Abstract
T-lineage committed precursor thymocytes are screened by a fate-determination process mediated via T cell receptor (TCR) signals for differentiation into distinct lineages. However, it remains unclear whether any antecedent event is required to couple TCR signals with the transcriptional program governing lineage decisions. Here we show that Bcl11b, known as a T-lineage commitment factor, is essential for proper expression of ThPOK and Runx3, central regulators for the CD4-helper/CD8-cytotoxic lineage choice. Loss of Bcl11b results in random expression of these factors and, thereby, lineage scrambling that is disconnected from TCR restriction by MHC. Initial Thpok repression by Bcl11b prior to the pre-selection stage is independent of a known silencer for Thpok, and requires the last zinc-finger motif in Bcl11b protein, which by contrast is dispensable for T-lineage commitment. Collectively, our findings shed new light on the function of Bcl11b in priming lineage-specifying genes to integrate TCR signals into subsequent transcriptional regulatory mechanisms.
Similar content being viewed by others
Introduction
Early thymocyte progenitors (ETPs) retain their developmental potential to become non-T- lymphoid cells, but, upon exposure to the thymic microenvironment, the ETP expression program is dramatically altered to commit them to the T-lymphoid lineage1, 2. Previous studies demonstrated that a final T-lymphoid lineage commitment occurs at a developmental checkpoint during the transition from the CD4−CD8− double negative (DN)2a to the DN2b stage, where expression of the transcription factor Bcl11b is induced3, 4. Bcl11b-deficiency results in a developmental arrest of early T cell progenitors at the DN2a stage along with a concomitant acquisition of myeloid-lineage/natural killer (NK) cell gene signatures5. Bcl11b thus serves as a T-lineage commitment factor that eliminates developmental potential to non-T lymphoid cells at the DN2 checkpoint3,4,5. Beside its function during early thymocyte development, Bcl11b continues to be expressed during T lymphocyte differentiation5, 6 and modulates the development of many lymphoid subsets, including natural killer T (NKT)7, regulatory T (Treg)8 and type2 innate lymphoid (ILC2) cells9,10,11.
After commitment to the T-lineage, another checkpoint, known as β-selection, selectively expands DN3 cells that successfully express functional TCRβ chain after V(D)J recombination of the Tcrb locus. Thymocytes that have passed β-selection become αβT-lineage-restricted CD4+CD8+ double positive (DP) precursor thymocytes, which express complete αβTCR complexes for the first time during T cell development. DP precursors are then subjected to additional positive and negative selections that enrich precursors with TCRs recognizing antigen-MHC complexes with intermediate affinity but eliminate precursors expressing self-reactive TCRs, respectively12. Positively selected thymocytes differentiate into distinct T cell subsets with distinct functionalities via the activation of specific developmental programs. For instance, positive selection signaled via TCR engagement by MHC class-II (MHC-II) and class-I (MHC-I) guides precursors to differentiate into CD4+CD8− helper or CD4−CD8+ cytotoxic T cells through the induction of key transcription factors, ThPOK and Runx3, respectively13, 14. Thus, DP precursors must be ready to integrate TCR signals, translating them into the appropriate developmental program. However, an important gap in our understanding of these processes is how TCR signals are coupled to mechanisms that control the expression of lineage-specifying genes, and it remains unclear whether preceding events are required for this coupling.
One such lineage-specifying transcription factor, Zbtb7b, also known as T-helper-inducing POZ/Krueppel− like factor (ThPOK), is a member of the BTB-POZ zinc-finger transcription factor family15 and is encoded by Zbtb7b, hereafter referred to as the Thpok gene. Previous genetic studies for ‘gain and loss’ of ThPOK function demonstrated that its presence or absence in post-selection thymocytes is a major determinant of the CD4-helper (ThPOK+) versus CD8-cytotoxic (ThPOK–) lineage dichotomy16,17,18. Consistent with these findings, expression of the Thpok gene is restricted to MHC-II selected thymocytes16 in positively selected thymocytes. Accordingly, Thpok regulation has been recognized as an ideal model to study how TCR signals are coupled with the transcriptional program that establishes the identity of CD4+ helper T cells. Such studies identified a transcriptional silencer in Thpok, hereafter denoted as Thpok silencer (Sth), which is an essential cis-acting element restricting Thpok expression to post-selection thymocytes in the helper lineage19, 20. In addition to Sth, there are at least three enhancers in the Thpok locus20. Among them, the thymic enhancer (TE), located upstream of the Sth, acts first to initiate Thpok expression21, which is subsequently upregulated through the activity of a proximal enhancer (PE) locating 1.8 kb downstream of the proximal P2-promoter22. While factors that regulate Sth activity, such as Runx family proteins19 and MAZR23, 24, have been identified, the factors involved in the activation of TE and PE remain poorly characterized. Gata325 and Tcf1/Lef126 regulate Thpok expression, but primarily do so by targeting other regulatory regions such as general T-lymphoid enhancer, the known third enhancer. In contrast to Thpok regulation, very little is known about transcription factors that orchestrate CD8+ T cell-specific expression of Runx3 from its distal P1-promoter27. Signals emanating from the IL-7 cytokine receptor have been shown to activate Runx3 28, 29; however, the intermediaries and their cis-regulatory targets in Runx3 remain to be established30.
Here we report two mechanisms by which Bcl11b governs the transcriptional program dissecting helper versus cytotoxic lineage commitment: Sth-independent repression of Thpok in DN thymocytes and enhancer-dependent Runx3 repression in CD4-lineage cells. Deletion of Bcl11b in thymocytes at post-β-selection stage causes chaotic Thpok and Runx3 expression, inducing random differentiation of both MHC-I and MHC-II selected cells into the helper and cytotoxic subsets. Along with earlier requirement for Bcl11b prior to DP stage in later Foxp3 activation, we conclude that lineage-specifying genes are ‘primed’ by Bcl11b before or during transition to the DP stage to represent an essential event for coupling TCR signals to expression programs for differentiating into the appropriate T-effector subsets.
Results
Bcl11b binds to the Thpok locus
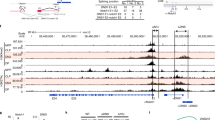
Based on our assumption that proteins bound to Thpok silencer (Sth) should mediate coupling of TCR signals during positive selection with release of Thpok expression, we attempted to purify protein complexes by in vitro capture with an oligo-nucleotide harboring core Sth sequences. Consistent with prior chromatin immunoprecipitation (ChIP) assays31, Bcl11b protein was efficiently enriched by affinity purification with core Sth sequences, but to lesser extent with mutant Sth sequences (Fig. 1a). Bcl11b also associated with other known Sth binding proteins, Runx1 (Fig. 1b). Furthermore, reduction of Bcll1b dosage to the half (Bcl11b +/−) in the Runx mutant mice, in which combined mutations of Runx1 and Runx3 genes attenuated Sth-mediated Thpok repression32, resulted in an increase of CD8+ subset de-repressing a Thpok gfp reporter22 (Fig. 1c), indicating genetic interaction between two factors in the regulation of Sth function. Our ChIP sequencing (ChIP-seq) assay of total thymocytes also detected that Bcl11b associated with Sth and proximal enhancer (PE) as well as intronic regions downstream from distal P1-promoter in the Thpok locus (Fig. 1d). Bcl11b also bound to an upstream regulatory element (URE)33 in the Sfpi-1 locus, which encodes a myeloid transcription factor, PU.1, and is a putative target of Bcl11b to eliminate myeloid potential during T-lineage commitment (Fig. 1d). A more global analysis revealed the Runx recognition motif (5′-ACCPuCA-3′) as the second-ranked sequence enriched at Bcl11b-bound regions in thymocytes (Fig. 1e) and Runx bindings coincided with ~ 85 % of Bcl11b-bound regions (Fig. 1e), which is consistent with previous observation using neuronal cells34. However, Bcl11b binding to Sth was Runx-independent, as determined using primary CD4+ T cells in which two Runx sites within the Sth were mutated by a knock-in on the Thpok gfp reporter allele35, whereas ThPOK binding to the Thpok silencer requires Runx-binding (Fig. 1f). Our analytical ChIP assay in CD4+ and CD8+ T cell subset detected that Bcl11b binding at PE was greater in Thpok-expressing CD4+ T cells, while fivefold more Runx was bound to Sth in ThPOK-negative cells (Fig. 1g). These results suggest that Bcl11b directly regulates activity of Thpok locus through binding to the silencer and enhancer elements.
