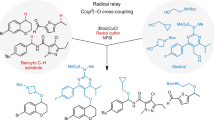
Abstract
Construction of C–C bonds via alkoxy radical-mediated remote C(sp3)–H functionalization is largely unexplored, as it is a formidable challenge to directly generate alkoxy radicals from alcohols due to the high bond dissociation energy (BDE) of O–H bonds. Disclosed herein is a practical and elusive metal-free alcohol-directed heteroarylation of remote unactivated C(sp3)–H bonds. Phenyliodine bis(trifluoroacetate) (PIFA) is used as the only reagent to enable the coupling of alcohols and heteroaryls. Alkoxy radicals are readily generated from free alcohols under the irradiation of visible light, which trigger the regioselective hydrogen-atom transfer (HAT). A wide range of functional groups are compatible with the mild reaction conditions. Two unactivated C–H bonds are cleaved and one new C–C bond is constructed during the reaction. This protocol provides an efficient strategy for the late-stage functionalization of alcohols and heteroaryls.
Similar content being viewed by others
Introduction
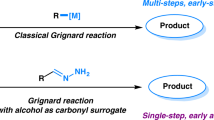
Direct functionalization of unactivated C(sp3)–H bonds represents one of the most intriguing and advanced technologies in synthetic chemistry but is still facing enormous challenges with reactivity and selectivity1,2,3,4. Radical-mediated hydrogen-atom transfer (HAT) renders an efficient entry to cleave the inert C(sp3)–H bonds that allows for subsequent substitutions5,6,7,8. Besides the classic Hofmann–Löffler reaction, recently new breakthroughs mediated by N-centered radicals have been achieved with the photoredox catalysis9,10,11,12,13,14,15,16,17,18,19. Alcohols are important and readily available chemicals. The radical-mediated late-stage functionalization of the C–H bonds of alcohols affords an ideal approach to the preparation of complex alcohol derivatives. According to the bond dissociation energy (BDE)20,21,22, the C–H bonds proximal to hydroxyl group are in higher reactivity than the distal ones (Fig. 1a)23,24,25,26,27,28. Therefore, the selective functionalization of the less reactive remote C–H bonds in alcohols is a formidable challenge.
The HAT process triggered by alkoxy radicals has long been developed in order to gain the good chemo-/regio-selectivities29,30. After several decades, however, this method is still underexplored, which is mainly ascribed to the difficult homolysis of the alcoholic O–H bonds with high BDEs (ca. 105 kcal mol−1) to generate alkoxy radicals from free alcohols. On the other hand, alkoxy radicals are prone to induce the β-C–C bond fragmentation under harsh reaction conditions31,32,33,34,35,36,37,38,39. Consequently, alcohols are often elaborated to other surrogates, e.g., nitrite esters40,41,42, peroxy compounds43,44,45,46, sulfonates47,48,49, lead(IV) alkoxides50,51,52, hypohalites53,54,55,56,57,58,59, N-alkoxylpyridine-2-thiones60,61, and N-alkoxyphthalimides62,63,64, for the formation of alkoxy radicals by heat or ultraviolet irradiation. Nevertheless, these compounds are sometimes hard to handle or synthesize. In this scenario, seeking a general and mild strategy to generate alkoxy radicals from free alcohols is highly desirable.
Recently, we disclosed a tertiary-alcohol-directed heteroarylation of remote C(sp3)–H bonds by a sequence of HAT and intramolecular heteroaryl migrations (Fig. 1b)65. Alkoxy radicals were directly obtained from alcohols in the presence of iridium complex irradiated by visible light. The tertiary alcohol substrates were well designed to suit for the intramolecular mode. Soon after, Zuo et al. reported a CeCl3-catalyzed amination of remote sp3 C–H bonds of alcohols66. Only primary alcohols were applied to generate alkoxy radicals (Fig. 1c). Concerning the values and ubiquity of heteroaryl moieties in drugs and bioactive molecules, the intermolecular heteroarylation of alcohols is more significant and valuable than the intramolecular mode. Herein we report our findings in the regioselectively intermolecular heteroarylation of alcohols to furnish the Minisci-type products. This reaction demonstrates a wide scope of both alcohols and heteroaryls. Notably, all types (1°, 2°, and 3°) of alcohols are apt to afford the alkoxy radicals that trigger the regioselective heteroarylation of C(sp3)–H bonds. The metal-free conditions are mild and operationally simple, providing a practical strategy for the late-stage functionalization of alcohols and heteroaryls (Fig. 1d).
