Abstract
Objectives
DRAKO (NCT02850263) is a 24-month, prospective, non-interventional, multi-centre cohort study which enroled patients diagnosed with centre-involving diabetic macular oedema (DMO). The study aims to evaluate standard of care with intravitreal aflibercept (IVT-AFL) treatment in the UK. This analysis describes the anti-vascular endothelial growth factor (anti-VEGF) treatment-naive patient cohort after 12-month follow-up.
Methods
Study eyes were treated with IVT-AFL as per local standard of care. The mean change in best-corrected visual acuity (BCVA) and central subfield thickness (CST) from baseline at 12 months were measured and stratified by baseline factors. The number of injections and safety data were also evaluated.
Results
A total of 507 patients were enroled from 35 centres. Mean (SD) baseline BCVA was 71.4 (12.0) letters and CST was 448.7 (88.7) µm, with 63.1% of patients presenting with baseline BCVA ≥ 70 letters (mean 78.1). Mean (SD) change in BCVA of 2.5 (12.2) letters and CST of −119.1 (116.4) µm was observed at month 12. A 7.3 letter gain was observed in patients with baseline BCVA < 70 letters. Mean number (SD) of injections in year one was 6.4 (2.1). No significant adverse events were recorded.
Conclusion
Year one results indicated that IVT-AFL was an effective treatment for DMO in standard of care UK clinical practice, maintaining or improving visual acuity in treatment-naive patients with good baseline visual acuity, despite some patients being undertreated versus the summary of product characteristics. These results also demonstrated the clinical importance and meaningful impact of diabetic retinopathy screening in the UK.
Similar content being viewed by others
Introduction
Diabetes, including its associated complications, is an escalating healthcare problem, with treatment estimated to cost around 10% of the UK’s entire National Health Service (NHS) budget [1]. The global prevalence of diabetes is projected to increase from the current 9.3% to 10.2% in 2030, affecting 578.4 million adults between the ages of 20–79 years, further increasing the burden on health systems [2]. Diabetic macular oedema (DMO), a microvascular complication of diabetes that can occur at any stage of retinopathy, is the most common cause of visual acuity (VA) loss in patients with diabetes and the most frequent cause of blindness in young and mid-aged adults in the developed world. One in four diabetic patients can expect to develop DMO in their lifetime [3,4,5,6].
In recent years, the use of anti-vascular endothelial growth factor (anti-VEGF) treatments for DMO has seen outcomes for many patients improve. Intravitreal injections of anti-VEGF therapies have shown superiority over laser photocoagulation of the macula in several clinical trials [7,8,9,10]. Subsequently, intravitreal VEGF inhibitors have become the first-line therapy of choice for management of vision loss from DMO.
Intravitreal aflibercept (IVT-AFL, Eylea) is an anti-VEGF treatment with an innovative fusion protein design. Current IVT-AFL standard of care treatment for DMO patients is a 2 mg dose of five initial monthly injections followed by a proactive bi-monthly treatment regimen in year one, with no requirement for monitoring between injections. In year two, the injection interval may be extended dependent on functional and anatomical outcomes, consequently reducing burden on patients and their care providers (IVT-AFL summary of product characteristics [SmPC] recommended posology for DMO treatment) [11].
Results from the two pivotal IVT-AFL Phase 3 trials in DMO, VIVIDDMO and VISTADMO, demonstrated significant superiority for the IVT-AFL treatment groups over laser in all endpoints at week 52 through to week 148, with similar efficacy in both treatment regimens [10, 12, 13].
The UK did not participate in these pivotal trials, and DRAKO represents the first UK-based prospective, non-interventional study to assess the effectiveness of standard of care IVT-AFL treatment in DMO patients. Such observational studies are valuable as they report and evaluate outcomes based on locally defined treatment practices, outside the rigorous clinical trial setting, enabling outcome characterisation within a more representative population and treatment environment.
The primary objectives of this study are to assess the mean change from baseline in best-corrected visual acuity (BCVA) and central subfield thickness (CST). In addition, by evaluating follow-up procedures and assessing treatment patterns in eyes affected by DMO treated with IVT-AFL in routine clinical practice, the study aims to inform best practice guidance in the UK upon completion of the follow-up period. Here we describe the month 12 outcomes of the anti-VEGF treatment-naive patient cohort.
