Abstract
Family history of hypertension (FH) is a nonmodifiable risk factor for hypertension. However, cardiorespiratory fitness (CRF) is a modifiable risk factor and might be important for preventing hypertension in both people with and without FH. The purpose of this study was to investigate the combined association of CRF and FH on the incidence of hypertension in normotensive male Japanese workers. A total of 6890 workers were included in this study. CRF was determined using a submaximal exercise test. A self-reported questionnaire was used to determine FH. Six groups were established, combining the two groups with and without FH (Yes, No) and the three CRF groups (Low, Moderate, High). The incidence of hypertension, defined as systolic blood pressure of ≥140 mmHg or diastolic blood pressure of ≥90 mmHg, or self-reported physician-diagnosed hypertension was evaluated. During the observation period of 101,212 man-years (median 17 years, minimum 1 year, maximum 23 years), 2210 workers developed hypertension. There were 3860 participants who had FH (56%). Compared with the Yes-Low CRF group, the HRs (95% CI) for hypertension were 66% lower in the No-High CRF group (0.34 [0.28–0.40]), 47% lower in the No-Low CRF group (0.53 [0.46–0.61]), and 24% lower in the Yes-High CRF group (0.76 [0.67–0.86]). FH and CRF did not show a significant interaction (p for interaction = 0.181). The combination of FH and CRF showed a clear association with the risk of incident hypertension, and moderate to high levels of CRF might be equally beneficial for preventing hypertension for both people with and without FH.
Similar content being viewed by others
Introduction
The World Health Organization (WHO) has reported that hypertension is an important risk factor for cardiovascular disease and in 2014, the prevalence of hypertension worldwide among adults aged 18 and over was approximately 22% [1]. In the 2014 Japanese National Health and Nutrition Survey, the percentage of the population aged 20 years or older with hypertension (systolic blood pressure ≥140/diastolic blood pressure ≥90 or taking antihypertensive medication) was 35% [2]. In Japan, the medical costs associated with hypertensive disorders in 2014 amount to 1.8 trillion yen (approx. $16.6 billion) [3]; therefore, preventing hypertension is an important issue.
It is known that the incidence of hypertension is high in groups with a family history of hypertension (FH) compared with those without FH [4]. Moreover, at any given age, blood pressure is higher among those with a positive family history [5]. However, when cardiorespiratory fitness (CRF), an objective indicator of the quantity of physical activity, is high, the incidence of hypertension is low [6,7,8,9], and at a given age, the blood pressure is lower among persons with high CRF than in those with low CRF [10,11,12]. In a cohort study (over an average 5-year period) of the relationship between the incidence of hypertension and combinations of FH and CRF in predominantly Caucasian individuals [13], the incidence of hypertension was low in participants with high CRF, even when there was FH. This suggests the possible importance of high CRF due to the role of habitual physical activity in preventing hypertension, particularly since FH is a nonmodifiable risk factor. However, we have not found any studies that have investigated the relationship between the incidence of hypertension and combinations of FH and CRF in Japanese individuals, who differ from Caucasian individuals with respect to genetic background and living environment. Accordingly, the purpose of this study was to investigate the combined association of CRF and FH on the incidence of hypertension in normotensive male Japanese workers. We hypothesized that the incidence of hypertension is low in Japanese individuals with high CRF, even in the presence of FH.
Methods
Design and study population
This was a cohort study investigating the relationship between CRF and health outcomes [7, 14, 15]. The participants were workers in the Tokyo area who, under the Japanese Safety and Health law, receive an annual medical examination.
From April 1986 to March 1987, 9221 workers participated in the study; the workers performed an exercise test and received an annual medical examination. Due to the small number of female participants in this study (n = 791), women were excluded from the analysis. Workers were also excluded (n = 24) when estimating CRF was not possible because they could not continue the exercise test for at least 4 min. A total of 1102 workers who had a history of hypertension (n = 1090), heart disease (n = 9) or stroke (n = 3) in March 1987 were excluded from the cohort in this study. In addition, 414 workers with missing data on potential confounding factors were excluded. Ultimately, 6890 workers were included in this study and were followed until March 2010.
This study was approved by the Research Ethics Committee of the National Institutes of Biomedical Innovation, Health and Nutrition.