Results
Reaction conditions survey
With 4-methyl quinoline 1a and n-pentanol 2a as model substrates, we set about investigating the reaction parameters (Table 1). After a brief survey, it was found that the only use of stoichiometric amounts of hypervalent iodine(III) reagent phenyliodine bis(trifluoroacetate) (PIFA) could promote the reaction under the irradiation of blue light-emitting diodes (LEDs) (entry 1). While varying the light source to compact fluorescent light (CFL) bulb decreased the yield (entry 2), the reaction did not proceed with the use of green LEDs or in dark (entry 3). The reaction also took place at 50 °C without light irradiation albeit in a low yield (entry 4), suggesting that this is not a photocatalytic process and the light may just input energy into the reaction. Increasing the amount of PIFA slightly improved the outcome (entry 5), but using too much PIFA significantly inhibited the reaction (entry 6). Other hypervalent iodine(III) reagents were also examined. Surprisingly, the use of (diacetoxyiodo)benzene (PIDA) almost turned off the reaction (entry 8). The use of 1,2-dichloroethane (DCE) or MeCN instead of dichloromethane (DCM) delivered comparable yields (entries 9 and 10), but other solvents did not work efficiently (entries 11–14). The Suárez’s conditions (PIDA and I2)56,57,58,59, which were often applied to the intramolecular cyclization reactions via the generation of alkoxy radicals from hypohalite intermediates, were not suitable for our reaction (entry 15). This result clearly illustrated that the current reaction underwent a non-trivial pathway rather than the formation of hypohalite intermediates. The yield was further elevated to 90% by treating the reaction with high-intensive blue LEDs (entry 16). However, it was found that the reaction yield was going down along with reducing the amount of alcohols (entries 17–20).
Scope of heteroaryls and alcohols
With the optimized reaction conditions in hand, we turned to examine the generality of protocol (Fig. 2). Firstly, a variety of heteroaryls were tested. The electronic properties of heteroaryls did not have much influence on the reaction, as both electron-donating (e.g., Me, OMe) and electron-withdrawing (e.g., Cl, CN, CO2Et) groups were well tolerated (3b–3f). While the reaction could take place at both the ortho- and para-positions of quinoline (3g) or pyridine (3k), the reaction of isoquinoline solely preferred the 1-position (3h–3j). The example of 3i was noteworthy, since the presence of bromide reserved a platform for the late-stage products manipulation by cross-coupling reactions. The conversion of N,N-bidentate ligand dtbpy to 3n provided a valuable tactic for the ligand modification. Other heteroaryls, such as phenanthridine (3o), acridine (3p), pyrazine (3q–3s), pyrimidine (3t), quinoxaline (3u), and 4-hydroxyquinazoline (3v) were also proved to be suitable substrates, indicating a broad scope of heteroaryls. While the pyrazine product 3r was formed along with another regio-isomer (~10%), the product 3s was delivered in a unique regioselectivity, which was determined by the heteronuclear multiple bond correlation (HMBC) analysis. Significantly, the method could be applied to complex molecules, such as voriconazole and eszopiclone, affording the corresponding products in good regioselectivities (3w and 3x). Next, a number of alcohols were investigated. The alkoxy radical-mediated 1,5-HAT exclusively occurred even in the presence of more reactive benzylic C–H bonds (3z), showing the outstanding regioselective control. It might be attributed to that the 1,5-HAT via six-membered cyclic transition state is more kinetically favorable in this reaction. A variety of Minisci-type products were readily furnished. It should be noted that the current Minisci reactions involving C(sp3)–H activation are largely dependent upon the inherent BDEs of C–H bonds, and scarcely discuss about the regioselective control67,68,69,70. The olefin moiety, which is generally susceptive to radical process, remained intact in the reaction (3aa). The C–H bonds adjacent to heteroatoms were also readily functionalized (3ab and 3ac). Remarkably, the reaction with cyclic C–H bonds proceeded stereoselectively, leading to the thermodynamically favored trans-product (3ad). In contrast, the reaction with linear C–H bonds delivered the diastereomer mixtures in a 2:1 or 1:1 ratio (3ae and 3af). In addition to primary and secondary C–H bonds, the congested tertiary C–H bonds were also readily transformed (3ag and 3ah), forming the new quaternary all-carbon centers. The applicability was further spread from primary to secondary and tertiary alcohols. Both of them were suitable precursors for the generation of alkoxy radicals to accomplish the distal C–H bond heteroarylation (3ai–3al). This method provides an efficient approach for alcohol derivatization. For instance, the heteroaryl groups could be directly introduced to the δ-position of cycloalkanols (3am and 3an). The regioselectivities were unambiguously determined by the HMBC experiments.