Materials and methods
Study design
DRAKO (NCT02850263) is a prospective, non-interventional, multi-centre, non-comparative cohort study conducted in 35 NHS hospitals throughout the UK (Supplementary Table 1). Patients were enroled from July 2016 through to April 2018 and followed up for 24 months. To reduce selection bias, patients were enroled in a consecutive manner. The study was approved by the North West Liverpool East Research Ethics Committee (16/NW/0238) and was conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent. All treatment decisions, including the decision to treat with IVT-AFL, were made as per local standard of care, independently of study participation. The study included two patient cohorts; Cohort 1 comprised anti-VEGF treatment-naive patients and Cohort 2 comprised patients who previously received anti-VEGF treatment other than IVT-AFL for DMO. Study size was calculated based on an assumed 10% drop-out rate at 24 months, allowing an estimate of change from baseline in BCVA letters within 95% confidence intervals (CI) of ±1.2 letters and CST of ±13.9 μm. This was based on a standard deviation (SD) of 13 letters for the mean change in BCVA and 150 µm for CST, conservative estimates based on SD observed in other recent studies involving aflibercept in DMO [10, 14]. Sample sizes of 450 and 225 were calculated for Cohorts 1 and 2 respectively.
Study population and treatment
Adult patients with a confirmed diagnosis of type 1 or type 2 diabetes and DMO with central involvement, defined as the centre subfield area on optical coherence tomography, were enroled if they met the eligibility criteria as defined in Table 1. Inclusion of patients based on a CST of ≥400 µm in the study eye was removed as an eligibility criterion via protocol amendment (21 February 2017) as this threshold is not applied in Scotland and removal enabled a more accurate representation of the UK-treated population. One of the two participating Scottish sites (University Hospital Hairmyres) was active prior to approval of the protocol amendment. The study eye was defined as the eye for which the IVT-AFL treatment was initiated or, if both eyes were affected, the eye with worse baseline VA. Data were collected for the fellow eye if DMO diagnosis was confirmed. No eligibility restriction was mandated for patient baseline BCVA letter score.
At study initiation, details of the local standard of care IVT-AFL treatment protocol was recorded for each centre (Supplementary Table 2). Data collection, including type of visit; diabetic management (diabetic retinopathy measured using the English National Screening Committee or Scottish Diabetic Retinopathy Grading Scheme classifications, and haemoglobin A1c (HbA1c) assessment), anatomical and functional assessments; treatment provided from the baseline visit; and all subsequent routine outpatient visits were recorded. The month 12 visit was nominated by the site (for data collection purposes) and defined as 12 months ±1 month from the patient’s baseline visit.
Patients who dropped out of the study for any reason were not replaced.
Outcome measures
Primary outcomes were the mean change from baseline in Early Treatment Diabetic Retinopathy Study (ETDRS) letters measured by BCVA with refraction and the mean change in CST as determined by spectral domain optical coherence tomography (as per local practice) at month 12. Secondary objectives included mean change in BCVA and CST stratified by pre-defined baseline factors; percentage gain and loss of at least 5, 10 or 15 letters; and adherence to IVT-AFL DMO SmPC in year one. Due to the observational design of the study, target changes in these parameters were not defined.
Statistical analysis
Interim analysis was conducted upon completion of the 12-month follow-up period for the treatment-naive patient cohort and are reported here.
Descriptive statistics were used to summarise the quantitative variables. Categorical variables were summarised using frequency distributions and percentages and data stratified by pre-defined baseline covariate subgroups; age (18–35, 36–50, 51–65, >65 years), BCVA (<35, 35–49, 50–69, ≥70 letters) and CST (<400, ≥400 µm).
All patients with a baseline IVT-AFL injection and at least one post-baseline assessment of BCVA or CST were included in the analysis. Two sub-populations were analysed, and 95% CI were calculated. The more stringent sub-population, defined as the per protocol window population (PPW), included patients with available BCVA or CST data at baseline and the nominated month 12 visit (12 months ± 1 month). The less stringent sub-population was defined as the full analysis set (FAS) and included patients with BCVA or CST available at baseline and at least one follow-up visit; missing values were imputed based on the last observation carried forward method, the visit immediately preceding the month 12 visit was used for analysis. Adherence to SmPC was measured by defining acceptable ‘windows’ between injections. For the first five injections, it was 25–38 days; after the fifth injection, it was 46–66 days.
