Abstract
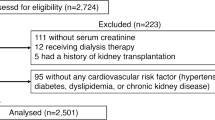
Atherosclerosis and arterial stiffness are phenotypes of atherosclerotic vascular damage. Atherosclerosis originates from endothelial vascular damage and forms focal morphological lesions; arterial stiffness originates from diffuse medial-layer damage in the arterial tree. Thus, the two phenomena reflect different facets of atherosclerotic vascular damage, and they both gradually progress. We conducted a subanalysis to compare the long-term effects of febuxostat on atherosclerosis and arterial stiffness in the PRIZE study (a multicenter, prospective, randomized, open-label, blinded-endpoint clinical trial to examine the effect of febuxostat on carotid atherosclerosis). Among 514 study participants, arterial stiffness parameters (brachial-ankle pulse wave velocity or cardio-ankle vascular index) were obtained at baseline, 12 months, and 24 months in 100 subjects. Among them, 48 subjects were allocated to the control group (i.e., nonpharmacological lifestyle modification for hyperuricemia), and 52 subjects were allocated to the febuxostat treatment group. While the decrease in serum uric acid was greater in the febuxostat group than in the control group, the adjusted percentage decrease in arterial stiffness parameters at month 24 was greater in the febuxostat group than in the control group, with a mean between-group difference (febuxostat − control) of −5.099% (95% confidence interval (CI) −10.009% to −0.188%, p = 0.042). Thus, long-term treatment with febuxostat may exert beneficial effects on arterial stiffness without improving carotid atherosclerosis. A long-term study to examine the effect of febuxostat on cardiovascular outcomes related to increased arterial stiffness is warranted.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kleber ME, Delgado G, Grammer TB, Silbernagel G, Huang J, Krämer BK, et al. Uric acid and cardiovascular events: a Mendelian Randomization Study. J Am Soc Nephrol. 2015;26:2831–8.
White J, Sofat R, Hemani G, Shah T, Engmann J, Dale C, et al. Plasma urate concentration and risk of coronary heart disease: a Mendelian randomisation analysis. Lancet Diabetes Endocrinol. 2016;4:327–36.
Ma M, Wang L, Huang W, Zhong X, Li L, Wang H, et al. Meta-analysis of the correlation between serum uric acid level and carotid intima-media thickness. PLoS ONE. 2021;16:e0246416 https://doi.org/10.1371/journal.pone.0246416.
Becker MA, Schumacher HR Jr, Wortmann RL, MacDonald PA, Eustace D, Palo WA, et al. Febuxostat compared with allopurinol in patients with hyperuricemia and gout. N Engl J Med. 2005;353:2450–61.
Tanaka A, Taguchi I, Teragawa H, Ishizaka N, Kanzaki Y, Tomiyama H, et al. Febuxostat does not delay progression of carotid atherosclerosis in patients with asymptomatic hyperuricemia: a randomized, controlled trial. PLoS Med. 2020;17:e1003095 https://doi.org/10.1371/journal.pmed.1003095.
Bos D, Arshi B, van den Bouwhuijsen QJA, Ikram MK, Selwaness M, Vernooij MW, et al. Atherosclerotic carotid plaque composition and incident stroke and coronary events. J Am Coll Cardiol. 2021;77:1426–35.
Libby P. The changing landscape of atherosclerosis. Nature 2021;592:524–33.
Chirinos JA, Segers P, Hughes T, Townsend R. Large-artery stiffness in health and disease: JACC state-of-the-art review. J Am Coll Cardiol. 2019;74:1237–63.
Tomiyama H, Yamashina A. Non-invasive vascular function tests: their pathophysiological background and clinical application. Circ J. 2010;74:24–33.
Tomiyama H, Shiina K. State of the art review: brachial-ankle PWV. J Atheroscler Thromb. 2020;27:621–36.
Kinoshita M, Yokote K, Arai H, Iida M, Ishigaki Y, Ishibashi S, et al. Japan Atherosclerosis Society (JAS) guidelines for prevention of atherosclerotic cardiovascular diseases 2017. J Atheroscler Thromb. 2018;25:846–984.
