Abstract
Supports can widely affect or even dominate the catalytic activity, selectivity, and stability of metal nanoparticles through various metal-support interactions (MSIs). However, underlying principles have not been fully understood yet, because MSIs are influenced by the composition, size, and facet of both metals and supports. Using Ru/TiO2 supported on rutile and anatase as model catalysts, we demonstrate that metal-support interfacial compatibility can critically control MSI modes and catalytic performances in CO2 hydrogenation. Annealing Ru/rutile-TiO2 in air can enhance CO2 conversion to methane resulting from enhanced interfacial coupling driven by matched lattices of RuOx with rutile-TiO2; annealing Ru/anatase-TiO2 in air decreases CO2 conversion and converts the product into CO owing to strong metal-support interaction (SMSI). Although rutile and anatase share the same chemical composition, we show that interfacial compatibility can basically modify metal-support coupling strength, catalyst morphology, surface atomic configuration, MSI mode, and catalytic performances of Ru/TiO2 in heterogeneous catalysis.
Similar content being viewed by others
Introduction
Supported metal nanoparticles (NPs) dominate practical catalysts in producing bulk and fine chemicals, reducing environmental emissions, energy conversions, etc.1,2,3. Their catalytic performances (activity, selectivity, and stability) not only depend on the composition, size, shape, and ligation state of metal NPs, but are also highly affected or even dominated by supports4,5,6,7,8,9,10,11. Metal-support interaction (MSI) has therefore become a central topic in heterogeneous catalysis1,3,12,13. Typical MSI modes include strong metal-support interaction (SMSI), interfacial charge transfer, interfacial perimeter, spillover, etc.1. In particular, SMSI phenomena, featuring in encapsulating metallic NPs (such as Pt, Au, Pd, Rh, Ru, and Ni) by reducible oxide supports like TiO2, can widely modify catalytic activity and selectivity of hydrogenation reactions9,14,15,16,17,18,19,20. Although various MSI forms have been reported, the underlying tuning principles still remain elusive, because all structural factors can affect MSIs, such as the composition, size, and shape of metals, the composition, phase, facet, and size of supports, as well as adsorbates and reaction atmospheres1,12,21,22,23,24,25. Moreover, their simultaneous interactions extremely complicate MSI phenomena and challenge the study on the mechanisms. Exploring the principles dominating support effects and MSIs is therefore crucial for rational design, optimization, and understanding of heterogeneous catalysis.
Among all structural factors, metal-support interface should play the primary role, because all MSI modes occur based on the direct contacts of catalysts with supports26,27. For solid–solid contacts, coupling strength is the most fundamental parameter determining the property and stability of their interfaces, which is thermodynamically described with adhesion energy (Φadh)28,29. The relative value of Φadh to bulk cohesion energy (Φcoh) basically determines interfacial contact angles and thermal stability of supported particles30. In catalysis science, adhesion energy widely controls the morphologies and sintering rates of supported-metal NPs31,32. Many post-treatment methods such as thermal annealing and reduction–oxidation cycles can modify interfacial adhesion and catalytic performances1,9,16,22,33,34. Furthermore, at the atomic scale, interfacial coupling occurs through forming chemical bonds, thus, interfacial bonding strength basically determines metal-support adhesions35. For example, Campbell et al. theoretically studied the trends in the adhesion energies of metal NPs on various oxide surfaces, and found that higher metal oxophilicity and more active surface-oxygen atoms could lead to stronger metal-support adhesions29. Senftle et al. further revealed that interfacial binding strengths between single-metal atoms and oxide supports depended on the oxophilicity of supported metals and reducibility of oxide supports36. Despite these understandings on the surface stability of metal NPs on oxides, the structure–function relationships on how interfacial structure features modify MSI modes and catalytic performances are still not fully revealed yet9,33,34,37.
At the atomic scale, strong catalyst-support contacts result from interfacial bonds. The strength parameters, macroscale adhesion energy, and microscale bonding energy can be correlated following:
where EIB and Ns denote the average energy and surface density of interfacial bonds, and k is a coefficient. Thus, Φadh can be enhanced by increasing EIB or Ns. Both EIB and Ns further depend on the atomic configurations of contacting surfaces, because the positions of interfacial atoms intrinsically affect the length, angle, and density of interfacial bonds. Therefore, interfacial configurations of catalysts and supports are intrinsic structural factors controlling catalyst states and MSI effects. The matching degree of interfacial configurations is also referred to as interfacial compatibility, which measures the strength of interfacial bonding and adhesion27,30,35. Catalysts weakly wet supports at misfit interfaces with low interfacial compatibilities, which leads to ready phase separations at the interfaces and catalyst sintering. While high interfacial compatibility can increase both the strength and density of interfacial bonds13,35. The highest interfacial compatibility exists in epitaxial interfaces between lattice-matched materials. In general, lattice misfit less than 5% can form epitaxial overlayers with zero contact angles. This principle has widely guided the growth of semiconductor and oxide heterogeneous structures with minimized interfacial defects38. However, the mechanism of how metal-support contacts and their interfacial compatibilities affect MSI modes and catalytic performances has been rarely revealed yet.