Safety was assessed on the safety set, which included all patients who provided written informed consent. Adverse events were listed using the Medical Dictionary for Regulatory Activities coding system.
Analysis was performed using SAS® software, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patient disposition and baseline characteristics
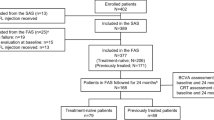
DRAKO enroled 507 anti-VEGF treatment-naive patients. A 2.0% drop-out rate was observed in year one, the reasons for drop-out were as follows; patient withdrawal (4), death (3), patient ineligibility (2) and change in centre (1). Further patients were excluded from the analysis due to unavailability of baseline and/or 12-month data. Patients were divided into PPW (n = 388) and FAS (n = 488) sub-populations for analysis based on pre-defined stringency criteria (Fig. 1). The PPW and FAS sub-populations demonstrated comparable trends throughout the analysis and results for the PPW sub-population are reported unless stated otherwise.
The mean age at baseline was 62.9 years, and 63.1% of patients were male (Table 2). Most patients were white (76.8%), and 85.8% had been diagnosed with type 2 diabetes. Over half of patients had fellow eye involvement. Patients demonstrated a suboptimal level of glycaemic control with mean (SD) HbA1c of 66.1 (20.5) mmol/mol. Background to pre-proliferative retinopathy was recorded for the majority of patients regardless of classification applied. On average, patients had been diagnosed with DMO 1.3 (SD 2.4) years before their baseline visit. Of the primary endpoint assessments, BCVA was recorded for 91.0% of patients and CST for 99.5% of patients. The mean (SD) baseline BCVA and CST were 71.4 (12.0) letters and 448.7 (88.7) µm, respectively (Table 3).
Treatment of DMO
Local standard of care IVT-AFL treatment was administered across all sites, with 11 different treatment posologies recorded at study initiation (Supplementary Table 2). All patients received at least one IVT-AFL injection in the study eye. The mean number (SD) of injections was 6.4 (2.1) and 6.3 (2.2) for the PPW and FAS sub-populations, respectively, compared with eight to nine injections if treatment was conducted as per the SmPC. At site initiation, 27 (77.1%) and 21 (60.0%) of the 35 participating sites confirmed their intention to adhere to DMO SmPC for the five initial monthly injections and for 12 months respectively as per their local standard of care protocol. This equated to 334 patients (86.1%) from the 27 sites intending to administer five initial injections and 211 (54.4%) from the 21 sites intending to adhere to SmPC throughout year one. However, in practice, 117 (30.2%) of patients received five initial IVT-AFL injections and 13 (3.4%) patients adhered to SmPC in year one.
Effect of treatment on functional and anatomical outcomes
The mean (95% CI) improvement in BCVA at month 12 was 2.5 letters (±1.3 letters) and 1.9 letters (±1.0 letters) for the PPW and FAS sub-populations, respectively (Table 3). The relative increase in BCVA letters varied based on initial VA. Patients with baseline BCVA of less than 35 letters benefitted from a mean increase of 23.6 letters (n = 7), whereas the mean BCVA for patients presenting with BCVA of 70 letters or more remained stable (Fig. 2A and Supplementary Table 3). Similarly, patients aged 18–35 years experienced a larger increase in mean BCVA letters than those aged 65 years and older (4.3 and 1.2 letters, respectively; Supplementary Table 3). A letter gain of five or more was observed in 40.2% of patients, whereas an equivalent letter loss was noted in 16.5% patients (Fig. 2B). When evaluated by adherence to SmPC, patients receiving five initial monthly injections obtained a mean (SD) letter gain of 4.2 (10.9). The small proportion of patients for whom treatment continued as per SmPC in year one experienced a 1.1 (7.4) letter gain (Supplementary Table 4).
