, arabinose, and xylose)39, 44,45,46,47,48,49. Central to these pathways is the pairing enzymes from the two largest paralog sets, mandelate racemase-like enzymes and NAD(P)-dependent dehydrogenases, which catalyze a dehydration reaction followed by an oxidation reaction resulting in a flow of electrons for respiration. A previous SAR202 pangenome analysis of variation in COG copy number found significant positive correlations in the numbers of copies of the same seven COGs (Supplementary Table 3) in SAR202 group I genomes relative to other SAR202 genomes12. The phylogenetically correlated distributions of these paralog expansions support our prediction that they play coordinated metabolic roles.
a Growth of strain JH545 on various carbon compounds that were predicted to require COG4948 enzymes for metabolization. Carbon compounds were added to the ASW5x media (Supplementary Table 1) at a final concentration of 250 μM. Carbon mixture-only, no additional carbon compounds were added, except for carbon mixture already included in the ASW5x media. The incubation was performed in dark at 20 °C. All experiments were performed in triplicates. Data are presented as mean values ± SEM. Note that the error bars are hidden when they are shorter than the size of the symbols. Source data are provided as a Source data file. b Putative non-phosphorylative catabolic pathways of l-fucose and l-rhamnose inferred from the SAR202 genome data. The number of proteins assigned to the COGs is indicated within parentheses next to the COG IDs at each step.
We reconstructed non-phosphorylative pathways for the oxidation of two sugars relevant to marine environments, fucose and rhamnose50, 51, in the genomes of the cultured SAR202 strains based on several previous studies46,47,52,53,54,55 (Fig. 5b). Although both rhamnose and fucose are known to be degraded via pathways involving phosphorylation in many organisms, these kinase-dependent pathways were not found in the genomes. This suggests that the reconstructed non-phosphorylative pathways shown in Fig. 5b might serve as the only catabolic routes for both sugars in these strains. In the pathway reconstruction, l-fucose and l-rhamnose share the same COG annotation at each step, yielding pyruvate and lactate or lactaldehyde as final products. Seven of the eight COGs represented in the reconstructed pathways for fucose and rhamnose catabolism were from the abundant paralog sets described above, ranging from 9 to 80 variants of each COG (Supplementary Table 3).
We tested the growth response of JH545 cells to external metabolites that were predicted to be substrates for the catabolic pathways proposed above. In these experiments we used a defined medium based on artificial seawater, to which we added the same cofactors and organic compounds that were used for the original isolation of the strains. We did not examine whether the tested substrates could serve as sole carbon sources because of the very low growth rate of the cells and because cells adapted to oligotrophic ecosystems often exhibit reduced metabolic flexibility in comparison to copiotrophic cells when challenged with simplified carbon mixtures. All substrates tested, including ʟ-fucose, ʟ-rhamnose, their lactone and acid forms, and ascorbate enhanced the growth of strain JH545 in artificial seawater media (Fig. 5a). With increases in growth rates, cell densities in late exponential phase (~35 days of incubation) were more than 10 times higher in substrate-amended cultures, although the cultures reached similar densities in stationary phase. Ascorbate was tested because an ascorbate degradation pathway requiring a COG4948 enzyme39,48 was nearly complete in the JH545 genome (Supplementary Fig. 9).
SAR202 isolates represent a cell type that is common in the euphotic zone but relatively more abundant in the dark ocean
The vertical distribution of species-level population represented by JH545 (hereafter, JH545 population) in marine metagenomes followed patterns previously observed for several SAR202 group I members12. In metagenomes from Tara Oceans and station ALOHA, the relative abundance of the JH545 population was higher in the mesopelagic zone (200–1000 m) compared to the euphotic zone (surface to 200 m) (Mann–Whitney U test, P = 1.78 × 10−10, one-sided; Fig. 6c). In metagenomes from several marine trenches, the relative abundance of the JH545 population generally increased with increasing depth above the hadopelagic zone (below 6000 m), where their relative abundance declined (Fig. 6a, b). Given the overall decline of cell numbers with increasing water depth56,57,58, the JH545 population is likely found throughout the ocean water column, reaching its highest concentration in the epipelagic zone but increasing in relative abundance in the dark ocean. This inference is also consistent with recent studies reporting higher SAR202 cell abundance in the euphotic zone compared with the aphotic zone in the Atlantic Ocean and Fram Strait as measured by FISH-based cell counting12,59. The likely high absolute abundances of the JH545 population observed in the euphotic zones seemingly conflict with the observations of growth inhibition by light-dark cycles and death in continuous light of the cultured strains upon exposure to broad spectrum white light (Fig. 2b, c). To reconcile these observations, we hypothesize that JH545 uses its two copies of heliorhodopsin to regulate functions that are negatively impacted by light, mitigating its susceptibility to inhibition34, and we note that the action spectrum for light inhibition has not been determined and the cells could be sensitive to frequencies that are normally absorbed in the water column. Regardless, our findings indicate that the challenge of culturing these cells might in part be explained by their sensitivity to light.
a, c Relative abundances are plotted for various stations and depths from the four marine trenches with water depths of more than 6000 m (a) and station ALOHA and TARA Oceans stations (c). For TARA Oceans, only the stations with samples from water depths of both less than 100 m and more than 250 m were used. The dotted horizontal line in (c) indicates a depth of 200 m, the boundary between epipelagic (euphotic) and mesopelagic zones. Bubble colors in (c) correspond to the oceans where the sampling stations are located, following the legend at the right: SO, the Southern Ocean; IO, the Indian Ocean; SAO and NAO, the South and North Atlantic Ocean, respectively; SPO and NPO, the South and North Pacific Ocean, respectively. b Data used in (a) re-plotted to show the change in relative abundances as a function of depth. Regression line (in blue) and confidence intervals (95%; gray shading) were drawn by “geom_smooth” function of the ggplot2 R package with “gam” method (generalized additive model). Source data are provided as a Source data file.