Using Ru/TiO2 as a model catalyst, here we demonstrate the critical role of interfacial compatibility in controlling MSI modes, surface atomic states, and catalytic activity and selectivity in CO2 hydrogenation reaction. Rutile (R–TiO2) and anatase (A–TiO2) are used as supports to vary interfacial structures. RuO2 shares the same lattice structure with R–TiO2, which can lead to a high interfacial compatibility to form epitaxial overlayers39. The interfacial RuOx species can act as anchoring layers to strengthen interfacial bonding between Ru and R–TiO2. For CO2 hydrogenation reaction, we find that both catalytic activity and selectivity can be oppositely modified by annealing Ru/TiO2 catalysts in air. On R–TiO2, Ru shows enhanced activity and dominant selectivity of CH4; on A–TiO2, Ru shows decreased activity and dominant selectivity of CO (Fig. 1a). Such opposite catalytic performances clearly indicate that interfacial compatibility can vary MSI modes—Ru/A–TiO2 shows normal SMSI effect, while Ru/R–TiO2 displays strong interfacial coupling.
a Summary scheme of varied activity and selectivity by direct H2 reduction and after annealing in air. Ru/TiO2–H2 refers to directly reduced catalysts by H2, while Ru/TiO2–air–H2 refers to catalysts by annealing in air at 400 °C and further reduction by H2. b Temperature-dependent CO2 conversions. c Temperature-dependent CH4 selectivity. d Temperature-dependent CO2 conversions of 2% Ru/R–TiO2 catalysts on other R–TiO2 supports that were prepared from TiN and TiCl3, and P25 TiO2. Temperature-dependent CO2 conversions by annealing (e) 2% Ru/R–TiO2–H2 and (f) 4% Ru/R–TiO2–H2 catalysts in Ar and air. Ru/R–TiO2–H2–air–H2 means the catalyst was first reduced by H2 at 300 °C, then annealed in air at 400 °C, and reduced with H2 at 300 °C again.
Results
Support effects on activity and selectivity of Ru/TiO2 for CO2 hydrogenation
All Ru/TiO2 catalysts were prepared following the same protocol to minimize the interference from synthetic conditions. To stabilize the surfaces and prevent further sintering during catalyst preparations and reactions, the supports were first annealed in air at 500 °C for 10 h (Supplementary Fig. 1). The annealed supports and RuCl3 were uniformly mixed in water, rapidly frozen with liquid nitrogen, and dried in a freezing drier (Supplementary Fig. 2). The fully mixed RuCl3–TiO2 precursors were reduced by H2 at 300 °C either directly (Ru/TiO2–H2) or after pre-annealing in air at 400 °C (Ru/TiO2–air–H2). CO2 hydrogenation was conducted at normal pressure with gas hourly space velocity (GHSVs) of 12,000 mL·g−1·h−1, in which the reaction gas composed of 60 vol% H2/15 vol% CO2/25 vol% Ar. At normal pressure, the products of CO2 hydrogenation are CH4 and CO. Therefore, CO2 conversion denotes catalytic activity, and the ratio of n(CH4)/(n(CO) + n(CH4)) denotes selectivity, while the varied activity and selectivity further reflect different support effects and MSI modes5,12,40,41,42,43.
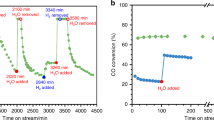
Annealing RuCl3–TiO2 precursors in air can effectively modify catalytic performances of Ru/TiO2 catalysts (Fig. 1a, b). Figure 1b presents temperature-dependent CO2 conversions by 2% Ru/TiO2. Ru/R–TiO2–H2 and Ru/A–TiO2–H2 show similar CO2 conversions between 120 °C and 320 °C, suggesting that R–TiO2 and A–TiO2 apply similar support effects on directly reduced Ru/TiO2 catalysts. However, the conversions dramatically differentiate by pre-annealing RuCl3–TiO2 precursors in air at 400 °C. Ru/R–TiO2–air–H2 displays an enhanced catalytic performance, with CO2 conversion at 300 °C increasing from 31.4% to 89.2%; Ru/A–TiO2–air–H2 shows a highly decreased activity, with CO2 conversion at 300 °C reducing from 29.4% to 1.7%. At each reaction temperature, Ru/R–TiO2–air–H2 shows the highest CO2 conversions among four catalysts. The results indicate that R–TiO2 and A–TiO2 apply opposite support effects on the activity of Ru NPs.
In addition to activity, annealing RuCl3–TiO2 precursors in air can also dramatically modify the catalytic selectivity of Ru on R–TiO2 and A–TiO2 (Fig. 1c). CH4 dominates both the products of Ru/R–TiO2–H2 and Ru/A–TiO2–H2 between 200 °C and 320 °C (>98%). However, the product on Ru/A–TiO2–air–H2 converts into 100% CO between 220 °C and 320 °C, while on Ru/R–TiO2–air–H2 is still 100% CH4 between 140 °C and 320 °C. The different products show another effect of supports on the catalytic performances of Ru NPs18,41.
The enhancement effect of air-annealing on the activity of Ru/R–TiO2 is a general trend. We verified the phenomena by varying the loading amounts of Ru, rutile supports, and post-processing procedures. The activities of 1% Ru/R–TiO2 and 4% Ru/R–TiO2 can also be enhanced by pre-annealing in air, showing the same trend with 2%-Ru/R–TiO2 (Supplementary Fig. 3). Moreover, the activities of 2% Ru/R–TiO2 supported on other R–TiO2 materials prepared using TiCl3 and TiN (Fig. 1d) as precursors, and P25 TiO2, a commercial TiO2 product composed of 4/5 anatase and 1/5 rutile, can also be effectively enhanced by pre-annealing in air. It is noted that CO2 conversions by Ru/R–TiO2–H2 and Ru/R–TiO2–air–H2 are almost the same on these three supports, confirming the stable reproducibility of this enhancement effect.
We further annealed 2% Ru/R–TiO2–H2 (Figs. 1e) and 4% Ru/R–TiO2–H2 (Fig. 1f) catalysts at 400 °C in 25-sccm air or Ar flows, and then reduced with H2 at 300 °C. CO2 conversion by the annealed catalyst in Ar (2% Ru/R–TiO2–H2–Ar–H2) is similar to that of 2% Ru/R–TiO2–H2. In contrast, CO2 conversions by the annealed catalyst in air (2% Ru/R–TiO2–H2–air–H2) notably increase between 160 °C and 320 °C. In particular, CO2 conversion increases from 1.9% to 8.8% at 200 °C, and 31.5% to 48.7% at 300 °C. This promotion effect can also be supported by the reduced apparent activation energies (Ea, Supplementary Fig. 4). Ea of 2% Ru/R–TiO2–H2 is 66.5 kJ·mol−1, while the values are 52.3 kJ·mol−1 and 40.4 kJ·mol−1 for 2% Ru/R–TiO2–H2–Ar–H2 and 2% Ru/R–TiO2–H2–air–H2, respectively5. The enhancement effect on 4% Ru/R–TiO2–H2 is more apparent than 2% Ru/R–TiO2 between 140 °C and 320 °C after annealing in Ar and air. This is because Ru nanoparticles on 4% Ru/R–TiO2–H2 are larger, and annealing can more effectively increase their contacts with R–TiO2 supports.