At month 12, CST was reduced, with mean (95% CI) change from baseline determined as –119.1 µm (±11.6 µm) and –117.7 µm (±10.1 µm) for the PPW and FAS sub-populations, respectively (Table 3). Baseline CST was <400 µm for 16.5% of patients. Mean (SD) CST change was observed as –42.5 (76.3) µm and –134.3 (117.0) µm for patients with initial CST measure of <400 µm and ≥400 µm, respectively (Fig. 2C and Supplementary Table 5). A larger improvement in BCVA was observed at month 12 for patients presenting with a baseline CST of ≥400 µm (mean [SD] BCVA letter change was 1.4 [9.2] and 2.7 [12.6] for patients with CST of <400 µm and ≥400 µm, respectively; see Supplementary Table 3).
Adverse events
The safety set comprised data from the full treatment-naive patient cohort (n = 507). During the first 12 months of treatment, a total of 3129 injections were administered to the study eye. Non-ocular treatment emergent adverse events (TEAE) were reported for 33.7% (n = 171) of patients and ocular TEAEs for 13.2% (n = 67) of patients. A total of 107 ocular TEAEs were captured. Reported TEAEs (percentage of patients) included vitreous haemorrhage (3.0%), cataract (1.2%), retinal haemorrhage (1.2%), retinal detachment (0.2%) and vitreous detachment (0.2%). Adverse events reported as associated with treatment were primarily related to the procedure rather than aflibercept. The safety profile was in line with other published studies, and no new safety concerns were identified. Further details on reported TEAEs are outlined in Supplementary Table 6.
Discussion
DRAKO is the first prospective, multi-centre study evaluating standard of care IVT-AFL treatment for DMO in the UK. The study enroled 507 treatment-naive patients across 35 contributing centres and it was noted that the majority of patients with DMO are diagnosed and treated at a high level of VA, demonstrating the effectiveness of the diabetic retinopathy screening programme in the UK.
The mean baseline BCVA for DRAKO (71.4 letters) was substantially higher than measures reported in pivotal clinical trials (VIVIDDMO and VISTADMO aflibercept cohorts reported mean baseline BCVA of 59.8 letters and 59.1 letters, respectively) and in other prospective observational DMO studies (APOLLON and POLARIS) [10, 15, 16]. Of note, two-thirds of DRAKO patients had baseline BCVA of 70 letters or more (mean 78.1). APOLLON, a French IVT-AFL treatment-based study enroled 77 patients between September 2016 and July 2019, reported mean baseline VA 62.7 letters in the treatment-naive cohort [15]. However, eligibility was limited to patients with baseline VA ≤20/40 (≤70 ETDRS letters). POLARIS, a European-based, ranibizumab study that recruited 125 patients within the UK cohort from September 2012 to January 2015 reported a mean baseline VA of 60.3 letters [16]. When comparing baseline VA in these UK cohorts, a clinically relevant difference of 11 letters in baseline VA was observed between DRAKO and POLARIS. Although these letter changes are not directly comparable given that DRAKO captured BCVA with refraction and POLARIS documented non-refracted VA, these measures are nonetheless highly correlated. This may be a further indication of a shift towards earlier screening and treatment of DMO within the NHS in more recent years, particularly as DRAKO observed a mean time from diagnosis of DMO to baseline treatment of 1.3 years, suggesting that patients are often being diagnosed with DMO in advance of the condition being considered clinically significant. Indeed, these results suggest that the National Institute for Health and Care Excellence (NICE) Technical Appraisal Guidance on the threshold for treatment initiation (central retinal thickness of at least 400 µm) may be encouraging early intervention to maintain or improve patient VA in areas of the UK where these guidelines apply (England, Wales and Northern Ireland).
DRAKO patients experienced a mean gain of 2.5 letters and a mean reduction in CST of 119.1 µm at month 12. Trends in letter gains and losses demonstrated that 40.2% and 20.1% patients observed at least a 5- or 10-letter gain respectively within the first year. Whereas, 16.5% and 9.8% patients observed at least a 5- or 10-letter loss respectively within the same period. A 5-letter gain in the context of the high observed baseline BCVA scores may be clinically relevant in most patients. In addition, stratification by baseline BCVA demonstrated that better gains were achieved in those with worse baseline BCVA. Of note, for patients with a baseline BCVA ≤ 69 letters (mean 58.7 letters), the mean gain at month 12 was 7.3 letters.