Medical examination
In Japan, under the Industrial Safety and Health Law, all workers, including the participants in this study, are obligated to undergo an annual medical examination organized by their employers. The medical examinations included measuring height, weight, and resting blood pressure. Bodyweight was measured using scales for routine weighing, wearing light clothing, and without footwear. Height and weight measurements were used to calculate body mass index (BMI [kg/m2]). Resting blood pressure was measured in a seated position using an automatic sphygmomanometer. Those who answered “yes” at least once to the question “FH within two degrees of kinship: yes or no” in the self-administered questionnaire at the medical examinations from 1987 to 2009 were assessed as having FH. Alcohol consumption habits (nondrinker, 1–40 g alcohol daily, ≥41 g daily) and cigarette smoking habits (never smoker, past smoker, 1–20 a day, ≥21 a day) were also included in the questionnaire.
Assessment of cardiorespiratory fitness
CRF was determined using a submaximal exercise test, with a cycle ergometer. The submaximal exercise test comprised a maximum of three 4-min stages, with the workload being increased from stage to stage. The workload in the first stage was, by age group, 98, 86, 74, and 61 watts for 19–29, 30–39, 40–49, and 50–60 years of age, respectively. Heart rate was measured using an electrocardiogram. The target heart rate was set to 85% of the estimated maximum heart rate (220−age [years]), and the workload was increased by 37 watts at each stage until reaching the target heart rate or until reaching the third stage. Maximal oxygen uptake was estimated as CRF in each participant from the heart rate obtained in the last minute of the final stage, using the Åstrand–Ryhming nomogram [16] and Åstrand age correction factors [17] commonly employed for estimating CRF [18].
Diagnosis of hypertension
Hypertension was defined as present when (1) systolic blood pressure was ≥140 mmHg or diastolic blood pressure was ≥90 mmHg or when (2) the individual himself reported in the interview with the nurse that he had started treatment for hypertension [19, 20]. In this study, resting blood pressure was measured in a seated position after >3 min of resting, and several measurements were made when the measured value was high. However, because the measurements were made in the workplace during a medical examination, there was a possibility that this could have affected the examination and temporarily elevated the blood pressure. Accordingly, to determine more accurately whether workers were suffering from hypertension, they were assessed as having hypertension when systolic blood pressure exceeded 140 mmHg or when diastolic blood pressure exceeded 90 mmHg at least twice in 6 years, if not treated for hypertension. The year in which systolic blood pressure initially exceeded 140 mmHg or diastolic blood pressure exceeded 90 mmHg was considered to be the first year of hypertension. If treatment for hypertension had started before the first year that blood pressure exceeded these thresholds, the year treatment started was recorded as the first year of hypertension.
Statistical analysis
We first classified the study participants into two groups, determined by the presence or absence of FH. Participants were also classified into tertiles for CRF (with tertiles determined separately for age groups ≤24 years, 25–29 years, 30–34 years, 35–39 years, 40–44 years, 45–49 years, and ≥50 years) and classified into three groups based on age-specific tertiles of CRF (Low, Moderate, High). Finally, six groups were established, combining the two groups with and without FH (Yes, No) and the three CRF groups (Yes-Low, Yes-Moderate, Yes-High, No-Low, No-Moderate, No-High). The physical characteristics of the six groups were compared at the beginning of the follow-up period. Continuous variables were expressed as the mean ± standard deviation (SD), and categorical variables were expressed as percentages.
We evaluated the association between FH and CRF with the incidence of hypertension. Cox proportional hazards regression analysis was carried out with the incidence of hypertension as the dependent variable and FH (2 groups) or CRF (3 groups) as the independent variables, and age-adjusted hazard ratios (HRs) and 95% confidence intervals (95% CI) were calculated. Considering potential confounding factors, HRs adjusted for BMI (continuous variable), cigarette smoking habits (never smoker, past smoker, 1–20 cigarettes per day, ≥21 cigarettes per day), and alcohol consumption habits (none, 1–40 g per day, ≥41 g per day) were determined by questionnaires. In addition, to evaluate whether a linear dose–response relationship existed between the incidence of hypertension and CRF, CRF was entered as an ordinal variable into the model to calculate p for trend. We additionally evaluated the associations between the incidence of hypertension and the six combined FH and CRF groups. Cox proportional hazards regression analysis was carried out with the incidence of hypertension as the dependent variable and the six groups as independent variables, and HRs adjusted for age and potential confounding factors and 95% CI were calculated. Furthermore, to confirm the presence or absence of FH and CRF effect modifications, we included an FH (yes, no) and CRF (continuous variable) product term into the model to confirm the presence or absence of interactions.