Mechanistic studies
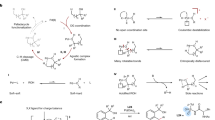
To shed light on the mechanistic pathways, a set of experiments were carried out. Replacement of the hydroxyl group by ether entirely inhibited the reaction, verifying that the reaction was enabled by free alcohols (Fig. 3a). Mixing 2a with PIFA in d-chloroform immediately led to a metastable intermediate 4, which was speculated by crude 1H NMR. Then irradiating the mixture with blue LEDs resulted in the formation of 2a and trifluoroacetate thereof presumably via the alkoxy radical intermediate (Fig. 3b). The intermediate 4 displayed weak absorption from 420 to 450 nm, suggesting the possibility of energy transfer from blue LEDs to 4 (Fig. 3c). Finally, the radical clock experiment unambiguously provided support for the proposed radical pathway (Fig. 3d). The PIFA-promoted reaction of 1a with 5 afforded the ring-opened product 6 in 70% yield as a 4:1 mixture of E and Z isomers.
The proposed mechanism was depicted on the basis of experimental observations (Fig. 4). Initially, the mixture of 2a and PIFA results in the dialkoxyiodo benzene 4. Homolysis of 4 induced by visible-light irradiation leads to the alkoxy radical I that triggers the subsequent 1,5-HAT to generate the alkyl radical II. Meanwhile, PhI and iodanyl radical are cogenerated in the reaction. Nucleophilic addition of II to the quinoline salt III affords the radical cation IV, which is then single-electron oxidized by excess PIFA or the in situ formed iodanyl radical to afford the final product 3a.
Discussion
In summary, we have described a practical and metal-free protocol of alcohol-directed remote C(sp3)–H functionalization. The combinational use of PIFA and visible-light irradiation offers a non-trivial and mild tactic for the direct generation of alkoxy radicals from free alcohols. This strategy is expected to significantly facilitate the alkoxy radical-mediated transformations. A vast array of heteroaryls and alcohols have proven to be suitable substrates. The protocol makes a complement to the classic Minisci reactions, and may find wide use in medicinal synthesis owing to the easy operation and metal-free conditions.
Methods
General procedure for heteroarylation of remote C(sp3)–H bonds
Heteroaryl 1 (0.4 mmol) and alcohol 2 (2.0 mmol) were loaded in a reaction vial, which was subjected to evacuation/flushing with N2 three times. Then DCM (2.0 mL) followed by PIFA (0.92 mmol) was added to the mixture. The reaction was irradiated with 100 W blue LEDs and kept at room temperature (rt) under fan cooling. After the reaction completion monitored by TLC, the mixture was quenched by addition of aq. KOH until pH > 8 and then extracted with ethyl acetate (3 × 10 mL). The combined organic extracts were washed by brine, dried over Na2SO4, filtered, concentrated, and purified by flash column chromatography on silica gel (eluent: ethyl acetate/petroleum ether) to give the desired product 3.
Data availability
The authors declare that all other data supporting the findings of this study are available within the article and Supplementary Information files, and also are available from the corresponding author on reasonable request.