Binding of Bcl11b to regulatory regions in the Thpok gene. a DNA pull-down assay showing in vitro Bcl11b binding to wild-type (Wt) Thpok silencer (Sth) core sequences, but not efficiently to mutant (Mut.) sequences. One representative of two experiments. b Co-immune precipitation assay showing interaction of Bcl11b with Runx1. One representative of two experiments. c Flow cytometry showing an increase of CD8+ T cells de-repressing Thpok-GFP upon reduction of Bcl11b dosage in Runx mutant mice. One representative of two independent experiments. d Bcl11b ChIP-seq tracks at the Thpok, Thpok gfp:ΔTESPE, and Sfpi1 genes in total thymocytes along with Runx ChIP-seq (GSE90794) tracks at the Thpok gene (top) for reference. Gene structure, transcriptional orientation and conservations in mammals (Cons.) in the Thpok gene are indicated. Positions of Thpok silencer (Sth), two enhancers (TE and PE), distal P1-promoter (P1) and proximal enhancer (PE) in the Thpok gene, and upstream regulatory element (URE) in the Sfpi1 gene are indicated as arrowheads. e Co-occupancy by Runx at Bcl11b-bound genome regions. Runx recognition site (5′-ACCPuCA-3′) was listed among the top two common sequences for Bcl11b-bound regions. f ChIP-PCR assay for binding of Runx, Bcl11b, and ThPOK to Wt and Runx site mutated (RSM) Sth regions in CD4+ T cells from Thpok +/gfp:241-401RM mice. Lanes 1, 2, and 3 are input, control IgG and antibody-against the indicated transcription factor, respectively. One representative of two independent experiments. g ChIP-qPCR analyses showing binding of Runx and Bcl11b to the Sth and PE in CD4+ (white bars) and CD8+ (gray bars) T cells. Combined data from three independent ChIP experiments is shown. **P < 0.01 (unpaired t-test)
Impaired Thpok regulation in Bcl11b mutant mice
Because there is a developmental arrest at the DN2a stage when Bcl11b function is completely lacking, it is necessary to utilize a Bcl11b fl allele to examine the function of Bcl11b during T cell development beyond the DN stage. We therefore attempted to generate our own Bcl11b fl allele. However, our first trial unexpectedly generated a mutant Bcl11b allele, hereafter referred as to a Bcl11b m, which produced a truncated Bcl11b protein due to a frameshift mutation incorporated into the targeting vector during its construction (Fig. 2a and Supplementary Fig. 1a−c). Despite its loss of the final zinc-finger motif, truncated mutant Bcl11b protein could bind to the Sth and an intronic region in the Thpok, as well as to the URE in Sfpi-1 (Fig. 2b). Similar to Bcl11b null animals (Bcl11b −/−), homozygous Bcl11b m mice died as neonates (Supplementary Fig. 1d). However, in contrast to the null animals, which completely lack CD4+CD8+ DP thymocytes and thereby have small thymus36, we noticed that total thymocyte number is only slightly reduced in Bcl11b m/m neonates (Supplementary Fig. 1e) and, more strikingly, the DP population was present in those mice (Supplementary Fig. 1f). In addition, Bcl11b m/m progenitors gave rise to ILC2 cells (Supplementary Fig. 1g), a subset that does not develop from Bcl11b −/− progenitors9, 10. These results indicated that the mutant Bcl11b m is a hypomorphic allele, thereby hereafter its product is referred to as hypomorphic Bcl11b protein (Bcl11bHM).
CD4-skewed development by impaired Thpok regulation from hypomorphic Bcl11b m/m progenitors. a Expression of truncated Bcl11b protein in the neonatal thymus from the hypomorphic Bcl11b m allele detected by an antibody recognizing the N- (left), but not the C- (right), terminus of Bcl11b. One representative of two experiments. b Summary of ChIP assay for binding of Wt (white bars) and hypomorphic (black bars) Bcl11b to indicated regions in total neonatal thymocytes. c−e Flow cytometry analyzing the expression of CD4, CD8, and Thpok-GFP during T cell development in Rag1-deficient hosts c and irradiated MHC-II deficient hosts e reconstituted with fetal liver cells with the indicated genotypes. Data are representative of at least two independent experiments. Numbers in dot plots indicate the percentage of cells in each quadrant. Histograms showing CD40L expression 2 days after in vitro TCR stimulation of peripheral CD4+, CD8+ and CD4−CD8− DN cells differentiated in Rag1-deficient mice from Bcl11b +/+and Bcl11b m/m fetal liver cells d. Data are representative of two independent experiments
We next investigated the T cell development of Bcl11b m/m progenitors that also harbor a Thpok gfp reporter allele. For this purpose, we transferred Bcl11b m/m:Thpok +/gfp or control Thpok +/gfp fetal liver progenitors into T cell-deficient Rag1 −/− hosts, allowing differentiation of the progenitors to proceed for two months. As shown in Fig. 2c, Thpok-GFP expression was absent in pre-selection thymocytes (defined as CD24hiTCRβlo) from control cells. In sharp contrast, nearly all pre-selection thymocytes derived from the transferred Bcl11b m/m progenitors expressed Thpok-GFP. While the two major T cell subsets, CD4+ helper and CD8+ cytotoxic, were differentiated from control Bcl11b +/+ progenitors, Bcl11b m/m progenitors gave rise to predominantly CD4+ T cells with a helper-related signature (e.g., CD40L induction) in mature thymocyte and peripheral T cell populations (Fig. 2c, d). To address whether a re-direction of MHC-I selected cells to the CD4+ lineage contributed to this CD4-skewing, we generated chimeras using irradiated MHC-II deficient hosts expressing the Ly9.1 marker, in which only differentiation of MHC-I selected cells was supported. Strikingly, we continued to observe skewing of CD4 differentiation in Ly9.1− donor-derived populations from Bcl11b m/m, but not Bcl11b +/+, progenitors (Fig. 2e). The shift to CD4 dominant differentiation was not observed when Bcl11b m/m :Thpok gfp/gfp progenitors lacking ThPOK expression were transferred (Fig. 2c). Along with de-repression of Thpok-GFP in remaining CD8+ T cells (Fig. 2c), we conclude that aberrant Thpok expression redirects MHC class I-selected Bcl11b m/m thymocytes to the CD4+ lineage. After these initial characterizations of the hypomorphic Bcll11b m allele, we generated a true conditional mutant (Bcl11b fl) allele and a mutant allele that retains the identical frameshift mutation to the Bcl11b m allele but lacks the loxP sequence in its 3′UTR, which we referred to as the Bcl11b HM allele (Supplementary Fig. 1a).
Silencer-independent Thpok repression by Bcl11b
To further investigate how Thpok expression was de-regulated in Bcl11b m/m cells, we examined promoter usages. Prior studies have shown that Thpok is transcribed from distal P1- and proximal P2-promoters20, however, P2-derived transcripts were detected specifically in T-lineage cells and require PE activity (Fig. 3a, b). Given the loss of Thpok-GFP expression in peripheral T cells upon removal of both TE and PE (Supplementary Fig. 2a), TE is likely to drive P1-promoter activity in T cells. In contrast, Thpok transcript in Bcl11b m/m pre-selection thymocytes was transcribed only from the P1-promoter (Fig. 3c). To understand the underlying mechanisms for unusual P1-promoter activation without P2-promoter activation in Bcl11b m/m thymocytes, we examined Thpok-GFP expression from the Thpok gfp:ΔTESPE reporter allele, in which all three T cell-specific regulatory elements, Sth, TE, and PE, were removed from the Thpok gfp allele by sequential gene targeting (Fig. 3d). In Bcl11b wild-type cells, Thpok expression from the enhancer-deficient Thpok gfp:ΔTESPE reporter allele was derived solely from the P1-promoter (Supplementary Fig. 2b), and was detected only in cells after the post-selection stages (Fig. 3d). In contrast, despite deletion of the Sth silencer, Thpok-GFP expression from the Thpok gfp:ΔTESPE reporter allele was initiated in Bcl11b m/m thymocytes at DN3 stage and its expression level was increased during transition to DP stage (Fig. 3d). These data revealed the presence of undefined Sth-independent mechanism by which Bcl11b represses Thpok expression in DN3 as well as in DP thymocytes.
Sth-independent Thpok repression by Bcl11b in pre-selection DP thymocytes. a Quantitative RT-PCR showing amount of promoter-specific Thpok transcripts in splenic CD4+ T cells and splenic B220+ B lymphocytes. Summary of three measurements. b Quantitative RT-PCR for Thpok-gfp expression in CD4+ T cells from mutant reporter alleles, Thpok gfp:ΔTE and Thpok gfp:ΔPE, lacking the thymic enhancer (TE) or proximal enhancer (PE), respectively. Levels of P1- and P2 promoter-specific transcripts relative to those from the control Thpok gfp allele harboring intact enhancers is shown. One representative of two independent experiments. c Promoter-specific Thpok transcripts in Bcl11b m/m pre-selection DP thymocytes and CD4+ T cells relative to those in control CD4+ T cells. Summary of three measurements. d Histograms showing Thpok-GFP expression from the Thpok gfp:ΔTESPE allele in the indicated cell subsets differentiated in Rag1-deficient recipients from control (dotted line) and Bcl11b m/m (solid line) fetal liver cells. Expression pattern of Thpok gfp is shown as reference (shaded). Schematic structure of the Thpok gfp:ΔTESPE allele is included (upper panel). One representative of two independent experiments. e ATAC-seq tracks at the Thpok locus in CD24hiTCRβloCD4+CD8+ pre-selection thymocytes of 6-week-old Bcl11b +/+ mice, newborn Bcl11b +/fl :Lck-cre, newborn Bcl11b HM/fl :Lck-cre and newborn Bcl11b HM/HM :Lck-cre mice. ATAC-seq results in immature B cells (GSM2123187) is shown as reference. DP thymocyte- and B lymphocyte-specific peaks are marked with blue and red arrowheads, respectively. Position of the P1 promoter is indicated with black arrowhead
To further address activation status of Thpok locus in Bcl11b mutant cells, we examined chromatin accessibility in DP thymocytes prepared from a Bcl11b HM/fl :Lck-Cre and Bcl11b HM/HM :Lck-Cre mice by ATAC-seq37. Interestingly, the Thpok locus in Bcl11b HM/HM pre-selection thymocytes retained open chromatin signatures around the P1-promoter, resembling those seen in B cells expressing P1-derived transcripts (Fig. 3e). ChIP-seq analyses for Bcl11b binding in thymocytes of Thpok gfp:ΔTESPE/gfp:ΔTESPE mice detected association of Bcl11b at intronic regions nearby the P1-promoter (Fig. 1d), which retained accessibility in the Bcl11b HM/HM cells and whose sequences are evolutionally conserved (Fig. 1d). Similar open structure was observed in DP cells of Bcl11b HM/fl :Lck-Cre mice. These observations indicate that truncation of Bcl11b C-terminal sequences interferes, most specifically, with normal patterns of Thpok expression at early thymocyte differentiation, reconfiguring its regulome to mimic expression patterns found in B cells. However, RNA-seq did not detect significant elevation of B cell signature genes in Bcl11b m/m pre-selection thymocytes (Supplementary Fig. 3a, b), suggesting that Bcll1b might be involved in modulating chromatin accessibility at the restricted loci such as Thpok.