Oyama J, Tanaka A, Sato Y, Tomiyama H, Sata M, Ishizu T, et al. Rationale and design of a multicenter randomized study for evaluating vascular function under uric acid control using the xanthine oxidase inhibitor, febuxostat: the PRIZE study. Cardiovasc Diabetol. 2016;15:87 https://doi.org/10.1186/s12933-016-0409-2.
Shimamoto K, Ando K, Fujita T, Hasebe N, Higaki J, Horiuchi M, et al. Japanese Society of Hypertension Committee for Guidelines for the Management of Hypertension. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2014). Hypertens Res. 2014;37:253–390. https://doi.org/10.1038/hr.2014.20
Yamashina A, Tomiyama H, Takeda K, Tsuda H, Arai T, Hirose K, et al. Validity, reproducibility, and clinical significance of noninvasive brachial-ankle pulse wave velocity measurement. Hypertens Res. 2002;25:359–64. https://doi.org/10.1291/hypres.25.359.
Shirai K, Hiruta N, Song M, Kurosu T, Suzuki J, Tomaru T, et al. Cardio-ankle vascular index (CAVI) as a novel indicator of arterial stiffness: theory, evidence and perspectives. J Atheroscler Thromb. 2011;18:924–38. https://doi.org/10.5551/jat.7716.
Stein JH, Korcarz CE, Hurst RT, Lonn E, Kendall CB, Mohler ER, et al. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: a consensus statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force. Endorsed by the Society for Vascular Medicine. J Am Soc Echocardiogr. 2008;21:93–111.
Lundby-Christensen L, Almdal TP, Carstensen B, Tarnow L, Wiinberg N. Carotid intima-media thickness in individuals with and without type 2 diabetes: a reproducibility study. Cardiovasc Diabetol. 2010;9:40. pmid:20727128
Tomiyama H, Shiina K, Vlachopoulos C, Iwasaki Y, Matsumoto C, Kimura K, et al. Involvement of arterial stiffness and inflammation in hyperuricemia-related development of hypertension. Hypertension. 2018;72:739–45. https://doi.org/10.1161/HYPERTENSIONAHA.118.11390.
Canepa M, Viazzi F, Strait JB, Ameri P, Pontremoli R, Brunelli C, et al. Longitudinal association between serum uric acid and arterial stiffness: results from the Baltimore Longitudinal Study of Aging. Hypertension. 2017;69:228–35. https://doi.org/10.1161/HYPERTENSIONAHA.116.08114.
Deng G, Qiu Z, Li D, Fang Y, Zhang S. Effects of allopurinol on arterial stiffness: a meta-analysis of randomized controlled trials. Med Sci Monit. 2016;22:1389–97.
Desideri G, Rajzer M, Gerritsen M, Nurmohamed MT, Giannattasio C, Tausche AK, et al. Effects of intensive urate lowering therapy with febuxostat in comparison with allopurinol on pulse wave velocity in patients with gout and increased cardiovascular risk: the FORWARD study. Eur Heart J Cardiovasc Pharmacother. 2021:pvaa144. https://doi.org/10.1093/ehjcvp/pvaa144.
Kario K, Nishizawa M, Kiuchi M, Kiyosue A, Tomita F, Ohtani H, et al. Comparative effects of topiroxostat and febuxostat on arterial properties in hypertensive patients with hyperuricemia. J Clin Hypertens. 2021;23:334–44.
Goicoechea M, Vinuesa SG, Verdalles U, Ruiz-Caro C, Ampuero J, Rincón A, et al. Effect of allopurinol in chronic kidney disease progression and cardiovascular risk. Clin J Am Soc Nephrol. 2010;5:1388–93.
Hosoya T, Ohno I, Nomura S, Hisatome I, Uchida S, Fujimori S, et al. Effects of topiroxostat on the serum urate levels and urinary albumin excretion in hyperuricemic stage 3 chronic kidney disease patients with or without gout. Clin Exp Nephrol. 2014;18:876–84.