We propose that the vertical distribution of SAR202 group I members represented by the JH545 population can be reconciled with their metabolic features that we report here. The compounds that enhanced the growth of JH545 (e.g., fucose and rhamnose) and related compounds that we predict JH545 and SAR202 group I bacteria might also utilize are found in the surface ocean largely as monomers in polysaccharides produced by phytoplankton51. The JH545 genome had a limited repertoire of glycoside hydrolases (GHs) and lacked representative GH families annotated as fucosidase and rhamnosidase (e.g., GH29, GH95, GH141, GH78, and GH106)41, 50,60. SAR202 group I may scavenge monomeric compounds and their metabolic products that diffuse into the water column during degradation of polysaccharides by other taxa (e.g., Bacteroidia, Gammaproteobacteria, and Verrucomicrobiota) (ref. 51 and references therein), many of which are capable of much more rapid growth and would have a competitive advantage as specialists when polysaccharides are available. In the dark ocean, where polysaccharides are depleted, however, paralogous gene expansions may provide group I with a competitive advantage by allowing them to use a diverse range of sugars, sugar acids, and related compounds, leading to the observed increase in relative abundance. Piezotolerance of SAR202 cells would also give them a competitive advantage in the dark ocean61. We note, however, that there are other SAR202 groups (e.g., group III) that are known to contribute substantially to the high relative abundance of the SAR202 clade in the deepest ocean regions. The isolation and characterization of a wider diversity of SAR202, especially lineages typical of the dark ocean, could propel future research that aims to reconstruct carbon chemistry and ecology in the dark ocean.
Evolutionary diversifications of paralogous enzymes in SAR202 cells have been proposed to benefit these cells by expanding the range of organic compounds they can metabolize8,12. To explain the non-phosphorylative pathways for sugar oxidation we report, and the possibility of a much larger set of parallel pathways in the same cells, we hypothesize that SAR202 group I cells have evolved to exploit relatively rare carbohydrate compounds that are not harvested by taxa specializing in more common carbohydrate types. Carbohydrates are structurally complex because of the chirality of monomers, the diversity of the linkages they form, and modifications such as O-methylation and sulfation. In this scenario, SAR202 exploits a niche, harvesting a class of compounds that are recalcitrant because of their low abundance and structural diversity. This combination of features, i.e., high molecular complexity and low concentrations of individual molecular species, is associated with the molecular diversity hypothesis62, a leading explanation for the sequestration of ocean carbon in dissolved molecules with millennial turnover times. Our findings are not contrary to this hypothesis; they instead suggest a class of compounds that would accumulate for the same reasons if a particular cell type, such as SAR202 group I, had not evolved a unique mechanism to harvest them. It should be noted that carbon pools in the dark ocean could also be affected by other SAR202 groups. For example, SAR202 group III, which is known to be more abundant than group I in the dark ocean, has been suggested to contribute to the degradation and transformation of recalcitrant organic matter using an expanded repertoire of flavin-dependent monooxygenases8,12.
In summary, we describe the accomplished cultivation of the abundant and ubiquitous marine bacterial SAR202 clade, a monophyletic superorder within the class Dehalococcoidia of the phylum Chloroflexota. The SAR202 strains grew very slowly, and their growth was further inhibited by exposure to light; these properties might explain why they have not been cultivated previously. We show that these cells contain paralog expansions and that these paralogs can be arranged into non-phosphorylative pathways for the catabolism of sugars and their lactone and acid forms. We show that fucose and rhamnose, predicted substrates of these pathways, enhanced the growth of the SAR202 isolate.
We explored the physiology of a superorder of bacteria that have been implicated in the oxidation of a variety of forms of semi-labile organic carbon, and it seems likely that further studies of the diverse cells in this clade will contribute to a more mechanistic understanding of the ocean carbon cycle. Cell cultures of novel prokaryotes provide opportunities to study a wide range of cellular properties that cannot be determined from genomes alone. Interactions of these slowly growing cells with organic carbon exometabolites, and their unexplained sensitivity to light, are promising avenues for future work. Studies of SAR202 archaella function and integration with the cell envelope of SAR202 may provide clues into the ecology and cell architecture of these unusual cells. The findings we report provided surprising insights into challenges that long-stalled SAR202 cultivation. Whether the diversity of not-yet-cultured lineages of SAR202 cells inhabiting the dark ocean can be explained by similar properties remains to be seen.
Methods
Sample collection and cultivation
Seawater samples used for high-throughput culturing (HTC) based on dilution-to-extinction and amplicon analysis were collected from a depth of 10 m at two stations (GR1 and GR3) in the West Sea of Korea (Yellow Sea) in October 2017 (Supplementary Fig. 1a). Physicochemical properties of the water samples are presented in Supplementary Fig. 1b. The total prokaryotic number was determined by counting 4′,6-diamidino-2-phenylindole (DAPI)-stained cells using an epifluorescence microscope (Nikon 80i, Nikon) after filtration using a 0.2-μm pore-sized polycarbonate membrane filter (Millipore). Culture media for HTC (LNHM) was prepared using a seawater sample collected from the East Sea (depth, 10 m) in 2016. In brief, the seawater sample was filtered using a 0.2-μm pore-sized polyethersulfone membrane filter (Pall), autoclaved (1.5 h), sparged with CO2 (8 h), aerated (24 h), and amended with carbon sources, macronutrients (nitrogen and phosphorus sources), trace metals, vitamins, and amino acids (Supplementary Table 1). The seawater samples were then diluted with culture media to a concentration of 5 cells mL−1 and dispensed (1 mL per well) into 48-well microplates (BD Falcon). The plates were incubated at 20 °C for 1 month in dark or under LED light (Philips; correlated color temperature, 3000 K; light intensity, ~155 μmol photons m–2 s–1) with a light/dark cycle of 14:10 h. The microbial growth in each well was screened by flow cytometry (GUAVA EasyCyte Plus flow cytometer and Guava CytoSoft (v5.3), Millipore) after staining with SYBR Green I (Life Technologies) and recorded as growth-positive when more than 5.0 × 104 cells mL−1 were detected. Cultures from growth-positive wells were used for further analyses and stored as glycerol stock (10%, v/v) at −80 °C.