Bcl11b is needed for lineage fidelity by MHC-restriction
Contrary to CD4-skewing from Bcl11b m/m progenitor, a previous study reported emergence of CD8+ T cells following Bcl11b inactivation at the DP stage using a Cd4-Cre driver. We reasoned that the apparent discrepancy could result from a stage-specific requirement for Bcl11b in regulating Thpok gene. We therefore next examined effect of Bcl11b inactivation by Cd4-Cre driver on Thpok expression. Although Thpok-GFP was uniformly de-repressed in all Bcl11b m/m pre-selection thymocytes (Fig. 2c), expression of this reporter was variegated and detected in only half of the pre-selection thymocytes from Bcl11b fl/fl :Cd4-Cre mice (Fig. 4a). We also observed that CD4+CD8+ and CD4−CD8+ subsets were present in mature thymocytes from Bcl11b fl/fl :Cd4-Cre mice (Fig. 4a, b). It is noteworthy that, after positive selection, levels of Thpok-GFP in CD4+CD8− cells differentiated from Bcl11b m/m progenitors or in Bcl11b fl/fl :Cd4-Cre mice were lower than those in control counterparts (Fig. 2c and Fig. 4a middle), a shift that was also reflected in attenuated P2-Thpok mRNA (Fig. 3c). Thus, loss of Bcl11b function disrupts not only Thpok repression, but also distinct mechanisms for its activation. These results suggests that, in a portion of pre-selection thymocytes in the Bcl11b fl/fl :Cd4-Cre mice, the kinetics of Thpok de-repression are delayed compared to Bcl11b m/m progenitors, which is combined later with low Thpok expression levels, allowing the cells to differentiate into CD4+CD8+ and CD4−CD8+ subsets.
Lineage scrambling resulting from inactivation of Bcl11b at the DP stage due to sporadic Thpok and Runx3 expression. a Flow cytometry analyzing the expression of CD4, CD8, Thpok-GFP, and Runx3-tdTomato by various thymocyte subsets in mice with the indicated genotypes. The histogram in the middle showing Thpok-GFP expression in CD4+CD8− mature thymocytes from control (open) and Bcl11b-deficient (shaded) cells. b Graph showing summary of percentage of each subset in mature (CD24−TCRβhi) thymocytes population of mice with indicated genotype. c Flow cytometry analyzing the expression of CD4, CD8, Thpok-GFP and Runx3-tdTomato in lymph node T cells of mice with the indicated genotypes. One representative of at least independent mice. d Flow cytometry analyzing the expression of CD4, CD8, Thpok-GFP and Runx3-tdTomato in pre-selection DP thymocytes and lymph node T cells differentiated in Rag1-KO hosts from Bcl11b +/+ and Bcl11b m/m fetal livers harboring Thpok gfp and Runx3 tdTomato reporter alleles. One representative of two independent experiments. e, f Representative flow cytometry e analyzing differentiation of MHC-I and MHC-II selected cells in MHC-II and MHC-I deficient (MHC-II0 and MHC-I0) backgrounds, respectively, along with a graph f showing a statistical summary of the percentages and absolute numbers in each cell subset in CD24−TCRβhi mature thymocytes population of control (white circles) and Bcl11b fl/fl :Cd4-Cre (filled circles) mice. Partial re-directed differentiation occurred in both MHC-I and -II selected cells. *P < 0.05, **P < 0.01, and ***P < 0.001(unpaired t-test)
Since previous studies showed release of Runx3 repression in CD4+ T cells that differentiated under low levels of ThPOK27, 38, we next examined Runx3 expression using a Runx3-tdTomato reporter allele that monitors distal P1-promoter activity, which is CD8-linege specific and serves as a major source for Runx3 protein expression in T cells27. Importantly, we found that most mature thymocytes from Bcl11b fl/fl :Cd4-Cre mice co-expressed Runx3-tdTomato and Thpok-GFP, regardless of their CD4/CD8 expression profiles (Fig. 4a), whereas expression of these genes was mutually exclusive between CD4+CD8− and CD4−CD8+ cells from control mice. Contrary to a previous report showing Runx3 de-repression in CD4+CD8+ DP thymocytes of Bcl11b fl/fl :Cd4-Cre mice31, Runx3-tdTomato was not detectably expressed in CD24hiTCRβlo pre-selection thymocytes (Fig. 4a). Co-expression of Thpok-GFP and Runx3-tdTomato was observed also in CD4+CD8− and CD4−CD8+ cells in the peripheral T cell pool of Bcl11b fl/fl :Cd4-Cre mice (Fig. 4c), as well as in CD4+ T cells differentiated from Bcl11b m/m progenitors (Fig. 4d). Thus, lineage-specific expression of two major factors, ThPOK and Runx3, which drive the CD4/CD8 lineage dichotomy, requires Bcl11b, or more specifically, Bcl11b function mediated by its C-terminal sequences.
We next examined the MHC specificity of T cells differentiated in Bcl11b fl/fl :Cd4-Cre mice by crossing with MHC-I or MHC-II deficient mice. In mature thymocytes and peripheral T cell populations, a significant proportion of both MHC-I- and MHC-II-specific cells in Bcl11b fl/fl :Cd4-Cre mice expressed surface markers for the alternative lineage, CD8 and CD4, respectively (Fig. 4e, f). We therefore conclude that a substantial number of both MHC-I and MHC-II selected thymocytes failed in their specification for the appropriate developmental pathway, presumably due to spurious expression of ThPOK and Runx3 following positive selection. Ultimately, the chaotic expression of these factors resulted in “lineage scrambling”, unlinking DP precursor thymocyte commitment from MHC specificity.
Functions of Bcl11b in Runx3 regulation
To understand how Bcl11b regulates Runx3 expression, we examined whether genomic regions around the Runx3 locus were occupied by Bcl11b, Runx, and ThPOK factors. In addition to the P1-promoter, Bcl11b and Runx bound at two regions (−39 and −21 kb), which were also bound by ThPOK (Fig. 5a) that is essential for Runx3 repression in CD4+ T cells22, 27. To examine potential of these regions as cis-regulatory elements for Runx3 regulation, we removed the regions separately or in combination from the Runx3-tdTomato allele using CRISPR/Cas9 technology39 (Supplementary Fig. 4). Levels of Runx3-tdTomato expression in CD4−CD8+ SP thymocytes were reduced upon removal of either the −39 or −21 kb region, but not a control region of similar length positioned at −40 kb region (Fig. 5a). Expression of the Runx3 reporter was further attenuated following combined deletion of the −39 and −21 kb regions. These observations indicated essential regulatory functions for these two regions in driving proper levels of Runx3 expression in CD8+ T cells. The residual expression when both regions were removed also indicated the presence of other enhancer(s) in the Runx3 locus.