Geometric states of Ru NPs on TiO2 supports
Given that R–TiO2 and A–TiO2 share the same chemical compositions, supports and catalysts were annealed and prepared following the same procedures, and reaction conditions were controlled the same, Ru–TiO2 interfacial interactions should dominate the opposite support effects. RuO2 shares the same lattice structure with R–TiO2, and their lattice misfit is less than 3.0%, thus RuO2 can form epitaxial overlayers on R–TiO2 with zero contact angles39. Figure 2a, b and Supplementary Fig. 5 present transmission electron microscopy (TEM) and scanning TEM (STEM) images of RuO2/R–TiO2 prepared by annealing RuCl3–R–TiO2 mixture at 400 °C in air. RuO2 encapsulates R–TiO2 nanorods as epitaxial overlayers and forms core–shell structures. Such epitaxial structures can enhance the activity and stability of RuO2 in catalytic oxidation reactions, such as Deacon reaction44. In contrast, RuO2 supported on A–TiO2 are NPs rather than epitaxial overlayers due to their different lattice types (Supplementary Fig. 6). Therefore, RuO2 shows a much higher interfacial compatibility with R–TiO2 than A–TiO2—this intrinsically determines their opposite support effects on the catalytic performances of Ru NPs.
The different interfacial compatibilities modify the surface states of Ru NPs on TiO2 supports first. For Ru/R–TiO2–air–H2, Ru can still partly show epitaxial structures after H2 reduction (Fig. 2c). In particular, after CO2 hydrogenation reaction, Ru presents as flat NPs on Ru/R–TiO2–air–H2 as shown by the images of STEM (Fig. 2d), elemental mapping (Fig. 2e), and high-resolution TEM (Fig. 2f). In contrast, for Ru/R–TiO2–H2, Ru/A–TiO2–H2, and Ru/A–TiO2–air–H2, Ru exists as NPs with average sizes around 2.0–3.0 nm after reactions (Fig. 2g, i, Supplementary Fig. 7). Specifically, size distributions of Ru nanoparticles on Ru–TiO2–air–H2 and Ru–TiO2–H2 are 2.4 ± 0.4 nm and 2.5 ± 0.5 nm, respectively (Supplementary Fig. 7). The high interfacial compatibility can intrinsically increase interfacial coupling strength, and modify the chemical and surface states of Ru species, MSI modes, and catalytic performances of Ru/TiO2 catalysts.
Interfacial bonding states of Ru/TiO2 catalysts
We use H2 temperature-programmed reduction (H2-TPR) to probe the effects of interfacial compatibility on Ru–TiO2 coupling strengths (Fig. 3a). H2-TPR is an effective method to characterize the reducibility of oxides and their interfacial interaction strengths with supports37,45. We prepared RuO2/TiO2 materials by annealing RuCl3–TiO2 mixtures at 400 °C in air. H2-TPR results show that RuO2/A–TiO2 can be reduced between 110 °C and 175 °C; the peaks at 128 °C, 150 °C, and 300 °C correspond to surface RuO2, interfacial RuOx, and surface A–TiO2 species, respectively. While RuO2/R–TiO2 can be reduced between 100 °C and 290 °C, and shows three states at 138, 185, and 270 °C, corresponding to surface RuO2, interfacial RuOx species45. The higher reduction temperature indicates that RuOx is more stable on R–TiO2, which further confirms the stronger interfacial coupling due to matched lattices. Furthermore, X-ray diffraction (XRD) patterns of Ru/A–TiO2 display clear Ru signals, while no Ru peaks appear on Ru/R–TiO2–air–H2 (Fig. 3b, Supplementary Fig. 8). The results indicate higher dispersions of Ru on R–TiO2, agreeing with TEM results.
We use X-ray photoelectron spectroscopy (XPS) to probe the chemical states of Ru. Figure 3c presents Ru-3d and C-1s lines of 2% Ru/TiO2 catalysts after catalytic reactions, in which C-1s is set at 284.8 eV. Ru-3d3/2 spectra show both metallic (Ru0) and oxidized (Ruδ+) states, in which oxidized states mainly locate at Ru–TiO2 interfaces. Ruδ+/Ru0 ratio is 1.3 for Ru/R–TiO2–H2 and 1.5 for Ru/A–TiO2–H2 (Table 1). The approximately identical Ruδ+/Ru0 ratios suggest that directly-reduced Ru NPs show similar interactions with R–TiO2 and A–TiO2, agreeing with their catalytic performances. However, pre-annealing in air can dramatically alter the states of Ru. Ruδ+/Ru0 ratio increases to 2.2 for Ru/R–TiO2–air–H2, indicating increased interfacial contacts. This trend can be reproduced on other Ru/R–TiO2 catalysts of varied R–TiO2 supports and loading ratios of Ru (Supplementary Figs. 9 and 10, Supplementary Table 1). In contrast, Ruδ+/Ru0 ratio decreases to 0.2 for Ru/A–TiO2–air–H2, and Ruδ+ state is very weak. The opposite trends of Ruδ+/Ru0 ratio also agree with their opposite catalytic performances.