Although these trends reflect those seen in both IVT-AFL treatment randomised clinical trials (VIVIDDMO and VISTADMO) and the APOLLON observational study, the overall 12-month BCVA letter gains observed in these studies are higher than in DRAKO (gain of 10.7, 10.7 and 7.8 letters, respectively) [10, 15]. This variation is likely due to the protocol-driven higher injection numbers, particularly in the pivotal trials (patients received five initial monthly doses followed by IVT-AFL treatment every 8 weeks) and the increased opportunity for larger BCVA gains from a lower baseline as a result of implementing a maximum BCVA inclusion criteria which was not mandated for DRAKO.
Previous reports have highlighted the positive association of injection frequency and functional outcomes for patients, with an emphasis on the initial injections [17]. The DRAKO study authors also advocate the administration of full initial dosing, with the subgroup of patients receiving five initial monthly injections obtaining a mean letter gain of 4.2, although this benefit was not extended to year one SmPC compliance, potentially due to the low patient numbers or high baseline BCVA. The reasons for the lower than intended number of injections administered were not collected and changes in standard of care treatment protocols at participating sites were not re-assessed after site initiation. Consequently, it was not possible to determine whether this was due primarily to capacity constraints within the NHS or other factors, such as clinical prioritisation, patient influences or perhaps a more relaxed treatment approach in response to favourable baseline metrics. Of note, APOLLON results showed a higher rate of patients receiving five initial doses of IVT-AFL (51.9%) than DRAKO, which may also have contributed to the higher letter gain reported.
Overall, DRAKO patients with a baseline BCVA of ≥70 letters maintained their vision. Those with baseline BCVA of 70–78 letters (n = 139) gained 1.3 letters, and those with baseline BCVA > 78 (n = 94) experienced a loss of 1.8 letters. This distinction may be important in efforts to preserve ‘driving vision’, which is categorised in the UK as a BCVA of 70 letters or more in the patient’s better eye. The results for patients with BCVA 78 letters or more are comparable to those of the Diabetic Retinopathy Clinical Research Retina Network Protocol V study. Whilst no control group was included in the DRAKO study design, the reduced benefits observed in this subgroup support the conclusion of Protocol V that a reasonable strategy for patients with high VA may be observation without treatment unless VA worsens, although there were few patients with >400 µm CST in Protocol V (9% patients in the aflibercept group) [18]. Analysis of the DRAKO year 2 results may provide further clarity on this clinically important point.
These DRAKO observations are supported by several retrospective registry studies, which demonstrated the real-world effectiveness of IVT-AFL for DMO treatment [19,20,21]. Notably, fewer injections were recorded compared to clinical trials, and functional gains were not as pronounced and were related to baseline measures. In the UK-based, single-centre retrospective cohort study conducted by Lukic et al. [21]., 99 eyes were evaluated and a mean of 6.9 injections were administered. The baseline VA (59.7 ETDRS letters) in this study was much lower than DRAKO, so although the overall letter gain (9.9 letters) was higher than that observed in DRAKO, in the 34% of eyes with a baseline VA ≥69, the mean gain was 2.6 letters.
A similar ‘ceiling’ effect was observed in the CST subgroup analysis. Baseline CST was stratified above or below 400 µm to align with the NICE Technology Appraisal Guidance. The reduction in CST for the baseline ≥400 µm subgroup was much greater than for the <400 µm subgroup, but the mean CST at month 12 was similar for both subgroups. The results suggest minimal difference in terms of BCVA outcomes between the two subgroups, with a marginally lower letter gain attained in the <400 µm subgroup, although patient numbers were limited (n = 52).
The study has some limitations often inherent in non-interventional studies such as, inconsistent treatment administration and non-defined functional eligibility metrics. However, the prospective study design and wide range of sites contributing enables treatment effects to be monitored across a diverse population that aims to be representative of the UK as a whole. In addition, the large number of anti-VEGF treatment-naive patients enroled, and the high proportion of primary endpoint data collected, strengthen the observations made during the study.
In conclusion, year one results indicate that IVT-AFL is an effective treatment for DMO in real-world UK clinical practice, maintaining or improving VA. Vision gains observed in DRAKO were lower than in the pivotal clinical trials and several observational studies [10, 14,15,16], perhaps reflecting better baseline vision in UK clinical practice and/or a failure to adhere to the SmPC for all patients. In addition, DRAKO demonstrated the effectiveness of diabetic retinopathy screening in the UK, with almost two-thirds of patients presenting with good baseline vision.