Roles of Bcl11b in ThPOK-mediated repression of Runx3. a Runx and Bcl11b ChIP-seq tracks at the Runx3 locus using total thymocytes, along with a ThPOK ChIP-chip track using CD4 SP thymocytes. Schematic structure of mutant Runx3 tdTomato reporter alleles harboring several types of deletion (bottom panel, left). Histograms (bottom panel, right) showing Runx3-tdTomato expression in CD4−CD8+ thymocytes from indicated mutant alleles (solid line) and non-transgenic allele (dotted line). Data shown are representative of at least three independent mice. b Amount of Socs1 and Socs3 mRNA in CD4 SP thymocytes and CD4+ T cells from mice of indicated genotypes. Summary of three independent experiments. Mean ± SD **P < 0.01 (unpaired t-test). c ChIP-qPCR analyzing ThPOK binding to the Sth in Thpok and the −39E and −21E regions in the Runx3 using chromatin from CD4+ T cells of Wt (white bars) and Bcl11b f/f :Cd4-Cre (gray bars) mice. Combined data of three independent ChIP experiments. Means ± SD. d Flow cytometry analyzing the effect of a Thpok transgene on Cd8 and Runx3 expression. Dot plots showing CD4/CD8 expression in splenic T cells. Histograms at the right show Runx3 tdTomato reporter expression in Wt (white) and Bcl11b-deficient (gray) CD4+CD8− subset. One representative of three individual mice. e Histograms showing CD8 and Runx3-tdTomato expression in control CD4−CD8+, Bcl11b-deficient CD4−CD8+ and CD4+CD8− T cells after retroviral transduction of Wt (open) and the non-functional HD mutant ThPOK (shaded). One representative of two independent experiments
SOCS family proteins were proposed to mediate ThPOK-dependent repression of Runx3 following thymocyte positive selection29. However, expression of Socs1 and Socs3 was not reduced in Bcl11b-deficient CD4+ T cells expressing Runx3 (Fig. 5b). In Bcl11b-deficient cells, ThPOK bindings to −39 and −21 enhancers were also observed, albeit to a lesser extent, presumably due to lower ThPOK expression levels (Fig. 5c). We then tested whether ectopic ThPOK expression restored Runx3 repression in CD4+ T cells. Although transgenic ThPOK prevented the development of CD8+ T cells in Bcl11b fl/fl :Cd4-Cre mice, Runx3-tdTomato was not repressed as much in CD4+CD8− splenic T cells (Fig. 5d). Consistent with this observation, retroviral ThPOK transduction into peripheral CD8+ T cells reduced Runx3-tdTomato expression in control cells, but not in Bcl11b-deficient cells, while CD8 expression was reduced in both cells, as reported previously40 (Fig. 5e). These results indicated that full Runx3 repression by ThPOK required Bcl11b after ThPOK bound to Runx3 enhancers. In contrast, ThPOK could repress Cd8 expression in the absence of Bcl11b.
Bcl11b regulates Thpok and Runx3 expression
To address at which developmental stages Bcl11b is necessary for appropriate Thpok and Runx3 expression, we inactivated the Bcl11b gene during maturation of MHC-II selected cells using a Thpok-Cre driver41. While CD4 and CD8 expression on peripheral T cells remained unchanged in Bcl11b fl/fl :Thpok-Cre mice, the expression of Thpok-GFP and Runx3-tdTomato in CD4+ T cells was decreased or increased, respectively (Fig. 6a, b). When Bcl11b was inactivated in differentiated CD8+ T cells by retroviral Cre transduction, de-repression of Thpok-GFP was also observed (Fig. 6c). These findings indicated that Bcl11b was necessary to maintain lineage-specific expression of both Thpok and Runx3 after primary commitment of post-selection thymocytes to CD4/CD8 lineages.
Bcl11b function requires its C-terminal zinc-finger motif. a Flow cytometry analyzing the expression of CD4, CD8, Thpok-GFP, and Runx3-tdTomato in peripheral T cells from Bcl11b +/+ :Thpok-Cre and Bcl1b fl/fl :Thpok-Cre mice harboring Thpok gfp and Runx3 tdTomato reporter genes. Inactivation of Bcl11b during the maturation of CD4-lineage cells by a Thpok-Cre transgene resulted in impaired Thpok-GFP expression, as well as Runx3-tdTomato de-repression. b RT-qPCR analyses showing relative amounts of Thpok (black bars) and P1-Runx3 (white bars) mRNA in splenic CD4+CD8− T cells of Bcl11b +/+ :Thpok-Cre and Bcl11b fl/fl :Thpok-Cre mice. Results in splenic CD4−CD8+ T cells of Bcl11b +/+:Thpok-Cre are shown as references. Data were summary of three experiments. Means ± SD *P < 0.05 (unpaired t-test). c Histograms showing Thpok-GFP expression from CD4+ and CD8+ T cells of Bcl11b +/+ :Thpok +/gfp (white bars) and Bcl11b fl/fl :Thpok +/gfp (gray bars) mice 4 days after retroviral Cre transduction. One representative of three experiments. d Histograms showing Runx3-tdTomato expression 3 days after retroviral transduction of Wt and hypomorphic (HM) Bcl11b into peripheral CD4+CD8− Runx3-tdTomato + cells from Bcl1b fl/fl :Runx3 +/tdTomato :Thpok-Cre mice. Expression of Runx3-tdTomato in activated control CD4+ T cells (dotted line) after transduction with an empty vector (open) are shown as controls. e Histograms showing Runx3-tdTomato expression after retroviral transduction of mutant Bcl11b proteins lacking the C-terminal zinc-finger motif are shown as in d. The dotted line represents Runx3-tdTomato expression after transduction of Wt Bcl11b. One representative of at least two experiments d, e
We also tested the converse scenario, namely, whether retroviral Bcl11b transduction restored Runx3-tdTomato repression in differentiated CD4+ cells of Bcl11b fl/fl :Thpok-Cre mice. Levels of Runx3-tdTomato was reduced upon Bcl11b transduction compared with empty vector controls (Fig. 6d); however, transduction with a vector encoding the Bcl11bHM protein repressed Runx3-tdTomato to lesser extent than wild-type Bcl11b (Fig. 6d). To gain more insights into the domainal structure of Bcl11b, we tested the activities of other mutants generated by sequential deletion on its C-terminus (Supplementary Fig. 5). While a Bcl11b mutant retaining the final zinc-finger domain (Bcl11b-Δ10) and wild-type Bcl11b equally repressed Runx3-tdTomato expression, other mutants lacking the zinc-finger structure showed weaker activity (Fig. 6e). These results not only confirmed that loss of the last zinc-finger motif, rather than acquisition of aberrant sequences, attenuated Bcl11b function in the Bcl11bHM protein, but the new data also indicated that the last zinc-finger motif is essential for Bcl11b function in preserving lineage-specific Runx3 expression.
In this regard, we compared structures of Bcl11 family proteins in other species. There are two zinc-finger clusters in Bcl11b, double zinc-fingers in its central region and triple zinc-fingers at its C-terminus. Comparative genomics analyses revealed that some species, such as the Ascidians (C. intestinalis) and Worm (C. elegans), lacked the middle zinc-finger motif in Bcl11 orthologues, whereas the triplet zinc-finger structure at the C-terminal was highly conserved during evolution (Supplementary Fig. 6), suggesting that C-terminal triplet zinc-fingers could be involved in ancestral and conserved function of Bcl11 family proteins.
Stage-specific requirement for Bcl11b in Foxp3 transcription
Additional analyses of T cell development from Bcl11b m/m progenitors detected a striking defect in the development of Foxp3+ regulatory T cells (Treg) (Fig. 7a). In line with this finding, we were unable to induce Foxp3 expression in the mutant CD4+ T cells even under potent Treg differentiation conditions in vitro (Fig. 7b). Contrary to this finding, a previous study reported the emergence of Foxp3+ Treg cells following Bcl11b inactivation at the DP stage using a Cd4-Cre driver8. This apparent discrepancy could result from a direct and stage-specific requirement for Bcl11b in activating Foxp3, as we observed in Thpok regulation by Bcl11b. Recent work reported that the CNS3 region in the Foxp3 gene is essential to initiate Foxp3 activation by poising the Foxp3 promoter in an active state prior to the DP stage42, 43. Our ChIP-seq detected Bcl11b binding to CNS3 in total thymocytes (Fig. 7c) and our ChIP-qPCR detected similar binding of both Bcl11b and Bcl11bHM proteins to CNS3 (Fig. 7d). Since we recently observed that binding of the genome organizer, SATB1, to CNS3 is essential for Foxp3+ Treg development in the thymus41, 44, we examined SATB1 association with CNS3 in Bcl11b mutant thymocytes. Consistent with development of Foxp3+ cells, SATB1 binding to CNS3 was unaffected in total thymocytes from Bcl11b fl/HM :Cd4-Cre mice. On the contrary, SATB1 recruitment was significantly decreased in those cells from Bcl11b fl/HM :Lck-Cre mice (Fig. 7e). These results suggested that priming of the Foxp3 locus by Bcl11b prior to the DP stage is essential for activation of Foxp3 gene via promoting SATB1 recruitment to the CNS3 enhancer.
Regulation of Foxp3 expression by Bcl11b. a Expression of Foxp3 in CD4+CD8− mature thymocytes differentiated in Rag1-KO hosts from Bcl11b +/+ and Bcl11b m/m fetal liver progenitors. A representative histogram (left) along with a summary of five independent hosts (right) are shown. ***P < 0.001 (unpaired t-test). b Histogram showing Foxp3 induction from CD62L+CD4+ T cells after in vitro culture under Treg differentiation condition. One representative of two experiments. c Bcl11b ChIP-seq tracks at the Foxp3 gene in DP thymocytes. Gene structure, transcriptional orientation and position of conserved non-coding sequence (CNS) 3 are indicated. d Summary of ChIP-qPCR assay for binding of Wt (white bars) and hypomorphic (black bars) Bcl11b to CNS2 and CNS3 regions in the Foxp3 locus in neonatal total thymocytes. e ChIP-qPCR for binding of SATB1 to the CNS3 enhancer in the Foxp3 gene. Chromatin was collected from DP thymocyte precursors using mice with the indicated genotypes. Combined data from three independent ChIP experiments are shown. **P < 0.01 (unpaired t-test)
Discussion
In this study, we demonstrate that a hypomorphic Bcl11b protein (Bcl11bHM), lacking only its final zinc-finger domain, supports T-lineage commitment and ILC2 development. In contrast, the hypomorphic Bcl11b mutation fails to control the expression of transcription factors, including ThPOK, Runx3, and Foxp3, which are important for specification of mature T lineages (CD4-helper, CD8-cytotoxic, and Treg, respectively). Importantly, our findings dissect the functional architecture of Bcll1b, revealing independent regions that are responsible for early T-lineage commitment versus transcriptional regulation of lineage-specifying genes during positive selection.