Another feature of Ru/A–TiO2–air–H2 lies in the shift of Ru0-3d5/2 from 280.5 to 279.4 eV, a binding energy even lower than that of Ru foil (280.1 eV, Supplementary Fig. 11). This phenomenon is usually ascribed to the occurrence of SMSI effect46. This can also be supported by the catalytic performances. For CO2 hydrogenation, SMSI and catalyst sizes can highly affect activity and selectivity5,21,22,45. Both SMSI effect and size reduction can convert the product from CH4 to CO43,47,48. For example, Paraskevi reported that 3-nm Ru NPs showed the highest turnover frequency (TOF) on TiO2, and bigger NPs favor CH45. In our system, pre-annealing increases the size of Ru NPs on A–TiO2 from 2.1 nm to 2.8 nm, but the activity and CH4 selectivity both dramatically decrease. The results indicate that SMSI effect should account for this change, because SMSI can generally decrease the activity and selectivity of CO2 methanation18. For example, for Rh/TiO2, adsorbates can induce SMSI to decrease the activity and selectivity for CO2 methanation; Ru/TiO2 also shows SMSI effect in reduction reactions18,49,50. Therefore, varied interfacial compatibilities can vary MSI modes of Ru/TiO2–air–H2 catalysts: Ru/R–TiO2–air–H2 shows enhanced interfacial coupling bridged by RuOx layers, while Ru/A–TiO2–air–H2 shows SMSI effect.
Surface atomic states of Ru NPs probed with CO-DRIFTS
Different MSI modes can vary the exposed surface atomic states of Ru NPs. We characterize surface Ru sites using diffuse-reflectance infrared Fourier transform spectroscopy (DRIFTS) at 25 °C with CO as the probe (Fig. 4). Figure 4a schemes the possible adsorption configurations of CO on Ru/TiO2. For Ru/A–TiO2–H2, 2140 and 2080 cm−1 result from multi-carbonyl-adsorption modes of CO on Ru sites with low coordination numbers (Ru(CO)x, x = 2, 3), while the modes appear at 2138 cm−1 and 2075 cm−1 for Ru/R–TiO2–H251. The broad peaks from 1900 to 2070 cm−1 result from top-absorption modes of CO on Ru NPs (Ru–CO) and at the interface between Ru and TiO2 (Ruif–CO)41,42. For Ru/R–TiO2–air–H2, the proportion of 1950 derived from Ruif–CO was higher than directly reduced, agreeing with the XPS results. The bands at 2030 and 2015 cm−1 belong to CO linearly adsorbed on Ru surfaces with A–TiO2 and R–TiO2 (Ru–CO), while the peak at 2105 cm−1 on A–TiO2 corresponds to Ru nanoclusters (Run+–CO). These Ru nanoclusters catalyze the formation of CO (<2%) above 240 °C (Fig. 1c).
After pre-annealing, the total intensities decrease, and the relative intensities of the peaks also change. In particular, Ru(CO)x mode on low coordinated Ru (RuLC) sites decreases for Ru/R–TiO2–air–H2, while it almost disappears for Ru/A–TiO2–air–H2. These changes mean that the ratios of RuLC sites decrease after annealing. Usually, the ratio of low coordinated surface atoms decreases with increased particle sizes. The result of Ru/A–TiO2–air–H2 is consistent with this trend, as shown by increased sizes of Ru NPs from 2.1 nm to 2.8 nm (Supplementary Fig. 7). The projected sizes of Ru NPs for Ru/R–TiO2–air–H2 and Ru/R–TiO2–H2 are 2.4 ± 0.4 nm and 2.5 ± 0.5 nm (Supplementary Fig. 7), which are almost the same. This suggests that the decreased ratios of RuLC sites did not result from increased particle sizes. TEM images show that Ru NPs display flatter shapes for Ru/R–TiO2–air–H2, owing to the stronger affinity of Ru with rutile supports bridged by interfacial RuOx species (Fig. 2d–f). The greater curvature radius of flatter particles can lead to more ordered arrangement of surface atoms and increase the number of surrounding atoms. We also characterize the metal dispersion of Ru NPs with CO pulse adsorption. Metal dispersions of Ru (Dco) are 33.6% and 31.1% for Ru/R–TiO2–air–H2 and Ru/R–TiO2–H2, respectively, which are almost the same (Supplementary Table 2). The results thus indicate that size and surface area does not play critical roles in enhancing the catalytic performances of Ru NPs on R–TiO2 supports. Therefore, such different surface atomic configurations of Ru nanoparticles directly modify their catalytic performances in CO2 methanation.
Reaction mechanisms probed with operando FTIR
We further use operando Fourier transform-infrared spectroscopy (operando FTIR) to reveal CO2 hydrogenation mechanisms on these Ru/TiO2 catalysts (Fig. 5). The measurements were performed from 25 to 300 °C in 20-sccm gas flows of 60 vol%H2/15 vol%CO2/25 vol% Ar. For Ru/R–TiO2 (Fig. 5a), the absorptions at 3017 and 1303 cm−1 result from C to H bonds of CH4, 1436 and 1560 cm−1 from adsorbed formate species (*HCO2), 1950, 2075 from adsorbed CO species (*CO), and 1360 cm−1 from adsorbed carbonate (*HCO3)5,41,42,52.
At 25 °C, both Ru/R–TiO2–air–H2 and Ru/R–TiO2–H2 show weak adsorption peaks, but the reactions can obviously occur above 160 °C. At 160 °C, *HCO3, *HCO2, and *CO appear on both catalysts, meaning CO2 is first activated as *HCO3 and *HCO2, and further reduced into *CO8,41. The difference lies in 2075 and 3017 cm−1. At 160 °C, CH4 appears on Ru/R–TiO2–air–H2 but not on Ru/R–TiO2–H2, indicating that Ru/R–TiO2–air–H2 is more active for CO2 methanation. This agrees with the enhanced catalytic results. Many researches have figured out that *CO and formate are two possible intermediates in thermal CO2 hydrogenation reactions. In our results, stepwise-increasing reaction temperatures can lead to similar changes of *HCO2 on the two samples (Fig. 5a). This suggests that formate is not likely the intermediate, or at least not linked to the distinctly different activity on rutile. Some previous reports have also concluded that *CO hydrogenation is the rate-determining step in the CO2 hydrogenation on Ru/TiO241. Two catalysts show different adsorption modes of *CO, more obvious multicarbonyl Ru(CO)n species at 2070 cm−1 on Ru/R–TiO2–H2, which adsorbed on RuLC of the surfaces of Ru nanoparticles. It is inactive at low temperatures because H2 cannot effectively reduce it at low temperatures51.