Change history
05 September 2018
This Article was originally published without the accompanying Peer Review File. This file is now available in the HTML version of the Article; the PDF was correct from the time of publication.
References
Chen, X., Engle, K. M., Wang, D. H. & Yu, J. Q. Palladium(II)-catalyzed C–H activation/C–C cross-coupling reactions: versatility and practicality. Angew. Chem. Int. Ed. 48, 5094–5115 (2009).
Lyons, T. W. & Sanford, M. S. Palladium-catalyzed ligand-directed C–H functionalization reactions. Chem. Rev. 110, 1147–1169 (2010).
Newhouse, T. & Baran, P. S. If C–H bonds could talk: selective C–H bond oxidation. Angew. Chem. Int. Ed. 50, 3362–3374 (2011).
Yamaguchi, J., Yamaguchi, A. D. & Itami, K. C–H bond functionalization: emerging synthetic tools for natural products and pharmaceuticals. Angew. Chem. Int. Ed. 51, 8960–9009 (2012).
Robertson, J., Pillai, J. & Lush, R. K. Radical translocation reactions in synthesis. Chem. Soc. Rev. 30, 94–103 (2001).
Chiba, S. & Chen, H. sp3 C–H oxidation by remote H-radical shift with oxygen- and nitrogen-radicals: a recent update. Org. Biomol. Chem. 12, 4051–4060 (2014).
Hu, X. Q., Chen, J. R. & Xiao, W. J. Controllable remote C–H bond functionalization by visible-light photocatalysis. Angew. Chem. Int. Ed. 56, 1960–1962 (2017).
Yan, M., Lo, J. C., Edwards, J. T. & Baran, P. S. Radicals: reactive intermediates with translational potential. J. Am. Chem. Soc. 138, 12692–12714 (2016).
Choi, G. J., Zhu, Q., Miller, D. C., Gu, C. J. & Knowles, R. R. Catalytic alkylation of remote C–H bonds enabled by proton-coupled electron transfer. Nature 539, 268–271 (2016).
Chu, J. C. K. & Rovis, T. Amide-directed photoredox-catalysed C–C bond formation at unactivated sp3 C–H bonds. Nature 539, 272–275 (2016).
Chen, D.-F., Chu, J. C. K. & Rovis, T. Directed γ-C(sp3)–H alkylation of carboxylic acid derivatives through visible light photoredox catalysis. J. Am. Chem. Soc. 139, 14897–14900 (2017).
Shu, W. & Nevado, C. Visible-light-mediated remote aliphatic C–H functionalizations through a 1,5-hydrogen transfer cascade. Angew. Chem. Int. Ed. 56, 1881–1884 (2017).
Dauncey, E. M., Morcillo, S. P., Douglas, J. J., Sheikh, N. S. & Leonori, D. Photoinduced remote functionalisations by iminyl radical promoted C–C and C–H bond cleavage cascades. Angew. Chem. Int. Ed. 57, 744–748 (2018).
Jiang, H. & Studer, A. α-Aminoxy-acid-auxiliary-enabled intermolecular radical γ-C(sp3)–H functionalization of ketones. Angew. Chem. Int. Ed. 57, 1692–1696 (2018).
Yoon, T. P., Ischay, M. A. & Du, J. Visible light photocatalysis as a greener approach to photochemical synthesis. Nat. Chem. 2, 527–532 (2010).
Narayanam, J. M. R. & Stephenson, C. R. J. Visible light photoredox catalysis: applications in organic synthesis. Chem. Soc. Rev. 40, 102–113 (2011).
Xuan, J. & Xiao, W. J. Visible-light photoredox catalysis. Angew. Chem. Int. Ed. 51, 6828–6838 (2012).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Xuan, J., Zhang, Z. G. & Xiao, W. J. Visible-light-induced decarboxylative functionalization of carboxylic acids and their derivatives. Angew. Chem. Int. Ed. 54, 15632–15641 (2015).
Laarhoven, L. J. J., Mulder, P. & Wayner, D. D. M. Determination of bond dissociation enthalpies in solution by photoacoustic calorimetry. Acc. Chem. Res. 32, 342–349 (1999).
