Abstract
Reverse intersystem crossing (RISC), the uphill spin-flip process from a triplet to a singlet excited state, plays a key role in a wide range of photochemical applications. Understanding and predicting the kinetics of such processes in vastly different molecular structures would facilitate the rational material design. Here, we demonstrate a theoretical expression that successfully reproduces experimental RISC rate constants ranging over five orders of magnitude in twenty different molecules. We show that the spin flip occurs across the singlet–triplet crossing seam involving a higher-lying triplet excited state where the semi-classical Marcus parabola is no longer valid. The present model explains the counterintuitive substitution effects of bromine on the RISC rate constants of previously unknown molecules, providing a predictive tool for material design.
Similar content being viewed by others
Introduction
Electronic spin-flip processes in molecular excited states have attracted increasing interest for optoelectronics1,2,3, photocatalytic synthesis4,5,6, and biomedical applications7,8,9. A relevant example is reverse intersystem crossing (RISC), the uphill transition of a non-emissive triplet excited state to an emissive singlet excited state. This process leads to E-type delayed fluorescence, also known as thermally activated delayed fluorescence (TADF), and allows an internal charge-to-photon conversion efficiency of nearly 100% in organic light-emitting diodes10. Although materials have typically been discovered experimentally, a fundamental understanding of RISC kinetics and strategy for predicting the rate constants may open vast opportunities for theory-driven materials discovery.
RISC kinetics are often considered in the framework of Marcus theory11,12,13,14. If the spin–orbit coupling HSO between the initial triplet and final singlet excited states is weak, meaning that the spin-flip only occurs on the crossing seam between their potential energy surfaces (PESs) (Fig. 1a), the RISC rate constant (kRISC) follows a Marcus-like nonadiabatic expression:
where ћ is the reduced Planck constant, kB is the Boltzmann constant, T is the temperature, λ is the reorganization energy, and EA is the activation energy to reach the crossing seam. In the case of simple parabolic PESs with equal force constants, which is a crucial assumption of Marcus theory, EA can be analytically expressed as
with ΔEST as the adiabatic singlet–triplet energy difference. A key implication of Eqs. (1) and (2) is that kRISC can be predicted from the equilibrium geometries, which correspond to the easily computable local minima on the PESs of the initial triplet state and final singlet state. However, this understanding of RISC becomes more complicated if the spin-flip process involves an energetically higher-lying excited state as an intermediate15,16,17,18,19 (Fig. 1b). Since Eq. (2) does not include information on the key intermediate involved in the actual spin-flip process, recent calculations using the equilibrium geometries only provided qualitative justification of the experimental kRISC for a handful of TADF molecules20. Herein, we explicitly compute singlet–triplet crossing seams to quantitatively predict kRISC for vastly different structures both from the literature and previously unknown molecules. Rigorous comparisons to experimental data reported over the last decade allowed a general understanding of the RISC kinetics governed by the singlet–triplet crossing seam involving a higher-lying triplet excited state.
Results
Computation of kRISC
To understand and predict the RISC kinetics, we first focused on twenty different TADF molecules reported in the literature (Fig. 2a). These molecules are characterized as donor–acceptor systems in which electron-rich donor units, aryl amines, are covalently bound to electron-deficient acceptor units, such as heterocycles, aryl nitriles, ketones, boranes, sulfones, alkynes, or phosphine oxides. We collected literature values for the steady-state and transient photoluminescence data of these molecules and estimated their kRISC values from differential rate equations of the population densities of their singlet and triplet excited states (see Supplementary Information Section 1 for details). The experimental kRISC values varied substantially, by five orders of magnitude, from 102 to 107 s–1.
Directly computing kRISC from Eq. (1) requires the minimum-energy seam of the crossing (MESX), the energetically most accessible geometry on the singlet–triplet crossing seam hypersurface21,22, as well as equilibrium excited-state geometries. To obtain the MESX for each molecule, we employed a constrained optimization algorithm using the gradient projection method23, which minimizes the mean energy of the singlet and triplet states (ES + ET)/2 while simultaneously fulfilling the crossing condition of the energy difference ES − ET = 0. ES and ET were calculated at the level of time-dependent density functional theory (TDDFT) within the Tamm–Dancoff approximation24 (see the Methods for details).