For Ru/A–TiO2 catalysts, the modes at 3014 cm−1 result from C to H bonds of CH4, 2008 cm−1 from *CO, and 1352 cm−1 from *HCO3 (Fig. 5b). This suggests that the reaction routes are the same with Ru/R–TiO2, agreeing with our catalytic results in fixed-bed reactors. While the reversal peaks at 1644 and 1344 cm−1 originate from the desorption of *H2O, *OH, and *H on catalyst surfaces during the reaction53. For Ru/A–TiO2–air–H2, the intensity at 2008 cm−1 sharply drops compared with Ru/A–TiO2–air–H2, in line with CO-DRIFTS results in Fig. 4c. The highly decreased Ru–CO mode at 2008 cm−1 indicates reduced exposure of surface Ru sites, which further supports the occurrence of SMSI, and agrees with the low catalytic activity (Fig. 1b). While SMSI effect can effectively convert the product from CH4 into CO12,17,18,48.
Discussion
Our results show that rutile and anatase TiO2 supports can dramatically modify the morphology, surface atomic configuration, MSI mode, and catalytic performances of Ru catalysts for CO2 hydrogenation reaction, although they share the same chemical compositions. Ru NPs adhere stronger with R–TiO2 than A–TiO2, which disagrees with the trend that more active surface-oxygen atoms lead to stronger interfacial adhesion28,29. Instead, this confirms that interfacial compatibility plays critical roles in controlling the metal-support adhesion strength and MSI modes of Ru/TiO2 catalysts.
For Ru/TiO2 catalysts, RuO2 shares the same lattice structure with R–TiO2, thus, annealing RuCl3–R–TiO2 precursor can incorporate Ru atoms into the surface lattices of R–TiO2 to form epitaxial RuOx species (Fig. 6a, b). Such interfacial RuOx species can act as anchoring layers to strongly bind Ru nanoparticles onto R–TiO2 supports, which yields flat shapes with low contact angles and larger curvatures. This morphology can decrease the ratio of undercoordinated surface sites (Fig. 6a), and further modifies CO adsorptions and reaction routes. At the atomic scale, Ruδ+ atoms can adequately occupy Ti sites, thus bonding to R–TiO2 substrate with maximized bonding strength and density. While Ru0 atoms of Ru NPs can further bond to such Ruδ+ sites through Ru–Ru metallic bonds (Fig. 6b). This kind of binding features can minimize interfacial defects of strain, dislocation, and vacancies, thus can further highly enhance metal-support adhesion and suppress interfacial phase separation.
The lattice type of A–TiO2 (I41/amd) is different from that of RuO2 (P42/mnm), thus their interfacial atomic configurations do not match. Such misfit interfaces can form defects like edge dislocation and vacancy, which reduces interfacial adhesion and stability. Figure 6c–d schemes atomic interfacial contact of Ru nanoparticle on A–TiO2 (101) surface. Some Ru atoms bond to surface oxygen atoms through Ru–O bonds, but their bonding lengths and strengths vary, depending on their atomic positions. Moreover, some atoms cannot effectively bond to surface-oxygen atoms due to misfit positions, and these sites form dislocations (Fig. 6d). Therefore, Ru NPs weakly adhere A–TiO2 surfaces, and appear as spherical particles. This morphology yields more undercoordinated Ru sites. While the higher reducibility of surface-oxygen atoms can further drive the occurrence of SMSI effect, which further modifies CO adsorption, catalytic activity, and selectivity.
In summary, we have demonstrated that interfacial compatibility can critically control the interfacial coupling strength, surface atomic configurations, MSI modes, and catalytic performances of Ru/TiO2 catalysts by varying interfacial adhesion strengths. For CO2 methanation, enhanced interfacial coupling of Ru/R–TiO2 can increase catalytic activity and CH4 selectivity, while SMSI effect on Ru/A–TiO2 can highly decrease catalytic activity and convert the product from CH4 into CO. This is because Ru NPs can strongly adhere to R–TiO2 supports and form flat particles with larger curvatures, in which interfacial RuOx species act as anchoring layers with R–TiO2; while Ru/A–TiO2 show classic SMSI effect due to lattice misfit and higher reducibility of surface oxygen atoms. Therefore, interfacial compatibility is a critical structural feature that can intrinsically modulate MSI modes and catalytic performances, which might be realized through designing interfacial atomic configurations, introducing anchoring layers, thermal annealing, and oxidation treatments. This work paves the way to improve catalytic performances through engineering interfacial compatibilities between metal NPs and supports.
Methods
Chemicals
Titanium tetrachloride (TiCl4, 99.5%), titanium trichloride (15.0~20% TiCl3 basis in 30% HCl), TiO2 (anatase, 25 nm), sodium hydroxide (NaOH, >98%), and sodium chloride (NaCl, 99.6%) were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Ethanol (>99.7%), hydrochloric acid (HCl, 36–38%), and nitric acid (HNO3, 68%) were purchased from Sinopharm Chemical Reagent Beijing Co., Ltd. Ruthenium trichloride hydrate (RuCl3·xH2O, 37.5–41% Ru) and titanium nitride (TiN, 99%) were purchased from Beijing Innochem Science & Technology Co., LTD.