Blanksby, S. J. & Ellison, G. B. Bond dissociation energies of organic molecules. Acc. Chem. Res. 36, 255–263 (2003).
Xue, X.-S., Ji, P., Zhou, B. & Cheng, J.-P. The essential role of bond energetics in C–H activation/functionalization. Chem. Rev. 117, 8622–8648 (2017).
Yi, H. et al. Recent advances in radical C–H activation/radical cross-coupling. Chem. Rev. 117, 9016–9085 (2017).
Guo, S., Kumar, P. S. & Yang, M. Recent advances of oxidative radical cross-coupling reactions: direct α-C(sp3)–H bond functionalization of ethers and alcohols. Adv. Synth. Catal. 359, 2–25 (2017).
Zhang, S.-Y., Tu, Y.-Q., Fan, C.-A., Zhang, F.-M. & Shi, L. Iron-catalyzed C(sp3)–C(sp3) bond formation through C(sp3)–H functionalization: a cross-coupling reaction of alcohols with alkenes. Angew. Chem. Int. Ed. 48, 8761–8765 (2009).
He, T., Yu, L., Zhang, L., Wang, L. & Wang, M. Direct C2-alkylation of azoles with alcohols and ethers through dehydrogenative cross-coupling under metal-free conditions. Org. Lett. 13, 5016–5019 (2011).
Zhou, W. et al. Metal-free oxidative functionalization of C(sp3)–H bond adjacent to oxygen and radical addition to olefins. Org. Lett. 17, 1160–1163 (2015).
Jeffrey, J. L., Terrett, J. A. & MacMillan, D. W. C. O–H hydrogen bonding promotes H-atom transfer from α C–H bonds for C-alkylation of alcohols. Science 349, 1532–1536 (2015).
Hartung, J. Stereoselective construction of the tetrahydrofuran nucleus by alkoxyl radical cyclizations. Eur. J. Org. Chem. 2001, 619–632 (2001).
Čeković, Ž. Reactions of δ-carbon radicals generated by 1,5-hydrogen transfer to alkoxyl radicals. Tetrahedron 59, 8073–8090 (2003).
Ren, R. & Zhu, C. Radical-mediated ring-opening functionalization of cyclobutanols: a shortcut to γ-substituted ketones. Synlett 27, 1139–1144 (2016).
Wu, X. & Zhu, C. Recent advances in ring-opening functionalization of cycloalkanols by C–C σ-bond cleavage. Chem. Rec. 18, 587–598 (2018).
Zhao, H., Fan, X., Yu, J. & Zhu, C. Silver-catalyzed ring-opening strategy for the synthesis of β- and γ-fluorinated ketones. J. Am. Chem. Soc. 137, 3490–3493 (2015).
Ren, R., Zhao, H., Huan, L. & Zhu, C. Manganese-catalyzed oxidative azidation of cyclobutanols: regiospecific aynthesis of alkyl azides by C–C bond cleavage. Angew. Chem. Int. Ed. 54, 12692–12696 (2015).
Ren, R., Wu, Z., Xu, Y. & Zhu, C. C–C bond-forming strategy by manganese-catalyzed oxidative ring-opening cyanation and ethynylation of cyclobutanol derivatives. Angew. Chem. Int. Ed. 55, 2866–2869 (2016).
Jia, K., Zhang, F., Huang, H. & Chen, Y. Visible-light-induced alkoxyl radical generation enables selective C(sp3)–C(sp3) bond cleavage and functionalizations. J. Am. Chem. Soc. 138, 1514–1517 (2016).
Yayla, H. G., Wang, H., Tarantino, K. T., Orbe, H. S. & Knowles, R. R. Catalytic ring-opening of cyclic alcohols enabled by PCET activation of strong O–H bonds. J. Am. Chem. Soc. 138, 10794–10797 (2016).
Guo, J. et al. Photocatalytic C–C bond cleavage and amination of cycloalkanols by cerium(III) chloride complex. Angew. Chem. Int. Ed. 55, 15319–15322 (2016).