Proportional hazards assumption was assessed by visually checking the log-minus-log plot; no violation was observed. SPSS Statistics version 22 (IBM Japan, Tokyo, Japan) was used for statistical analysis. A statistically significant difference was considered to be present when the two-tailed p value was <0.05.
Results
The average age (SD) of the participants at baseline was 37.3 years (±9.8 years). The average follow-up period was 14.7 years (minimum 1 year, maximum 23 years). During the observation period of 101,212 man-years, 2210 workers were newly diagnosed with hypertension. A total of 3860 participants reported FH (56%). The baseline characteristics of participants in the 6 groups, categorized by FH and CRF, are shown in Table 1. BMI was lower across levels of CRF, with or without FH. When comparing systolic blood pressure and diastolic blood pressure within the same level of CRF, values were higher in participants with FH. Irrespective of whether FH was present, both systolic blood pressure and diastolic blood pressure were lower across increasing CRF levels. The percentage of smokers increased as the level of CRF decreased, irrespective of FH, and alcohol consumption was reported by a higher proportion of participants with FH, irrespective of the level of CRF.
The HRs of incident hypertension according to either FH or CRF are shown in Table 2. The HR for hypertension was significantly lower in participants without FH (HR 0.50 [95% CI, 0.46–0.55]) than in participants with FH. In addition, there was a significant inverse trend for incident hypertension across increasing CRF levels, with HRs (95% CI) of 1.0 (referent), 0.87 (0.79–0.96), and 0.72 (0.65–0.81) for low, moderate, and high CRF, respectively (p for trend <0.001).
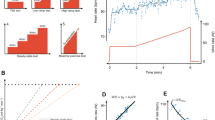
The HRs for hypertension in the six groups of participants cross-classified by FH and CRF are presented in Fig. 1. In both FH groups, there was a strong trend for decreased incidence of hypertension across CRF levels. For the group with FH, HRs (95% CI) were 1.0 (referent), 0.88 (0.79–0.99), and 0.76 (0.67–0.86) for low, moderate, and high CRF, respectively. For the group with no FH, HRs (95% CI) were 0.53 (0.46–0.62), 0.45 (0.38–0.52), and 0.34 (0.28–0.40) for low, moderate, and high CRF, respectively. There was a significant inverse trend for incident hypertension in the six groups (p for trend < 0.001). FH and CRF did not show a significant interaction (p for interaction = 0.181).
Hazard ratios of incident hypertension according to combined family history of hypertension and cardiorespiratory fitness. Multivariable model was adjusted for age (continuous variable), BMI (continuous variable), smoking habit (never smoker, past smoker, 1–20 cigarettes per day, 21≥ cigarettes per day), and drinking habit (none, 1–40 g per day, ≥41 g per day)
We also classified hypertension as systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg in the year of the earliest episode (Supplementary Tables 1 and 2). We performed a whole new set of analysis using the 1987 FH data instead of using the data from 1987 to 2009 (Supplementary Tables 3 and 4).
Discussion
In this cohort study following normotensive male Japanese workers, the risk of developing hypertension was significantly higher in participants with FH than without FH. Importantly, the risk of developing hypertension was significantly lower in participants across increasing CRF levels, regardless of whether FH was present or not. Moreover, the combination of FH and CRF showed a clear association with the risk of hypertension, and even participants with FH showed a lower risk of hypertension when the level of CRF was high. FH and CRF did not show a significant interaction. Therefore, these findings suggest that moderate to high levels of CRF might be equally beneficial for preventing hypertension in both people with and without FH.
Earlier studies have reported that there is a high risk of hypertension and high blood pressure in individuals with a parental history of hypertension. Wang et al. [4] reported that compared with individuals whose parents did not have hypertension, the risk of hypertension was 1.5 times higher in individuals with a maternal history of hypertension, 1.8 times higher in individuals with a paternal history of hypertension, and 2.4 times higher in individuals with a history of hypertension in both parents. In a report on Japanese participants, Mitsumata et al. [5] reported that the age-blood pressure curve has an upward shift of 5.3 mmHg for systolic blood pressure and an upward shift of 2.8 mmHg for diastolic blood pressure in participants with parental hypertension compared with participants without parental hypertension. Moreover, a recent study showed that among young to middle-aged normotensive individuals, arterial stiffness, which is a major factor causing elevated blood pressure, is already high in people with parental hypertension [21]. These studies indicate that FH increases the risk of hypertension. In this study, the HR for hypertension was also lower in the participants without FH than in participants with FH (HR = 0.50), showing similar results to prior studies.