For the MESX geometries of the twenty molecules shown in Fig. 2a, TDDFT predicts nonzero HSO of 0.17–3.61 cm–1 with fairly small EA of 0.11–0.32 eV, corresponding to kRISC of 102–107 s–1 calculated using Eq. (1) at T of 300 K. Figure 2b compares the theoretical kRISC values to the experimental values, demonstrating that the present model successfully reproduces the experimental rates. The mean absolute logarithmic error (MALE) reaches only 0.23, whereas a larger MALE of 1.2, corresponding to an error of 1.2 orders of magnitude, is observed for the values based on the conventional model shown in Fig. 1a and the parabolic approximation of Eq. (2) (see Supplementary Fig. 1 for the errors for each molecule). These results thus demonstrate the importance of the explicit computation of the singlet–triplet crossing seams for quantitatively predicting kRISC.
Mechanism of the RISC
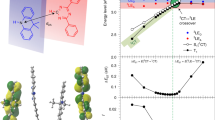
Closer inspection of the data further reveals that the lowest singlet excited state (S1) does not cross the lowest triplet state (T1) and instead crosses the higher-lying triplet state (T2) at the obtained MESX geometry, consistent with the model shown in Fig. 1b. This feature explains the larger errors for the parabolic approximation, which does not account for any higher-lying excited states. We attribute the uncrossed S1 and T1 to a nonzero exchange interaction between the singlet and triplet states, which leads to T1 always lying below S1 if the two states have the same electronic configuration25. In accordance with El-Sayed’s rule26, a large change in the orbital angular momentum between S1 and T2 consisting of different electronic configurations induces an effective HSO and thus enables spin flipping via the MESX. These results are consistent with the RISC picture anticipated based on recent theoretical and experimental studies using ACRXTN16 and 4CzIPN19. It must be stressed that S1–T2 MESX is present in every molecule examined in this quantitative study despite their wide variety of excited-state electronic configurations, including intramolecular charge transfer (CT) states and locally excited (LE) states of π–π* and n–π* on either donor or acceptor units, illustrating the generality of RISC via S1–T2 crossing in organic donor–acceptor molecules.
Prediction of the effects of bromination on kRISC
To further validate the present RISC model of Eq. (1), we computed kRISC of brominated analogues of representative TADF materials ACRXTN and 3-PXZ-XO: 3-(2,7-dibromo-9,9-dimethylacridan-10-yl)xanthone (Br-ACRXTN) and 3-(3,7-dibromo-phenoxazin-10-yl)xanthone (Br-3-PXZ-XO) (Fig. 3a, b). Although heavy halogen atoms such as bromine are well known to induce large HSO and thus facilitate ISC27, the calculations predict that the electrophilic bromination of ACRXTN counterintuitively decreases kRISC from 1.3 × 106 s–1 to 7.1 × 105 s–1. In contrast, the bromination of 3-PXZ-XO leads to a more than hundredfold increase in kRISC from 2.7 × 105 s–1 to 4.2 × 107 s–1. Indeed, subsequent synthesis and characterization confirm the predicted opposite trend; the brominations of ACRXTN and 3-PXZ-XO caused the experimental kRISC to decrease from 1.0 × 106 s–1 to 8.7 × 105 s–1 and to increase from 1.7 × 105 s–1 to 2.6 × 107 s–1, respectively (see Supplementary Fig. 2 and Table S1 for details). To the best of our knowledge, kRISC of over 107 s–1 for Br-3-PXZ-XO is the highest value ever reported for an organic TADF material28. This high kRISC reflects its fast transient photoluminescence decay with a delayed fluorescence lifetime of 490 ns (Fig. 3c), which is considerably shorter than typical values of several microseconds29. We also note that both brominated molecules exhibit similar blueshifts in their broad, unstructured CT emissions compared to the corresponding nonbrominated analogues (Fig. 3d), and this shift is attributed to the electron-withdrawing effects of the bromine atoms on the donor units, destabilizing the CT states between the donor and acceptor units (i.e., increasing the energy of the CT states).
a, b Synthetic routes to Br-ACRXTN (a) and Br-3-PXZ-XO (b). c, d Transient photoluminescence decays (c) and steady-state photoluminescence spectra (d) of ACRXTN, Br-ACRXTN, 3-PXZ-XO, and Br-3-PXZ-XO in a solid-state host matrix, 2,8-bis(diphenylphosphoryl)dibenzo[b,d]furan (PPF), at a doping concentration of 5 wt%.