The distinct effects on expression of Thpok and Foxp3 genes of the germline hypomorphic mutation when compared with a DP stage-specific inactivation of its gene indicate that Bcl11b acts on these two genes at an early developmental stage, presumably at the DN to DP transition, to control appropriate expression of these lineage-specifying transcription factors at later stage. For Thpok, while a half of precursors still repressed Thpok gene after conditional loss of Bcl11b expression, hypomorphic Bcl11b mutation caused Thpok expression in all precursors that retains non-T cells like open chromatin structure around the P1-promoter. This observation indicates that early Thpok repression at the DN to DP transition by Bcl11b involves chromatin closing. Our results using the Thpok gfp:ΔTESPE mutant reporter allele lacking Sth silencer revealed that Bcl11b regulates such chromatin closing independently of the Sth, which is essential to silence Thpok expression at later stage specifically in cytotoxic T cells35. Given that the Sth is sufficient to repress reporter transgenes driven by heterologous enhancer/promoter in DP thymocytes20 and is essential to prevent expression of the endogenous gene in Bcl11b-sufficient DP cells19, both Sth-independent and Sth-dependent pathways operate to repress Thpok in precursor DP thymocytes. Considering that ectopic ThPOK expression in DP precursor thymocytes not only disturbs helper/cytotoxic lineage choice16 but also increases a risk for lymphomagenesis45, it might be beneficial to have two independent mechanisms to secure no leaky ThPOK expression in DP precursors. It is noteworthy that Sth-independent Thpok repression was reversible, since Thpok-GFP expression from the Thpok gfp:ΔTESPE allele was induced in mature T cells, suggesting that irreversible epigenetic changes such as DNA methylation are unlikely to be involved in early Sth-independent Thpok repression by Bcl11b. Given that Sth could stably silence Thpok transcript, particularly from the P2-promoter, in CD8-lineage cells35, mechanisms of Sth-dependent and Sth-independent Thpok repression could be different.
The precise involvement of Bcl11b in Sth-independent Thpok repression remains elusive. As detected by ChIP-seq, Bcl11b covers regions around the P1-promoter in an Sth-independent manner. Along with evolutional conservation in sequences of intronic region just downstream of the P1-promoter, there might exist uncharacterized regulatory element(s) that suppresses P1-promoter activity upon expression of Bcl11b. Binding of mutant Bcl11bHM protein to such intronic region suggest that the C-terminal zinc-finger motif in Bcl11b is necessary to close P1-promoter region after Bcl11b binding to putative regulatory element(s). Alternatively, Bcl11b may be involved indirectly in the Sth-independent Thpok repression through an uncharacterized intermediate factor, however, the similar gene expression profiles between wild-type and Bcl11b m/m precursors disfavors this possibility.
De-repression of Thpok in mature CD8+ populations that emerge from Bcl11b fl/fl :Cd4-Cre precursors suggests that Bcl11b is required also for Sth-mediated Thpok repression. Thus, Bcll1b regulates Thpok repression by both Sth-independent and Sth-dependent mechanisms. Based on our data, we presume that Bcl11b acts initially to inhibit P1-promoter activity in an Sth-independent manner. Either sequentially or simultaneously, Bcl11b primes three T cell specific regulatory regions, Sth and two enhancers, TE and PE. Upon such priming, Sth initiates to suppress TE and PE activity, thereby removal of Sth results in Thpok de-repression in Bcl11b-sufficent precursors in which TE/PE enhancers also becomes functional after Bcl11b-mediated priming. In this regard, an Sth-independent mechanism alone is likely to be insufficient for extinguishing TE/PE enhancer activities. Thus, we assume that pre-conditioning of T cell specific regulatory regions by Bcl11b is an essential process for dissecting TCR signals that arise from engagement by distinct MHCs, and for converting them into establishment of “on” and “off” Thpok expression status for inducing helper- or cytotoxic-fate via inactivation or maintaining Sth activity, respectively.
Bcl11b similarly primes the Foxp3 locus for later activation upon exposure to agonistic selection signals46. A recent study showed that the pioneering enhancer, CNS343, confers a poised state to the Foxp3 promoter in precursor cells in response to TCR stimuli42. In the present study, we show that binding of SATB1 to CNS3 in precursors, an essential process for activation of Foxp3 gene in the thymus41, 44, requires Bcl11b-mediated priming before transition to the DP stage. Whether common Bcl11b-mediated mechanisms operate to prime the Thpok and Foxp3 loci remains unclear; however, we establish that the C-terminal zinc-finger motif of Bcl11b is involved in both processes. Collectively, our results reveal a set of novel functions for the T-lineage commitment factor Bcl11b, which “pre-conditions” precursors for integration of environmental cues, specifically TCR signals, into a developmental program that dissects effector lineages and shapes the primary T cell pool.
Another striking finding from our studies is that the lack of Bcl11b inhibits both the positive and negative regulatory regions near Thpok. One possible model to explain how Bcl11b controls multiple regulatory regions would be its function as a scaffold to facilitate communication between the distal elements and the Thpok promoters. Using insertion ChIP technology47, we recently observed that the Sth and PE regions were positioned in close proximity during differentiation into CD8-lineage cells (I.T. manuscript in preparation). Thus, it is possible that Bcl11b is involved in shaping promoter-regulatory elements communications on the Thpok locus.
Contrary to Thpok regulation, Bcl11b is merely involved in repression of Runx3 in CD4+ T cells22, 27. Prior studies invoked an indirect mechanism for Runx3 repression by ThPOK via the induction of SOCS family proteins29. However, our finding that ThPOK associates with functional Runx3 enhancers raises the possibility that ThPOK directly antagonizes enhancer function. Indeed, ThPOK bound to the enhancers even in Bcl11b-deficient cells that were de-repressing Runx3 albeit normal level of Socs family gene expression. These data suggest that the antagonistic action of ThPOK against Runx3 expression requires Bcl11b after its binding to the Runx3 enhancers. It is conceivable that Bcl11b assists ThPOK in preventing the formation of looping between the enhancers and the P1-Runx3 promoter. Nevertheless, it will be important in future studies to examine whether lineage-specific chromatin structures are formed in Thpok and Runx3 loci and whether Bcl11b regulates these topologies.
The C2H2 zinc-finger motif not only serves as a DNA binding domain that recognizes specific sequences, but also as a docking module for RNA and proteins48. We found that the Bcl11bHM protein retains its ability to associate with regulatory regions, indicating that its final zinc finger is dispensable for DNA binding. However, it remains possible that the other four zinc-fingers domains have compensatory functions in this regard. There are two zinc-fingers clusters in mammalian Bcl11 family proteins, double zinc-fingers in its central portion and a trio of zinc-fingers at its C-terminus. Although there are Bcl11-related proteins in lower species that lacks the central dual zinc-finger domain, a trio of zinc-fingers at the C-terminus is evolutionally well conserved. Our results indicate that this domain is essential for gene regulation, presumably through modulating topological structures. Bcl11a has been shown to play a key role in the switch from fetal γ- to adult β-globin in human49. It is conceivable that common Bcl11 family-dependent mechanisms play a key role in developmentally programmed switching of regulatory regions by modulating topological structures through the C-terminal triplet zinc-finger stretch. Isolation of interacting molecules with it will further elucidate regulatory mechanism by Bcl11 family.