In cohort studies carried out previously to investigate the relationship between CRF and incidence of hypertension, it has been reported that high CRF is associated with a low risk of hypertension [6,7,8,9]. In this study, there was also a clear negative dose–response relationship between CRF and the incidence of hypertension, confirming that participants with moderate to high CRF show a lower risk of hypertension. One study reported the relationship between the incidence of hypertension and the combination of FH and CRF [13]. Most of the participants in this prior study were Caucasian individuals. When compared with participants with low fitness and FH, participants with high fitness and no FH showed the lowest risk of hypertension (41%). To our knowledge, the present study is the first long-term cohort study to investigate the relationship between the incidence of hypertension and FH and CRF in a Japanese population. Although this study was carried out with Japanese participants who differed from Caucasian individuals with respect to genetic background and living environment, our results were similar to studies involving Caucasians.
The high risk of hypertension in participants with FH is considered to be the result of a mixture of genetic and environmental factors [22]. Regarding genetic factors, genes associated with hypertension, such as ATP2B1, have been identified as the result of studies with Japanese participants [23]. The contribution of genetic factors associated with hypertension has been estimated at 30–70% [4, 24]. Some of the genes associated with blood pressure have been identified and are shared by people of Asian, European, and North American origin. However, it has been reported that there are ethnic differences regarding the incidence of mutations and effects on blood pressure [25] and that there are ethnic differences in the incidence of hypertension [26]. Moreover, regarding lifestyle habits, Japanese people tend to have a higher salt intake [2], which is reported to be associated with elevated blood pressure and the risk of hypertension [27, 28]. On the other hand, the incidence of obesity is higher among Caucasian individuals than among Japanese individuals, and obesity is a risk factor for hypertension [29]. This study has shown that for Japanese individuals with FH, the incidence of hypertension is lower in individuals with moderate to high CRF, as is the case with Caucasian individuals despite differing genetic backgrounds and living environments. This study suggests the importance of maintaining moderate to high CRF to prevent hypertension. The mechanism explaining the relationship between moderate to high CRF and a low risk of hypertension is likely to be due to the ability of physical activity to decrease blood pressure [30]; additionally, regular physical activity improves CRF [31, 32]. Therefore, it appears that in participants with moderate to high CRF, elevation in blood pressure is suppressed by physical activity [33], lowering the risk of hypertension. Because the progression rate of arterial stiffness is slower [34], wave reflection is low [35], and vascular endothelial function is enhanced [36, 37] in individuals with high CRF, these factors likely contribute to a lower incidence of hypertension. Therefore, it appears that the risk of hypertension was low in the group with a high fitness level and no FH because of fewer genetic factors predisposing them to hypertension combined with the suppression of elevated blood pressure through physical activity.
There are some limitations associated with this study. In this study, a self-reported questionnaire was used to determine family history. The question asked was simple, “FH within two degrees of kinship: yes or no”, but the validity of the questionnaire was not investigated. In addition, blood pressure measurement in this study was carried out during medical examinations in the workplace and that environment may have influenced the measurements. However, we endeavored to minimize errors in assessing hypertension by conservatively assessing that a participant developed hypertension only when systolic blood pressure exceeded 140 mmHg or diastolic blood pressure exceeded 90 mmHg at least twice in 6 years. Moreover, because the participants in this study were young to middle-aged Japanese males, the results may not be generalizable to other groups. Future studies are needed that include women and other age groups and races/ethnicities. Finally, CRF was estimated by a submaximal exercise test, which is slightly more prone to error compared to the direct method. However, a previous study reported a high correlation coefficient between estimated (our procedure) and directly measured values (r = 0.92) [38]. Therefore, we assume that the estimated values are sufficiently accurate.
In conclusion, this study showed that the combination of FH and CRF is associated with a higher risk of hypertension. The risk of hypertension was low when the level of CRF was high, even in the case of Japanese participants with FH. This suggests that increasing CRF with habitual physical activity is important for preventing hypertension given that FH remains a nonmodifiable risk factor. On the other hand, the group with FH and low CRF can be considered a high-risk group, and hence, physical activity interventions targeting this group may be an effective strategy for preventing cardiovascular disease in the future.
References
World Health Organization. Global status report on noncommunicable diseases 2014. 2014; http://www.who.int/nmh/publications/ncd-status-report-2014/en/. Accessed 19 April 2018.
Ministry of Health Labour and Welfare. The National Health and Nutrition Survey in Japan, 2014. 2014; http://www.mhlw.go.jp/bunya/kenkou/eiyou/h26-houkoku.html. Accessed 19 April 2018.