Syntheses of rutile-type TiO2 nanorods
R–TiO2 nanorods were prepared using three methods:
-
(1)
TiCl4 (2.5 mL) was slowly added into ice water (30 mL) under vigorous stirring. After being stirred for 15 min, the solution was transferred into a 40-mL Teflon-lined stainless-steel autoclave. Then the solution was heated at 170 °C for 14 h in an oven. After cooling down to room temperature, the products were collected by centrifugation and washed 4 times with deionized water, and dried at 60 °C for 12 h54.
-
(2)
TiCl3 (5 mL) was first mixed with 30 mL of 1.0 M NaCl solution. After being transferred into a 40 mL Teflon-lined stainless-steel autoclave, the solution was heated at 200 °C for 6 h.
-
(3)
TiN (0.5 g) was dispersed into 30 mL of 4.0 M HNO3 solution, then was transferred into a 40 mL Teflon-lined stainless-steel autoclave. The mixture was heated at 180 °C for 24 h55.
Syntheses of Ru/TiO2 catalysts
All Ru/TiO2 catalysts were prepared following the same procedure, in which only TiO2 supports were changed. (1) TiO2 supports were annealed in air at 500 °C for 10 h to stabilize their surfaces. (2) Then 1.0 g of the annealed TiO2 supports and a certain amount of RuCl3 were mixed in 10 mL of H2O under sonication for 30 min. (3) The mixtures were rapidly frozen with liquid nitrogen, and further dried in a freeze drier. (4) As-obtained powders were calcined at 400 °C for 4 h in air or directly reduced with H2 to prepare Ru/TiO2 catalysts.
Characterization
Power X-ray diffraction (XRD) data were collected on a Bruker D8 diffractometer using Cu Kα radiation (1.5418 Å), which was operated at 40 kV and 40 mA with a scanning rate of 6 degree/min. High-resolution transmission electron microscopy (HRTEM), high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM), and element mapping were performed on FEI Tecnai F30 transmission electron microscope (TEM) under an acceleration voltage of 300 kV. Size distributions of Ru nanoparticles were obtained through measuring at least 100 particles. X-ray photoelectron spectroscopy (XPS) data were collected on Thermo Scientific ESCALAB 250Xi system using Al Kα line as the X-ray source. The spectra were calibrated by with C1s peak at 284.8 eV.
H2-TPR
H2 temperature-programmed reduction (H2-TPR) and CO pulse adsorption were performed on Autochem1 II 2920 instrument. Before TPR measurement, the samples were annealed in Ar at 300 °C for 60 min, then cooled down to 50 °C. The signals were recorded online with a thermal conductivity detector (TCD), as the reactor was heated to 800 °C at a heating rate of 10 °C /min under 10% H2–90% Ar flows.
DRIFT
In situ diffuse-reflectance infrared Fourier transform (DRIFT) spectra of CO adsorption were performed on Thermo Fisher Nicolet iS50 with a resolution of 4 cm−1 at 25 °C. Prior to CO adsorption, the sample was treated in H2 flow at 300 °C for 1 h and then cooled down. Prior to collecting the background spectrum, the sample was purged with Ar for 30 min. Then 5% CO/Ar flow (20 sccm) was introduced into the reactor until saturated adsorptions. DRIFT spectra were collected until no gas-phase CO could be detected with Ar purging.
Operando FT-IR study of reaction mechanisms
The measurements were performed on Thermo Fisher Nicolet iS50. Prior to collecting the spectra, the samples were treated in H2 flow at 300 °C for 1 h and then purging with Ar for 1 h. The background spectrum was collected, until the reactor cooling down to 25 °C. Subsequently, the feed gas (n(H2):n(CO2):n(Ar)=15:60:25) was introduced, then the IR spectra were collected at certain temperatures after being stabilized for 30 min.
Catalytic test of CO2 hydrogenation
CO2 hydrogenation reaction was carried out in a fixed-bed reactor made of stainless steel. First, the catalysts were in situ reduced at 300 °C for 1 h with 25-sccm pure H2 before catalytic testing. After cooling down to 100 °C, the gas was switched to the reaction gas with molar ratio of n(H2):n(CO2):n(Ar) = 60:15:25. The reaction pressure was controlled at 1 atm, and the gas hourly space velocities (GHSVs) were 12,000 mL·g−1·h−1. The outgassing gas compositions were detected using an online gas chromatography (GC) equipped with a TCD detector after the reactions were stabilized for 25 min at specific temperatures.
The CO2 conversion (XCO2) was calculated according to the following equation:
where nin(CO2) and nout(CO2) refer to the molar number of the CO2 before or after the reaction, respectively, the Ain(CO2) and Ain(Ar) refer to the chromatographic peak areas of the CO2 and Ar in the reaction gas, and the Aout(CO2) and Aout(Ar) refer to the GC area after reactions.
The reaction rate (v) was calculated following:
where GHSVs refer to the gas hourly space velocities and mRu refers to the mass of the Ru. Under this condition, the products are CO and CH4, so the selectivity of CO and CH4 (SCO and SCH4) meets
where the fCO/CH4 refers to the relative correction factors of CO–CH4 obtained by the calibrating gas; the ACO and ACH4 refer to the chromatographic peak areas of CO and CH4, respectively.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The data supporting the findings of the study are available within the paper and its Supplementary Information. Source data are provided with this paper in excel format. Source data are provided with this paper.
References
van Deelen, T. W., Hernández Mejía, C. & de Jong, K. P. Control of metal-support interactions in heterogeneous catalysts to enhance activity and selectivity. Nat. Catal. 2, 955–970 (2019).
Zhou, W. et al. New horizon in C1 chemistry: breaking the selectivity limitation in transformation of syngas and hydrogenation of CO2 into hydrocarbon chemicals and fuels. Chem. Soc. Rev. 48, 3193–3228 (2019).
Liu, L. & Corma, A. Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles. Chem. Rev. 118, 4981–5079 (2018).