Methods
Mice
Runx1 Δ446 mice50, Runx3 fl mice51, Bcl11b +/− mice36, Thpok gfp mice22, Thpok gfp:241-401RM mice35 and Thpok transgenic mice19 have been described. Lck-Cre mice, and Cd4-Cre mice were from Dr J. Takeda, and Dr C. Wilson, respectively. β2m-deficient mice (stock No:002070), Rag1-deficient mice (Stock No:002216) were from Jackson laboratory, and I-Aβ deficient mice and Il2rg –/–:Rag1 –/– were from Taconic. In order to generate a Thpok gfp:ΔTESPE allele, TE/Sth and PE regions were sequentially removed from the Thpok gfp reporter allele by using homologous recombination in ES cells. In order to construct the target vector for Bcl11b fl allele, genomic fragment isolated from the phage library (Stratagene) was used as starting material. The neomycin-resistance gene (neo r) flanked with two loxP sequences was cut out from pL2-Neo(2) vector and thymidine kinase (TK) gene was isolated from pNT vector. A 5′ short homology region was amplified by PCR and was ligated with the neo r fragment, followed by sequential ligation to add 3′ homology region. The third loxP sequences placed in the 3′ untranslated region (UTR) were derived from synthetic oligo-nucleotide. The single nucleotide deletion that causes a frameshift mutation in the Bcl11b m allele was accidentally incorporated into the target vector at some point during above sequential ligation steps. To construct a real target vector for Bcl11b fl mutation, correct exon 4 sequences were replaced in the Bcl11b m target vector. A fragment that harbors the identical Bcl11b m single nucleotide deletion but lacks the third loxP sequences was used to construct the target vector for Bcl11b HM allele. In order to generate Runx3 tdTomato reporter allele, we constructed the targeting vector by replacing YFP cDNA fragment with cDNA fragment encoding tdTomato fluorescent protein in pRx31-KIN cassette vector27, which was used to generate the Runx3 YFP allele27. Those targeting vectors were transfected into M1 ES cells as previously described22. ES clones underwent homologous recombination were identified by PCR with appropriate primers sets. In order to delete −39 kb and −21 kb genomic regions from the Runx3 tdTomato reporter allele by CRISPR/Cas9 technology52, 53, we selected two single guide RNA (sgRNA) target sequences that flanks target genomic regions, which were shown in Supplementary Fig. 3. Custom sgRNA, in which CRISPR RNAs was fused to a normally trans-encoded tracrRNA, were transcribed from a T7 promoter in the pUC18 vector by in vitro transcription with MEGAshortscript T7 kit (Life Technologies, AM1354) and both the Cas9 mRNA and the sgRNAs were purified using MEGAclear kit (Life Technologies, AM1908). The sgRNAs and mRNA encoding Cas9 were co-injected into cytoplasm of fertilized eggs that were prepared by in vitro fertilization with Runx3 tdTomato/tdTomato sperm and wild-type oocytes. Cas9-mediated double-stranded DNA breaks resolved by non-homologous end joining (NHEJ) ablated the intervening sequences. Founder offspring that had deletion of the target genome region were crossed to wild-type mice, and F1 founders that harbor both deletion mutation and Runx3 tdTomato allele were selected for establishing mouse line and were analyzed. In order to generate allele harboring double deletions at −39 kb and −21 kb regions, sgRNA pair used for generation of Runx3 Δ21E mutation were injected into eggs obtained by in vitro fertilization between Runx3 tdTomato:Δ39LD/+ sperm and wild-type oocytes. RNA injection was performed by the Animal Facility Group at RIKEN, IMS. All mice were maintained in the specific pathogen free animal facility at the RIKEN IMS, and all animal procedures were in accordance with institutional guidelines for animal care and with the protocol (28-017) approved by the safety section in RIKEN Yokohama Campus.
DNA pull-down assay
In order to analyze binding of Bcl11b to Thpok silencer (Sth) core sequences in vitro, nuclear extract was prepared from 10 × 107 total thymocytes and were mixed with 10 μg of biotin-labeled synthetic oligo-nucleotides probe (Eurofins Genomics) in 400 μl of affinity purification (AP) buffer (20 mM HEPES-KOH (pH 7.5), 80 mM KCl, 10% Glycerol, 0.1% Triton X, 0.5 mM PMSF, 1× cOmplete protease inhibitor Cocktail tablets (Roche)) on ice for 3 h, followed by mixture with 100 μl of Dynabeads M-280 Streptavidin (Thermo Fisher Scientific) for additional 3 h on ice. After one time wash with AP buffer, beads were incubated with 500 pmol of mutant non-labeled oligo-nucleotide for 30 min at 4 °C, and were washed with AP buffer three times. Captured protein complexes by beads were then released into 50 μl of SDS sample buffer and were applied for western blot analyses. Sequences for oligo-nucleotides probes used were as follows:
Wt:5′-TGGCAGCCACCGCCTCTTCAGGTGGGTTGGGCGGTCGCGGTAGGGGTTCTGGGGGGCGGCGGGAGGGAGGGGGCTGCGGTCTGAG-3′
Mutant:5′-TGGCAGCCACCGCCTCTTCAGCACCCATGGGCGCAGGCGGTAGGGGTTCTGGGGGGCGGCGGGAGGGAGGGGGCACGCCACTGAG-3′.
Co-immune precipitation and western blot
Nuclear extract was prepared from 10 × 106 total thymocytes with NE-PER kit (Thermo Fisher Scientific) in the presence of cOmplete protease inhibitor cocktail tablets (Roche), followed by incubation with 2 μg of anti-Bc111b (A300-383A, Bethyl Laboratories) antibody, which was pre-mixed with 50 μl of Dynabeads ProteinA, overnight at 4 °C with gentle rotation. Antibody bound proteins were released into 20 μl of SDS sample buffer and was loaded onto SDS-PAGE gel. In western blot, anti-Runx1 serum54 and antibodies that reacts with N-terminal sequences (A300-383A, Bethyl Laboratories) and C-terminal sequences (A300-385A, Bethyl Laboratories) of Bcl11b were used to detect protein.
Chromatin immunoprecipitation
For ChIP-seq, 3 × 107 thymocytes that were freshly prepared from C57BL/6 mice at 4−6 weeks old were washed once with PBS supplemented with 1% FCS, cOmplete protease inhibitor cocktail (Roche) and 1 mM 4-(2-Aminoethyl) benzenesulfonyl fluoride hydrochloride (Sigma-Aldrich), and were cross-linked by incubation in a 1% paraformaldehyde solution for 10 min with gentle rotation at RT. The reaction was stopped by addition of glycine to 0.15 M. Cells were then washed with ice-cold PBS containing 1% FCS for 10 min with gentle rotation at 4 °C, and were lysed in Lysis Buffer 1 (50 mM HEPES pH 7.5, 140 mM NaCl, 1 mM EDTA, 10% Glycerol, 0.5% NP-40, 0.25% Triton X-100) supplemented with cOmplete protease inhibitor cocktail tablets (Roche) for 10 min at 4 °C with gentle rotation. Nuclei were pelleted and were washed by Lysis Buffer 2 (10 mM Tris-HCl pH 8.0, 200 mM NaCl, 1 mM EDTA and 0.5 mM EGTA) supplemented with cOmplete protease inhibitor (Roche). Pelleted chromatin was resuspended in 300 μl of Lysis Buffer 3 (10 mM Tris-HCl pH 8.0, 100 mM NaCl, 1 mM EDTA, 0.5 mM EGTA, 0.1% Sodium deoxycholate and 0.5% N-laurylsarcosine sodium salt), and was sonicated using a model XL2000 ultrasonic cell disruptor (MICROSON) at output level 6 for 15 s for 10 times. After removing debris by centrifugation, 30 μl of 10% Triton X-100 (Nacalai tesque) was added to 270 μl of supernatant (final 1%) and sonicated chromatin was incubated overnight at 4 °C with 5 μg of anti-Bcl11b rabbit polyclonal antibody (A300-383A, Bethyl Laboratories) that was pre-conjugated with 50 μl of Dynabeads M-280 Sheep anti-Rabbit IgG (Thermo Fisher Scientific). After washing beads with ChIP-RIPA (50 mM HEPES (pH 7.6), 500 mM LiCl, 1 mM EDTA, 1% NP-40, 0.7% sodium deoxycholate) and TE buffer supplemented with 50 mM NaCl, immunoprecipitates were eluted from beads into 100 μl of elution buffer (50 mM Tris-HCl pH 8.0, 10 mM EDTA, 1% SDS) by incubation for 15 min at 65 °C with vigorous shaking. Eluted immunoprecipitates were then incubated at 65 °C overnight for reverse-crosslinking. Input DNA and ChIP DNA were treated with RNAse A (Thermo Fisher Scientific) at 37 °C for 1 h, followed by incubation with Proteinase K (Thermo Fisher Scientific) at 55 °C in the presence of 6 mM CaCl2 for one hour. DNA was purified by Phenol/Chloroform extraction for ChIP-seq or ChIP DNA Clean and Concentrator kit (ZYMO RESEARCH) for ChIP-qPCR. For ChIP-seq analysis, purified DNA was subjected to re-sonication with a Covaris S220 to produce DNA fragments with an average size of 200 bp, and was used for library construction with NEBNext ChIP-seq Library Prep Master Mix set for Illumina Kit (NEB). Sequencing was performed by the RIKEN IMS sequence facility with Illumina HiSeq 1500 or at Hiroshima University using a GAIIx (illumina). To detect ThPOK binding regions in the murine Runx3 locus, we performed ChIP-on-chip experiment. Chromatin DNA isolated from 10 × 106 CD4+ SP thymocytes from FH-ThPOK mice22 was immunoprecipitated with anti-Flag (M2; Sigma-Aldrich), and were hybridized against custom microarrays generated by Agilent that tiled through the murine Runx3 locus up to ~ 565 kb upstream and ~ 66 kb downstream by means of 60-nucleotide probes. Probe hybridization and scanning of oligo-nucleotide array data were done according to manufacturer’s protocol (Agilent). For analytical ChIP, we used 5 μg of rabbit anti-Bcl11b antibody (A300-383A, Bethyl Laboratories), 5 μg of rabbit anti-ThPOK polyclonal antibody27 and 5 μg of rabbit anti-SATB1 polyclonal antibody (ab70004, Abcam). Quantitative PCR was performed using the StepOnePlus Real-Time PCR system (Applied Biosystems) with SYBR Green detection system. Primers sequences for quantitative PCR are listed in the Supplementary Table 1.
Flow cytometry analyses
Thymus, spleen, and lymph nodes were removed from mice at 4−8 weeks of age, and were mashed through a 70 μm cell strainer to make single-cell suspensions. Cells were stained with following antibodies purchased from BD-Bioscience: CD4 (RM4-5), CD8 (53-6.7), CD24 (M1/69), CD25 (PC61), CD154 (MR1), IL1RL1 (DJ8), ScaI (E13-161.7), KLRG1 (2F1), and TCRβ (H57-597). Foxp3 staining buffer Set (00-5523-00) and anti-mouse FoxP3 antibody (FJK-16s) from affymetirx eBioscience was used to stain Foxp3. Antibodies were used at a concentration of 2.5 μg ml−1. Multi-color flow cytometry analysis was performed using a FACSCANTO II (BD-Bioscience) and data were analyzed using FlowJo (Tree Star) software. Cell subsets were sorted using a FACSAria II (BD Biosciences) by cell sorting facility at RIKEN IMS.
