Abstract
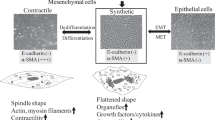
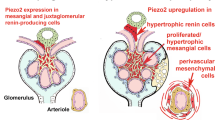
We have shown that complement 3 (C3) is upregulated in cardiovascular and renal organs, which induces the synthetic phenotype and exaggerates the growth of mesenchymal cells from spontaneously hypertensive rats (SHRs). However, the mechanisms of the upregulation of C3 have remained unclear. In the present study, we investigated the role of TWIST1, a transcription factor that regulates mesodermal embryogenesis, in the upregulation of C3 in glomerular mesangial cells (GMCs) from SHRs and Wistar–Kyoto (WKY) rats. Immunocytochemical staining and western blot analysis showed that the expression of TWIST1 in GMCs from SHRs was higher than that in GMCs from WKY rats in vivo and in vitro. Real-time PCR analysis showed increases in the expression of Twist1 mRNA with attenuated expression of miR-151-3p in GMCs from SHRs compared to that in cells from WKY rats. Chromatin immunoprecipitation assays showed increases in TWIST1 binding to the C3 promoter in GMCs from SHRs compared to that in cells from WKY rats. Transfection of Twist1 cDNA by a lentiviral vector increased the expression of C3 mRNA in GMCs from WKY rats. TWIST1 siRNA significantly decreased the mRNA expression of C3 and osteopontin in GMCs from SHRs. These results indicate that the increases in TWIST1 expression, attenuation of miR-151-3p, and strong binding of TWIST1 upregulate C3 gene expression in GMCs from SHRs. The enhanced TWIST1-C3 system induces the synthetic phenotype of mesenchymal tissue that may be associated with cardiovascular and renal remodeling in hypertension.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Fukuda N, Satoh C, Hu WY, Soma M, Kubo A, Kishioka H, et al. Production of angiotensin II by homogeneous cultures of vascular smooth muscle cells from spontaneously hypertensive rats. Arterioscler Thromb Vasc Biol. 1999;19:1210–7.
Hu WY, Fukuda N, Kanmatsuse K. Growth characteristics, angiotensin II-generation, and microarray-determined gene expression in vascular smooth muscle cells from young spontaneously hypertensive rats. J Hypertens. 2002;20:1323–33.
Fukuda N, Hu WY, Satoh C, Nakayama M, Kishioka H, Kubo A, et al. Contribution of synthetic phenotype on the enhanced angiotensin II-generating system in vascular smooth muscle cells from spontaneously hypertensive rats. J Hypertens. 1999;17:1099–107.
Hu WY, Fukuda N, Satoh C, Jian T, Kubo A, Nakayama M, et al. Phenotypic modulation by fibronectin enhances the angiotensin II-generating system in cultured vascular smooth muscle cells. Arterioscler Thromb Vasc Biol. 2000;20:1500–5.
Lin ZH, Fukuda N, Jin XQ, Yao EH, Ueno T, Endo M, et al. Complement 3 is involved in the synthetic phenotype and exaggerated growth of vascular smooth muscle cells from spontaneously hypertensive rats. Hypertension. 2004;44:1–6.
Yao EH, Fukuda N, Ueno T, Tsunemi A, Endo M, Matsumoto K. Complement 3 activates KLF5 gene in vascular smooth muscle cells. Biochem Biophys Res Commun. 2008;367:468–73.
Wan J, Fukuda N, Endo M, Tahira Y, Yao E, Matsuda H, et al. Complement 3 is involved in changing the phenotype of human glomerular mesangial cells. J Cell Physiol. 2007;213:495–501.
Ikeda K, Fukuda N, Ueno T, Endo M, Kobayashi N, Soma M, et al. Role of complement 3a in the growth of mesangial cells from stroke-prone spontaneously hypertensive rats. Clin Exp Hypertens. 2014;36:58–63.
Chen L, Fukuda N, Matsumoto T, Abe M. Role of complement 3 in the pathogenesis of hypertension. Hypertens Res. 2020;43:255–62.