Jia, K., Pan, Y. & Chen, Y. Selective carbonyl-C(sp3) bond cleavage to construct ynamides, ynoates, and ynones by photoredox catalysis. Angew. Chem. Int. Ed. 56, 2478–2481 (2017).
Barton, D. H. R., Beaton, J. M., Geller, L. E. & Pechet, M. M. A new photochemical reaction. J. Am. Chem. Soc. 82, 2640–2641 (1960).
Barton, D. H. R. & Beaton, J. M. A synthesis of aldosterone acetate. J. Am. Chem. Soc. 82, 2641 (1960).
Barton, D. H. R., Beaton, J. M., Geller, L. E. & Pechet, M. M. A new photochemical reaction. J. Am. Chem. Soc. 83, 4076–4083 (1961).
Čeković, Ž. & Green, M. M. Formation of remote double bonds by ferrous sulfate–cupric acetate promoted decomposition of alkyl hydroperoxides. J. Am. Chem. Soc. 96, 3000–3002 (1974).
Čeković, Ž., Dimitrijević, L., Djokić, G. & Srnić, T. Remote functionalisation by ferrous ion–cupric ion induced decomposition of alkyl hydroperoxides. Tetrahedron 35, 2021–2026 (1979).
Čeković, Ž. & Ĉvetković, M. Functionallization of the δ-carbon atom by the ferrous ion induced decomposition of alkyl hydroperoxides in the presence of cupric salts. Tetrahedron Lett. 23, 3791–3794 (1982).
Kundu, R. & Ball, Z. T. Copper-catalyzed remote sp3 C–H chlorination of alkyl hydroperoxides. Org. Lett. 12, 2460–2463 (2010).
Beckwith, A. L. J., Hay, B. P. & Williams, G. M. Generation of alkoxyl radicals from O-alkyl benzenesulphenates. J. Chem. Soc. Chem. Commun. 0, 1202–1203 (1989).
Čeković, Ž. & Petrović, G. Alkylation of remote non-activated δ-carbon atoms: addition of δ-carbon radicals, generated by 1,5-hydrogen transfer in alkoxy radical intermediates, to activated olefins. Tetrahedron 55, 1377–1390 (1999).
Petrović, G., Saičić, R. N. & Čeković, Ž. Synthesis of acetyl scopine. Intramolecular reactions of N-carbethoxy nortropine-3α-benzenesulfenate. Synlett. 1999, 635–637 (1999).
Heusler, K. & Kalvoda, J. Intramolecular free-radical reactions. Angew. Chem. Int. Ed. Engl. 3, 525–538 (1964).
Mihailović, M. L. & Čeković, Ž. Intramolecular oxidative cyclization of alcohols with lead tetraacetate. Synthesis. 1970, 209–224 (1970).
Kalvoda, J. & Heusler, K. Die Hypojodit-reaktion (Verfahren zur intramolekularen substitution an nicht-aktivierten C-Atomen). Synthesis. 1971, 501–526 (1971).
Walling, C. R. & Padwa, A. Positive halogen compounds. VII. Intramolecular chlorinations with long chain hypochlorites. J. Am. Chem. Soc. 85, 1597–1601 (1963).
Walling, C. & Bristol, D. delta-Chloro alcohols and tetrahydrofurans from primary and secondary alkyl hypochlorites. J. Org. Chem. 37, 3514–3516 (1972).
Walling, C. R. & Clark, T. Reactions of primary and secondary alkoxy radicals derived from hypochlorites. J. Am. Chem. Soc. 96, 4530–4534 (1974).
Concepción, J. I., Francisco, C. G., Hernández, R., Salazar, J. A. & Suárez, E. Intramolecular hydrogen abstraction. Iodosobenzene diacetate, an efficient and convenient reagent for alkoxy radical generation. Tetrahedron Lett. 25, 1953–1956 (1984).
Martín, A., Salazar, J. A. & Suárez, E. Synthesis of chiral spiroacetals from carbohydrates. Tetrahedron Lett. 36, 4489–4492 (1995).
Martín, A., Salazar, J. A. & Suárez, E. Synthesis of chiral spiroacetals from carbohydrates. J. Org. Chem. 61, 3999–4006 (1996).