In vitro T cell culture
Purified T cells were cultured in custom ordered Dulbecco’s Modified Eagle Medium (D-MEM, KOHJIN BIO) supplemented with 10% heat inactivated FBS (Hyclone). 1.0 × 106 cells were stimulated in 24-well plates pre-coated with 2 μg ml−1anti-CD3e antibody (553058, BD Bioscience) with 2 μg ml−1 soluble anti-CD28 antibody (553295, BD Bioscience) for two days. For induction of Treg differentiation, 10 ng ml−1 TGFβ1 (7666-MB, R&D systems), 5 μg ml−1 anti-IL4 (554433, BD Biosciences) and 5 μg ml−1 anti-IFNγ (554409, BD Biosciences) were added throughout the culture.
Reconstitution of T cell development in host mice
Total liver cells with either Bcl11b +/+ or Bcl11b m/m genotype were prepared from 13.5 to 14.5 dpc embryos, and were suspended in 400 μl of D-MEM supplemented with 10% heat inactivated FBS (Hyclone). 200 μl of these fetal liver cells were intravenously injected into sub-lethally irradiated (6.5 Gy, Gammacell 40 Exactor, MDS Nordion) Rag1-deficeint or I-Aβ deficient mice that expressing Ly9.1. For ILC2 reconstitution, Il2rg –/–:Rag1 –/– mice were used as recipients. Host mice were supplied with antibiotics (1 mg ml−1 neomycin and 100 unit ml−1 polymyxin B) containing water for first 2 weeks, and were analyzed 8 weeks after the fetal liver cell injection. In I-Aβ deficient host mice, donor derived lymphocytes were identified as Ly9.1-negative cells.
RNA isolation, RT-qPCR and RNA-seq
After total cellular RNA extraction by using Trizol reagent (Thermo Fisher Scientific), samples were incubated with RNase-free DNase I (Thermo Fisher Scientific) before cDNA synthesis with the SuperScriptIII First Strand Synthesis System (Invitrogen). Quantitative RT-PCR was performed using the StepOnePlus Real-Time PCR system (Applied Biosystems) with an internal fluorescent TaqMan probe, Universal ProbeLibrary (Roche) or SYBR Green detection system. Primers sequences and probe sequences for quantitative RT-PCR are listed in the Supplementary Table 2. Primer sequence to measure total Thpok mRNA was previously described16. For RNA-seq analyses, purified 1.0 × 106 cells were used to prepare mRNA and 200−300 ng total RNA were used for the library construction with SureSelect Strand Specific RNA-Seq LibraryPreparation kit (Agilent Technologies) according to the manufacturer’s protocol. Sequencing was performed by genomics facility at RIKEN IMS with Illumina HiSeq 1500.
Assay of transposase-accessible chromatin sequencing
Assay of transposase-accessible chromatin sequencing (ATAC-seq) samples were prepared from 25,000 sorted cells. The transposase reactions were carried as previously described55 with 12 or 13 total PCR cycles. Amplified DNA fragments were purified with QIAGEN MinElute PCR Purification Kit and size-selected twice with Agencourt AMPure XP. Libraries were quantified with KAPA Library Quantification Kit for Illumina Sequencing Platforms (KAPA Biosystems), and size distribution was checked on a Bioanalyzer (Agilent High Sensitivity DNA chip, Agilent Technologies). Libraries were sequenced on Illumina HiSeq 2500.
Retroviral transduction into T cells and T cell culture
Retroviral vectors encoding Cre recombinase or ThPOK in pMSCV-GFP vector was previously described56. Retroviral vectors encoding mutant Bcl11b proteins were generated by insertion of corresponding cDNA into pMSCV-GFP vector after conformation of sequences of PCR amplified fragment. Those vectors were transfected into Plat-E packaging line (a gift from Dr T. Kitamura at the University of Tokyo) by FuGENE6 Reagent (Promega, E2691) and supernatant collected two days after transfection were used for transduction into activated T cells by in vitro TCR stimulation with immobilized anti-CD3 and soluble anti-CD28 by spin infection at 2400 r.p.m. for 60 min in the presence of 4 μg of polybrene (Sigma-Aldrich). Transdcuced T cells were identified according to GFP expression for FACS analyses and RNA extraction.
Data and statistical analyses
Sequences retrieved in ChIP-seq experiments and ATAC-seq were aligned on the mouse genome (mm9) using the bowtie2 (http://bowtie-bio.sourceforge.net/index.shtml) with default parameters, and accumulated reads were normalized using total mapped reads. RNA-seq reads were mapped using tophat2 (https://ccb.jhu.edu/software/tophat/index.shtml) to the mouse genome and gene expression was estimated with fragments per mapped reads per kilobase (FPKM) values calculated using cufflinks (http://cole-trapnell-lab.github.io/cufflinks/) and gene annotation provided by iGenome. Primary component analysis (PCA) was carried out using our in-house program with FPKM values of gene sets whose annotation was provided by Gene Ontology database (http://geneontology.org/). Statistical analysis was performed by F-test and unpaired t-test with or without Welch’s correction using using GraphPAd Prism6 (Graphpad software). Figures display means and SD.
Comparative genomics analyses
Homologous sequences of Bcl11 family were searched with human Bcl11b protein sequences as a query against sequences listed at the Ensemble genome browser 85 and Genebank with “TBLASTN” algorithm. As for the Japanese lamprey search, Bcl11 protein sequences for human, mouse, and elephant shark were used as queries to search the Japanese lamprey genome assembly (http://jlampreygenome.imcb.a-star.edu.sg/) with “TBLASTN” algorithm. Genomic regions that showed high similarity were extracted and searched against the non-redundant protein database at NCBI to confirm their identity. Alignments of C-terminal sequences of Bcl11b orthologue of each species were performed by a computer program Clustal Omega (http://www.clustal.org/omega/) with default options for amino acid sequences using zinc-finger regions obtained proposed by BLASTP algorithm and NCBI conserved domain database (https://www.ncbi.nlm.nih.gov/Structure/cdd/wrpsb.cgi).
Data availability
RNA-seq, ChIP-seq and ATAC-seq data that support the findings of this study have been deposited with accession GSE90134, GSE90949, and GSE90989, respectively. All other relevant data are available from the authors.
References
Yui, M. A. & Rothenberg, E. V. Developmental gene networks: a triathlon on the course to T cell identity. Nat. Rev. Immunol. 14, 529–545 (2014).
Yang, Q., Jeremiah Bell, J. & Bhandoola, A. T-cell lineage determination. Immunol. Rev. 238, 12–22 (2010).
Li, L., Leid, M. & Rothenberg, E. V. An early T cell lineage commitment checkpoint dependent on the transcription factor Bcl11b. Science 329, 89–93 (2010).
Ikawa, T. et al. An essential developmental checkpoint for production of the T cell lineage. Science 329, 93–96 (2010).
Li, P. et al. Reprogramming of T cells to natural killer-like cells upon Bcl11b deletion. Science 329, 85–89 (2010).
Kueh, H. Y. et al. Asynchronous combinatorial action of four regulatory factors activates Bcl11b for T cell commitment. Nat. Immunol. 17, 956–965 (2016).
Albu, D. I. et al. Transcription factor Bcl11b controls selection of invariant natural killer T-cells by regulating glycolipid presentation in double-positive thymocytes. Proc. Natl Acad. Sci. USA 108, 6211–6216 (2011).
Vanvalkenburgh, J. et al. Critical role of Bcl11b in suppressor function of T regulatory cells and prevention of inflammatory bowel disease. J. Exp. Med. 208, 2069–2081 (2011).
Walker, J. A. et al. Bcl11b is essential for group 2 innate lymphoid cell development. J. Exp. Med. 212, 875–882 (2015).
Yu, Y. et al. The transcription factor Bcl11b is specifically expressed in group 2 innate lymphoid cells and is essential for their development. J. Exp. Med. 212, 865–874 (2015).
Califano, D. et al. Transcription factor Bcl11b controls identity and function of mature type 2 innate lymphoid cells. Immunity 43, 354–368 (2015).
Hogquist, K. A. & Jameson, S. C. The self-obsession of T cells: how TCR signaling thresholds affect fate ‘decisions’ and effector function. Nat. Immunol. 15, 815–823 (2014).
Singer, A., Adoro, S. & Park, J. H. Lineage fate and intense debate: myths, models and mechanisms of CD4- versus CD8-lineage choice. Nat. Rev. Immunol. 8, 788–801 (2008).
Taniuchi, I. & Ellmeier, W. Transcriptional and epigenetic regulation of CD4/CD8 lineage choice. Adv. Immunol. 110, 71–110 (2011).
Ellmeier, W. & Taniuchi, I. The role of BTB-zinc finger transcription factors during T cell development and in the regulation of T cell-mediated immunity. Curr. Top Microb. Immunol. 381, 21–49 (2014).