Ministry of Health Labour and Welfare. Estimates of National Medical Care Expenditure 2014. 2014; http://www.mhlw.go.jp/toukei/saikin/hw/k-iryohi/14/. Accessed 19 April 2018.
Wang NY, Young JH, Meoni LA, Ford DE, Erlinger TP, Klag MJ. Blood pressure change and risk of hypertension associated with parental hypertension: the Johns Hopkins Precursors Study. Arch Intern Med. 2008;168:643–8.
Mitsumata K, Saitoh S, Ohnishi H, Akasaka H, Miura T. Effects of parental hypertension on longitudinal trends in blood pressure and plasma metabolic profile: mixed-effects model analysis. Hypertension. 2012;60:1124–30.
Blair SN, Goodyear NN, Gibbons LW, Cooper KH. Physical fitness and incidence of hypertension in healthy normotensive men and women. JAMA. 1984;252:487–90.
Sawada S, Tanaka H, Funakoshi M, Shindo M, Kono S, Ishiko T. Five year prospective study on blood pressure and maximal oxygen uptake. Clin Exp Pharmacol Physiol. 1993;20:483–7.
Lee DC, Sui X, Church TS, Lavie CJ, Jackson AS, Blair SN. Changes in fitness and fatness on the development of cardiovascular disease risk factors hypertension, metabolic syndrome, and hypercholesterolemia. J Am Coll Cardiol. 2012;59:665–72.
Carnethon MR, Gidding SS, Nehgme R, Sidney S, Jacobs DR Jr., Liu K. Cardiorespiratory fitness in young adulthood and the development of cardiovascular disease risk factors. JAMA. 2003;290:3092–3100.
Liu J, Sui X, Lavie CJ, Zhou H, Park YM, Cai B, et al. Effects of cardiorespiratory fitness on blood pressure trajectory with aging in a cohort of healthy men. J Am Coll Cardiol. 2014;64:1245–53.
Diez-Fernandez A, Sanchez-Lopez M, Nieto JA, Gonzalez-Garcia A, Miota-Ibarra J, Ortiz-Galeano I, et al. Relationship between cardiorespiratory fitness and blood pressure in young adults: a mediation analysis of body composition. Hypertens Res. 2017;40:511–5.
Yang Y, Lau PW, Wang J, Dong B, Wu L, Quach B, et al. Associations among cardiorespiratory endurance, body mass index and blood pressure in Han Chinese children: results from the 2010 Chinese National Survey On Students’ Constitution and Health. Hypertens Res. 2016;39:799–804.
Shook RP, Lee DC, Sui X, Prasad V, Hooker SP, Church TS, et al. Cardiorespiratory fitness reduces the risk of incident hypertension associated with a parental history of hypertension. Hypertension. 2012;59:1220–4.
Sawada SS, Lee IM, Muto T, Matuszaki K, Blair SN. Cardiorespiratory fitness and the incidence of type 2 diabetes: prospective study of Japanese men. Diabetes Care. 2003;26:2918–22.
Sawada SS, Lee IM, Naito H, Noguchi J, Tsukamoto K, Muto T, et al. Long-term trends in cardiorespiratory fitness and the incidence of type 2 diabetes. Diabetes Care. 2010;33:1353–7.
Astrand PO, Ryhming I. A nomogram for calculation of aerobic capacity (physical fitness) from pulse rate during sub-maximal work. J Appl Physiol. 1954;7:218–21.
Astrand I. Aerobic work capacity in men and women with special reference to age. Acta Physiol Scand Suppl. 1960;49:1–92.
Astrand PO, Rodahl K, Dahl H, Stromme S, editors. Textbook of work physiology: physiological bases of exercise. Human Kinetics Pub: Champaign, IL, USA; 2003.
Shimamoto K, Ando K, Fujita T, Hasebe N, Higaki J, Horiuchi M, et al. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2014). Hypertens Res. 2014;37:253–390.
James PA, Oparil S, Carter BL, Cushman WC, Dennison-Himmelfarb C, Handler J, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA. 2014;311:507–20.
Andersson C, Quiroz R, Enserro D, Larson MG, Hamburg NM, Vita JA, et al. Association of parental hypertension with arterial stiffness in nonhypertensive offspring: The Framingham Heart Study. Hypertension. 2016;68:584–9.
Wang X, Xu X, Su S, Snieder H. Familial aggregation and childhood blood pressure. Curr Hypertens Rep. 2015;17:509.