Matsubu, J. C., Yang, V. N. & Christopher, P. Isolated metal active site concentration and stability control catalytic CO2 reduction selectivity. J. Am. Chem. Soc. 137, 3076–3084 (2015).
Panagiotopoulou, P. Hydrogenation of CO2 over supported noble metal catalysts. Appl. Catal. A 542, 63–70 (2017).
Chen, S. et al. Morphology-engineered highly active and stable Ru/TiO2 catalysts for selective CO methanation. Angew. Chem. Int. Ed. 58, 10732–10736 (2019).
Zhu, Y. et al. Selective activation of C–OH, C–O–C, or C═C in furfuryl alcohol by engineered Pt sites supported on layered double oxides. ACS Catal. 10, 8032–8041 (2020).
Zhang, J., Deo, S., Janik, M. J. & Medlin, J. W. Control of molecular bonding strength on metal catalysts with organic monolayers for CO2 reduction. J. Am. Chem. Soc. 142, 5184–5193 (2020).
Macino, M. et al. Tuning of catalytic sites in Pt/TiO2 catalysts for the chemoselective hydrogenation of 3-nitrostyrene. Nat. Catal. 2, 873–881 (2019).
Neumann, S. et al. Effects of particle size on strong metal–support interactions using colloidal “Surfactant-Free” Pt nanoparticles supported on Fe3O4. ACS Catal. 10, 4136–4150 (2020).
Mitchell, S., Qin, R. X., Zheng, N. F. & Perez-Ramirez, J. Nanoscale engineering of catalytic materials for sustainable technologies. Nat. Nanotechnol. 16, 129–139 (2021).
Kattel, S., Liu, P. & Chen, J. G. Tuning selectivity of CO2 hydrogenation reactions at the metal/oxide interface. J. Am. Chem. Soc. 139, 9739–9754 (2017).
Ro, I., Resasco, J. & Christopher, P. Approaches for understanding and controlling interfacial effects in oxide-supported metal catalysts. ACS Catal. 8, 7368–7387 (2018).
Zhang, J. et al. Wet-chemistry strong metal-support interactions in Titania-supported Au catalysts. J. Am. Chem. Soc. 141, 2975–2983 (2019).
Li, S. et al. Tuning the selectivity of catalytic carbon dioxide hydrogenation over Iridium/Cerium oxide catalysts with a strong metal-support interaction. Angew. Chem. Int. Ed. 56, 10761–10765 (2017).
Xu, J. et al. Influence of pretreatment temperature on catalytic performance of rutile TiO2-supported ruthenium catalyst in CO2 methanation. J. Catal. 333, 227–237 (2016).
Li, J. et al. Enhanced CO2 methanation activity of Ni/anatase catalyst by tuning strong metal–support interactions. ACS Catal. 9, 6342–6348 (2019).
Matsubu, J. C. et al. Adsorbate-mediated strong metal-support interactions in oxide-supported Rh catalysts. Nat. Chem. 9, 120–127 (2017).
Du, X. et al. Size-dependent strong metal-support interaction in TiO2 supported Au nanocatalysts. Nat. Commun. 11, 5811 (2020).
Zhang, Y. et al. Structure sensitivity of Au-TiO2 strong metal-support interactions. Angew. Chem. Int. Ed. 60, 12074–12081 (2021).
Vogt, C. et al. Unravelling structure sensitivity in CO2 hydrogenation over Nickel. Nat. Catal. 1, 127–134 (2018).
Parastaev, A. et al. Boosting CO2 hydrogenation via size-dependent metal–support interactions in Cobalt/Ceria-based catalysts. Nat. Catal. 3, 526–533 (2020).
Chen, A. et al. Structure of the catalytically active Copper–Ceria interfacial perimeter. Nat. Catal. 2, 334–341 (2019).
Melaet, G. et al. Evidence of highly active cobalt oxide catalyst for the fischer-tropsch synthesis and CO2 hydrogenation. J. Am. Chem. Soc. 136, 2260–2263 (2014).
Yao, Y. et al. Engineering the electronic structure of single atom Ru sites via compressive strain boosts acidic water oxidation electrocatalysis. Nat. Catal. 2, 304–313 (2019).
Cargnello, M. et al. Control of metal nanocrystal size reveals metal-support interface role for ceria catalysts. Science 341, 771–773 (2013).
Suchorski, Y. et al. The role of metal/oxide interfaces for long-range metal particle activation during CO oxidation. Nat. Mater. 17, 519–522 (2018).
Tosoni, S. & Pacchioni, G. Trends in adhesion energies of Gold on MgO(100), Rutile TiO2(110), and CeO2(111) surfaces: a comparative DFT study. J. Phys. Chem. C 121, 28328–28338 (2017).
Hemmingson, S. L. & Campbell, C. T. Trends in adhesion energies of metal nanoparticles on oxide surfaces: understanding support effects in catalysis and nanotechnology. ACS Nano 11, 1196–1203 (2017).
Farmer, J. A. & Campbell, C. T. Ceria maintains smaller metal catalyst particles by strong metal-support bonding. Science 329, 933–936 (2010).
Wan, Q., Hu, S., Dai, J., Chen, C. & Li, W.-X. First-principles kinetic study for ostwald ripening of late transition metals on TiO2(110). J. Phys. Chem. C 123, 1160–1169 (2018).
Zhu, B., Qi, R., Yuan, L. & Gao, Y. Real-time atomistic simulation of the Ostwald ripening of TiO2 supported Au nanoparticles. Nanoscale 12, 19142–19148 (2020).
Hernandez Mejia, C., van Deelen, T. W. & de Jong, K. P. Activity enhancement of cobalt catalysts by tuning metal-support interactions. Nat. Commun. 9, 4459 (2018).
Freakley, S. J. et al. Palladium-Tin catalysts for the direct synthesis of H2O2 with high selectivity. Science 351, 965–968 (2016).