The notable retardation of kRISC by bromination of ACRXTN is due to a decrease in HSO from 0.88 cm–1 to 0.72 cm–1 at the S1–T2 MESX geometries. This counterintuitive substitution effect of bromine on HSO can be rationalized by two factors. First, the S1–T2 spin flipping in Br-ACRXTN is compensated by a smaller change in the orbital angular momentum than that in ACRXTN (Fig. 4a, b). This is due to an increase in the occupation of the CT state in T2 from 28% to 57% upon bromination, which leads to both S1 and T2 having similar CT states with small HSO according to El-Sayed’s rule. Additionally, the resulting change in the orbital angular momentum of Br-ACRXTN involves the n orbital of the carbonyl oxygen on the acceptor unit rather than bromine on the donor unit (Fig. 4b), suggesting that the heavy atom effect plays a minor role in determining HSO between S1 and T2. In contrast, Br-3-PXZ-XO has a perceivable contribution from the bromine atom to the orbital angular momentum change between S1 of the CT state and T2 of the LE π–π* state on the donor unit (Fig. 4c, d). Such circumstances are indeed consistent with the heavy atom effect of bromine being responsible for the increase in HSO from 1.2 cm–1 to 3.5 cm–1 and thus for the high kRISC, over 107 s–1, in Br-3-PXZ-XO.
Discussion
Figure 5 displays the impact of varying HSO and EA on kRISC. While the existing organic TADF molecules exhibit kRISC smaller than 108 s–1, the theory predicts that even kRISC of 109 s–1, corresponding to a time constant of 1.0 ns, can be achieved with HSO less than 10 cm–1; for example, HSO of 7.7 cm–1 for EA of 0.10 eV and HSO of 2.9 cm–1 for EA of 0.05 eV at T of 300 K. These HSO are an order of magnitude smaller than those of iridium-containing phosphors and could be achieved by exploiting heavy atom effects of nonmetals in periods 3 and 430,31. However, we have shown that such heuristic approaches sometimes lead to the retardation of HSO, in part because of their more pronounced effects on the excited-state electronic configurations at the S1–T2 MESX geometries. Thus, for material design, a priori computational screening is essential, and the RISC model presented here allows for it.
In summary, we have presented a RISC kinetic model that successfully predicts the experimental rates for a wide variety of organic TADF molecules. Our results suggest that explicitly computing the crossing seam between the singlet and triplet excited states leads to more reliable predictions than those obtained by the conventional approach using the Marcus parabolic approximation because the RISC in these molecules involves higher-lying triplet excited states. The presented model is thus a viable tool for theory-driven materials discovery with a relevant exemplar exhibiting a high kRISC of 2.6 × 107 s–1.
We envisage that further computational screenings of vast chemical space will facilitate the discovery of materials exploiting the spin-flipping process for various photochemical applications. We also anticipate the possible existence of materials that may not follow the model and provide platforms to discover spin-flipping mechanisms different from the presented one.
Methods
Computation
The geometries of the singlet–triplet MESXs, where the square energy difference (ES − ET)2 and the mean energy (ES + ET)/2 are minimized, were obtained by the gradient projection method23 using a composed gradient vector G for the nuclear coordinates Q:
where
In Eq. (3), the first term contains the difference gradient vector v to minimize the energy difference. The second term is responsible for minimizing the mean energy, while the projection matrix P ensures the orthogonality between the two terms of the composed gradient vector. The excited-state energy and gradient were calculated using linear-response TDDFT with the LC-BLYP functional32 and the 6–31+G(d) basis set within the Tamm–Dancoff approximation24. The range-separated parameters for the LC-BLYP functional were non-empirically optimized for each molecule to incorporate a reasonable amount of exact exchange33,34. The geometry optimization of the MESXs was performed with the GRRM17 program35, which refers to the energy and gradient calculated by the Gaussian 16 program36. The HSO values were calculated perturbatively using the Breit–Pauli spin–orbit Hamiltonian with an effective charge approximation implemented in the PySOC program37 interfaced to the Gaussian 16 program. The EA values were calculated as the electronic energy difference between the MESX and the equilibrium T1. The λ values were calculated as the difference between the T1 electronic energies at the equilibrium T1 and S1 geometries.
Chemical synthesis
The synthetic procedures and characterization data of the compounds are detailed in Supplementary Information Section 1.
Photoluminescence measurements
Steady-state photoluminescence spectra were acquired using a Fluoromax-4 spectrophotometer (HORIBA) with 370 nm photoexcitation from a Xe arc lamp. Transient photoluminescence decay measurements were performed by time-correlated single photon counting under a flow of N2 using a Fluorolog-3 fluorescence lifetime spectrometer (HORIBA) with a 370 nm LED excitation source. The absolute PL quantum yields were determined under a flow of N2 using a C9920 integrating sphere system (Hamamatsu Photonics). The method for determining the experimental kRISC values is detailed in Supplementary Information Section 1.