Martín, A., Pérez-Martín, I. & Suárez, E. Intramolecular hydrogen abstraction promoted by amidyl radicals. Evidence for electronic factors in the nucleophilic cyclization of ambident amides to oxocarbenium ions. Org. Lett. 7, 2027–2030 (2005).
Beckwith, A. L. J. & Hay, B. P. Generation of alkoxy radicals from N-alkoxypyridinethiones. J. Am. Chem. Soc. 110, 4415–4416 (1988).
Hartung, J. & Gallou, F. Ring closure reactions of substituted 4-pentenyl-1-oxy radicals. The stereoselective synthesis of functionalized disubstituted tetrahydrofurans. J. Org. Chem. 60, 6706–6716 (1995).
Kim, S., Lee, T. A. & Song, Y. Facile generation of alkoxy radicals from N-alkoxyphthalimides. Synlett. 1998, 471–472 (1998).
Zhang, J., Li, Y., Zhang, F., Hu, C. & Chen, Y. Generation of alkoxyl radicals by photoredox catalysis enables selective C(sp3)–H functionalization under mild reaction conditions. Angew. Chem. Int. Ed. 55, 1872–1875 (2016).
Wang, C., Harms, K. & Meggers, E. Catalytic asymmetric C–H functionalization under photoredox conditions by radical translocation and stereocontrolled alkene addition. Angew. Chem. Int. Ed. 55, 13495–13498 (2016).
Wu, X. et al. Tertiary-alcohol-directed functionalization of remote C(sp3)–H bonds by sequential hydrogen atom and heteroaryl migrations. Angew. Chem. Int. Ed. 57, 1640–1644 (2018).
Hu, A. et al. δ-Selective functionalization of alkanols enabled by visible-light-induced ligand-to-metal charge transfer. J. Am. Chem. Soc. 140, 1612–1616 (2018).
Antonchick, A. P. & Burgmann, L. Direct selective oxidative cross-coupling of simple alkanes with heteroarenes. Angew. Chem. Int. Ed. 52, 3267–3271 (2013).
Jin, J. & MacMillan, D. W. C. Direct α-arylation of ethers through the combination of photoredox-mediated C–H functionalization and the Minisci reaction. Angew. Chem. Int. Ed. 54, 1565–1569 (2015).
Quattrini, M. C. et al. Versatile cross-dehydrogenative coupling of heteroaromatics and hydrogen donors via decatungstate photocatalysis. Chem. Commun. 53, 2335–2338 (2017).
Liu, W., Yang, X., Zhou, Z.-Z. & Li, C.-J. Simple and clean photo-induced methylation of heteroarenes with MeOH. Chem 2, 688–702 (2017).
Acknowledgements
C.Z. is grateful for the financial support from Soochow University, the National Natural Science Foundation of China (21722205), the Project of Scientific and Technologic Infrastructure of Suzhou (SZS201708), and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Contributions
X.W. and C.Z. conceived and designed the experiments; X.W. carried out most of the experiments; X.W., H.Z., N.T., Z.W., D.W., M.J., Y.X., M.W. and C.Z. analyzed the data; C.Z. wrote the paper; C.Z. directed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, X., Zhang, H., Tang, N. et al. Metal-free alcohol-directed regioselective heteroarylation of remote unactivated C(sp3)–H bonds. Nat Commun 9, 3343 (2018). https://doi.org/10.1038/s41467-018-05522-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-05522-9
This article is cited by
-
Photoinduced generation of alkyl and phthalimide nitrogen radicals from N-hydroxyphthalimide esters for the synthesis of benzophenone-type bioisosteres
Science China Chemistry (2024)
-
Tuning the reactivity of alkoxyl radicals from 1,5-hydrogen atom transfer to 1,2-silyl transfer
Nature Communications (2021)
-
Photo-mediated selective deconstructive geminal dihalogenation of trisubstituted alkenes
Nature Communications (2020)
-
Site-selective remote C(sp3)–H heteroarylation of amides via organic photoredox catalysis
Nature Communications (2019)
-
Visible light-induced direct α C–H functionalization of alcohols
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.