Zhou X, Fukuda N, Matsuda H, Endo M, Wang X, Saito K, et al. Complement 3 activates the renal renin-angiotensin system by induction of epithelial-to-mesenchymal transition of the nephrotubulus in mice. Am J Physiol Ren Physiol. 2013;305:F957–7.
Negishi E, Fukuda N, Otsuki T, Katakawa M, Komatsu K, Chen L, et al. Involvement of complement 3 in the salt-sensitive hypertension by activation of renal renin-angiotensin system in spontaneously hypertensive rats. Am J Physiol Ren Physiol. 2018;315:F1747–58.
Castanon I, Baylies MK. A Twist in fate: evolutionary comparison of Twist structure and function. Gene. 2002;287:11–22.
Chen HF, Wu KJ. Endothelial transdifferentiation of tumor cells triggered by the Twist1-Jagged1-KLF4 axis: relationship between cancer stemness and angiogenesis. Stem Cells Int. 2016;2016:6439864.
Yang MH, Wu MZ, Chiou SH, Chen PM, Chang SY, Liu CJ, et al. Direct regulation of TWIST by HIF-1훼 promotes metastasis. Nat Cell Biol. 2008;10:295–305.
Lovisa S, LeBleu VS, Tampe B, Sugimoto H, Vadnagara K, Carstens JL, et al. Epithelial-to-mesenchymal transition induces cell cycle arrest and parenchymal damage in renal fibrosis. Nat Med. 2015;21:998–1009.
Cho MS, Rupaimoole R, Choi HJ, Noh K, Chen J, Hu Q, et al. Complement Component 3 is regulated by TWIST1 and mediates epithelial mesenchymal transition. J Immunol. 2016;196:1412–8.
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, et al. 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map format and SAMtools. Bioinformatics. 2009;25:2078–9.
Matsumoto K, Hatano M. Production of interleukin 1 in glomerular cell cultures from rats with nephrotoxic serum nephritis. Clin Exp Immunol. 1989;75:123–8.
Ding S, Zhu Y, Liang Y, Huang H, Xu Y, Zhong C. Circular RNAs in vascular functions and diseases. Adv Exp Med Biol. 2018;1087:287–97.
Yeh TH, Huang TT, Yeh TS, Chen YR, Hsu KW, Yin PH, et al. miR-151-3p targets TWIST1 to repress migration of human breast cancer cells. PLoS One. 2016;11:e0168171.
Wei H, Li Z, Wang X, Wang J, Pang W, Yang G, et al. microRNA-151–3p regulates slow muscle gene expression by targeting ATP2a2 in skeletal muscle cells. J Cell Physiol. 2015;230:1003–12.
Vincentz JW, Barnes RM, Rodgers R, Firulli BA, Conway SJ, Firulli AB. An absence of Twist1 results in aberrant cardiac neural crest morphogenesis. Dev Biol. 2008;320:131–9.
Acknowledgements
The authors would like to thank Akiko Tsunemi and Mayumi Katakawa for their technical support.
Funding
This study was supported by a Grant-in-Aid for Scientific Research from MEXT 2015-2018 (15K09300).
Author information
Authors and Affiliations
Contributions
Conceptualization, NF, MA; Methodology, NF; Validation, TO; Investigation, TO, LC; Data Curation, TU; Writing—Original Draft Preparation, TO; Writing—Review and Editing, NF, MA; Visualization, TO, NF; Supervision, NF, MA; Project Administration, NF, MA; Funding Acquisition, NF, MA. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Otsuki, T., Fukuda, N., Chen, L. et al. TWIST1 transcriptionally upregulates complement 3 in glomerular mesangial cells from spontaneously hypertensive rats. Hypertens Res 45, 66–74 (2022). https://doi.org/10.1038/s41440-021-00750-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-021-00750-x
Keywords
This article is cited by
-
Update on Hypertension Research in 2021
Hypertension Research (2022)