Data availability
The data that support the findings of this study available in this published Article and its Supplementary Information, or from the corresponding authors upon reasonable request.
References
Baldo, M. A. et al. Highly efficient phosphorescent emission from organic electroluminescent devices. Nature395, 151–154 (1998).
Rao, A. et al. The role of spin in the kinetic control of recombination in organic photovoltaics. Nature500, 435–439 (2013).
Einzinger, M. et al. Sensitization of silicon by singlet exciton fission in tetracene. Nature571, 90–94 (2019).
Theriot, J. C. et al. Organocatalyzed atom transfer radical polymerization driven by visible light. Science352, 1082–1086 (2016).
Lim, C.-H. et al. Intramolecular charge transfer and ion pairing in N,N-diaryl dihydrophenazine photoredox catalysts for efficient organocatalyzed atom transfer radical polymerization. J. Am. Chem. Soc.139, 348–355 (2017).
Singh, V. K. et al. Highly efficient organic photocatalysts discovered via a computer-aided-design strategy for visible-light-driven atom transfer radical polymerization. Nat. Catal.1, 794–804 (2018).
Henderson, B. W. & Dougherty, T. J. How does photodynamic therapy work? Photochem. Photobiol.55, 145–157 (1992).
Xiong, X. et al. Thermally activated delayed fluorescence of fluorescein derivative for time-resolved and confocal fluorescence imaging. J. Am. Chem. Soc.136, 9590–9597 (2014).
Zhen, X. et al. Ultralong phosphorescence of water-soluble organic nanoparticles for in vivo afterglow imaging. Adv. Mater.29, 1606665 (2017).
Uoyama, H., Goushi, K., Shizu, K., Nomura, H. & Adachi, C. Highly efficient organic light-emitting diodes from delayed fluorescence. Nature492, 234–238 (2012).
Marcus, R. A. Electron transfer reactions in chemistry: theory and experiment (Nobel lecture). Angew. Chem. Int. Ed.32, 1111–1121 (1993).
Samanta, P. K., Kim, D., Coropceanu, V. & Brédas, J.-L. Up-conversion intersystem crossing rates in organic emitters for thermally activated delayed fluorescence: impact of the nature of singlet vs triplet excited states. J. Am. Chem. Soc.139, 4042–4051 (2017).
Olivier, Y. et al. Nature of the singlet and triplet excitations mediating thermally activated delayed fluorescence. Phys. Rev. Mater.1, 075602 (2017).
Olivier, Y., Sancho-Garcia, J.-C., Muccioli, L., D’Avino, G. & Beljonne, D. Computational design of thermally activated delayed fluorescence materials: the challenges ahead. J. Phys. Chem. Lett.9, 6149–6163 (2018).
Chen, X.-K., Zhang, S.-F., Fan, J.-X. & Ren, A.-M. Nature of highly efficient thermally activated delayed fluorescence in organic light-emitting diode emitters: nonadiabatic effect between excited states. J. Phys. Chem. C.119, 9728–9733 (2015).
Marian, C. M. Mechanism of the triplet-to-singlet upconversion in the assistant dopant ACRXTN. J. Phys. Chem. C.120, 3715–3721 (2016).
Gibson, J., Monkman, A. P. & Penfold, T. J. The importance of vibronic coupling for efficient reverse intersystem crossing in thermally activated delayed fluorescence molecules. ChemPhysChem17, 2956–2961 (2016).
Etherington, M. K., Gibson, J., Higginbotham, H. F., Penfold, T. J. & Monkman, A. P. Revealing the spin–vibronic coupling mechanism of thermally activated delayed fluorescence. Nat. Commun.7, 13680 (2016).
Noda, H. et al. Critical role of intermediate electronic states for spin-flip processes in charge-transfer-type organic molecules with multiple donors and acceptors. Nat. Mater.18, 1084–1090 (2019).
Park, I. S., Matsuo, K., Aizawa, N. & Yasuda, T. High-performance dibenzoheteraborin-based thermally activated delayed fluorescence emitters: molecular architectonics for concurrently achieving narrowband emission and efficient triplet–singlet spin conversion. Adv. Funct. Mater.28, 1802031 (2018).
Koga, N. & Morokuma, K. Determination of the lowest energy point on the crossing seam between two potential surfaces using the energy gradient. Chem. Phys. Lett.119, 371–374 (1985).