Saag KG, Whelton A, Becker MA, MacDonald P, Hunt B, Gunawardhana L. Impact of febuxostat on renal function in gout patients with moderate-to-severe renal impairment. Arthritis Rheumatol. 2016;68:2035–43.
Ponticelli C, Podestà MA, Moroni G. Hyperuricemia as a trigger of immune response in hypertension and chronic kidney disease. Kidney Int. 2020;98:1149–59.
Cicalese S, Scalia R, Eguchi S. Xanthine oxidase inhibition as a potential treatment for aortic stiffness in hypertension. Am J Hypertens. 2019;32:234–6.
Matesanz N, Lafuente N, Azcutia V, Martín D, Cuadrado A, Nevado J, et al. Xanthine oxidase-derived extracellular superoxide anions stimulate activator protein 1 activity and hypertrophy in human vascular smooth muscle via c-Jun N-terminal kinase and p38 mitogen-activated protein kinases. J Hypertens. 2007;25:609–18. PMID: 17278978.
Kondo M, Imanishi M, Fukushima K, Ikuto R, Murai Y, Horinouchi Y, et al. Xanthine oxidase inhibition by febuxostat in macrophages suppresses angiotensin II-induced aortic fibrosis. Am J Hypertens. 2019;32:249–56.
Amer MS, Khater MS, Omar OH, Mabrouk RA, Mostafa SA. Association between Framingham risk score and subclinical atherosclerosis among elderly with both type 2 diabetes mellitus and healthy subjects. Am J Cardiovasc Dis. 2014;4:14–9.
Wang TJ, Wollert KC, Larson MG, Coglianese E, McCabe EL, Cheng S, et al. Prognostic utility of novel biomarkers of cardiovascular stress: the Framingham Heart Study. Circulation. 2012;126:1596–604. https://doi.org/10.1161/CIRCULATIONAHA.112.129437.
Laurent S, Chatellier G, Azizi M, Calvet D, Choukroun G, Danchin N, et al. SPARTE Study: normalization of arterial stiffness and cardiovascular events in patients with hypertension at medium to very high risk. Hypertension. 2021;78:983–95. https://doi.org/10.1161/HYPERTENSIONAHA.121.17579.
Kojima S, Matsui K, Hiramitsu S, Hisatome I, Waki M, Uchiyama K, et al. Febuxostat for Cerebral and CaRdiorenovascular Events PrEvEntion StuDy. Eur Heart J. 2019;40:1778–86. https://doi.org/10.1093/eurheartj/ehz119.
Tsuchihashi T. Which is more important, xanthine oxidase activity or uric acid itself, in the risk for cardiovascular disease? Hypertens Res 2021;44:1543–45.
Guo QH, Muhammad IF, Borné Y, Sheng CS, Persson M, Wang JG, et al. Difference in the risk profiles of carotid-femoral pulse wave velocity: results from two community-based studies in China and Sweden. J Hum Hypertens. 2020;34:207–13.
Shirai K, Utino J, Otsuka K, Takata M. A novel blood pressure-independent arterial wall stiffness parameter; cardio-ankle vascular index (CAVI). J Atheroscler Thromb. 2006;13:101–7. https://doi.org/10.5551/jat.13.101.
Kubozono T, Miyata M, Ueyama K, Nagaki A, Otsuji Y, Kusano K, et al. Clinical significance and reproducibility of new arterial distensibility index. Circ J. 2007;71:89–94. https://doi.org/10.1253/circj.71.89.
Acknowledgements
The authors thank the members of the Data and Safety Monitoring Board (Hiroyuki Daida, MD, PhD (Juntendo University Graduate School of Medicine, Tokyo, Japan); Junya Ako, MD, PhD, (Kitasato University School of Medicine, Sagamihara, Japan); Kazuo Kitagawa, MD, PhD (Tokyo Women’s Medical University, Tokyo, Japan)) and the Clinical Events Committee (Wataru Shimizu, MD, PhD (Nippon Medical School, Tokyo, Japan); Yoshio Kobayashi, MD, PhD (Chiba University Graduate School of Medicine, Chiba, Japan) and Masaharu Ishihara (Hyogo College of Medicine, Nishinomiya, Japan)). We also thank Yasunori Sato, PhD (Keio University School of Medicine, Tokyo, Japan), and Shusuke Tani, PhD (Nouvelle Place Inc., Tokyo, Japan), for their statistical support, and we thank Mikiko Kagiyama (Saga University, Saga, Japan) and Itsuka Suzuki (Nouvelle Place Inc., Tokyo, Japan) for secretarial assistance.