Phylogenetic analysis and classification of 16S rRNA gene sequences
Phylogenetic analyses of growth-positive cultures were based on PCR amplification and sequencing of 16S rRNA genes. DNA templates for PCR were prepared from growth-positive cultures using the InstaGeneTM Matrix (Bio-Rad) according to the manufacturer’s instructions. The amplification of 16S rRNA genes were performed by PCR using 27F and 1492R primers, followed by Sanger sequencing with 800R and 518F primers (Macrogen Inc., Korea). Taxonomic classification of 610 strains obtained from the HTC experiments was carried out using the “classify.seqs” command of the Mothur software package (v1.39.5)63 using the SILVA database SSURef NR99 (release 132)64 as a reference. For more refined taxonomic and phylogenetic analysis of the SAR202 isolates, the 16S rRNA gene sequences were aligned using the SINA online aligner (v1.2.11, http://www.arb-silva.de/aligner)65, imported into ARB program66, and inserted using the ARB parsimony into the guide tree of SILVA database SSURef NR99 (release 132)64. After manual curation, the aligned sequences of the isolates and their phylogenetic relatives were exported with “ssuref:bacteria” filter. Maximum-likelihood phylogenetic trees were constructed using RAxML67 (v8.2.12) with GTRGAMMA method including 100 bootstrap replicates and visualized using the MEGA software (v7.0)68.
Microbial community analysis
Two liters of each seawater sample (GR1 and GR3) were filtered through a 0.2-μm pore-size polyethersulfone membrane filter (Supor, Pall). DNA was extracted directly from the membrane filters using DNeasy PowerWater Kit (Qiagen) according to the manufacturer’s instructions. The V4-V5 regions of 16S rRNA genes were amplified using fusion primers that were designed based on universal primers, 518F and 926R69. The pooled PCR products were sequenced on Illumina MiSeq platform (300-bp, paired-end; Chunlab Inc.). The analyses of the 16S rRNA gene amplicon sequences were performed using QIIME270 after primer trimming with cutadapt (v2.7)71.
Culture experiments
The SAR202 strains were grown and maintained using LNHM5x at 20 °C in dark. Growth experiments were performed using LNHM, LNHM5x, and ASW5x. The detailed recipes of the media are presented in Supplementary Table 1. Bacterial growth was monitored by flow cytometry, and the purity and identity of cultures were regularly determined by sequencing the amplified 16S rRNA genes and by microscopic examination.
The growth of strain JH545 was monitored at temperatures ranging from 4 to 37 °C in dark. Growth substrate tests were performed using ʟ-rhamnose, ʟ-rhamnono-1,4-lactone, ʟ-rhamnonate, ʟ-fucose, ʟ-fucono-1,4-lactone, ʟ-fuconate, and ascorbate. All tested compounds were purchased from Sigma-Aldrich. These chemicals were added to ASW5x media at a final concentration of 250 μM. Cultures were incubated in dark at 20 °C.
For the initial examination of the effect of light exposure on SAR202 growth (Fig. 2b), cells were inoculated in LNHM at an initial cell density of ~1.0 × 104 cells mL−1 and incubated in a chamber equipped with LED (3000 K; Phillips), with a light/dark cycle of 14:10 h. Light intensity was ~155 μmol photons m−2 s−1. To test the effect of light intensity on SAR202 growth (Fig. 2c), a custom-made equipment with a LED-array (3000 K) and a light dimmer was utilized, and the light intensities were set at ~45 (weak light), ~89 (medium light), and ~134 (strong light) μmol photons m−2 s−1. During the test, the lights continuously remained on. All experiments included control samples that were constantly kept in dark by wrapping the tubes or flasks with aluminum foil. Light intensity was measured using a digital light meter (TES-1335; TES).
Morphological characterization
Cell morphology was observed by transmission electron microscopy (TEM; CM200, Philips) and scanning electron microscopy (SEM; S-4300 and S-4300SE, Hitachi). To prepare samples for TEM, 20 mL of culture prefixed with 2.5% glutaraldehyde were filtered using a 0.2-μm pore-size polycarbonate membrane on which formvar/carbon-coated copper grids were placed, followed by staining of the grids with uranyl acetate (2%). For thin-section TEM, centrifuged cells from 800 mL of culture were subjected to primary fixation with Karnovsky’s solution (2% paraformaldehyde, 2.5% glutaraldehyde), post-fixation with 2% OsO4, en-bloc staining with 0.5% uranyl acetate, sequential dehydration with 30, 50, 70, 80, 90, and 100% ethanol, and embedding with resin (EMBed-812, Electron Microscopic Science). Finally, sectioning was performed using a diamond knife. Thin sections were stained with uranyl acetate (2%). For SEM analysis, 100 mL of culture was concentrated by centrifugation, fixed with 2.5% glutaraldehyde, post-fixed with 1% OsO4, dehydrated with 30, 50, 70, 80, 90, and 100% ethanol, and chemically dried using hexamethyldisilazane (Sigma-Aldrich). Treated samples were gently mounted on a cover glass and coated with a thin layer of carbon.
