Abstract
Arterial hypertension is associated with increased morbidity and mortality and research in the field is highly dynamic. This summary reviews the most important clinical trials published in 2022 and early 2023. Findings on new pharmacological approaches to treat resistant hypertension are presented and new knowledge about the optimal timing of the antihypertensive medication intake is discussed. It is focused on optimal blood pressure treatment targets and the problem of treatment and guideline inertia is acknowledged. Information about pregnancy-related hypertension is presented and blood pressure control following percutaneous thrombectomy after ischemic stroke is discussed. Finally, novel clinical data on device-based approaches to treat hypertension are summarized.

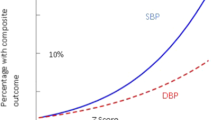
The hypertension trials update summarizes the most important clincal trials on hypertension research in 2022 and early 2023. CTD – chlorthalidone, CV – cardiovascular, HCT – hydrochlorothiazide, SBP – systolic blood pressure, RDN – renal denervation *depicts systolic blood pressure only.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dicker D, Nguyen G, Abate D, Abate KH, Abay SM, Abbafati C, et al. Global, regional, and national age-sex-specific mortality and life expectancy, 1950–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1684–735. https://doi.org/10.1016/S0140-6736(18)31891-9
Forouzanfar MH, Liu P, Roth GA, Ng M, Biryukov S, Marzcak L, et al. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mm Hg, 1990-2015. JAMA. 2017;317:165–82. https://doi.org/10.1001/jama.2016.19043
Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M, et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur Heart J. 2018;39:3021–104. https://doi.org/10.1093/eurheartj/ehy339
Carey RM, Calhoun DA, Bakris GL, Brook RD, Daugherty SL, Dennison-Himmelfarb CR et al. Resistant hypertension: detection, evaluation, and management: A scientific statement from the American Heart Association. Hypertension. 2018;72. https://doi.org/10.1161/HYP.0000000000000084
Carey RM, Sakhuja S, Calhoun DA, Whelton PK, Muntner P. Prevalence of apparent treatment-resistant hypertension in the United States. Hypertension. 2019;73:424–31. https://doi.org/10.1161/HYPERTENSIONAHA.118.12191
Daugherty SL, Powers JD, Magid DJ, Tavel HM, Masoudi FA, Margolis KL, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation. 2012;125:1635–42. https://doi.org/10.1161/CIRCULATIONAHA.111.068064
Myat A, Redwood SR, Qureshi AC, Spertus JA, Williams B. Resistant hypertension. BMJ. 2012;345:e7473. https://doi.org/10.1136/bmj.e7473
Williams B. Resistant hypertension: an unmet treatment need. Lancet. 2009;374:1396–8. https://doi.org/10.1016/S0140-6736(09)61600-7
Sarafidis PA, Bakris GL. Resistant hypertension: an overview of evaluation and treatment. J Am Coll Cardiol. 2008;52:1749–57. https://doi.org/10.1016/j.jacc.2008.08.036
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Dennison Himmelfarb C, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults. J Am Coll Cardiol. 2018;71:e127–e248. https://doi.org/10.1016/j.jacc.2017.11.006
Götzinger F, Kunz M, Lauder L, Böhm M, Mahfoud F. Arterial hypertension - clinical trials update 2022. Hypertens Res. 2022;45:1140–6. https://doi.org/10.1038/s41440-022-00931-2
Esler MD, Krum H, Sobotka PA, Schlaich MP, Schmieder RE, Böhm M. Renal sympathetic denervation in patients with treatment-resistant hypertension (The Symplicity HTN-2 Trial): a randomised controlled trial. Lancet. 2010;376:1903–9. https://doi.org/10.1016/S0140-6736(10)62039-9