Tabara Y, Kohara K, Kita Y, Hirawa N, Katsuya T, Ohkubo T, et al. Common variants in the ATP2B1 gene are associated with susceptibility to hypertension: the Japanese Millennium Genome Project. Hypertension. 2010;56:973–80.
Kupper N, Willemsen G, Riese H, Posthuma D, Boomsma DI, de Geus EJ. Heritability of daytime ambulatory blood pressure in an extended twin design. Hypertension. 2005;45:80–85.
Kato N. Ethnic differences in genetic predisposition to hypertension. Hypertens Res. 2012;35:574–81.
Carson AP, Howard G, Burke GL, Shea S, Levitan EB, Muntner P. Ethnic differences in hypertension incidence among middle-aged and older adults: the multi-ethnic study of atherosclerosis. Hypertension. 2011;57:1101–7.
Intersalt: an international study of electrolyte excretion and blood pressure. Results for 24h urinary sodium and potassium excretion. Intersalt Cooperative Research Group. BMJ. 1988;297:319–28.
Takase H, Sugiura T, Kimura G, Ohte N, Dohi Y. Dietary sodium consumption predicts future blood pressure and incident hypertension in the Japanese normotensive general population. J Am Heart Assoc. 2015;4:e001959.
Williams PT. Increases in weight and body size increase the odds for hypertension during 7 years of follow-up. Obesity. 2008;16:2541–8.
Dickinson HO, Mason JM, Nicolson DJ, Campbell F, Beyer FR, Cook JV, et al. Lifestyle interventions to reduce raised blood pressure: a systematic review of randomized controlled trials. J Hypertens. 2006;24:215–33.
Blair SN, Kampert JB, Kohl HW 3rd, Barlow CE, Macera CA, Paffenbarger RS Jr, et al. Influences of cardiorespiratory fitness and other precursors on cardiovascular disease and all-cause mortality in men and women. JAMA. 1996;276:205–10.
Garber CE, Blissmer B, Deschenes MR, Franklin BA, Lamonte MJ, Lee IM, et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43:1334–59.
Pescatello LS, Franklin BA, Fagard R, Farquhar WB, Kelley GA, Ray CA. American College of Sports Medicine position stand. Exercise and hypertension. Med Sci Sports Exerc. 2004;36:533–53.
Gando Y, Murakami H, Kawakami R, Yamamoto K, Kawano H, Tanaka N, et al. Cardiorespiratory fitness suppresses age-related arterial stiffening in healthy adults: a 2-year longitudinal observational study. J Clin Hypertens. 2016;18:292–8.
Meyer J, Elmenhorst J, Giegerich T, Oberhoffer R, Muller J. Controversies in the association of cardiorespiratory fitness and arterial stiffness in children and adolescents. Hypertens Res. 2017;40:675–8.
Pahkala K, Heinonen OJ, Simell O, Viikari JS, Ronnemaa T, Niinikoski H, et al. Association of physical activity with vascular endothelial function and intima-media thickness. Circulation. 2011;124:1956–63.
Buscemi S, Canino B, Batsis JA, Buscemi C, Calandrino V, Mattina A, et al. Relationships between maximal oxygen uptake and endothelial function in healthy male adults: a preliminary study. Acta Diabetol. 2013;50:135–41.
Teräslinna P, Ismail A, MacLeod D. Nomogram by Astrand and Ryhming as a predictor of maximum oxygen intake. J Appl Physiol. 1966;21:513–5.
Acknowledgements
The authors thank the study participants and the physicians and medical staff of Tokyo Gas Company for assisting with data collection. We also thank Benjamin Howe for helpful comments.
Funding
This work was supported by JSPS KAKENHI (JP16K13037, YG).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Gando, Y., Sawada, S.S., Kawakami, R. et al. Combined association of cardiorespiratory fitness and family history of hypertension on the incidence of hypertension: a long-term cohort study of Japanese males. Hypertens Res 41, 1063–1069 (2018). https://doi.org/10.1038/s41440-018-0117-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-018-0117-2
Keywords:
This article is cited by
-
Independent and joint associations of estimated cardiorespiratory fitness and its dynamic changes and obesity with the risk of hypertension: A prospective cohort
Journal of Human Hypertension (2024)
-
The association of cardiorespiratory fitness and the risk of hypertension: a systematic review and dose–response meta-analysis
Journal of Human Hypertension (2022)
-
The Chester step test is a valid tool to assess cardiorespiratory fitness in adults with hypertension: reducing the gap between clinical practice and fitness assessments
Hypertension Research (2019)