The DRAKO data set contains a rich source of real-world ocular-, treatment- and resource-related data that will mature further upon completion of the study. It is anticipated that this will provide a benchmark for better understanding of DMO IVT-AFL outcomes and for shaping how DMO patient care pathways are defined in the future.
Summary
What was known before
-
The effectiveness of intravitreal aflibercept (IVT-AFL) for treatment of diabetic macular oedema (DMO) patients has been demonstrated in several pivotal clinical trials (VIVID and VISTA) and non-UK focused observational studies (APOLLON), although such investigations primarily focused on patients with baseline visual acuity of <73 letters.
-
Retrospective registry-based studies of anti-vascular endothelial growth factor (anti-VEGF) treatments have reported lower injection frequency and functional gains than randomised clinical trials.
-
The scope for improvement of functional and anatomical parameters in response to anti-VEGF treatment is closely associated with baseline values.
What this study adds
-
DRAKO is the first prospective non-interventional study to evaluate IVT-AFL treatment of DMO patients across the UK, demonstrating the effectiveness of this treatment to maintain or improve patient outcomes across a diverse range of local standard of care protocols, despite often observing undertreatment compared with locally defined treatment plans.
-
DRAKO indicates the effectiveness of the diabetic retinopathy screening programme in the UK where patients with DMO are being identified and treated at a high level of VA, thereby preserving patient vision.
References
Written by Editor. Diabetes U. K. Cost of diabetes. 2019 https://www.diabetes.co.uk/cost-of-diabetes.html. Accessed 19 Nov 2020.
International Diabetes Federation Diabetes Atlas ninth edition. 2019 https://diabetesatlas.org/upload/resources/2019/IDF_Atlas_9th_Edition_2019.pdf. Accessed 26 Nov 2020.
Fong DS, Aiello LP, Ferris FL 3rd, Klein R. Diabetic retinopathy. Diabetes Care. 2004;27:2540–53.
Musat O, Cernat C, Labib M, Gheorghe A, Toma O, Zamfir M, et al. Diabetic macular edema. Rom J Ophthalmol. 2015;59:133–6.
Moss SE, Klein R, Klein BE. The 14-year incidence of visual loss in a diabetic population. Ophthalmology. 1998;105:998–1003.
Stefánsson E, Bek T, Porta M, Larsen N, Kristinsson JK, Agardh E. Screening and prevention of diabetic blindness. Acta Ophthalmol Scand. 2000;78:374–85.
Nguyen QD, Brown DM, Marcus DM, Boyer DS, Patel S, Feiner L, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119:789–801.
Mitchell P, Bandello F, Schmidt-Erfurth U, Lang GE, Massin P, Schlingemann RO, et al. The RESTORE study: ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011;118:615–25.
Do DV, Nguyen QD, Boyer D, Schmidt-Erfurth U, Brown DM, Vitti R, et al. One-year outcomes of the da Vinci Study of VEGF Trap-Eye in eyes with diabetic macular edema. Ophthalmology. 2012;119:1658–65.
Korobelnik JF, Do DV, Schmidt-Erfurth U, Boyer DS, Holz FG, Heier JS, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014;121:2247–54.
Eylea 40mg/ml solution of injection in a vial summary of product characteristics. 2012. https://www.medicines.org.uk/emc/product/2879/smpc#gref. Accessed 19 Nov 2020.
Brown DM, Schmidt-Erfurth U, Do DV, Holz FG, Boyer DS, Midena E, et al. Intravitreal aflibercept for diabetic macular edema: 100-week results from the VISTA and VIVID studies. Ophthalmology. 2015;122:2044–52.
Heier JS, Korobelnik JF, Brown DM, Schmidt-Erfurth U, Do DV, Midena E, et al. Intravitreal aflibercept for diabetic macular edema: 148-week results from the VISTA and VIVID studies. Ophthalmology. 2016;123:2376–85.
Diabetic Retinopathy Clinical Research Network, Wells JA, Glassman AR, Ayala AR, Jampol LM, Aiello LP, Antoszyk AN, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. N Engl J Med. 2015;372:1193–203.