Campbell, C. T. & Starr, D. E. Metal adsorption and adhesion energies on MgO(100). J. Am. Chem. Soc. 124, 9212–9218 (2002).
O’Connor, N. J., Jonayat, A. S. M., Janik, M. J. & Senftle, T. P. Interaction trends between single metal atoms and oxide supports identified with density functional theory and statistical learning. Nat. Catal. 1, 531–539 (2018).
Saadun, A. J. et al. Epitaxially directed Iridium nanostructures on Titanium dioxide for the selective hydrodechlorination of dichloromethane. ACS Catal. 10, 528–542 (2019).
O’Sullivan, M. et al. Interface control by chemical and dimensional matching in an oxide heterostructure. Nat. Chem. 8, 347–353 (2016).
Xiang, G. et al. Ultrathin 2D nanolayer of RuO2 effectively enhances charge separation in the photochemical processes of TiO2. Small 11, 4469–4474 (2015).
Sakpal, T. & Lefferts, L. Structure-dependent activity of CeO2 supported Ru catalysts for CO2 methanation. J. Catal. 367, 171–180 (2018).
Li, X. et al. Controlling CO2 hydrogenation selectivity by metal-supported electron transfer. Angew. Chem. Int. Ed. 59, 19983–19989 (2020).
Chen, S. et al. Raising the COx methanation activity of a Ru/gamma-Al2O3 catalyst by activated modification of metal-support interactions. Angew. Chem. Int. Ed. 59, 22763–22770 (2020).
Su, X., Yang, X. F., Huang, Y., Liu, B. & Zhang, T. Single-atom catalysis toward efficient CO2 conversion to CO and formate products. Acc. Chem. Res. 52, 656–664 (2019).
Xiang, G., Shi, X., Wu, Y., Zhuang, J. & Wang, X. Size effects in atomic-level epitaxial redistribution process of RuO2 over TiO2. Sci. Rep. 2, 801 (2012).
Guo, Y. et al. Low-temperature CO2 methanation over CeO2-supported Ru single atoms, nanoclusters, and nanoparticles competitively tuned by strong metal–support interactions and H-spillover effect. ACS Catal. 8, 6203–6215 (2018).
Xu, M. et al. Insights into interfacial synergistic catalysis over Ni@TiO2-x catalyst toward water-gas shift reaction. J. Am. Chem. Soc. 140, 11241–11251 (2018).
Chen, X. et al. Theoretical Insights and the corresponding construction of supported metal catalysts for highly selective CO2 to CO conversion. ACS Catal. 7, 4613–4620 (2017).
Aitbekova, A. et al. Low-temperature restructuring of CeO2-supported Ru nanoparticles determines selectivity in CO2 catalytic reduction. J. Am. Chem. Soc. 140, 13736–13745 (2018).
Zhang, Y. et al. Tuning reactivity of Fischer-Tropsch synthesis by regulating TiOx overlayer over Ru/TiO2 nanocatalysts. Nat. Commun. 11, 3185 (2020).
Zhang, Y. et al. Ru/TiO2 catalysts with size-dependent metal/support interaction for tunable reactivity in Fischer–Tropsch synthesis. ACS Catal., 10, 12967–12975 (2020).
Loveless, B. T., Buda, C., Neurock, M. & Iglesia, E. CO chemisorption and dissociation at high coverages during CO hydrogenation on Ru catalysts. J. Am. Chem. Soc. 135, 6107–6121 (2013).
Abdel-Mageed, A. M., Widmann, D., Olesen, S. E., Chorkendorff, I. & Behm, R. J. Selective CO methanation on highly active Ru/TiO2 catalysts: identifying the physical origin of the observed activation/deactivation and loss in selectivity. ACS Catal. 8, 5399–5414 (2018).
Panayotov, D. A. & John T. Yates, J. Spectroscopic detection of hydrogen atom spillover from Au nanoparticles supported on TiO2: use of conduction band electrons. J. Phys. Chem. C 11, 2959–2964 (2007).
Li, L. et al. Sub-10 nm rutile titanium dioxide nanoparticles for efficient visible-light-driven photocatalytic hydrogen production. Nat. Commun. 6, 5881 (2015).
Zhao, Z. et al. Effect of defects on photocatalytic activity of rutile TiO2 nanorods. Nano Res. 8, 4061–4071 (2015).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21801012 to G.X.) and the National Key Research and Development Program of China (2018YFA0702002 to L.W.).
Author information
Authors and Affiliations
Contributions
G.X. initiated, designed, and supervised this work, and wrote the manuscript. J.Z. prepared the materials and performed catalytic tests. Z.G. carried out DRIFTS experiments. T.Z., Z.L. and W.Z. helped with preparing the materials and catalytic experiments. G.X., X.L., and L.W. supported this research. All the authors discussed the results and commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Leon Lefferts and the other, anonymous, reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhou, J., Gao, Z., Xiang, G. et al. Interfacial compatibility critically controls Ru/TiO2 metal-support interaction modes in CO2 hydrogenation. Nat Commun 13, 327 (2022). https://doi.org/10.1038/s41467-021-27910-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-021-27910-4
This article is cited by
-
Thermally stable Ni foam-supported inverse CeAlOx/Ni ensemble as an active structured catalyst for CO2 hydrogenation to methane
Nature Communications (2024)
-
Enhancing alkyne semi-hydrogenation through engineering metal-support interactions of Pd on oxides
Nano Research (2024)
-
Recent Progress on the Hydrodeoxygenation of Lignin-Derived Pyrolysis Oil Using Ru-Based Catalysts
Korean Journal of Chemical Engineering (2024)
-
Highly active Ru/TiO2 nanostructures for total catalytic oxidation of propane
Environmental Science and Pollution Research (2023)
-
Enriching the surface oxygen as efficient anchoring site of highly dispersed Ru for enhanced hydrogenolysis activity
Nano Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.