Author contributions
All authors helped conceptualize this trial. KS and HT wrote the draft of the article. HY performed statistical analysis. All authors confirmed the data collection and study selection criteria. All authors enrolled patients and performed study quality assessment. All authors approved the final version of the manuscript.
Funding
This work was funded by Teijin Pharma Limited, Japan (to KN). The funder of the trial had no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
KS and HT were supported by Omron, Asahi Calpis Wellness and Teijin Pharma. AT has received honoraria from Boehringer Ingelheim and research funding from GlaxoSmithKline. KK received significant research grants from Teijin Pharma and Fukuda Denshi. HT has received honoraria from Abbott Medical Japan, Bayer, Boehringer Ingelheim, Daiichi Sankyo, Kowa, Ono, Mitsubishi Tanabe, and Takeda. YF has received research grants from Sanofi KK and Shionogi & Co. Ltd; honoraria from the Public Health Research Foundation, AstraZeneca KK, Eisai Co. Ltd, and Kowa Pharmaceutical Co Ltd; and research grants and honoraria from MSD KK, Otsuka Pharmaceutical Co Ltd, Daiichi Sankyo Co Ltd, Sumitomo Dainippon Pharma Co Ltd, Teijin Pharma Ltd, Bayer Yakuhin, Ltd, Mochida Pharmaceutical Co Ltd, Astellas Pharma Inc., Sanwa Kagaku Kenkyusho Co Ltd, Takeda Pharmaceutical Co Ltd, Mitsubishi Tanabe Pharma Corp, Pfizer Japan Inc., Ono Pharmaceutical Co Ltd, and AstraZeneca KK. TI has received research grants from Ono, Daiichi Sankyo, and Teijin; has carried out joint research with Canon Medical systems and PIA; and is an employee of Tsukuba Echo Core Laboratory. KN has received honoraria from MSD, Astella, AstraZeneca, Novartis Pharma, Ono Pharmaceutical, Daiichi Sankyo, Mitsubishi Tanabe Pharma, Eli Lilly Japan, Boehringer Ingelheim Japan, and Takeda Pharmaceutical; research grants from Asahi Kasei, Astellas, Mitsubishi Tanabe Pharma, Teijin Pharma, Terumo, Boehringer Ingelheim Japan, Eli Lilly and Company, Mochida Pharmaceutical, and Fuji Yakuhin; and scholarships from Daiichi Sankyo Healthcare, Teijin Pharma, Medtronic, and Bayer Yakuhin.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A list of members and their affiliations appears in the Supplementary Information.
Supplementary information
Rights and permissions
About this article
Cite this article
Shiina, K., Tomiyama, H., Tanaka, A. et al. Differential effect of a xanthine oxidase inhibitor on arterial stiffness and carotid atherosclerosis: a subanalysis of the PRIZE study. Hypertens Res 45, 602–611 (2022). https://doi.org/10.1038/s41440-022-00857-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-022-00857-9
Keywords
This article is cited by
-
2023 update and perspectives
Hypertension Research (2024)
-
Is uric acid a causal risk factor of arterial stiffness in patients with hypertension?
Hypertension Research (2024)
-
Clinical effects of a selective urate reabsorption inhibitor dotinurad in patients with hyperuricemia and treated hypertension: a multicenter, prospective, exploratory study (DIANA)
European Journal of Medical Research (2023)
-
Preface–special issue on hypertension in Asia: the second story
Hypertension Research (2022)
-
Uric acid, xanthine oxidase, and vascular damage: potential of xanthine oxidoreductase inhibitors to prevent cardiovascular diseases
Hypertension Research (2022)