Bhatt DL, Vaduganathan M, Kandzari DE, Leon MB, Rocha-Singh K, Townsend RR, et al. Long-term outcomes after catheter-based renal artery denervation for resistant hypertension: final follow-up of the randomised SYMPLICITY HTN-3 Trial. Lancet. 2022;400:1405–16. https://doi.org/10.1016/S0140-6736(22)01787-1
Esler MD, Böhm M, Sievert H, Rump CL, Schmieder RE, Krum H, et al. Catheter-based renal denervation for treatment of patients with treatment-resistant hypertension: 36 month results from the SYMPLICITY HTN-2 randomized clinical trial. Eur Heart J. 2014;35:1752–9. https://doi.org/10.1093/eurheartj/ehu209
Freeman MW, Bond M, Murphy B, Hui J, Isaacsohn J. Results from a phase 1, randomized, double-blind, multiple ascending dose study characterizing the pharmacokinetics and demonstrating the safety and selectivity of the aldosterone synthase inhibitor baxdrostat in healthy volunteers. Hypertens Res. 2023;46:108–18. https://doi.org/10.1038/s41440-022-01070-4
Freeman MW, Halvorsen YD, Marshall W, Pater M, Isaacsohn J, Pearce C, et al. Phase 2 trial of baxdrostat for treatment-resistant hypertension. New Eng J Med. 2023;388:395–405. https://doi.org/10.1056/NEJMoa2213169
Fagard RH, Thijs L, Staessen JA, Clement DL, de Buyzere ML, de Bacquer DA. Night–day blood pressure ratio and dipping pattern as predictors of death and cardiovascular events in hypertension. J Hum Hypertens. 2009;23:645–53. https://doi.org/10.1038/jhh.2009.9
Kario K. Morning surge in blood pressure and cardiovascular risk. Hypertension. 2010;56:765–73. https://doi.org/10.1161/HYPERTENSIONAHA.110.157149
Kario K, Hoshide S, Mizuno H, Kabutoya T, Nishizawa M, Yoshida T, et al. Nighttime hemodynamic phenotype. A novel risk factor for cardiovascular disease, especially heart failure: the practitioner-based nationwide JAMP study. Clin Res Cardiol. 2023;112:98–110. https://doi.org/10.1007/s00392-022-02051-w
Hermida RC, Calvo C, Ayala DE, Fernández JR, Covelo M, Mojón A, et al. Treatment of non-dipper hypertension with bedtime administration of valsartan. J Hypertens. 2005;23:1913–22. https://doi.org/10.1097/01.hjh.0000182522.21569.c5
Ho CLB, Chowdhury EK, Doust J, Nelson MR, Reid CM. The effect of taking blood pressure lowering medication at night on cardiovascular disease risk. A systematic review. J Hum Hypertens. 2021;35:308–14. https://doi.org/10.1038/s41371-020-00469-1
Mackenzie IS, Rogers A, Poulter NR, Williams B, Brown MJ, Webb DJ, et al. Cardiovascular outcomes in adults with hypertension with evening versus morning dosing of usual antihypertensives in the UK (TIME study): a prospective, randomised, open-label, blinded-endpoint clinical trial. Lancet. 2022;400:1417–25. https://doi.org/10.1016/S0140-6736(22)01786-X
SPRINT Research Group, Wright JT, Williamson JD, Whelton PK, Snyder JK, Sink KM, et al. A randomized trial of intensive versus standard blood-pressure control. N Enl J Med. 2015;373:2103–16. https://doi.org/10.1056/NEJMoa1511939
Jaeger BC, Bress AP, Bundy JD, Cheung AK, Cushman WC, Drawz PE, et al. Longer-term all-cause and cardiovascular mortality with intensive blood pressure control. JAMA Cardiol. 2022;7:1138 https://doi.org/10.1001/jamacardio.2022.3345
Agarwal R. Hydrochlorothiazide versus chlorthalidone: What is the difference. Circulation. 2022;146:1641–3. https://doi.org/10.1161/CIRCULATIONAHA.122.061029
Dorsch MP, Gillespie BW, Erickson SR, Bleske BE, Weder AB. Chlorthalidone reduces cardiovascular events compared with hydrochlorothiazide. Hypertension. 2011;57:689–94. https://doi.org/10.1161/HYPERTENSIONAHA.110.161505