Genome sequencing, assembly, and analyses
Genomic DNA of the SAR202 strains was extracted from cell pellets obtained by centrifugation of liquid cultures (~1 L), using DNeasy Blood & Tissue Kit (Qiagen), according to manufacturer’s instructions. The genomic DNA of strains JH545 and JH1073 was used for the construction of the 20-kb SMRTbell library, which was sequenced on the PacBio RS II platform (Pacific Biosciences). De novo assembly of raw sequencing reads was carried out by the RS_HGAP_Assembly.2 protocol of SMRT Analysis (v2.3.0), resulting in a single contig. The contig was circularized using Circlator (v1.5.5)72 and polished using the RS_Resequencing.1 protocol of SMRT Analysis to obtain the final error-corrected genome sequence. Genome sequencing of strains JH702 and JH639 was performed on the Illumina HiSeq platform (2 × 150 bp). Raw reads were trimmed using BBDuk with the following options: ktrim=r k=23 mink=11 hdist=1 tpe tbo ftm=5 qtrim=rl trimq=10 minlen=100. Assembly of Illumina sequencing data was performed using SPAdes v3.11.1 in a multi-cell mode with read error and mismatch correction73.
The genome sequences were submitted to the IMG-ER system for annotation. Prokka (v1.12)74 was also used for annotation. The predicted protein sequences were analyzed using BlastKOALA75 and KofamKOALA76 for metabolic pathway reconstruction based on KEGG Orthologs (KOs). Annotation by eggnog-mapper (v2.0.1)77 and hmmsearch (v3.3) against protein databases such as Pfam were also performed for more accurate and detailed functional annotation. Analysis of CAZymes was performed using dbCAN278. A map of metabolic pathways was created using BioRender (https://biorender.com). ANIb values between genomes were calculated using JSpeciesWS79. Genomic islands were predicted using IslandViewer 480. Genome comparison was visualized by BLAST Ring Image Generator (BRIG)81.
The SAR202 genome sequences of the present study and a previous study12 were classified using GTDB-Tk (v1.7.0), which indicated that the genomes belonged to ~10 different orders according to the GTDB. Representative genomes of these orders were used to construct a phylogenomic tree of the SAR202 clade. We used UBCG pipeline82 to obtain a concatenated alignment of core genes. A maximum-likelihood tree was constructed using RAxML (v8.2.12)67, with a PROTGAMMAAUTO option including 100 bootstrap iterations.
Analyses of the HeR and FlaB
Genomic regions around the HeR genes of the JH545 genome were visualized using Easyfig83. Multiple amino acid alignment of HeRs from JH545 and the first-characterized HeR (48C12) was performed using ClustalW84. The Clustal X color scheme of the Jalview (v2.11.1.3)85 was applied to visualize the alignment.
Various FlaB sequences were collected for phylogenetic analysis. In addition to literature searches, putative orthologs of JH545 FlaBs were searched using SHOOT86 (https://www.shoot.bio/), resulting in the retrieval of only archaeal FlaBs. Bacterial FlaBs were searched using BlastP in IMG-ER and NCBI (nr database) with JH545 FlaBs as queries. Additionally, AnnoTree was searched using K07325 (archaeal flagellin FlaB) as a query. The collected sequences were aligned using MUSCLE, followed by tree building using RAxML (v8.2.12) with the PROTGAMMAAUTO option. FlaBs from MAGs or SAGs affiliated with bacterial phyla other than Chloroflexota were removed from the analyses, as these FlaBs were the only ones found in their respective phyla, suggesting the possibility of contamination. Some archaeal FlaBs that are much longer than other sequences were also removed. Some genomes in which FlaB was found were used to visualize the gene map of the archaella gene cluster using Easyfig.
Analysis of COG4948 proteins
A total of 80 proteins were assigned to COG4948 by the IMG-ER annotation of the JH545 genome, which was also verified by the Conserved Domain search87 (v3.18) at NCBI. A search against Pfam-A database (by Pfam_Scan.pl; both available at https://ftp.ebi.ac.uk/pub/databases/Pfam/) showed that 74 COG4948 proteins had both PF02746 and PF13378 domains. The remaining six proteins had either PF13378 (four proteins) or PF02746 (two proteins) domains. The two Pfam domains were found only in 80 COG4948 proteins. Based on these results showing a correspondence between COG4948 and the two Pfam domains, we decided to use the two Pfam domains for approximate calculation of the proportion of COG4948 proteins in 47,894 species cluster-representative genomes of the GTDB (R202). The genomic faa files (gtdb_proteins_aa_reps_r202.tar.gz) were downloaded from the GTDB repository (https://data.gtdb.ecogenomic.org/) and searched using hmmsearch (v3.3) against the hmm files of the two Pfam domains, with “cut_tc” option. The approximate proportion of COG4948 proteins in each genome was calculated by dividing the number of hmmsearch hits by twice the number of CDS. The results were visualized using the R package ‘tidyverse’.
The 80 COG4948 protein sequences of JH545 were submitted to the Enzyme Similarity Tool (EFI-EST; https://efi.igb.illinois.edu/efi-est/)42 to generate an SSN. A total of 77,142 proteins in the UniProt database (v2020_02) that had the two Pfam domains were included in the SSN, which was constructed using BLAST with default options. The obtained SSN was explored using the Cytoscape (v3.7.2)88. After the application of a cutoff threshold of alignment score (larger than or equal to 150), clusters that included the 80 COG4948 proteins of the strain JH545 were retained for visualization.
Reconstruction of non-phosphorylative fucose and rhamnose degradation pathways is based on a KEGG pathway map (fructose and mannose metabolism; map00051), MetaCyc pathways (l-fucose degradation II/III and l-rhamnose degradation II/III), and several publications46,47,52,53,54,55. When necessary, proteins characterized in past reports were explored in the UniProt database or analyzed by CD-search at NCBI to ascertain their COG assignments.