Harvey, J. N. Understanding the kinetics of spin-forbidden chemical reactions. Phys. Chem. Chem. Phys.9, 331–343 (2007).
Bearpark, M. J., Robb, M. A. & Schlegel, H. B. A direct method for the location of the lowest energy point on a potential surface crossing. Chem. Phys. Lett.223, 269–274 (1994).
Hirata, S. & Head-Gordon, M. Time-dependent density functional theory within the Tamm–Dancoff approximation. Chem. Phys. Lett.314, 291–299 (1999).
Slater, J. C. The theory of complex spectra. Phys. Rev.34, 1293–1322 (1929).
El-Sayed, M. Spin–orbit coupling and the radiationless processes in nitrogen heterocyclics. J. Chem. Phys.38, 2834–2838 (1963).
Turro, N. J. Modern Molecular Photochemistry (University Science Books, 1991).
Wada, Y., Nakagawa, H., Matsumoto, S., Wakisaka, Y. & Kaji, H. Molecular design realizing very fast reverse intersystem crossing in purely organic emitter. Preprint at https://doi.org/10.26434/chemrxiv.9745289 (2019).
Liu, Y., Li, C., Ren, Z., Yan, S. & Bryce, M. R. All-organic thermally activated delayed fluorescence materials for organic light-emitting diodes. Nat. Rev. Mater.3, 18020 (2018).
de Sa Pereira, D. et al. The effect of a heavy atom on the radiative pathways of an emitter with dual conformation, thermally-activated delayed fluorescence and room temperature phosphorescence. J. Mater. Chem. C.7, 10481–10490 (2019).
Drummond, B. H. et al. Selenium substitution enhances reverse intersystem crossing in a delayed fluorescence emitter. J. Phys. Chem. C.124, 6364–6370 (2020).
Iikura, H., Tsuneda, T., Yanai, T. & Hirao, K. A long-range correction scheme for generalized-gradient-approximation exchange functionals. J. Chem. Phys.115, 3540–3544 (2001).
Baer, R., Livshits, E. & Salzner, U. Tuned range-separated hybrids in density functional theory. Annu. Rev. Phys. Chem.61, 85–109 (2010).
Sun, H., Zhong, C. & Bredas, J.-L. Reliable prediction with tuned range-separated functionals of the singlet–triplet gap in organic emitters for thermally activated delayed fluorescence. J. Chem. Theory Comput.11, 3851–3858 (2015).
Maeda, S. et al. Implementation and performance of the artificial force induced reaction method in the GRRM17 program. J. Comput. Chem.39, 233–251 (2018).
Frisch, M. J. et al. Gaussian 16 Rev. C.01. (Wallingford, CT).
Gao, X. et al. Evaluation of spin-orbit couplings with linear-response time-dependent density functional methods. J. Chem. Theory Comput.13, 515–524 (2017).
Acknowledgements
This work was supported by JST PRESTO (Grant No. JPMJPR17N1 for N.A. and JPMJPR16N8 for Y.H.), Grant-in-Aid for JSPS KAKENHI Grant (No. JP20K15252 for N.A.) and JST-ERATO (Grant No. JPMJER1903 for S.M. and Y.H.). The computations were partially performed at the computer cennter of Kyoto University and the HOKUSAI system at RIKEN.
Author information
Authors and Affiliations
Contributions
N.A. and Y.H. performed the theoretical calculations. N.A. synthesized the compounds and characterized their photoluminescence properties. S.M. and Y.-J.P. supervised the project. All authors contributed to the discussion, writing, and editing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review informationNature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aizawa, N., Harabuchi, Y., Maeda, S. et al. Kinetic prediction of reverse intersystem crossing in organic donor–acceptor molecules. Nat Commun 11, 3909 (2020). https://doi.org/10.1038/s41467-020-17777-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-17777-2
This article is cited by
-
A figure of merit for efficiency roll-off in TADF-based organic LEDs
Nature (2024)
-
Highly efficient multi-resonance thermally activated delayed fluorescence material toward a BT.2020 deep-blue emitter
Nature Communications (2024)
-
Understanding of complex spin up-conversion processes in charge-transfer-type organic molecules
Nature Communications (2024)
-
Confining donor conformation distributions for efficient thermally activated delayed fluorescence with fast spin-flipping
Nature Communications (2023)
-
Dielectric control of reverse intersystem crossing in thermally activated delayed fluorescence emitters
Nature Materials (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.