Ishani A, Cushman WC, Leatherman SM, Lew RA, Woods P, Glassman PA, et al. Chlorthalidone vs. Hydrochlorothiazide for hypertension–cardiovascular events. N Engl J Med. 2022;387:2401–10. https://doi.org/10.1056/NEJMoa2212270
Pathak A, Poulter NR, Kavanagh M, Kreutz R, Burnier M. Improving the management of hypertension by tackling awareness, adherence, and clinical inertia: a symposium report. Am J Cardiovasc Drugs. 2022;22:251–61. https://doi.org/10.1007/s40256-021-00505-6
Ali DH, Kiliç B, Hart HE, Bots ML, Biermans MCJ, Spiering W, et al. Therapeutic inertia in the management of hypertension in primary care. J Hypertens. 2021;39:1238–45. https://doi.org/10.1097/HJH.0000000000002783
Ferrari P, Hess L, Pechere-Bertschi A, Muggli F, Burnier M. Reasons for not intensifying antihypertensive treatment (RIAT). J Hypertens. 2004;22:1221–9. https://doi.org/10.1097/00004872-200406000-00024
Parati G, Kjeldsen S, Coca A, Cushman WC, Wang J. Adherence to single-pill versus free-equivalent combination therapy in hypertension. Hypertension.2021;77:692–705. https://doi.org/10.1161/HYPERTENSIONAHA.120.15781.
Mahfoud F, Kieble M, Enners S, Kintscher U, Laufs U, Böhm M, et al. Use of fixed-dose combination antihypertensives in Germany between 2016 and 2020: an example of guideline inertia. Clin Res Cardiol. 2023;112:197–202. https://doi.org/10.1007/s00392-022-01993-5
Schmieder RE, Wassmann S, Predel HG, Weisser B, Blettenberg J, Gillessen A, et al. Improved persistence to medication, decreased cardiovascular events and reduced all-cause mortality in hypertensive patients with use of single-pill combinations: results from the START-study. Hypertension. 2023;80:1127–35. https://doi.org/10.1161/HYPERTENSIONAHA.122.20810
Say L, Chou D, Gemmill A, Tunçalp Ö, Moller AB, Daniels J, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014;2:e323–e333. https://doi.org/10.1016/S2214-109X(14)70227-X
Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Obstet Gynecol. 2013;122:1122-31. https://doi.org/10.1097/01.AOG.0000437382.03963.88
Tita AT, Szychowski JM, Boggess K, Dugoff L, Sibai B, Lawrence K, et al. Treatment for mild chronic hypertension during pregnancy. N Eng J Med. 2022;386:1781–92. https://doi.org/10.1056/NEJMoa2201295
Feigin VL, Stark BA, Johnson CO, Roth GA, Bisignano C, Abady GG, et al. Global, regional, and national burden of stroke and its risk factors, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021;20:795–820. https://doi.org/10.1016/S1474-4422(21)00252-0
Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K et al. 2018 Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2018;49. https://doi.org/10.1161/STR.0000000000000158
Feske SK. Ischemic stroke. Am J Med. 2021;134:1457–64. https://doi.org/10.1016/j.amjmed.2021.07.027
Yang P, Song L, Zhang Y, Zhang X, Chen X, Li Y, et al. Intensive blood pressure control after endovascular thrombectomy for acute ischaemic stroke (ENCHANTED2/MT): a multicentre, open-label, blinded-endpoint, randomised controlled trial. Lancet. 2022;400:1585–96. https://doi.org/10.1016/S0140-6736(22)01882-7
Schlaich MP, Sobotka PA, Krum H, Lambert E, Esler MD. Renal sympathetic-nerve ablation for uncontrolled hypertension. N Engl J Med. 2009;361:932–4. https://doi.org/10.1056/NEJMc0904179
Parati G, Esler M. The human sympathetic nervous system: its relevance in hypertension and heart failure. Eur Heart J. 2012;33:1058–66. https://doi.org/10.1093/eurheartj/ehs041
Mahfoud F, Kandzari DE, Kario K, Townsend RR, Weber MA, Schmieder RE, et al. Long-term efficacy and safety of renal denervation in the presence of antihypertensive drugs (SPYRAL HTN-ON MED): a randomised, sham-controlled trial. Lancet. 2022;399:1401–10. https://doi.org/10.1016/S0140-6736(22)00455-X
Kandzari DE, Böhm M, Mahfoud F, Townsend RR, Weber MA, Pocock S, et al. Effect of renal denervation on blood pressure in the presence of antihypertensive drugs: 6-month efficacy and safety results from the SPYRAL HTN-ON MED proof-of-concept randomised trial. Lancet. 2018;391:2346–55. https://doi.org/10.1016/S0140-6736(18)30951-6
Kario K, Mahfoud F, Kandzari DE, Townsend RR, Weber MA, Schmieder RE, et al. Long-term reduction in morning and nighttime blood pressure after renal denervation: 36-month results from SPYRAL HTN-ON MED trial. Hypertens Res. 2023;46:280–8. https://doi.org/10.1038/s41440-022-01042-8
Azizi M, Saxena M, Wang Y, Jenkins JS, Devireddy C, Rader F, et al. Endovascular ultrasound renal denervation to treat hypertension. JAMA. 2023;329:651 https://doi.org/10.1001/jama.2023.0713
Mahfoud F, Mancia G, Schmieder RE, Ruilope L, Narkiewicz K, Schlaich M, et al. Cardiovascular risk reduction after renal denervation according to time in therapeutic systolic blood pressure range. J Am Coll Cardiol. 2022;80:1871–80. https://doi.org/10.1016/j.jacc.2022.08.802
Böhm M, Kario K, Kandzari DE, Mahfoud F, Weber MA, Schmieder RE, et al. Efficacy of catheter-based renal denervation in the absence of antihypertensive medications (SPYRAL HTN-OFF MED Pivotal): a multicentre, randomised, sham-controlled trial. Lancet. 2020;395:1444–51. https://doi.org/10.1016/S0140-6736(20)30554-7
Azizi M, Sanghvi K, Saxena M, Gosse P, Reilly JP, Levy T, et al. Ultrasound renal denervation for hypertension resistant to a triple medication pill (RADIANCE-HTN TRIO): a randomised, multicentre, single-blind, sham-controlled trial. Lancet. 2021;397:2476–86. https://doi.org/10.1016/S0140-6736(21)00788-1
Azizi M, Schmieder RE, Mahfoud F, Weber MA, Daemen J, Davies J, et al. Endovascular ultrasound renal denervation to treat hypertension (RADIANCE-HTN SOLO): a multicentre, international, single-blind, randomised, sham-controlled trial. Lancet. 2018;391:2335–45. https://doi.org/10.1016/S0140-6736(18)31082-1
Barbato E, Azizi M, Schmieder RE, Lauder L, Böhm M, Brouwers S et al. Renal denervation in the management of hypertension in adults. A clinical consensus statement of the ESC Council on Hypertension and the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J. Published online February 15, 2023. https://doi.org/10.1093/eurheartj/ehad054
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
FG received speaker honoraria from AstraZeneca and is supported by the German Heart Foundation/German Foundation of Heart Research. LL received speaker honoraria from Medtronic and ReCor Medical. MB is supported by Abbott, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Deutsche Forschungsgemeinschaft (DFG) (TRR 219 Project-ID 322900939, S-01, M-02 and M-06), Medtronic, Novartis, ReCor Medical, Servier, and Vifor. FM is supported by Deutsche Gesellschaft für Kardiologie (DGK), DFG (SFB TRR 219 Project-ID 322900939) and by the German Heart Foundation/German Foundation of Heart Research and has received scientific support and speaker honoraria from Bayer, Boehringer Ingelheim, Medtronic, and ReCor Medical. MK has nothing to declare.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Götzinger, F., Kunz, M., Lauder, L. et al. Arterial Hypertension—clinical trials update 2023. Hypertens Res 46, 2159–2167 (2023). https://doi.org/10.1038/s41440-023-01359-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-023-01359-y