He, X. et al. The zinc finger transcription factor Th-POK regulates CD4 versus CD8 T-cell lineage commitment. Nature 433, 826–833 (2005).
Sun, G. et al. The zinc finger protein cKrox directs CD4 lineage differentiation during intrathymic T cell positive selection. Nat. Immunol. 6, 373–381 (2005).
He, X., Park, K. & Kappes, D. J. The role of ThPOK in control of CD4/CD8 lineage commitment. Annu. Rev. Immunol. 28, 295–320 (2010).
Setoguchi, R. et al. Repression of the transcription factor Th-POK by Runx complexes in cytotoxic T cell development. Science 319, 822–825 (2008).
He, X. et al. CD4-CD8 lineage commitment is regulated by a silencer element at the ThPOK transcription-factor locus. Immunity 28, 346–358 (2008).
Muroi, S., Tanaka, H., Miyamoto, C. & Taniuchi, I. Cutting edge: fine-tuning of Thpok gene activation by an enhancer in close proximity to its own silencer. J. Immunol. 190, 1397–1401 (2013).
Muroi, S. et al. Cascading suppression of transcriptional silencers by ThPOK seals helper T cell fate. Nat. Immunol. 9, 1113–1121 (2008).
Sakaguchi, S. et al. The zinc-finger protein MAZR is part of the transcription factor network that controls the CD4 versus CD8 lineage fate of double-positive thymocytes. Nat. Immunol. 11, 442–448 (2010).
Sakaguchi, S. et al. MAZR and runx factors synergistically repress ThPOK during CD8+T cell lineage development. J. Immunol. 195, 2879–2887 (2015).
Wang, L. et al. Distinct functions for the transcription factors GATA-3 and ThPOK during intrathymic differentiation of CD4+ T cells. Nat. Immunol. 9, 1122–1130 (2008).
Steinke, F. C. et al. TCF-1 and LEF-1 act upstream of Th-POK to promote the CD4+ T cell fate and interact with Runx3 to silence Cd4 in CD8+ T cells. Nat. Immunol. 15, 646–656 (2014).
Egawa, T. & Littman, D. R. ThPOK acts late in specification of the helper T cell lineage and suppresses Runx-mediated commitment to the cytotoxic T cell lineage. Nat. Immunol. 9, 1131–1139 (2008).
Park, J. H. et al. Signaling by intrathymic cytokines, not T cell antigen receptors, specifies CD8 lineage choice and promotes the differentiation of cytotoxic-lineage T cells. Nat. Immunol. 11, 257–264 (2010).
Luckey, M. A. et al. The transcription factor ThPOK suppresses Runx3 and imposes CD4+ lineage fate by inducing the SOCS suppressors of cytokine signaling. Nat. Immunol. 15, 638–645 (2014).
Taniuchi, I. Views on helper/cytotoxic lineage choice from a bottom-up approach. Immunol. Rev. 271, 98–113 (2016).
Kastner, P. et al. Bcl11b represses a mature T-cell gene expression program in immature CD4+CD8+thymocytes. Eur. J. Immunol. 40, 2143–2154 (2010).
Seo, W. et al. Roles of VWRPY motif-mediated gene repression by Runx proteins during T-cell development. Immunol. Cell Biol. 90, 827–830 (2012).
Huang, G. et al. PU.1 is a major downstream target of AML1 (RUNX1) in adult mouse hematopoiesis. Nat. Genet. 40, 51–60 (2008).
Tang, B. et al. Genome-wide identification of Bcl11b gene targets reveals role in brain-derived neurotrophic factor signaling. PLoS ONE 6, e23691 (2011).
Tanaka, H. et al. Epigenetic Thpok silencing limits the time window to choose CD4+helper-lineage fate in the thymus. EMBO J. 32, 1183–1194 (2013).
Wakabayashi, Y. et al. Bcl11b is required for differentiation and survival of alphabeta T lymphocytes. Nat. Immunol. 4, 533–539 (2003).
Buenrostro, J. D., Giresi, P. G., Zaba, L. C., Chang, H. Y. & Greenleaf, W. J. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods 10, 1213–1218 (2013).
Wang, L. et al. The zinc finger transcription factor Zbtb7b repressesCD8-lineage gene expression in peripheral CD4+ T cells. Immunity 29, 876–887 (2008).
Zetsche, B. et al. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 163, 759–771 (2015).
Jenkinson, S. R. et al. Expression of the transcription factor cKrox in peripheral CD8 T cells reveals substantial postthymic plasticity in CD4-CD8 lineage differentiation. J. Exp. Med. 204, 267–272 (2007).
Kakugawa, K. et al. Essential roles of SATB1 in specifying T lymphocyte subsets. Cell Rep. 19, 1176–1188 (2017).
Feng, Y. et al. A mechanism for expansion of regulatory T-cell repertoire and its role in self-tolerance. Nature 528, 132–136 (2015).
Zheng, Y. et al. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature 463, 808–812 (2010).
Kitagawa, Y. et al. Guidance of regulatory T cell development by Satb1-dependent super-enhancer establishment. Nat. Immunol. 18, 173–183 (2017).
Lee, H. O. et al. Disregulated expression of the transcription factor ThPOK during T-cell development leads to high incidence of T-cell lymphomas. Proc. Natl. Acad. Sci. USA 112, 7773–7778 (2015).
Kronenberg, M. & Rudensky, A. Regulation of immunity by self-reactive T cells. Nature 435, 598–604 (2005).
Fujita, T. & Fujii, H. Direct identification of insulator components by insertional chromatin immunoprecipitation. PLoS ONE 6, e26109 (2011).
Iuchi, S. Three classes of C2H2 zinc finger proteins. Cell Mol. Life Sci. 58, 625–635 (2001).
Sankaran, V. G. et al. Developmental and species-divergent globin switching are driven by BCL11A. Nature 460, 1093–1097 (2009).
Nishimura, M. et al. VWRPY motif-dependent and -independent roles of AML1/Runx1 transcription factor in murine hematopoietic development. Blood 103, 562–570 (2004).
Naoe, Y. et al. Repression of interleukin-4 in T helper type 1 cells by Runx/Cbfβ binding to the Il4 silencer. J. Exp. Med. 204, 1749–1755 (2007).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Wang, H. et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell 153, 910–918 (2013).
Egawa, T., Tillman, R. E., Naoe, Y., Taniuchi, I. & Littman, D. R. The role of the Runx transcription factors in thymocyte differentiation and in homeostasis of naive T cells. J. Exp. Med. 204, 1945–1957 (2007).
Buenrostro, J. D., Wu, B., Chang, H. Y. & Greenleaf, W. J. ATAC-seq: a method for assaying chromatin accessibility genome-wide. Curr. Proto. Mol. Biol. 109, 21.29.1–21.29.9 (2015).
Mucida, D. et al. Transcriptional reprogramming of mature CD4+ helper T cells generates distinct MHC class II-restricted cytotoxic T lymphocytes. Nat. Immunol. 14, 281–289 (2013).
Acknowledgements
We thank C. Miyamoto and R. Chihara for mouse genotyping, N. Yoza for cell sorting, T. Ishikura for ES cell aggregation, Y. Iizuka for microinjection of RNA for CRISPR/Cas9 mediated genome editing. We are grateful to Dr Thomas Boehm for helpful suggestion on analyses of lamprey genome and Dr Eugene Oltz for critical reading of the manuscript. This work was supported by JSPS KAKENHI Grant Number JP 21229008 (I.T.), JP 26293109 (I.T.) and JP 22021045 (I.T.), the Uehara Memorial Foundation (I.T.), Takeda Science Foundation (I.T.) the National Institutes of Health grant R01AI 097244-01A1 (T.E.).
Author information
Authors and Affiliations
Contributions
S.K., H.T., W.S., K.K., Y.N., and I.T. performed experiments for phenotypic characterization of Bcl11b mutant mice. M.T. and K.M. performed reconstitution experiments. S.M. isolated ES cell clones. T.A.E. performed statistical and comparative genomics analyses. K.N. made mutant Bcl11b expression vectors. S.K., A.K., and T.I. performed sequencing of ChIP libraries. T.E. provided anti-ThPOK antibody. Y.L. and A.M. performed ATAC-seq. B.V. analyzed Japanese lamprey genome. Y.K. and R.K. provided Bcl11b germline KO mice. I.T. designed experiments, interpreted data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kojo, S., Tanaka, H., Endo, T.A. et al. Priming of lineage-specifying genes by Bcl11b is required for lineage choice in post-selection thymocytes. Nat Commun 8, 702 (2017). https://doi.org/10.1038/s41467-017-00768-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-017-00768-1
This article is cited by
-
Epigenetic reprogramming of T cells: unlocking new avenues for cancer immunotherapy
Cancer and Metastasis Reviews (2024)
-
Identification of key transcription factors and their functional role involved in Salmonella typhimurium infection in chicken using integrated transcriptome analysis and bioinformatics approach
BMC Genomics (2023)
-
Downregulation of chemokine receptor 9 facilitates CD4+CD8αα+ intraepithelial lymphocyte development
Nature Communications (2023)
-
TCF1 in T cell immunity: a broadened frontier
Nature Reviews Immunology (2022)
-
Human induced-T-to-natural killer cells have potent anti-tumour activities
Biomarker Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.