Korobelnik JF, Daien V, Faure C, Tadayoni R, Giocanti-Auregan A, Dot C, et al. Real-world outcomes following 12 months of intravitreal aflibercept monotherapy in patients with diabetic macular edema in France: results from the APOLLON study. Graefes Arch Clin Exp Ophthalmol. 2020;258:521–8.
Stefanickova J, Cunha-Vaz J, Ulbig M, Pearce I, Fernández-Vega Sanz A, Theodossiadis P, et al. A noninterventional study to monitor patients with diabetic macular oedema starting treatment with ranibizumab (POLARIS). Acta Ophthalmol. 2018;96:e942–9.
Ziemssen F, Schlottman PG, Lim JI, Agostini H, Lang GE, Bandello F. Initiation of intravitreal aflibercept injection treatment in patients with diabetic macular edema: a review of VIVID‑DME and VISTA‑DME data. Int J Retina Vitreous. 2016;2:16.
Baker CW, Glassman AR, Beaulieu WT, Antoszyk AN, Browning DJ, Chalam KV, et al. Effect of initial management with aflibercept vs laser photocoagulation vs observation on vision loss among patients with diabetic macular edema involving the center of the macula and good visual acuity: a randomized clinical trial. JAMA. 2019;321:1880–94.
Ciulla TA, Pollack JS, Williams DF. Visual acuity outcomes and anti-VEGF therapy intensity in diabetic macular oedema: a real-world analysis of 28 658 patient eyes. Br J Ophthalmol. 2021;105:216–21.
Bhandari S, Nguyen V, Fraser-Bell S, Mehta H, Viola F, Baudin F, et al. Ranibizumab or aflibercept for diabetic macular edema: comparison of 1-Year outcomes from the Fight Retinal Blindness! Registry. Ophthalmology. 2020;127:608–15.
Lukic M, Williams G, Shalchi Z, Sim D, Patel PJ, Keane PA, et al. Intravitreal aflibercept for diabetic macular oedema: Moorfields’ real-world 12-month visual acuity and anatomical outcomes. Eur J Ophthalmol. 2020;30:557–62.
Acknowledgements
The DRAKO study was sponsored by Bayer Plc. Medical writing support was provided by Andrea Parke of O4 Research and Apothecom Publications Ophthalmology, and was funded by Bayer Plc. The DRAKO Study Group on behalf of Bayer Plc. would like to thank the patients and investigators who have participated in the study.
Funding
This study was sponsored by Bayer Plc, Reading. The sponsor participated in the design of the study, conducting of the study, data collection, data management, data analysis, data interpretation and preparation, review and approval of the manuscript.
Author information
Authors and Affiliations
Consortia
Contributions
SS, FG, SPK, AK, JT and PS were participating investigators in the study, played an important role in interpreting the results and form part of the steering committee. They reviewed, revised, provided feedback and approval of the manuscript, approve the decision to submit for publication and agree to be accountable for all aspects of the work. HM, AN, MS and JN participated in the study design, reviewed, revised, provided feedback and approval of the manuscript, approve the decision to submit the manuscript for publication and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
SS received consultancy fees from Bayer, Allergan, Novartis Pharma AG, Roche, Boehringer Ingelheim, Optos, Apellis, Oxurion, Biogen, Oculis and Heidelberg Engineering. SS is Editor-in-Chief for the Journal Eye. Faruque Ghanchi has been a consultant and speaker for Novartis, Bayer, Allergan, Alimera and Roche; and has received travel grants from Bayer and Novartis. SPK received travel grants from Bayer, research support from Bayer and Novartis Pharma AG, and participated in advisory boards for Novartis Pharma AG and Polyphotopics. AK received travel support from Novartis, Bayer, and Allergan, and speaker fees from Allergan and Bayer. JT participated in advisory boards for Bayer and Novartis; received travel support from Bayer; and received research grants from Bayer, Novartis and Roche. PS attended advisory boards for Pfizer, Allergan, Boehringer, Roche and Bayer, and his department has received educational, research, and audit grants from Allergan, Boehringer Ingelheim, Novartis, and Bayer. HM is an employee of Bayer Plc. AN is an employee of Bayer Plc. MS is an employee of O4 Research. JN is an employee of Bayer Plc.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A list of members and their affiliations appears in the Supplementary Information.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sivaprasad, S., