Abstract
High blood pressure is the most significant risk factor of cardiovascular and cerebrovascular diseases worldwide. Blood pressure and its variability are recognized as risk factors. Thus, hypertension control should focus not only on maintaining optimal levels but also on achieving less variability in blood pressure. Psychosocial stress is known to contribute to the development and worsening of hypertension. Stress is perceived by the brain and induces neuroendocrine responses in either a rapid or long-term manner. Moreover, endothelial dysfunction and inflammation might be further involved in the modulation of blood pressure elevation associated with stress. White-coat hypertension, defined as high clinic blood pressure but normal out-of-office blood pressure, is the most popular stress-related blood pressure response. Careful follow-up is necessary for this type of hypertensive patients because some show organ damage or a worse prognosis. On the other hand, masked hypertension, defined as high out-of-office blood pressure but normal office blood pressure, has received considerable interest as a poor prognostic condition. The cause of masked hypertension is complex, but evidence suggests that chronic stress at the workplace or home could be involved. Chronic psychological stress could be associated with distorted lifestyle and mental distress as well as long-lasting allostatic load, contributing to the maintenance of blood pressure elevation. Stress issues are common in patients in modern society. Considering psychosocial stress as the pathogenesis of blood pressure elevation is useful for achieving an individual-focused approach and 24-h blood pressure control.
Similar content being viewed by others
Introduction
Hypertension is the most significant risk factor of cardiovascular morbidity and mortality [1, 2]. According to the global report of the World Health Organization, hypertension-related complications are estimated to cause 7.5 million deaths worldwide, comprising nearly 13% of annual mortality [3]. Of these deaths, over 45% were due to coronary heart disease and 50% were due to stroke [4]. Accumulated evidence has shown that consistently high blood pressure and its variability are risk factors of cardiovascular events [5,6,7,8,9]. Thus, hypertension control should focus not only on maintaining an optimal level of blood pressure but also on achieving less variability.
The etiology of high blood pressure is complex and multifactorial. Genes, the neuroendocrine system, organ failure, lifestyle and environmental factors are involved at the individual level [10,11,12,13]. Because of the development of satisfactory antihypertensive agents, the treatment of hypertension has considerably improved over the last decades [14,15,16]. However, hypertension and its cardiovascular complications are the most prevalent non-communicable diseases worldwide [3]. The number of people diagnosed with hypertension is increasing. This increase may be largely attributed to social or environmental factors, such as aging populations, increased obesity prevalence, air pollution due to industrialization, and exposure to various types of mental stress [13, 14, 17].
Stress-related increases in blood pressure include a large spectrum in terms of pathophysiology. This increase includes not only temporal blood pressure elevation induced by emotional stress but also long-lasting high blood pressure due to distorted lifestyle or mental distress derived from stress [18, 19]. Recent studies have shown that the stress-related blood pressure response is modified by endothelial function [20], inflammation [21,22,23], or immune function [24]. Because stress-induced blood pressure elevation could be deeply involved in the pathogenesis of both short- and long-term blood pressure variabilities, considering stress as a pathogenesis of high blood pressure seems necessary to individualize hypertension management and achieve a 24-h blood pressure control.
This article will provide an update on the mechanisms of stress-related blood pressure elevation. Stress generally represents a series of bodily reactions to various types of external and internal stimuli, such as physical, traumatic, psychological, biochemical, infectious and radiological stimuli. This article mainly focuses on psychosocial stress, which has received considerable attention as a cardiovascular risk that modern civilization brings to society [25]. The clinical significance of stress-related blood pressure elevation was subsequently reviewed for clinic and out-of-clinic settings.
Psychosocial stress is a relatively fixed risk factor for hypertension in the 2017 high blood pressure clinical practice guidelines of the USA because it is usually difficult to modify [26]. The latest systematic reviews on the clinical effectiveness of stress-reduction techniques in hypertensive patients have shown that such interventions appear to indicate some hypotensive effects, but the results require cautious interpretation due to major methodological limitations [27]. However, the recent largest interventional study conducted in China has demonstrated that 2-year psychological intervention could improve blood pressure control, health-related quality of life, and stroke prevalence in Chinese mine workers [28]. This finding suggests that adequate stress management could improve hypertension treatment.
Mechanisms of stress-induced blood pressure elevation
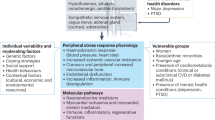
Mechanisms of stress-induced blood pressure elevation are complex and not completely understood. Figure 1 indicates a global schema of stress-induced blood pressure elevation. Psychological stress is often induced by environmental changes, which are perceived by the brain. Some are integrated in the neocortex, but others are sensed by the limbic system in a situation of “fight or flight” [29, 30]. Neuroendocrine stress responses occur if an individual feels afraid or anxious against such changes or if a demand exceeds an individual’s capability [31]. In humans, stress responses to the same situation largely differ among individuals because they are determined by the integration of personality characteristics, knowledge, previous experience, and skills [32]. Subsequent activation of the sympathetic nervous system and hypothalamic–pituitary–adrenocortical axis are the main characteristics of stress responses [31]. These systems promote adaptation to challenges, called “allostasis,” meaning the process of maintaining stability under environmental changes. However, these adaptive systems could be harmful if their function is inadequate or excessive [33]. For example, it has been reported that larger blood pressure increases in response to mental stress are associated with more severe vascular damage [34, 35], although there are some exceptions [36]. Moreover, stress-related blood pressure elevation is largely influenced by vascular condition or volume status. Increased arterial stiffness and a diminished natriuretic response are known to be associated with higher blood pressure response to acute psychological stress [37,38,39]. Insulin resistance may be further involved in daily life blood pressure variability through the augmentation of peripheral vascular resistance [40].
The mechanisms of long-lasting blood pressure elevation induced by chronic stress are much more complex and multifactorial than acute stress response. In chronic stress conditions, repeated and/or continuous activation of the sympathetic nervous system, hypothalamic–pituitary–adrenal axis, and renin–angiotensin–aldosterone system occur, leading to blood pressure elevation, increased shear stress, and endothelial damage [19, 20]. Neuroendocrine activation associated with chronic stress has been shown to trigger an inflammatory response through the stimulation of cytokines and acute phase reactant production [41], which can further impair endothelial function. The endothelium is well recognized as an endocrine homeostatic organ that regulates vascular tone and structure. Endothelial dysfunction leads to increased vasomotor tone, cellular hypertrophy, and remodeling, becoming a pro-atherosclerotic structure [42]. An altered endothelium also becomes a source of hypertensive substrates from the arterial wall, such as reactive oxygen species, thromboxane A2, endothelin, and prostaglandin H2 [43]. All these changes could contribute to chronic blood pressure elevation. Even worse, psycho-behavioral factors could be further involved in the maintenance of high blood pressure associated with chronic stress. Chronic psychological stress has been reported to increase the risk of unhealthy lifestyle behaviors, such as physical inactivity [39, 44], smoking [45], and heavy drinking [46], which are risk factors for hypertension. Associated mental distress, such as depression [47], could cause or worsen hypertension due to coexistence with autonomic or hypothalamic–pituitary–adrenal axis dysregulation [48, 49], distorted lifestyle [46], and insomnia [50].
Stress-induced blood pressure elevation in a clinic setting
Blood pressure measurement by a doctor is well known to trigger a defense reaction, which causes a rise in blood pressure [51, 52]. This phenomenon is called the “white-coat effect,” and it is clinically important because it may often lead to misdiagnosis [53]. Moreover, the temporal pressor response may reflect inherent hyper-reactivity to stress in some cases and be harmful in the long term [54]. In humans with mild hypertension, a doctor’s visit induces a sudden, marked, and prolonged pressor and tachycardic response accompanied by a significant increase in skin sympathetic nerve traffic and a significant decrease in muscle sympathetic nerve traffic [55]. Thus, the sympathetic nervous response induced by the doctor’s blood pressure measurement is not homogenous but rather differs among organs. This response might be reasonably understood in that the diencephalic area coordinates the systemic sympathetic response to an increase skeletal muscle blood flow to adjust the “fight or flight” situation [56, 57].
The clinic-ambulatory difference in blood pressure has been conventionally used as a surrogate measure of the white-coat effect. However, this difference has no or low correlation with the pressor response triggered by a doctor’s measurement of blood pressure or the true white-coat effect [58, 59]. Clinic-ambulatory difference in blood pressure is positively correlated with clinic blood pressure and negatively correlated with daytime ambulatory blood pressure [60]. This finding indicates that the clinic-ambulatory difference in blood pressure is not a pure measure of pressor response in the clinic but rather a variable largely affected by environmental factors outside the clinic [61]. These factors may include physical activity, workload, and smoking. In the Progetto Ipertensione Umbria Monitoraggio Ambulatoriale study on 1522 subjects, the clinic-ambulatory blood pressure difference was not correlated with left ventricular mass nor long-term cardiovascular morbidity, indicating that this measure had no clinical significance [60]. The absence of clinical significance was also confirmed by the study on ambulatory monitoring of blood pressure and lisinopril evaluation. This study reported that 12-month treatment-induced reductions in left ventricular mass index and those in the clinic-daytime or clinic-home differences in systolic and diastolic blood pressure showed no significant relationship [62].
Only a few studies have examined the clinical significance of the pure white-coat effect, which can be investigated by monitoring beat-to-beat blood pressure at rest and at a doctor’s visit (Fig. 2) [54, 58, 59]. The true white-coat effect is defined as the mean value of the pressor response or the maximum point triggered by the doctor’s measurement of blood pressure relative to measurement in the resting period. Lantelme et al. [59] examined the relationship between the pure white-coat effect and organ damage in 88 hypertensive patients. They found no difference in terms of cardiac mass, diastolic function, arterial distensibility, or renal function between the groups with high and low white-coat effects. In our study conducted in 75 hypertensive patients who were never treated, the pure white-coat effect was an independent predictor of left ventricular mass index in men and left ventricular diastolic function in women [54]. Thus, our data showed that the white-coat effect has clinical significance. Men showed a positive correlation between blood pressure response to the doctor’s visit and a stress-induced increase in blood pressure, whereas women did not. These data suggest that the white-coat effect correlates with blood pressure reactivity to real-life stressors in men; thus, it predicts left ventricular mass. In our study, clinic blood pressures and pure white-coat effects were similar between men and women, whereas the clinic-ambulatory difference in blood pressure in men was nearly half of that in women (14 ± 3/8 ± 2 vs. 28 ± 3/14 ± 2 mmHg, p < 0.03 for both). This finding indicates that an increase in daytime ambulatory blood pressures in men resulted in considerable underestimation of the white-coat effect. Therefore, it is plausible that the clinic-ambulatory difference in blood pressure could reflect the white-coat effect in subjects with truly normal ambulatory blood pressure, where the influence of pressor factors during ambulatory conditions would be minimized. Correlation between the systolic white-coat effect and clinic-ambulatory difference in systolic blood pressure was significant for women (r = 0.442, p = 0.007) but not for men (r = 0.364, p = 0.07) in our study.
Representative beat-to-beat recording of RR interval and systolic and diastolic blood pressures (SBP and DBP, respectively) during doctor’s visit and mental stress in a 52-year-old man. Blood pressure was measured in the finger using the Finapres device. The figure was adapted from Munakata et al. [54]
Patients with high clinic blood pressure but normal daytime ambulatory blood pressure were defined as having white-coat hypertension (WCH) [53]. The method for the treatment of these patients has been long debated because long-term follow-up studies and interventional studies were necessary. The majority of evidence derived from cross-sectional studies supports increased target organ damage in subjects with WCH compared with those with normotension [63,64,65,66], and the data were confirmed by meta-analysis [67, 68]. However, longitudinal studies seem to provide inconsistent results. Several observational event-based cohort studies [69,70,71] and meta-analyses [72,73,74] addressed the issue of the cardiovascular event risk of WCH. Most data showed little or no difference in the risk between untreated patients with WCH and their normotensive comparators [69, 70, 73, 74], but some data showed significant differences [71, 72]. One meta-analysis demonstrated that the incidence of stroke tended to increase in the WCH group. The corresponding hazard curve caught up with that of the ambulatory hypertension group by the 9th year of follow-up, raising some concerns about the long-term safety of WCH [72]. In this regard, Mancia et al. [71] provided novel evidence indicating different cardiovascular risks in individuals with stable or unstable WCH, that is, those in whom ambulatory blood pressure normality was associated with a persistent or non-persistent office blood pressure elevation at 2 consecutive visits, respectively. Compared with the normotensive group, the risk of cardiovascular and all-cause death was not significantly different in unstable WCH. In stable WCH, the risk was also increased when data were adjusted for baseline confounders, including ambulatory blood pressure (hazard ratio (HR) 16; p = 0.001 for cardiovascular death and 1.92; p = 0.02 for all-cause death). This study strongly suggests that stable WCH is a risk factor for cardiovascular events.
We should observe other points when considering cardiovascular risk in WCH. First, in a long-term follow-up study, WCH was associated with an increased risk of developing sustained hypertension compared with true normotension. The odds ratio (OR) was 2.86 in the Ohasama study (home BP based) [75] and 2.5 in the PAMELA study (both ABPM and home BP based) [76]. Second, WCH was associated with metabolic abnormality [77, 78] and with an increased risk of incident diabetes compared with normotension [79]. In the International Database on Ambulatory Blood Pressure in Relation to Cardiovascular Outcomes (IDACO) study, cardiovascular event risk was higher in WCH than in normotension in a diabetic population in contrast to a non-diabetic population [74]. Third, recent data showed that some organ damage depends more on clinic blood pressure than out-of-office blood pressure. Increased left atrium diameter is known to be associated with an elevated risk of cardiovascular morbidity and mortality. Bombelli et al. [80] evaluated new-onset left atrial enlargement and their correlates over a 10-year period in the PAMELA study. In multivariate analysis, office systolic blood pressure was an independent risk factor for left atrial enlargement, whereas home and ambulatory blood pressure were not. Thus, some organ damage depends more on temporal blood pressure elevation in the clinic rather than stable blood pressure in the out-of-office setting. The clinical characteristics of WCH are heterogeneous and could change over time with aging and subsequent environmental factors. Careful follow-up of the hypertension phenotype, metabolic risks, and organ damage is necessary in the management of WCH.
Whether home blood pressure could be substituted for daytime ambulatory blood pressure in the diagnosis of WCH is a controversial topic [81,82,83]. Home blood pressure measurement is much more feasible than ambulatory blood pressure monitoring [84], but these measurements monitor different aspects of blood pressure behavior [83]. In our recent study conducted in a worksite setting, of the 157 employees who showed normal clinic (<140/90 mmHg) and normal morning home blood pressures (<135/85 mmHg), 27 (17%) demonstrated high worksite blood pressure (≥135/85 mmHg) [85]. Thus, some subjects with normal home and clinic blood pressures show worksite hypertension. The percentage of worksite hypertension would be higher in WCH than in normotension because some patients with a greater white-coat effect demonstrate larger pressor reactivity to daily life stress as noted above. In other words, home blood pressure-based WCH could include true WCH and persistent hypertension with high worksite blood pressure (Fig. 3). In the International Database on Home Blood Pressure in Relation to Cardiovascular Outcome (IDHOCO) study, 6458 participants from five populations were followed up for 8.3 years [86]. Among the untreated subjects (n = 5007), cardiovascular risk was significantly higher in WCH than in normotensive subjects (adjusted HR 1.42, 95% confidence interval (CI) 1.06–1.91, p = 0.02). Therefore, in the home blood pressure-based study, cardiovascular event risk was higher in WCH than in normotension. The reasons for the difference in results between IDACO and IDHOCO studies are unclear. However, as speculated above, WCH patients defined by home blood pressure might include some degree of ambulatory hypertensive patients, leading to higher risk for this group than for the normotensive group. Examining worksite blood pressure either by ambulatory monitoring or by self-measurement is recommended to improve the diagnostic accuracy of WCH in subjects on the job.
Work environment as a profound modulator of blood pressure
Work may be the most influential factor for out-of-office blood pressure in subjects on the job [87]. The clinic-daytime blood pressure difference has been shown to be positively correlated with age [61]. Clinic blood pressures are higher than daytime blood pressures in elderly people aged over 60 years, unlike the differences in people aged 50 years or younger. Many people usually retire in their 60 s, suggesting that work conditions considerably influence blood pressure balance between clinic and out-of-clinic settings. In working people, blood pressures tend to be higher on a workday than on a day away from work [88, 89]. Moreover, the correlation between left ventricular mass and blood pressure is closer for blood pressure measured at work than for blood pressure measured at home or during sleep [90]. These data strongly suggest that work-related blood pressure increase is an important target for managing hypertension. In this section, our review focuses on the relationship between qualitative work stress, work hours, shift work, and blood pressures; all of these factors have received a great interest as a cause of cardiovascular diseases [91,92,93].
Work stress and blood pressure
The two internationally recognized theoretical models for examining the effects of job stress on blood pressure are demand–control support (DCS) [94, 95] and effort–reward imbalance (ERI) [96]. The DCS model indicates that workers experiencing high psychological demands (e.g., excessive workload, very hard or fast work, and conflicting demands) and low job control are associated with a higher risk of developing stress-related diseases [91]. Job control is a combination of skill discretion (e.g., learning new things, opportunities to develop skills, creativity, a variety of activities, and non-repetitive work) and decision authority (i.e., taking part in decisions affecting oneself, making one’s own decisions, having a say on the job and freedom as to how the work is accomplished). Poor social support has been introduced as a third component of the demand–control model [97]. This component refers to a lack of help and cooperation from supervisors and coworkers. The ERI model suggests that extrinsic efforts (i.e., pressure to work overtime, increasingly demanding work, constant time pressure, repeated interruptions) should be rewarded in various ways, such as financially (income), socially (respect and esteem), and organizationally (job security and promotion prospects) [96]. Workers are in an unhealthy condition if high extrinsic efforts are accompanied by low reward. A third component is overcommitment, which is a personal coping style that presents as being unable to withdraw from work obligations and being impatient and irritable [98]. Overcommitment has been considered to be an amplifier of the ERI effect.
Gilbert-Ouimet et al. [99] published a systematic review on the relationship between work stress and blood pressure in 2014, which included studies published from 1982 to 2011. Prospective cohorts and cross-sectional or case–control studies were considered. Cross-sectional or case–control studies that compare working and non-working conditions are important because blood pressure at work is closely related to organ damage, as described above. Sixty-four studies investigated the DCS model, twelve studies examined the ERI model, and two studies considered both models. Studies were conducted in 18 countries and included various working populations. Either clinic or ambulatory blood pressures were used, and changes in blood pressure or incident hypertension were evaluated as outcome measures. In the prospective studies, the follow-up period ranged from 6 weeks to 12 years.
DCS model: of the 40 cross-sectional studies on job strain, 16 demonstrated a significant pressor effect, namely, the difference in systolic blood pressure and diastolic blood pressure means ranging from +2 to +10.2 mmHg and +2 to +17.97 mmHg, respectively, and OR for hypertension ranging from 1.18 to 2.9. The remaining 24 studies showed no significant effects on blood pressures. If the data were analyzed by gender, worse effects were more frequent in men (blood pressure level: 6/18 studies, hypertension: 2/5 studies) than in women (blood pressure level: 1/10, hypertension: 0/7 studies). Of the 12 prospective studies on job strain, 9 demonstrated a significant pressor effect, namely, the difference in systolic blood pressure and diastolic blood pressure means ranging from +1.2 to +7.7 mmHg and +0.8 to +7 mmHg, respectively, and OR for hypertension ranging from 1.27 to 2.06. Worse effects were also more frequently observed in men than in women in studies on blood pressure level as an outcome measure (5/5 vs. 2/4 studies). Moreover, in one study on incident hypertension as an outcome measure, a significant effect was observed in men but not in women.
The method of blood pressure measurements could considerably influence the results. Overall, a higher percentage of studies using ambulatory blood pressure measures demonstrated more worse effects of job strain than those using office blood pressure measures (13/20 vs. 12/35 studies). This tendency was more evident in cross-sectional studies (9/15 vs. 7/27 studies) than in prospective studies (4/5 vs. 3/4 studies).
Some studies examined the relationship between job stress components, i.e., job demand or job control, and blood pressures. Significantly worse effects were observed for high psychological demands in 7/25 studies on blood pressure level and 2/7 studies on hypertension and for low social support in 1/9 studies on blood pressure levels. A significant protective effect was observed for high job control in 9/25 studies on blood pressure level and 3/6 studies on hypertension. Thus, not only job strain but also its components should be considered as measures of work-related stress. This concept was confirmed by our recent study conducted in Japanese male hospital employees [100]. We examined resting blood pressures and job stress components in 113 Japanese male hospital clerks (38.1 ± 4.4 years). Subjects were classified into normotensive (<130/85 mmHg, n = 83) and mildly elevated blood pressure (≥130/85 mmHg, n= 30) groups. Subjects with low job control demonstrated higher diastolic blood pressure than those with high job control (89.1 ± 2.1 vs. 82.3 ± 2.3 mmHg, p = 0.042) in the mildly elevated blood pressure group, even after adjustments for co-variates. Diastolic blood pressure did not differ between low and high job control subjects in the normotensive group. Systolic blood pressure did not differ between high and low job control subjects in both groups. Neither systolic blood pressure nor diastolic blood pressure differed between high- and low-demand groups in either group. Thus, low job control was independently related to high diastolic blood pressure in men with mildly elevated blood pressure but not in normotensive men. This result suggests higher stress blood pressure reactivity in the former [100].
ERI model: of the 11 cross-sectional studies with the ERI model, 7 demonstrated a significant pressor effect, namely, the difference in systolic blood pressure and diastolic blood pressure means ranging from +1.86 to +4.52 mmHg and +1.31 to +4.17 mmHg, respectively, and OR for hypertension ranging from 1.62 to 5.77. In addition, two cross-sectional studies evaluated the separate effect of effort and reward, but none of the studies observed significant effects. When the effects were analyzed by gender, six cross-sectional studies presented results separately for men and women. The worse effect was more frequent in men than in women (5/6 vs. 1/6 studies). In line with the results of the DCS model, adverse effects tended to be more frequently observed in studies conducted using ambulatory blood pressure measure than those using office blood pressure measure (3/4 vs. 5/8 studies). A significantly worse effect of overcommitment was observed in one out of three cross-sectional studies. This study observed a higher ambulatory systolic blood pressure mean among men, but no effects were noted among women.
Some new evidence after this review will also be discussed. In a Canadian prospective study including >3000 white-collar male and female workers, women with high ERI scores demonstrated a significant increase in systolic ambulatory blood pressure after 3 years. Moreover, a significant increase in incident hypertension was found among older women, whereas no significant effect was observed among men [101]. This trend among women was further confirmed over a 5-year observation period [102]. The double exposure of ERI and family obligations among women resulted in a significant rise in ambulatory blood pressure after 5 years [102]. The finding that ERI was associated with significantly increased risks of untreated hypertension [103] and masked hypertension is important [104].
Taken together, current evidence suggests that the deleterious effects of work stress on blood pressures are more frequently observed in studies using ambulatory blood pressure than those with clinic blood pressure either in cross-sectional or prospective studies. This finding is quite reasonable because work stress could increase blood pressure only at work, leading to masked hypertension. However, temporal blood pressure increases, even when restricted to work times, could lead to the development of organ damage if they are repeated over time. An examination of work-related blood pressure changes by ambulatory blood pressure monitoring in subjects involved in high-stress jobs is strongly recommended.
However, self-blood pressure measurement at the workplace may be more useful and practical than ambulatory blood pressure monitoring in some situations. Cardiovascular events have been reported to often occur on Monday morning compared with other week days among the working population [105, 106]. This effect has been attributed in part to high Monday morning blood pressure [105, 107]. However, repeating ambulatory blood pressure monitoring within a week is difficult. To address this issue, we conducted a multicenter study to examine the hypothesis of whether blood pressure is higher on Monday morning than on other days of the week [108]. We studied 207 treated hypertensive subjects working for 29 Rosai hospitals. Blood pressures and heart rate were measured with an automated device at standardized conditions 4 times a day: immediately after waking, 10:00 h, 16:00 h, and before going to bed on three days (Monday, Friday, and Saturday or Sunday). Job stress was examined using the Japanese version of the JDC model [109]. Urinary albumin excretion was examined as a measure of endothelial damage and future cardiovascular event risk [110]. Recorded data were automatically transferred to a data center through a wireless telephone. A significant interaction was found between the rate pressure product (systolic blood pressure × heart rate) and weekly and daily variations (Fig. 4). The rate pressure product was higher in the morning on Monday than at other times or on other days. This tendency was more evident in women than in men. The increase in rate pressure product on Monday morning was significantly correlated with urinary albumin excretion and job strain index in women. Thus, higher cardiovascular load partially due to work stress on Monday morning seemed to be associated with endothelial damage in treated hypertensive subjects. In our study, the compliance for self-measurement of blood pressure was satisfactory, and no patient dropout was noted. Self-measurement of blood pressure seemed to be suitable for examining work-related daily and weekly variations.
Double product elevation on Monday morning during work. A significant interaction (alternative action) was found between the double product (systolic BP × heart rate: mmHg × bpm) and weekly and circadian rhythms, particularly in women and in the overall population. In the graphs, small square, large square, and triangle represent the weekend (Saturday or Sunday), Monday, and Friday, respectively. The figure was modified from Kimura et al. [108]
Work hours and blood pressure
Long working hours are associated with an increased risk of cardiovascular disease [93], but their relationship with hypertension remains unclear. Long working hours are officially recognized as a causal factor for “Karoshi,” death from overwork, or work-related cardiovascular events in Japan, Chinese Taiwan, and Korea [111, 112]. However, to the best of our knowledge, no systematic review has been conducted on work hours and elevation of blood pressure. Thus, we surveyed studies on the relationship between work hours and blood pressure (Table 1).
The results were inconsistent among studies. Six studies reported a positive relationship between long work hours and blood pressure increase or increased risk of incident hypertension, whereas two studies demonstrated no relationship. Surprisingly, three studies from Japan reported an inverse relationship between work hours and incident or prevalent hypertension. The first pioneering work was conducted by Hayashi et al. [113]. They compared ambulatory blood pressures at normal control work periods and those at very long overtime work period in white-collar workers. Blood pressure was significantly higher when working overtime (average overtime of 96 h per month) than in a “control” period (average overtime of 43 h per month). Iwasaki et al. [114] showed significantly higher systolic blood pressure among salesmen aged 50–60 years who spent >61 h per week commuting and working than among those who spent <57 h. However, blood pressure did not differ between longer and shorter working subgroups younger than 50 years of age. Yang et al. [115] showed that in the US working population, individuals who worked 40, 41–50, and >50 h per week were 1.14-fold (95% CI, 1.01–1.28), 1.17-fold (95% CI, 1.04–1.33), and 1.29-fold (95% CI, 1.10–1.52) more likely to have self-reported hypertension, respectively, than individuals who worked 11–39 h per week. Although the study was cross-sectional, the findings were important because it included a large range of age groups and all occupational categories. Artazcoz et al. [116] showed in the Catalonian Health Survey that long work hours were associated with hypertension (adjusted OR, 2.25; 95% CI, 1.17–4.32) among female workers only in a cross-sectional study. Nakamura et al. [117] were the first to report a positive relationship between overtime work and blood pressure increase in a longitudinal study. The multivariate-adjusted mean for 1-year change in diastolic blood pressure in 611 male assembly-line workers was 1.5 mmHg (95% CI; 0.8–2.2) for <40.0 h per month, 2.3 mmHg (95% CI 1.3–3.2) for 40.0–79.9 h per month and 5.3 mmHg (95% CI 2.7–7.9) for ≥80.0 h per month (p for heterogeneity = 0.02). A largely similar pattern was observed for systolic blood pressure. No such relationship was noted for clerks or engineers/special technicians. This study showed that work type may modify the relationship between overtime work hours and blood pressure progression. Similarly, Yoo et al. [118] reported that the HR for incident hypertension significantly increased as the number of working hours per week increased in Korean wage workers. Compared with those working 40 h or less per week, the HR of subjects working 41–50 h per week was 2.20 (95% CI; 1.19–4.06), that of subjects working 51–60 h per week was 2.40 (95% CI, 1.07–5.39) and that of subjects working 61 h and over per week was 2.87 (95% CI, 1.33–6.20).
On the other hand, several Japanese studies have reported negative associations between work hours or overtime work and the incident or prevalent hypertension. Nakanishi et al. [119] showed in 941 hypertension-free (<140/90 mmHg) Japanese male white-collar workers that the relative risk for hypertension above the borderline level (140/90 mmHg) was 0.63 (95% CI: 0.43, 0.91) for those who worked 10.0–10.9 h per day and 0.48 (95% CI: 0.31, 0.74) for those who worked ≥11.0 h per day compared with those who worked <8.0 h per day. In this study, architects or research workers worked more overtime than clerks, and being an architect or a research worker (vs. being a clerk) was negatively associated with the slope of blood pressure. The authors speculated that the work type would be important to determining the perception of overwork or stress and its associated responses in blood pressure. A similar prospective study was also reported in Japanese male workers [120]. Imai et al. [121] conducted a large-scale cross-sectional study to examine long working hours and prevalent hypertension. Their participants consisted of 52,365 workers from four companies that provided both health-checkup data and self-reported data on overtime work. Logistic regression analysis was performed to determine the OR for hypertension in each category of overtime work (<45, 45–79, 80–99 or ≥100 h per month) with adjustments for co-variates. The adjusted ORs (95% CI) for hypertension were 1.00 (reference), 0.81 (0.75–0.86), 0.73 (0.62–0.86), and 0.58 (0.44–0.76) for <45, 45–79, 80–99, and ≥100 h/month, respectively. Two studies showed no relationship between work hours and blood pressure or incident hypertension [122, 123].
Thus, current evidence suggests that the relationship between work hours and blood pressure varies among studies and is not conclusive. The type of job and job satisfaction are assumed to largely modify the relationship.
Shift work and blood pressure
Shift workers play an important role in the maintenance of production, health-care, and service industries in many countries [92]. Approximately 15–30% of workers are employed as rotational shift workers, which include working day, afternoon, and night shifts [124]. The human body has an inherent biological circadian rhythm, which is programmed to be active during the day and to rest at night. Shift workers are forced to adapt their behavior against such an inherent rhythm and thus experience complex stress from psychological, physiological, and behavioral aspects [125]. Therefore, shift work is hazardous to one’s health [126, 127]. The risk of cardiovascular disease has been reported to be increased in shift workers compared with non-shift workers. A recent meta-analysis has shown that shift workers had a 26% higher risk of coronary artery disease than non-shift workers [92]. This finding might be mediated partially by an altered circadian blood pressure rhythm or a blood pressure elevation induced by shift work.
Circadian blood pressure profiles of rotating shift workers have been compared in several studies using ambulatory blood pressure monitoring. Some studies have shown that these profiles are identical or nearly the same between normal work and shift work days and concluded that circadian blood pressure change is largely determined by the cycle of physical or mental activity and sleep [128,129,130]. However, others reported an altered circadian blood pressure rhythm in shift workers. Chau et al. carefully examined the circadian blood pressure profile curve of morning, afternoon, and night shifts by Fourier series [131]. They found that high pressure periods did not coincide entirely with the times of activity. They concluded that factors other than the subject’s activity contribute to the circadian blood pressure profiles of shift workers. Ohira et al. [132] compared 24-h ambulatory blood pressure between 27 shift workers and 26 day workers. Even after adjustment for co-variates, the blood pressure during waking and work periods was higher in shift workers than in day workers, although sleep blood pressures were similar. This study showed greater pressor reactivity during work in shift workers than in day workers. To precisely examine the dynamic changes in blood pressure and autonomic nervous activity associated with shift work, we studied 18 healthy nurses engaged in a shift rotation system (day work, 8:15–17:15; evening work, 16:00–22:00; night work, 21:30–8:30) [133]. Blood pressure, heart rate, RR interval variability, and physical activity were measured for 24 h from the start of work during the night and day shifts. Systolic blood pressure and heart rate during work were lower during the night shift than the during day shift. Both parameters were still lower (p < 0.005 and p < 0.05) when they were measured outside of the hospital under waking conditions following a night shift than following a day shift, even though the levels of physical activity were similar. The high-frequency power spectrum of RR interval variability, a measure of cardiac vagal modulation, was greater not only during work but also during the awake period after a night shift compared with a day shift. Blood pressures during sleep were similar between day and night shifts. Thus, our data showed that waking blood pressure could be lower after a night shift due to altered autonomic regulation compared with day shift in subjects involved in rotating shift work. This finding was a new cause of the flattening of circadian blood pressure rhythms in rotating shift workers. Kitamura et al. [134] examined the changes in circadian blood pressure patterns in response to work shift changes in hypertensive patients. Ambulatory blood pressure monitoring was performed three times: the last day of a 4-day period of day shifts, the first day of a 4-day period of night shifts, and the fourth day of night shifts. Circadian rhythm was dipper on the day of a day shift and the last day of night shifts but was non-dipper on the first day of night shifts. Non-dipper patterns on the first day of night shifts were associated with high sleep blood pressure compared with pressures on day shifts, but awake blood pressure also tended to be lower in the night shift than in the day shift, confirming our results. The results suggest that the sympathetic modulation of awake and sleep blood pressures dynamically changes within a period of rotating shift. Later studies also confirmed these findings [135, 136]. Therefore, the results of the cross-sectional comparison of blood pressure between shift workers and non-shift workers might be inconsistent [131, 137,138,139,140]. Overall evidence suggests that rotating shift workers repeatedly suffer from a non-dipper pattern of circadian blood pressure rhythm partially due to altered sympathetic cardiovascular modulation. Repeated experiences of non-dipper circadian blood pressure rhythms are speculated to increase the risk of cardiovascular events [141].
How about the circadian blood pressure rhythm of subjects involved in fixed rotating shifts? Are they completely adjusted to environmental changes? Sternberg et al. [142] reported that the awake–sleep difference in mean systolic/diastolic ambulatory blood pressure was smaller in night workers than in day workers (7.9/7 vs. 15.5/13 mmHg). In another study evaluating 58 day-shift workers and 35 evening + night-shift workers, the evening + night workers had a significantly smaller decrease in systolic blood pressure during sleep than the day-shift workers. This result was largely attributed to higher sleep systolic blood pressure in the evening + night-shift group [143]. Urinary noradrenaline and adrenaline were higher during work than non-work in day-shift workers. However, in evening + night-shift workers, the difference was small and in the opposite direction, suggesting an altered sympathetic nervous rhythm of the evening + night-shift group. Kario et al. [144] examined the hypothesis of whether cardiovascular reactivity to acute stress and/or delayed recovery predicts greater diurnal blood pressure variation between day and fixed night-shift female workers. Night-shift workers demonstrated higher sleep systolic blood pressure than day-shift workers (105 vs. 99 mmHg), although the awake systolic blood pressure was similar between groups (118 vs. 116 mmHg). In the night-shift group, the sleep/awake systolic blood pressure ratio was negatively correlated with relative systolic blood pressure reactivity (r = −0.41, p = 0.02) and relative stress response of systolic blood pressure (r = −0.48, p = 0.006) induced by anger recall, but it was positively correlated with recovery rate (r = 0.34, p = 0.06). These correlations were not significant in the day-shift group. The sleep/awake systolic blood pressure ratio was inversely correlated with an exercise-induced systolic blood pressure increase in day-shift workers (r = −0.30, p = 0.03), but this association was not found in night-shift workers. These lines of evidence suggest that fixed night-shift workers never demonstrate normal circadian blood pressure rhythm but are associated with less dipping patterns of circadian blood pressure rhythms partially due to altered sympatho-adrenal modulation of the cardiovascular system.
Finally, the influence of long-term shift work on incident hypertension or blood pressure progression is discussed. Morikawa et al. [145] conducted a 5-year prospective study in a cohort of 1551 male manual workers and found that the OR for incident hypertension in shift workers relative to daytime workers was 3.6 in the group aged 18–29 years. Sakata et al. [146] reported a significant relationship between alternating shift work and hypertension based on a prospective cohort study of 5338 workers over a 10-year period. The OR of the onset of hypertension in shift workers for daytime workers was 1.10 and significant. Furthermore, in the same group, alternating shift work was significantly associated with the progression of mild hypertension to severe hypertension [147] and an increase in blood pressure in a cohort study over a 10-year period [148]. Notably, the effects of shift work on blood pressure increase were more pronounced than those of age or body mass index [148]. Thus, many Japanese studies confirmed that long-term shift work increases the risk of incident hypertension and blood pressure progression, but some exceptions have been reported in other countries [149, 150]. Therefore, the relationship between shift work and blood pressure progression might be modified by Japan-specific lifestyle factors, such as high salt intake. This important issue should be elucidated in the future.
Caregiving
In this section, the review focuses on home stress because not only work stress but also home stress considerably influences ambulatory blood pressures [151].
The world’s population is rapidly aging. The number of old people (aged 65 and over) was 562 million (8% of 7 billion total population) in 2012, and it increased by 595 million (to 8.5% of total population) in 2015 [152]. In Japan, life expectancy at birth is currently nearly the highest in the world. However, the duration of life with light or moderate disability increases with an increase in life expectancy at birth for both genders [153]. Thus, the number of elderly people who require caregiving is expected to markedly increase.
Family caregiving can be a positive and rewarding experience [154]. However, it is associated with a variety of deleterious health behaviors and outcomes [155, 156]. In a nationally representative sample of US adults, spousal caregiving is associated with a significant elevation in the risk of cardiovascular disease [157]. Hypertension may partially explain this connection between caregiving and an increase in cardiovascular disease risk.
In a cross-sectional study conducted in USA, Switzerland, and Japan, blood pressures were consistently higher in caregivers than in non-caregivers after adjustments for co-variates [158,159,160]. King et al. [158] compared ambulatory blood pressure and heart rate responses to the stress of work and caregiving in older women. Caregivers demonstrated a significant increase in systolic blood pressure when returning home to the care recipient, whereas systolic blood pressure declined when leaving work and returning home in non-caregivers. These observations strongly suggest that caregiving stress could be a cause of home hypertension. Only a few longitudinal studies have examined whether caregiving could be a risk of incident hypertension [161, 162]. Shaw et al. [161] compared the risk of incident hypertension between spousal caregivers of Alzheimer’s disease patients and non-caregiving controls (n = 47). Based on periodic 6-month assessments of blood pressure over 6 years, the risk for borderline hypertension was greater for caregivers than for controls (HR 4.86, p = 0.03). Capistrant et al. [162] examined this issue in a large, nationally representative sample of American older adults. Married, hypertension-free, health and retirement study respondents (n = 5708) were followed for up to 8 years. After adjusting for multiple co-variates, current caregiving significantly predicted hypertension incidence (RR = 1.36, 95% CI: 1.01–1.83). For long-term caregivers, significant evidence of the risk of hypertension onset was associated with caregiving (RR = 2.29, 95% CI: 1.17–4.49). Thus, available evidence strongly suggests a link between caregiving stress and the risk of incident hypertension and temporal blood pressure elevation.
Conclusion
This article reviewed the clinical significance of stress-related elevations in blood pressure in office and out-of-office settings separately. WCH was the most common office stress-related hypertension phenotype. Currently, there is no definite evidence indicating that untreated WCH is associated with increased cardiovascular event risk compared with normotension. However, WCH is associated with long-term risk of sustained hypertension and diabetes. Moreover, WCH demonstrates worse cardiovascular prognosis as compared with normotension in the diabetic population. The risk of WCH is never determined from temporal data but must be evaluated from long-term observation of hypertension phenotype and development of complication. Masked hypertension is a well-known poor prognostic hypertension phenotype. The cause of masked hypertension is complex, but stress at workplace or home could be involved. Chronic stress as well as allostatic load, distorted lifestyle, and mental distress could increase blood pressure.
Removal of such stress is difficult, and the evidence is limited if stress-reduction techniques have hypotensive effects. However, considering patients’ background and giving them an opportunity to present their concern help to establish rapport [163], which is essential to keep long-term compliance for hypertension treatment.
References
Bromfield S, Muntner P. High blood pressure: the leading global burden of disease risk factor and the need for worldwide prevention programs. Curr Hypertens Rep. 2013;15:134–6.
Forouzanfar MH, Liu P, Roth GA, Ng M, Biryukov S, Marczak L, Alexander L, Estep K, Abate KH, Akinyemiju TF, Ali R, Alvis-Guzman N, Azzopardi P, Banerjee A, Baernighausen T, Basu A, Bekele T, Bennett DA, Biadgilign S, Catala-Lopez F, Feigin VL, Fernandes JC, Fischer F, Gebru AA, Gona P, Gupta R, Hankey GJ, Jonas JB, Judd SE, Khang Y-H, Khosravi A, Kim YJ, Kimokoti RW, Kokubo Y, Kolte D, Lopez A, Lotufo PA, Malekzadeh R, Melaku YA, Mensah GA, Misganaw A, Mokdad AH, Moran AE, Nawaz H, Neal B, Ngalesoni FN, Ohkubo T, Pourmalek F, Rafay A, Rai RK, Rojas-Rueda D, Sampson UK, Santos IS, Sawhney M, Schutte AE, Sepanlou SG, Shifa GT, Shiue I, Tedla BA, Thrift AG, Tonelli M, Truelsen T, Tsilimparis N, Ukwaja KN, Uthman OA, Vasankari T, Venketasubramanian N, Vlassov VV, Vos T, Westerman R, Yan LL, Yano Y, Yonemoto N, Zaki MES, Murray CJL. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mmHg, 1990-2015. JAMA. 2017;317:165–82.
Mendis S, Davis S, Norrving B. Organizational update: the world health organization global status report on noncommunicable diseases 2014; one more landmark step in the combat against stroke and vascular disease. Stroke. 2015;46:e121–2.
Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, de Ferranti S, Després JP, Fullerton HJ, Howard VJ, Huffman MD, Judd SE, Kissela BM, Lackland DT, Lichtman JH, Lisabeth LD, Liu S, Mackey RH, Matchar DB, McGuire DK, Mohler ER 3rd, Moy CS, Muntner P, Mussolino ME, Nasir K, Neumar RW, Nichol G, Palaniappan L, Pandey DK, Reeves MJ, Rodriguez CJ, Sorlie PD, Stein J, Towfighi A, Turan TN, Virani SS, Willey JZ, Woo D, Yeh RW, Turner MB, American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics--2015 update: a report from the American Heart Association. Circulation. 2015;131:e29–322.
Kario K, Pickering TG, Umeda Y, Hoshide S, Hoshide Y, Morinari M, Murata M, Kuroda T, Schwartz JE, Shimada K. Morning surge in blood pressure as a predictor of silent and clinical cerebrovascular disease in elderly hypertensives: a prospective study. Circulation. 2003;107:1401–6.
Ohkubo T, Kikuya M, Metoki H, Asayama K, Obara T, Hashimoto J, Totsune K, Hoshi H, Satoh H, Imai Y. Prognosis of “masked” hypertension and “white-coat” hypertension detected by 24-h ambulatory blood pressure monitoring 10-year follow-up from the Ohasama study. J Am Coll Cardiol. 2005;46:508–15.
Mancia G, Bombelli M, Facchetti R, Madotto F, Corrao G, Trevano FQ, Grassi G, Sega R. Long-term prognostic value of blood pressure variability in the general population: results of the Pressioni Arteriose Monitorate e Loro Associazioni Study. Hypertension. 2007;49:1265–70.
Rothwell PM, Howard SC, Dolan E, O’Brien E, Dobson JE, Dahlöf B, Sever PS, Poulter NR. Prognostic significance of visit-to-visit variability, maximum systolic blood pressure, and episodic hypertension. Lancet. 2010;375:895–905.
Kikuya M, Ohkubo T, Metoki H, Asayama K, Hara A, Obara T, Inoue R, Hoshi H, Hashimoto J, Totsune K, Satoh H, Imai Y. Day-by-day variability of blood pressure and heart rate at home as a novel predictor of prognosis: the Ohasama study. Hypertension. 2008;52:1045–50.
Singh M, Singh AK, Pandey P, Chandra S, Singh KA, Gambhir IS. Molecular genetics of essential hypertension. Clin Exp Hypertens. 2016;38:268–77.
Waken RJ, de Las Fuentes L, Rao DC. A review of the genetics of hypertension with a focus on gene-environment interactions. Curr Hypertens Rep. 2017;19:23.
Hall JE, Granger JP, do Carmo JM, da Silva AA, Dubinion J, George E, Hamza S, Speed J, Hall ME. Hypertension: physiology and pathophysiology. Compr Physiol. 2012;2:2393–442.
Brook RD. The environment and blood pressure. Cardiol Clin. 2017;35:213–21.
Miura K, Nagai M, Ohkubo T. Epidemiology of hypertension in Japan: where are we now? Circ J. 2013;77:2226–31.
Falaschetti E, Chaudhury M, Mindell J, Poulter N. Continued improvement in hypertension management in England: results from the Health Survey for England 2006. Hypertension. 2009;53:480–6.
Egan BM, Zhao Y, Axon RN. US trends in prevalence, awareness, treatment, and control of hypertension, 1988-2008. JAMA. 2010;303:2043–50.
Cuffee Y, Ogedegbe C, Williams NJ, Ogedegbe G, Schoenthaler A. Psychosocial risk factors for hypertension: an update of the literature. Curr Hypertens Rep. 2014;16:483.
Huang CJ, Webb HE, Zourdos MC, Acevedo EO. Cardiovascular reactivity, stress, and physical activity. Front Physiol. 2013;4:314.
Ushakov AV, Ivanchenko VS, Gagarina AA. Psychological stress in pathogenesis of essential hypertension. Curr Hypertens Rev. 2016;12:203–14.
Puzserova A, Bernatova I. Blood pressure regulation in stress: focus on nitric oxide-dependent mechanisms. Physiol Res. 2016;65:S309–342.
Rohleder N. Stimulation of systemic low-grade inflammation by psychosocial stress. Psychosom Med. 2014;76:181–9.
Cheung BM, Ong KL, Tso AW, Leung RY, Xu A, Cherny SS, Sham PC, Lam TH, Lam KS. C-reactive protein as a predictor of hypertension in the Hong Kong Cardiovascular Risk Factor Prevalence Study (CRISPS) cohort. J Hum Hypertens. 2012;26:108–16.
Michishita R, Ohta M, Ikeda M, Jiang Y, Yamato H. An exaggerated blood pressure response to exercise is associated with nitric oxide bioavailability and inflammatory markers in normotensive females. Hypertens Res. 2016;39:792–8.
Marvar PJ, Vinh A, Thabet S, Lob HE, Geem D, Ressler KJ, Harrison DG. T lymphocytes and vascular inflammation contribute to stress-dependent hypertension. Biol Psychiatry. 2012;71:774–82.
Lagraauw HM, Kuiper J, Bot I. Acute and chronic psychological stress as risk factors for cardiovascular disease: insights gained from epidemiological, clinical and experimental studies. Brain Behav Immun. 2015;50:18–30.
Whelton PK, Carey RM, Aronow WS, Casey DE Jr., Collins KJ, Dennison Himmelfarb C, DePalma SM, Gidding S, Jamerson KA, Jones DW, MacLaughlin EJ, Muntner P, Ovbiagele B, Smith SC Jr., Spencer CC, Stafford RS, Taler SJ, Thomas RJ, Williams KA Sr., Williamson JD, Wright JT Jr.. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines. Hypertension. 2017;71:e13–115.
Nagele E, Jeitler K, Horvath K, Semlitsch T, Posch N, Herrmann KH, Grouven U, Hermanns T, Hemkens LG, Siebenhofer A. Clinical effectiveness of stress-reduction techniques in patients with hypertension: systematic review and meta-analysis. J Hypertens. 2014;32:1936–44.
Liu L, Li M, Song S, Shi A, Cheng S, Dang X, Chen H, Zhang H, Ziguli A, Cao L, Wang P, Luan H, Ma Y, Zhang S, Wang Z, Wang X, Gao R, Tian G. Effects of long-term psychological intervention on blood pressure and health-related quality of life in patients with hypertension among the Chinese working population. Hypertens Res. 2017;40:999–1007.
Ordaz S, Luna B. Sex differences in physiological reactivity to acute psychosocial stress in adolescence. Psychoneuroendocrinology. 2012;37:1135–57.
Dampney RA. Central neural control of the cardiovascular system: current perspectives. Adv Physiol Educ. 2016;40:283–96.
Selye H. Stress and the general adaptation syndrome. Br Med J. 1950;1:1383–92.
Keller A, Litzelman K, Wisk LE, Maddox T, Cheng ER, Creswell PD, Witt WP. Does the perception that stress affects health matter? The association with health and mortality. Health Psychol. 2012;31:677–84.
McEwen BS. Stress, adaptation, and disease. Allostasis and allostatic load. Ann N Y Acad Sci. 1998;840:33–44.
Jennings JR, Kamarck TW, Everson-Rose SA, Kaplan GA, Manuck SB, Salonen JT. Exaggerated blood pressure responses during mental stress are prospectively related to enhanced carotid atherosclerosis in middle-aged Finnish men. Circulation. 2004;110:2198–203.
Matthews KA, Zhu S, Tucker DC, Whooley MA. Blood pressure reactivity to psychological stress and coronary calcification in the Coronary Artery Risk Development in Young Adults Study. Hypertension. 2006;47:391–5.
Cardillo C, Degen C, De Felice F, Folli G. Relationship of stress testing blood pressure with electrocardiographic and fundoscopy indices of hypertensive end-organ damage. Clin Exp Hypertens A. 1992;14:469–88.
Lipman RD, Grossman P, Bridges SE, Hamner JW, Taylor JA. Mental stress response, arterial stiffness, and baroreflex sensitivity in healthy aging. J Gerontol A Biol Sci Med Sci. 2002;57:B279–84.
Harshfield GA, Dong Y, Kapuku GK, Zhu H, Hanevold CD. Stress-induced sodium retention and hypertension: a review and hypothesis. Curr Hypertens Rep. 2009;11:29–34.
Gerber M, Puhse U. Review article: do exercise and fitness protect against stress-induced health complaints? A review of the literature. Scand J Public Health. 2009;37:801–19.
Kidambi S, Kotchen JM, Krishnaswami S, Grim CE, Kotchen TA. Cardiovascular correlates of insulin resistance in normotensive and hypertensive African Americans. Metabolism. 2011;60:835–42.
Black PH, Garbutt LD. Stress, inflammation and cardiovascular disease. J Psychosom Res. 2002;52:1–23.
Schiffrin EL. The endothelium and control of blood vessel function in health and disease. Clin Invest Med. 1994;17:602–20.
Versari D, Daghini E, Virdis A, Ghiadoni L, Taddei S. Endothelium-dependent contractions and endothelial dysfunction in human hypertension. Br J Pharmacol. 2009;157:527–36.
Hamer M. Psychosocial stress and cardiovascular disease risk: the role of physical activity. Psychosom Med. 2012;74:896–903.
Aro S, Hasan J. Occupational class, psychosocial stress and morbidity. Ann Clin Res. 1987;19:62–8.
McQueen DV, Celentano DD. Social factors in the etiology of multiple outcomes: the case of blood pressure and alcohol consumption patterns. Soc Sci Med. 1982;16:397–418.
Artinian NT, Washington OG, Flack JM, Hockman EM, Jen KL. Depression, stress, and blood pressure in urban African-American women. Prog Cardiovasc Nurs. 2006;21:68–75.
Udupa K, Sathyaprabha TN, Thirthalli J, Kishore KR, Lavekar GS, Raju TR, Gangadhar BN. Alteration of cardiac autonomic functions in patients with major depression: a study using heart rate variability measures. J Affect Disord. 2007;100:137–41.
Malan L, Schutte CE, Alkerwi A, Stranges S, Malan NT. Hypothalamic-pituitary-adrenal-axis dysregulation and double product increases potentiate ischemic heart disease risk in a Black male cohort: the SABPA study. Hypertens Res. 2017;40:590–7.
Ulmer CS, Bosworth HB, Germain A, Lindquist J, Olsen M, Brancu M, VA Mid-Atlantic Mental Illness Research Education and Clinical Center Registry Workgroup, Beckham JC. Associations between sleep difficulties and risk factors for cardiovascular disease in veterans and active duty military personnel of the Iraq and Afghanistan conflicts. J Behav Med. 2015;38:544–55.
Mancia G, Bertinieri G, Grassi G, Parati G, Pomidossi G, Ferrari A, Gregorini L, Zanchetti A. Effects of blood-pressure measurement by the doctor on patient’s blood pressure and heart rate. Lancet. 1983;2:695–8.
Mancia G, Parati G, Pomidossi G, Grassi G, Casadei R, Zanchetti A. Alerting reaction and rise in blood pressure during measurement by physician and nurse. Hypertension. 1987;9:209–15.
Pickering TG, James GD, Boddie C, Harshfield GA, Blank S, Laragh JH. How common is white coat hypertension? JAMA. 1988;259:225–8.
Munakata M, Saito Y, Nunokawa T, Ito N, Fukudo S, Yoshinaga K. Clinical significance of blood pressure response triggered by a doctor’s visit in patients with essential hypertension. Hypertens Res. 2002;25:343–9.
Grassi G, Turri C, Vailati S, Dell’Oro R, Mancia G. Muscle and skin sympathetic nerve traffic during the “white-coat” effect. Circulation. 1999;100:222–5.
Adams DB, Baccelli G, Mancia G, Zanchetti A. Cardiovascular changes during preparation for fighting behaviour in the cat. Nature. 1968;220:1239–40.
Folkow B. Physiology of behaviour and blood pressure regulation in animals. In: Julius S, Bassett DR, editors. Handbook of hypertension: behavioral factors in hypertension. Amsterdam: Elsevier Science Publishers; 1987. p. 1–18.
Parati G, Ulian L, Santucciu C, Omboni S, Mancia G. Difference between clinic and daytime blood pressure is not a measure of the white coat effect. Hypertension. 1998;31:1185–9.
Lantelme P, Milon H, Vernet M, Gayet C. Difference between office and ambulatory blood pressure or real white coat effect: does it matter in terms of prognosis? J Hypertens. 2000;18:383–9.
Verdecchia P, Schillaci G, Borgioni C, Ciucci A, Porcellati C. Prognostic significance of the white coat effect. Hypertension. 1997;29:1218–24.
Conen D, Aeschbacher S, Thijs L, Li Y, Boggia J, Asayama K, Hansen TW, Kikuya M, Björklund-Bodegård K, Ohkubo T, Jeppesen J, Gu YM, Torp-Pedersen C, Dolan E, Kuznetsova T, Stolarz-Skrzypek K, Tikhonoff V, Schoen T, Malyutina S, Casiglia E, Nikitin Y, Lind L, Sandoya E, Kawecka-Jaszcz K, Mena L, Maestre GE, Filipovský J, Imai Y, O’Brien E, Wang JG, Risch L, Staessen JA. Age-specific differences between conventional and ambulatory daytime blood pressure values. Hypertension. 2014;64:1073–9.
Parati G, Ulian L, Sampieri L, Palatini P, Villani A, Vanasia A, Mancia G. Attenuation of the “white-coat effect” by antihypertensive treatment and regression of target organ damage. Hypertension. 2000;35:614–20.
Glen SK, Elliott HL, Curzio JL, Lees KR, Reid JL. White-coat hypertension as a cause of cardiovascular dysfunction. Lancet. 1996;348:654–7.
Muscholl MW, Hense HW, Brockel U, Doring A, Riegger GA, Schunkert H. Changes in left ventricular structure and function in patients with white coat hypertension: cross sectional survey. BMJ. 1998;317:565–70.
Hara A, Ohkubo T, Kikuya M, Shintani Y, Obara T, Metoki H, Inoue R, Asayama K, Hashimoto T, Harasawa T, Aono Y, Otani H, Tanaka K, Hashimoto J, Totsune K, Hoshi H, Satoh H, Imai Y. Detection of carotid atherosclerosis in individuals with masked hypertension and white-coat hypertension by self-measured blood pressure at home: the Ohasama study. J Hypertens. 2007;25:321–7.
Puato M, Palatini P, Zanardo M, Dorigatti F, Tirrito C, Rattazzi M, Pauletto P. Increase in carotid intima-media thickness in grade I hypertensive subjects: white-coat versus sustained hypertension. Hypertension. 2008;51:1300–5.
Cuspidi C, Rescaldani M, Tadic M, Sala C, Grassi G, Mancia G. White-coat hypertension, as defined by ambulatory blood pressure monitoring, and subclinical cardiac organ damage: a meta-analysis. J Hypertens. 2015;33:24–32.
Cuspidi C, Sala C, Tadic M, Rescaldani M, Grassi G, Mancia G. Is white-coat hypertension a risk factor for carotid atherosclerosis? A review and meta-analysis. Blood Press Monit. 2015;20:57–63.
Khattar RS, Senior R, Lahiri A. Cardiovascular outcome in white-coat versus sustained mild hypertension: a 10-year follow-up study. Circulation. 1998;98:1892–7.
Kario K, Shimada K, Schwartz JE, Matsuo T, Hoshide S, Pickering TG. Silent and clinically overt stroke in older Japanese subjects with white-coat and sustained hypertension. J Am Coll Cardiol. 2001;38:238–45.
Mancia G, Facchetti R, Grassi G, Bombelli M. Adverse prognostic value of persistent office blood pressure elevation in white coat hypertension. Hypertension. 2015;66:437–44.
Verdecchia P, Reboldi GP, Angeli F, Schillaci G, Schwartz JE, Pickering TG, Imai Y, Ohkubo T, Kario K. Short- and long-term incidence of stroke in white-coat hypertension. Hypertension. 2005;45:203–8.
Pierdomenico SD, Cuccurullo F. Prognostic value of white-coat and masked hypertension diagnosed by ambulatory monitoring in initially untreated subjects: an updated meta analysis. Am J Hypertens. 2011;24:52–8.
Franklin SS, Thijs L, Hansen TW, Li Y, Boggia J, Kikuya M, Björklund-Bodegård K, Ohkubo T, Jeppesen J, Torp-Pedersen C, Dolan E, Kuznetsova T, Stolarz-Skrzypek K, Tikhonoff V, Malyutina S, Casiglia E, Nikitin Y, Lind L, Sandoya E, Kawecka-Jaszcz K, Imai Y, Wang J, Ibsen H, O’Brien E, Staessen JA, International Database on Ambulatory Blood Pressure in Relation to Cardiovascular Outcomes Investigators. Significance of white-coat hypertension in older persons with isolated systolic hypertension: a meta-analysis using the International Database on Ambulatory Blood Pressure Monitoring in Relation to Cardiovascular Outcomes population. Hypertension. 2012;59:564–71.
Ugajin T, Hozawa A, Ohkubo T, Asayama K, Kikuya M, Obara T, Metoki H, Hoshi H, Hashimoto J, Totsune K, Satoh H, Tsuji I, Imai Y. White-coat hypertension as a risk factor for the development of home hypertension: the Ohasama study. Arch Intern Med. 2005;165:1541–6.
Mancia G, Bombelli M, Facchetti R, Madotto F, Quarti-Trevano F, Polo Friz H, Grassi G, Sega R. Long-term risk of sustained hypertension in white-coat or masked hypertension. Hypertension. 2009;54:226–32.
Weber MA, Neutel JM, Smith DH, Graettinger WF. Diagnosis of mild hypertension by ambulatory blood pressure monitoring. Circulation. 1994;90:2291–8.
Kario K, Pickering TG. White-coat hypertension or white-coat hypertension syndrome: which is accompanied by target organ damage? Arch Intern Med. 2000;160:3497–8.
Mancia G, Bombelli M, Facchetti R, Madotto F, Quarti-Trevano F, Grassi G, Sega R. Increased long-term risk of new-onset diabetes mellitus in white-coat and masked hypertension. J Hypertens. 2009;27:1672–8.
Bombelli M, Cuspidi C, Facchetti R, Sala C, Tadic M, Brambilla G, Re A, Villa P, Grassi G, Mancia G. New-onset left atrial enlargement in a general population. J Hypertens. 2016;34:1838–45.
Stergiou GS, Nasothimiou E, Giovas P, Kapoyiannis A, Vazeou A. Diagnosis of hypertension in children and adolescents based on home versus ambulatory blood pressure monitoring. J Hypertens. 2008;26:1556–62.
Bayo J, Cos FX, Roca C, Dalfo A, Martin-Baranera MM, Albert B. Home blood pressure self-monitoring: diagnostic performance in white-coat hypertension. Blood Press Monit. 2006;11:47–52.
Kang YY, Li Y, Huang QF, Song J, Shan XL, Dou Y, Xu XJ, Chen SH, Wang JG. Accuracy of home versus ambulatory blood pressure monitoring in the diagnosis of white-coat and masked hypertension. J Hypertens. 2015;33:1580–7.
Imai Y, Obara T, Asamaya K, Ohkubo T. The reason why home blood pressure measurements are preferred over clinic or ambulatory blood pressure in Japan. Hypertens Res. 2013;36:661–72.
Munakata M, Kimura G, Inoue N. Final report on “lifestyle-related disease research” (in Japanese). 3rd stage medical research on work-related disease. Japan Organization of Occupational Health and Safety; 2018. Tokyo: p. 50–5.
Stergiou GS, Asayama K, Thijs L, Kollias A, Niiranen TJ, Hozawa A, Boggia J, Johansson JK, Ohkubo T, Tsuji I, Jula AM, Imai Y, Staessen JA, International Database on Home Blood Pressure in Relation to Cardiovascular Outcome (IDHOCO) Investigators. Prognosis of white-coat and masked hypertension: International Database of Home Blood Pressure in Relation to Cardiovascular Outcome. Hypertension. 2014;63:675–82.
Pickering TG. Reflections in hypertension: work and blood pressure. J Clin Hypertens. 2004;6:403–5.
Pieper C, Warren K, Pickering TG. A comparison of ambulatory blood pressure and heart rate at home and work on work and non-work days. J Hypertens. 1993;11:177–83.
del Arco-Galan C, Suarez-Fernandez C, Gabriel-Sanchez R. What happens to blood pressure when on-call? Am J Hypertens. 1994;7:396–401.
Devereux RB, Pickering TG, Harshfield GA, Kleinert HD, Denby L, Clark L, Pregibon D, Jason M, Kleiner B, Borer JS, Laragh JH. Left ventricular hypertrophy in patients with hypertension: importance of blood pressure response to regularly recurring stress. Circulation. 1983;68:470–6.
Szerencsi K, van Amelsvoort LG, Viechtbauer W, Mohren DC, Prins MH, Kant I. The association between study characteristics and outcome in the relation between job stress and cardiovascular disease - a multilevel meta-regression analysis. Scand J Work Environ Health. 2012;38:489–502.
Torquati L, Mielke GI, Brown WJ, Kolbe-Alexander T. Shift work and the risk of cardiovascular disease. A systematic review and meta-analysis including dose-response relationship. Scand J Work Environ Health. 2017;44:229–38.
Kivimaki M, Jokela M, Nyberg ST, Singh-Manoux A, Fransson EI, Alfredsson L, Bjorner JB, Borritz M, Burr H, Casini A, Clays E, De Bacquer D, Dragano N, Erbel R, Geuskens GA, Hamer M, Hooftman WE, Houtman IL, Jöckel KH, Kittel F, Knutsson A, Koskenvuo M, Lunau T, Madsen IE, Nielsen ML, Nordin M, Oksanen T, Pejtersen JH, Pentti J, Rugulies R, Salo P, Shipley MJ, Siegrist J, Steptoe A, Suominen SB, Theorell T, Vahtera J, Westerholm PJ, Westerlund H, O’Reilly D, Kumari M, Batty GD, Ferrie JE, Virtanen M, IPD-Work Consortium. Long working hours and risk of coronary heart disease and stroke: a systematic review and meta-analysis of published and unpublished data for 603,838 individuals. Lancet. 2015;386:1739–46.
Schnall PL, Dobson M, Landsbergis P. Globalization, work, and cardiovascular disease. Int J Health Serv. 2016;46:656–92.
Karasek R, Baker D, Marxer F, Ahlbom A, Theorell T. Job decision latitude, job demands, and cardiovascular disease: a prospective study of Swedish men. Am J Public Health. 1981;71:694–705.
Siegrist J. Adverse health effects of high-effort/low-reward conditions. J Occup Health Psychol. 1996;1:27–41.
Johnson JV, Hall EM, Theorell T. Combined effects of job strain and social isolation on cardiovascular disease morbidity and mortality in a random sample of the Swedish male working population. Scand J Work Environ Health. 1989;15:271–9.
Siegrist J, Starke D, Chandola T, Godin I, Marmot M, Niedhammer I, Peter R. The measurement of effort-reward imbalance at work: European comparisons. Soc Sci Med. 2004;58:1483–99.
Gilbert-Ouimet M, Trudel X, Brisson C, Milot A, Vezina M. Adverse effects of psychosocial work factors on blood pressure: systematic review of studies on demand-control-support and effort-reward imbalance models. Scand J Work Environ Health. 2014;40:109–32.
Hattori T, Munakata M. Low job control is associated with higher diastolic blood pressure in men with mildly elevated blood pressure: the Rosai Karoshi study. Ind Health. 2015;53:480–8.
Gilbert-Ouimet M, Brisson C, Vezina M, Milot A, Blanchette C. Repeated exposure to effort-reward imbalance, increased blood pressure, and hypertension incidence among white-collar workers: effort-reward imbalance and blood pressure. J Psychosom Res. 2012;72:26–32.
Trudel X, Brisson C, Milot A, Masse B, Vezina M. Adverse psychosocial work factors, blood pressure and hypertension incidence: repeated exposure in a 5-year prospective cohort study. J Epidemiol Community Health. 2016;70:402–8.
Trudel X, Milot A, Gilbert-Ouimet M, Duchaine C, Guénette L, Dalens V, Brisson C. Effort-reward imbalance at work and the prevalence of unsuccessfully treated hypertension among white-collar workers. Am J Epidemiol. 2017;186:456–62.
Boucher P, Gilbert-Ouimet M, Trudel X, Duchaine CS, Milot A, Brisson C. Masked hypertension and effort-reward imbalance at work among 2369 white-collar workers. J Hum Hypertens. 2017;31:620–6.
Willich SN, Lowel H, Lewis M, Hormann A, Arntz HR, Keil U. Weekly variation of acute myocardial infarction. Increased Monday risk in the working population. Circulation. 1994;90:87–93.
Witte DR, Grobbee DE, Bots ML, Hoes AW. Excess cardiac mortality on Monday: the importance of gender, age and hospitalisation. Eur J Epidemiol. 2005;20:395–9.
Murakami S, Otsuka K, Kubo Y, Shinagawa M, Yamanaka T, Ohkawa S, Kitaura Y. Repeated ambulatory monitoring reveals a Monday morning surge in blood pressure in a community-dwelling population. Am J Hypertens. 2004;17:1179–83.
Kimura G, Inoue N, Mizuno H, Izumi M, Nagatoya K, Ohtahara A, Munakata M. Workplace hypertension co-operative study by 29 Rosai hospitals belonging to the Japan Organization of Occupational Health and Safety. Increased double product on Monday morning during work. Hypertens Res. 2017;40:671–4.
Inoue N, Otsui K, Yoshioka T, Suzuki A, Ozawa T, Iwata S, Takei A. A simultaneous evaluation of occupational stress and depression in patients with lifestyle-related diseases. Intern Med. 2016;55:1071–5.
Konno S, Munakata M. Moderately increased albuminuria is an independent risk factor of cardiovascular events in the general Japanese population under 75 years of age: the Watari study. PLoS ONE. 2015;10:e0123893.
Iwasaki K, Takahashi M, Nakata A. Health problems due to long working hours in Japan: working hours, workers’ compensation (Karoshi), and preventive measures. Ind Health. 2006;44:537–40.
Ke DS. Overwork, stroke, and karoshi-death from overwork. Acta Neurol Taiwan. 2012;21:54–9.
Hayashi T, Kobayashi Y, Yamaoka K, Yano E. Effect of overtime work on 24-hour ambulatory blood pressure. J Occup Environ Med. 1996;38:1007–11.
Iwasaki K, Sasaki T, Oka T, Hisanaga N. Effect of working hours on biological functions related to cardiovascular system among salesmen in a machinery manufacturing company. Ind Health. 1998;36:361–7.
Yang H, Schnall PL, Jauregui M, Su TC, Baker D. Work hours and self-reported hypertension among working people in California. Hypertension. 2006;48:744–50.
Artazcoz L, Cortes I, Borrell C, Escriba-Aguir V, Cascant L. Gender perspective in the analysis of the relationship between long workhours, health and health-related behavior. Scand J Work Environ Health. 2007;33:344–50.
Nakamura K, Sakurai M, Morikawa Y, Miura K, Ishizaki M, Kido T, Naruse Y, Suwazono Y, Nakagawa H. Overtime work and blood pressure in normotensive Japanese male workers. Am J Hypertens. 2012;25:979–85.
Yoo DH, Kang MY, Paek D, Min B, Cho SI. Effect of long working hours on self-reported hypertension among middle-aged and older wage workers. Ann Occup Environ Med. 2014;26:25.
Nakanishi N, Yoshida H, Nagano K, Kawashimo H, Nakamura K, Tatara K. Long working hours and risk for hypertension in Japanese male white collar workers. J Epidemiol Community Health. 2001;55:316–22.
Wada K, Katoh N, Aratake Y, Furukawa Y, Hayashi T, Satoh E, Tanaka K, Satoh T, Aizawa Y. Effects of overtime work on blood pressure and body mass index in Japanese male workers. Occup Med. 2006;56:578–80.
Imai T, Kuwahara K, Nishihara A, Nakagawa T, Yamamoto S, Honda T, Miyamoto T, Kochi T, Eguchi M, Uehara A, Kuroda R, Omoto D, Nagata T, Pham NM, Kurotani K, Nanri A, Akter S, Kabe I, Mizoue T, Sone T, Dohi S, Japan Epidemiology Collaboration on Occupational Health Study Group. Association of overtime work and hypertension in a Japanese working population: a cross-sectional study. Chronobiol Int. 2014;31:1108–14.