Metagenome fragment recruitment
The relative abundance of the JH545 population in marine metagenomes was estimated using CoverM (v0.6.1; https://github.com/wwood/CoverM). Metagenomes from several marine trenches, station ALOHA (collected in December, 2011), and some Tara Oceans stations were downloaded from SRA and quality-trimmed using BBduk (v38.86)89. Ribosomal RNA and tRNA genes of the JH545 genome were masked before the analyses. CoverM was run with the following options: --mapper bwa-mem --methods relative_abundance --min-read-aligned-length 50 --min-read-percent-identity 95. Note that only the JH545 genome was used as the reference genome. The threshold value for “--min-read-percent-identity” was set to 95%, an ANI value widely used for species demarcation, as we wanted to estimate the relative abundance of metagenome reads that could be regarded as being from the same species as JH545. The list of metagenome samples is provided in Supplementary Data 2.
Analysis of fatty acid composition
The analysis of cellular fatty acid methyl esters (FAMEs) was performed using the standard protocol provided by the MIDI/Hewlett-Packard Microbial Identification System. To extract FAMEs, cells of strain JH545 were harvested by centrifugation at 13,000 × g for 1 h at the end of the exponential growth phase in a 400 mL volume of liquid culture (ASW5x media). The extracted FAMEs were saponified and methylated before being analyzed using a gas chromatograph (Agilent 7890 GC) with TSBA6 database from the Sherlock Microbial Identification System (MIDI) version 6.1.
Proposal of ranks of the new taxa
Description of “Candidatus Lucifugimonas” gen. nov
Lucifugimonas (Lu.ci.fu.gi.mo’nas. L. fem. n. lux, lucis, light; L. fem. n. fuga, flight; L. fem. n. monas, a unit, monad; N.L. fem. n. Lucifugimonas, a monad that prefers dark habitats).
Aerobic, oligotrophic, and chemoheterotrophic. Gram-negative with a monoderm envelope. Cells are non-motile and dimorphic with short rods of ~0.8 × 0.4 µm and cocci of ~0.5 µm diameter. Do not form colonies on solid agar plates. Grows very slowly and reaches stationary phase at ~50 days of growth in artificial seawater medium. Light inhibits cellular growth. Major cellular fatty acids are summed feature 9 (C17:1 ω9c and/or 10-methyl C16:0), 10-methyl C18:0, and C16:0 (Supplementary Table 4). The genus “Candidatus Lucifugimonas” is assigned to SAR202 group Ia within the class Dehalococcoidia based on 16S rRNA gene phylogeny and whole genome phylogenomics. The type species of the genus is “Candidatus Lucifugimonas marina”.
Description of “Candidatus Lucifugimonas marina” sp. nov
Lucifugimonas marina (ma.ri’na. L. fem. adj. marina, marine, of the sea).
In addition to the properties given in the genus description, the species is described as follows. Growth occurs at temperatures between 10 and 25 °C, but not at 4 °C or below, nor at 30 °C or above. Optimum growth temperature is 15−20 °C. Grows only in seawater-based liquid medium or artificial seawater medium. Cellular growth is enhanced by fucose, fuconate, fucono-1,4-lactone, rhamnose, rhamnono-1,4-lactone, rhamnonate, and ascorbate. The type strain, JH545, was isolated from epipelagic seawater off the coast of Garorim Bay at Tea-An, South Korea. The length of the complete whole genome sequence of the type strain is 3.08 Mbp with 51.8% of the DNA G + C content. GenBank accession number of the type strain is CP046146. Besides the type strain, whole genome sequences of strains JH639, JH702, and JH1073 belonging to this species are also available under GenBank accession numbers WMBD00000000, WMBE00000000, and CP046147, respectively.
Description of “Candidatus Lucifugimonadaceae” fam. nov
Candidatus Lucifugimonadaceae (Lu.ci.fu.gi.mo.na.da.ce’ae. N.L. fem. n. Lucifugimonas, a bacterial genus; -aceae, ending to denote a family; N.L. fem. pl. n. Lucifugimonadaceae, the Lucifugimonas family).
The description is the same as with the genus “Candidatus Lucifugimonas”. The type genus is “Candidatus Lucifugimonas”. Equivalent to GTDB f__UBA1328 (R207).
Description of “Candidatus Lucifugimonadales” order. nov
Candidatus Lucifugimonadales (Lu.ci.fu.gi.mo.na.da’les. N.L. fem. n. Lucifugimonas, a bacterial genus; -ales, ending to denote a family; N.L. fem. pl. n. Lucifugimonadales, the Lucifugimonas order).
The description is the same as with the genus “Candidatus Lucifugimonas”. The type genus is “Candidatus Lucifugimonas”. Equivalent to GTDB o__UBA1151 (R207).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All relevant data supporting the findings of this study are available within the paper and its supplementary information and data files. The 16S rRNA gene sequences of 24 isolates in SAR202 group I generated in this study have been deposited in the GenBank database under accession numbers OQ689977 to OQ690000. The whole genome sequences generated in this study are available in the GenBank database under accession numbers CP046146 (JH545), CP046147 (JH1073), WMBD00000000 (JH639), and WMBE00000000 (JH702). The genomic data are also available in the IMG/M database under genome IDs 2901382945 (JH545), 2917498938 (JH1073), 2892960865 (JH639), and 2892963810 (JH702). Source data are provided with this paper.
References
Giovannoni, S. J., Rappé, M. S., Vergin, K. L. & Adair, N. L. 16S rRNA genes reveal stratified open ocean bacterioplankton populations related to the green non-sulfur bacteria. Proc. Natl Acad. Sci. USA 93, 7979–7984 (1996).
DeLong, E. F. et al. Community genomics among stratified microbial assemblages in the ocean’s interior. Science 311, 496–503 (2006).
Morris, R., Rappe, M., Urbach, E., Connon, S. & Giovannoni, S. J. Prevalence of the Chloroflexi-related SAR202 bacterioplankton cluster throughout the mesopelagic zone and deep ocean. Appl. Environ. Microbiol. 70, 2836–2842 (2004).
Schattenhofer, M. et al. Latitudinal distribution of prokaryotic picoplankton populations in the Atlantic Ocean. Environ. Microbiol. 11, 2078–2093 (2009).