Park J, Kim Y, Cho Y, Woo KH, Chung HK, Iwasaki K, Oka T, Sasaki T, Hisanaga N. Regular overtime and cardiovascular functions. Ind Health. 2001;39:244–9.
Pimenta AM, Beunza JJ, Bes-Rastrollo M, Alonso A, López CN, Velásquez-Meléndez G, Martínez-González MA. Work hours and incidence of hypertension among Spanish university graduates: the Seguimiento Universidad de Navarra prospective cohort. J Hypertens. 2009;27:34–40.
Boivin DB, Boudreau P. Impacts of shift work on sleep and circadian rhythms. Pathol Biol. 2014;62:292–301.
Reinberg A, Ashkenazi I. Internal desynchronization of circadian rhythms and tolerance to shift work. Chronobiol Int. 2008;25:625–43.
Scheer FA, Hilton MF, Mantzoros CS, Shea SA. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc Natl Acad Sci USA. 2009;106:4453–8.
Puttonen S, Harma M, Hublin C. Shift work and cardiovascular disease - pathways from circadian stress to morbidity. Scand J Work Environ Health. 2010;36:96–108.
Sundberg S, Kohvakka A, Gordin A. Rapid reversal of circadian blood pressure rhythm in shift workers. J Hypertens. 1988;6:393–6.
Baumgart P, Walger P, Fuchs G, Dorst KG, Vetter H, Rahn KH. Twenty-four-hour blood pressure is not dependent on endogenous circadian rhythm. J Hypertens. 1989;7:331–4.
Goto T, Yokoyama K, Araki T, Miura T, Saitoh H, Saitoh M, Satoh S. Identical blood pressure levels and slower heart rates among nurses during night work and day work. J Hum Hypertens. 1994;8:11–4.
Chau NP, Mallion JM, de Gaudemaris R, Ruche E, Siche JP, Pelen O, Mathern G. Twenty-four-hour ambulatory blood pressure in shift workers. Circulation 1989;80:341-7.
Ohira T, Tanigawa T, Iso H, Odagiri Y, Takamiya T, Shimomitsu T, Hayano J, Shimamoto T. Effects of shift work on 24-hour ambulatory blood pressure and its variability among Japanese workers. Scand J Work Environ Health. 2000;26:421–6.
Munakata M, Ichi S, Nunokawa T, Saito Y, Ito N, Fukudo S, Yoshinaga K. Influence of night shift work on psychologic state and cardiovascular and neuroendocrine responses in healthy nurses. Hypertens Res. 2001;24:25–31.
Kitamura T, Onishi K, Dohi K, Okinaka T, Ito M, Isaka N, Nakano T. Circadian rhythm of blood pressure is transformed from a dipper to a non-dipper pattern in shift workers with hypertension. J Hum Hypertens. 2002;16:193–7.
Su TC, Lin LY, Baker D, Schnall PL, Chen MF, Hwang WC, Chen CF, Wang JD. Elevated blood pressure, decreased heart rate variability and incomplete blood pressure recovery after a 12-hour night shift work. J Occup Health. 2008;50:380–6.
Lo SH, Liau CS, Hwang JS, Wang JD. Dynamic blood pressure changes and recovery under different work shifts in young women. Am J Hypertens. 2008;21:759–64.
Knutsson A, Akerstedt T, Jonsson BG. Prevalence of risk factors for coronary artery disease among day and shift workers. Scand J Work Environ Health. 1988;14:317–21.
Murata K, Yano E, Hashimoto H, Karita K, Dakeishi M. Effects of shift work on QTc interval and blood pressure in relation to heart rate variability. Int Arch Occup Environ Health. 2005;78:287–92.
Inoue M, Morita H, Inagaki J, Harada N. Influence of differences in their jobs on cardiovascular risk factors in male blue-collar shift workers in their fifties. Int J Occup Environ Health. 2004;10:313–8.
Sfreddo C, Fuchs SC, Merlo AR, Fuchs FD. Shift work is not associated with high blood pressure or prevalence of hypertension. PLoS ONE. 2010;5:e15250.
White WB. Ambulatory blood pressure monitoring: dippers compared with non-dippers. Blood Press Monit. 2000;5(Suppl 1):S17–23.
Sternberg H, Rosenthal T, Shamiss A, Green M. Altered circadian rhythm of blood pressure in shift workers. J Hum Hypertens. 1995;9:349–53.
Yamasaki F, Schwartz JE, Gerber LM, Warren K, Pickering TG. Impact of shift work and race/ethnicity on the diurnal rhythm of blood pressure and catecholamines. Hypertension. 1998;32:417–23.
Kario K, Schwartz JE, Gerin W, Robayo N, Maceo E, Pickering TG. Psychological and physical stress-induced cardiovascular reactivity and diurnal blood pressure variation in women with different work shifts. Hypertens Res. 2002;25:543–51.
Morikawa Y, Nakagawa H, Miura K, Ishizaki M, Tabata M, Nishijo M, Higashiguchi K, Yoshita K, Sagara T, Kido T, Naruse Y, Nogawa K. Relationship between shift work and onset of hypertension in a cohort of manual workers. Scand J Work Environ Health. 1999;25:100–4.
Sakata K, Suwazono Y, Harada H, Okubo Y, Kobayashi E, Nogawa K. The relationship between shift work and the onset of hypertension in male Japanese workers. J Occup Environ Med. 2003;45:1002–6.
Oishi M, Suwazono Y, Sakata K, Okubo Y, Harada H, Kobayashi E, Uetani M, Nogawa K. A longitudinal study on the relationship between shift work and the progression of hypertension in male Japanese workers. J Hypertens. 2005;23:2173–8.
Suwazono Y, Dochi M, Sakata K, Okubo Y, Oishi M, Tanaka K, Kobayashi E, Nogawa K. Shift work is a risk factor for increased blood pressure in Japanese men: a 14-year historical cohort study. Hypertension. 2008;52:581–6.
Virkkunen H, Harma M, Kauppinen T, Tenkanen L. Shift work, occupational noise and physical workload with ensuing development of blood pressure and their joint effect on the risk of coronary heart disease. Scand J Work Environ Health. 2007;33:425–34.
Gholami Fesharaki M, Kazemnejad A, Zayeri F, Rowzati M, Akbari H. Historical cohort study of shift work and blood pressure. Occup Med. 2014;64:109–12.
Kario K, James GD, Marion R, Ahmed M, Pickering TG. The influence of work- and home-related stress on the levels and diurnal variation of ambulatory blood pressure and neurohumoral factors in employed women. Hypertens Res. 2002;25:499–506.
He W, Goodkind D, Kowal P. An aging world: 2015. Washington: US Census Bureau; 2016.
Hashimoto S, Kawado M, Seko R, Murakami Y, Hayashi M, Kato M, Noda T, Ojima T, Nagai M, Tsuji I. Trends in disability-free life expectancy in Japan, 1995-2004. J Epidemiol. 2010;20:308–12.
Tarlow B, Wisniewski S, Belle S, Rubert M, Ory M, Gallagher-Thompson D. Positive aspects of caregiving. Res Aging. 2004;26:429–53.
Pinquart M, Sorensen S. Correlates of physical health of informal caregivers: a meta-analysis. J Gerontol B Psychol Sci Soc Sci. 2007;62:P126–37.
Schulz R, Beach SR. Caregiving as a risk factor for mortality: the caregiver health effects study. JAMA. 1999;282:2215–9.
Haley WE, Roth DL, Howard G, Safford MM. Caregiving strain and estimated risk for stroke and coronary heart disease among spouse caregivers: differential effects by race and sex. Stroke. 2010;41:331–6.
King AC, Oka RK, Young DR. Ambulatory blood pressure and heart rate responses to the stress of work and caregiving in older women. J Gerontol. 1994;49:M239–45.
von Kanel R, Mausbach BT, Patterson TL, Dimsdale JE, Aschbacher K, Mills PJ, Ziegler MG, Ancoli-Israel S, Grant I. Increased Framingham coronary heart disease risk score in dementia caregivers relative to non-caregiving controls. Gerontology. 2008;54:131–7.
Torimoto-Sasai Y, Igarashi A, Wada T, Ogata Y, Yamamoto-Mitani N. Female family caregivers face a higher risk of hypertension and lowered estimated glomerular filtration rates: a cross-sectional, comparative study. BMC Public Health. 2015;15:177.
Shaw WS, Patterson TL, Semple SJ, Dimsdale JE, Ziegler MG, Grant I. Emotional expressiveness, hostility and blood pressure in a longitudinal cohort of Alzheimer caregivers. J Psychosom Res. 2003;54:293–302.
Capistrant BD, Moon JR, Glymour MM. Spousal caregiving and incident hypertension. Am J Hypertens. 2012;25:437–43.
Butalid L, Verhaak PF, Boeije HR, Bensing JM. Patients’ views on changes in doctor-patient communication between 1982 and 2001: a mixed-methods study. BMC Fam Pract. 2012;13:80.
Acknowledgements
This study was supported by the grants and aid from the Japan Organization of Occupational Health and Safety. We would like to express his gratitude to Dr. Genjiro Kimura, a past president of Asahi Rosai Hospital, for his great contribution to the research on workplace hypertension conducted by 29 Rosai hospital groups, which hold an important framework of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Munakata, M. Clinical significance of stress-related increase in blood pressure: current evidence in office and out-of-office settings. Hypertens Res 41, 553–569 (2018). https://doi.org/10.1038/s41440-018-0053-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-018-0053-1
This article is cited by
-
Immune and inflammatory mechanisms in hypertension
Nature Reviews Cardiology (2024)
-
Microglia-derived TNF-α contributes to RVLM neuronal mitochondrial dysfunction via blocking the AMPK–Sirt3 pathway in stress-induced hypertension
Journal of Neuroinflammation (2023)
-
Structure and dynamics of the course of chronic non-infectious somatic diseases in patients during war events on the territory of Ukraine
BMC Public Health (2023)
-
Hypertension paradox in Japan: the road ahead
Hypertension Research (2023)
-
Investigation of the effect of music listened to by patients with moderate dental anxiety during restoration of posterior occlusal dental caries
Clinical Oral Investigations (2023)