Varela, M. M., Van Aken, H. M. & Herndl, G. J. Abundance and activity of Chloroflexi‐type SAR202 bacterioplankton in the meso‐and bathypelagic waters of the (sub) tropical Atlantic. Environ. Microbiol. 10, 1903–1911 (2008).
Wei, Z.-F., Li, W.-L., Huang, J.-M. & Wang, Y. Metagenomic studies of SAR202 bacteria at the full-ocean depth in the Mariana Trench. Deep Sea Res. Part I Oceanogr. Res. Pap. 165, 103396 (2020).
Sebastián, M. et al. Environmental gradients and physical barriers drive the basin‐wide spatial structuring of Mediterranean Sea and adjacent eastern Atlantic Ocean prokaryotic communities. Limnol. Oceanogr. 66, 4077–4095 (2021).
Landry, Z., Swan, B. K., Herndl, G. J., Stepanauskas, R. & Giovannoni, S. J. SAR202 genomes from the dark ocean predict pathways for the oxidation of recalcitrant dissolved organic matter. MBio 8, e00413–e00417 (2017).
Thrash, J. C. et al. Metabolic roles of uncultivated bacterioplankton lineages in the northern Gulf of Mexico “dead zone”. MBio 8, e01017–e01017 (2017).
Mehrshad, M., Rodriguez-Valera, F., Amoozegar, M. A., López-García, P. & Ghai, R. The enigmatic SAR202 cluster up close: shedding light on a globally distributed dark ocean lineage involved in sulfur cycling. ISME J. 12, 655 (2018).
Colatriano, D. et al. Genomic evidence for the degradation of terrestrial organic matter by pelagic Arctic Ocean Chloroflexi bacteria. Commun. Biol. 1, 90 (2018).
Saw, J. H. et al. Pangenomics analysis reveals diversification of enzyme families and niche specialization in globally abundant SAR202 bacteria. MBio 11, e02975–02919 (2020).
Shang, H., Rothman, D. H. & Fournier, G. P. Oxidative metabolisms catalyzed Earth’s oxygenation. Nat. Commun. 13, 1–9 (2022).
Lewis, W. H., Tahon, G., Geesink, P., Sousa, D. Z. & Ettema, T. J. Innovations to culturing the uncultured microbial majority. Nat. Rev. Microbiol. 19, 225–240 (2021).
Price, M. N., Deutschbauer, A. M. & Arkin, A. P. Filling gaps in bacterial catabolic pathways with computation and high-throughput genetics. PLoS Genet. 18, e1010156 (2022).
Steen, A. D. et al. High proportions of bacteria and archaea across most biomes remain uncultured. ISME J. 13, 3126–3130 (2019).
Carini, P. A. “cultural” renaissance: genomics breathes new life into an old craft. mSystems 4, e00092–00019 (2019).
Sung, Y., Ritalahti, K. M., Apkarian, R. P. & Löffler, F. E. Quantitative PCR confirms purity of strain GT, a novel trichloroethene-to-ethene-respiring Dehalococcoides isolate. Appl. Environ. Microbiol. 72, 1980–1987 (2006).
Löffler, F. E. et al. Dehalococcoides mccartyi gen. nov., sp. nov., obligately organohalide-respiring anaerobic bacteria relevant to halogen cycling and bioremediation, belong to a novel bacterial class, Dehalococcoidia classis nov., order Dehalococcoidales ord. nov. and family Dehalococcoidaceae fam. nov., within the phylum Chloroflexi. Int. J. Syst. Evol. Microbiol. 63, 625–635 (2013).
Sutcliffe, I. C. Cell envelope architecture in the Chloroflexi: a shifting frontline in a phylogenetic turf war. Environ. Microbiol. 13, 279–282 (2011).
Sutcliffe, I. C. A phylum level perspective on bacterial cell envelope architecture. Trends Microbiol. 18, 464–470 (2010).
Wasmund, K. et al. Genome sequencing of a single cell of the widely distributed marine subsurface Dehalococcoidia, phylum Chloroflexi. ISME J. 8, 383–397 (2014).
Fullerton, H. & Moyer, C. L. Comparative single-cell genomics of Chloroflexi from the Okinawa Trough deep-subsurface biosphere. Appl. Environ. Microbiol. 82, 3000–3008 (2016).
Maymo-Gatell, X., Chien, Y.-T., Gossett, J. M. & Zinder, S. H. Isolation of a bacterium that reductively dechlorinates tetrachloroethene to ethene. Science 276, 1568–1571 (1997).
Adrian, L., Szewzyk, U., Wecke, J. & Görisch, H. Bacterial dehalorespiration with chlorinated benzenes. Nature 408, 580–583 (2000).
Albers, S.-V. & Jarrell, K. F. The archaellum: an update on the unique archaeal motility structure. Trends Microbiol. 26, 351–362 (2018).
Jarrell, K. F., Albers, S.-V. & Machado, J. A comprehensive history of motility and Archaellation in Archaea. FEMS Microbes 2, xtab002 (2021).
Albers, S. V., Szabó, Z. & Driessen, A. J. Archaeal homolog of bacterial type IV prepilin signal peptidases with broad substrate specificity. J. Bacteriol. 185, 3918–3925 (2003).
Hug, L. A. et al. Community genomic analyses constrain the distribution of metabolic traits across the Chloroflexi phylum and indicate roles in sediment carbon cycling. Microbiome 1, 1–17 (2013).
Kale, V. et al. Litorilinea aerophila gen. nov., sp. nov., an aerobic member of the class Caldilineae, phylum Chloroflexi, isolated from an intertidal hot spring. Int. J. Syst. Evol. Microbiol. 63, 1149–1154 (2013).
Maurais, E. G., Iannazzi, L. C. & MacLea, K. S. Genome Sequence of Litorilinea aerophila, an Icelandic Intertidal Hot Springs Bacterium. Microbiol. Resour. Announc. 11, e01206–e01221 (2022).
Nakahara, N. et al. Aggregatilinea lenta gen. nov., sp. nov., a slow-growing, facultatively anaerobic bacterium isolated from subseafloor sediment, and proposal of the new order Aggregatilineales ord. nov. within the class Anaerolineae of the phylum Chloroflexi. Int. J. Syst. Evol. Microbiol. 69, 1185–1194 (2019).
Singh, M., Inoue, K., Pushkarev, A., Béjà, O. & Kandori, H. Mutation study of heliorhodopsin 48C12. Biochemistry 57, 5041–5049 (2018).
Bulzu, P.-A. et al. Heliorhodopsin evolution is driven by photosensory promiscuity in monoderms. mSphere 6, e00661–21 (2021).
Chazan, A. et al. Diverse heliorhodopsins detected via functional metagenomics in freshwater Actinobacteria, Chloroflexi and Archaea. Environ. Microbiol. 24, 110–121 (2022).
Pao, S. S., Paulsen, I. T. & Saier, M. H. Major facilitator superfamily. Microbiol. Mol. Biol. Rev. 62, 1–34 (1998).
Reddy, V. S., Shlykov, M. A., Castillo, R., Sun, E. I. & Saier, M. H. Jr The major facilitator superfamily (MFS) revisited. FEBS J. 279, 2022–2035 (2012).
Dang, S. et al. Structure of a fucose transporter in an outward-open conformation. Nature 467, 734–738 (2010).
Stack, T. M. et al. Characterization of an l-ascorbate catabolic pathway with unprecedented enzymatic transformations. J. Am. Chem. Soc. 142, 1657–1661 (2020).
Diallo, M. et al. L-Rhamnose metabolism in Clostridium beijerinckii strain DSM 6423. Appl. Environ. Microbiol. 85, e02656–02618 (2019).
Reisky, L. et al. A marine bacterial enzymatic cascade degrades the algal polysaccharide ulvan. Nat. Chem. Biol. 15, 803–812 (2019).
Gerlt, J. A. et al. Enzyme function initiative-enzyme similarity tool (EFI-EST): a web tool for generating protein sequence similarity networks. Biochim. Biophys. Acta - Proteins Proteom. 1854, 1019–1037 (2015).
Wichelecki, D. J. et al. Discovery of function in the enolase superfamily: D-mannonate and D-gluconate dehydratases in the D-mannonate dehydratase subgroup. Biochemistry 53, 2722–2731 (2014).
Rakus, J. F. et al. Evolution of enzymatic activities in the enolase superfamily: L-rhamnonate dehydratase. Biochemistry 47, 9944–9954 (2008).
Yew, W. S. et al. Evolution of enzymatic activities in the enolase superfamily: L-fuconate dehydratase from Xanthomonas campestris. Biochemistry 45, 14582–14597 (2006).
Watanabe, S., Saimura, M. & Makino, K. Eukaryotic and bacterial gene clusters related to an alternative pathway of nonphosphorylated L-rhamnose metabolism. J. Biol. Chem. 283, 20372–20382 (2008).
Watanabe, S. & Makino, K. Novel modified version of nonphosphorylated sugar metabolism—an alternative l‐rhamnose pathway of Sphingomonas sp. FEBS J. 276, 1554–1567 (2009).
Ghasempur, S. et al. Discovery of a novel L-lyxonate degradation pathway in Pseudomonas aeruginosa PAO1. Biochemistry 53, 3357–3366 (2014).
Kopp, D., Bergquist, P. L. & Sunna, A. Enzymology of alternative carbohydrate catabolic pathways. Catalysts 10, 1231 (2020).
Orellana, L. H. et al. Verrucomicrobiota are specialist consumers of sulfated methyl pentoses during diatom blooms. ISME J. 16, 630–641 (2022).
Arnosti, C. et al. The biogeochemistry of marine polysaccharides: sources, inventories, and bacterial drivers of the carbohydrate cycle. Annu. Rev. Mar. Sci. 13, 81–108 (2021).
Bae, J., Kim, S. M. & Lee, S. B. Identification and characterization of 2-keto-3-deoxy-L-rhamnonate dehydrogenase belonging to the MDR superfamily from the thermoacidophilic bacterium Sulfobacillus thermosulfidooxidans: implications to L-rhamnose metabolism in archaea. Extremophiles 19, 469–478 (2015).
Reinhardt, A., Johnsen, U. & Schönheit, P. l‐Rhamnose catabolism in archaea. Mol. Microbiol. 111, 1093–1108 (2019).
Watanabe, S. Characterization of l-2-keto-3-deoxyfuconate aldolases in a nonphosphorylating l-fucose metabolism pathway in anaerobic bacteria. J. Biol. Chem. 295, 1338–1349 (2020).
Sizikov, S. et al. Characterization of sponge‐associated Verrucomicrobia: microcompartment‐based sugar utilization and enhanced toxin–antitoxin modules as features of host‐associated Opitutales. Environ. Microbiol. 22, 4669–4688 (2020).
Arístegui, J., Gasol, J. M., Duarte, C. M. & Herndld, G. J. Microbial oceanography of the dark ocean’s pelagic realm. Limnol. Oceanogr. 54, 1501–1529 (2009).
Yokokawa, T., Yang, Y., Motegi, C. & Nagata, T. Large‐scale geographical variation in prokaryotic abundance and production in meso‐and bathypelagic zones of the central Pacific and Southern Ocean. Limnol. Oceanogr. 58, 61–73 (2013).
Herndl, G. J., Bayer, B., Baltar, F. & Reinthaler, T. Prokaryotic life in the deep ocean’s water column. Annu. Rev. Mar. Sci. 15, 461–483 (2023).
Cardozo-Mino, M. G., Fadeev, E., Salman-Carvalho, V. & Boetius, A. Spatial distribution of Arctic bacterioplankton abundance is linked to distinct water masses and summertime phytoplankton bloom dynamics (Fram Strait, 79° N). Front. Microbiol. 12, 658803 (2021).
Sichert, A. et al. Verrucomicrobia use hundreds of enzymes to digest the algal polysaccharide fucoidan. Nat. Microbiol. 5, 1026–1039 (2020).
Amano, C. et al. Limited carbon cycling due to high-pressure effects on the deep-sea microbiome. Nat. Geosci. 15, 1041–1047 (2022).
Kattner, G., Simon, M. & Koch, B. in Microbial Carbon Pump in the Ocean (eds. Jiao, N., Azam, F. & Sanders, S.) 60–61 (Science/AAAS, 2011).
Kozich, J. J., Westcott, S. L., Baxter, N. T., Highlander, S. K. & Schloss, P. D. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 79, 5112–5120 (2013).
Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41, D590–D596 (2012).
Pruesse, E., Peplies, J. & Glöckner, F. O. SINA: accurate high-throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics 28, 1823–1829 (2012).
Ludwig, W. et al. ARB: a software environment for sequence data. Nucleic Acids Res. 32, 1363–1371 (2004).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Kumar, S., Stecher, G. & Tamura, K. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 33, 1870–1874 (2016).
Parada, A. E., Needham, D. M. & Fuhrman, J. A. Every base matters: assessing small subunit rRNA primers for marine microbiomes with mock communities, time series and global field samples. Environ. Microbiol. 18, 1403–1414 (2016).
Bolyen, E. et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 37, 852–857 (2019).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).
Hunt, M. et al. Circlator: automated circularization of genome assemblies using long sequencing reads. Genome Biol. 16, 294 (2015).
Bankevich, A. et al. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 19, 455–477 (2012).
Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics 30, 2068–2069 (2014).
Kanehisa, M., Sato, Y. & Morishima, K. BlastKOALA and GhostKOALA: KEGG tools for functional characterization of genome and metagenome sequences. J. Mol. Biol. 428, 726–731 (2016).
Aramaki, T. et al. KofamKOALA: KEGG ortholog assignment based on profile HMM and adaptive score threshold. Bioinformatics 36, 2251–2252 (2020).
Huerta-Cepas, J. et al. Fast genome-wide functional annotation through orthology assignment by eggNOG-mapper. Mol. Biol. Evol. 34, 2115–2122 (2017).
Zhang, H. et al. dbCAN2: a meta server for automated carbohydrate-active enzyme annotation. Nucleic Acids Res. 46, W95–W101 (2018).
Richter, M., Rosselló-Móra, R., Oliver Glöckner, F. & Peplies, J. JSpeciesWS: a web server for prokaryotic species circumscription based on pairwise genome comparison. Bioinformatics 32, 929–931 (2015).
Bertelli, C. et al. IslandViewer 4: expanded prediction of genomic islands for larger-scale datasets. Nucleic Acids Res. 45, W30–W35 (2017).
Alikhan, N.-F., Petty, N. K., Zakour, N. L. B. & Beatson, S. A. BLAST Ring Image Generator (BRIG): simple prokaryote genome comparisons. BMC Genom. 12, 1 (2011).
Na, S.-I., Kim, Y. O., Yoon, S.-H., Ha, S.-M. & Chun, J. UBCG: up-to-date bacterial core gene set and pipeline for phylogenomic tree reconstruction. J. Microbiol. 56, 280–285 (2018).
Sullivan, M. J., Petty, N. K. & Beatson, S. A. Easyfig: a genome comparison visualizer. Bioinformatics 27, 1009–1010 (2011).
Thompson, J. D., Gibson, T. J. & Higgins, D. G. Multiple sequence alignment using ClustalW and ClustalX. Curr. Protoc. Bioinformatics 2.3.1–2.3.22 https://doi.org/10.1002/0471250953.bi0203s00 (2003).
Waterhouse, A. M., Procter, J. B., Martin, D. M., Clamp, M. & Barton, G. J. Jalview Version 2—a multiple sequence alignment editor and analysis workbench. Bioinformatics 25, 1189–1191 (2009).
Emms, D. M. & Kelly, S. SHOOT: phylogenetic gene search and ortholog inference. Genome Biol. 23, 85 (2022).
Lu, S. et al. CDD/SPARCLE: the conserved domain database in 2020. Nucleic Acids Res. 48, D265–D268 (2020).
Saito, R. et al. A travel guide to Cytoscape plugins. Nat. Methods 9, 1069 (2012).
Bushnell, B., Rood, J. & Singer, E. BBMerge—accurate paired shotgun read merging via overlap. PLoS ONE 12, e0185056 (2017).
Acknowledgements
This research was supported by High Seas Bioresources Program of Korea Institute of Marine Science & Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries (KIMST-20210646 to J.-C.C.) and National Research Foundation of Korea (NRF) grants (NRF-2022R1A2C3008502, NRF-2018R1A5A1025077, and NRF-2021M3A9I4021431 to J.-C.C.; NRF-2022R1A6A3A01087360 to Y.L.) funded by the Ministry of Sciences and Information and Communications Technology, Korea.
Author information
Authors and Affiliations
Contributions
J.-H.S. and J.-C.C. planned and designed the initial isolation project. Y.L. and J.-H.S. performed the experiments. Y.L., J.-H.S., and I.K. analyzed the data. Y.L., I.K., S.J.G., and J.-C.C. wrote and revised the manuscript. I.K. and J.-C.C. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Brett Baker and Yusuke Okazaki for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lim, Y., Seo, JH., Giovannoni, S.J. et al. Cultivation of marine bacteria of the SAR202 clade. Nat Commun 14, 5098 (2023). https://doi.org/10.1038/s41467-023-40726-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-023-40726-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.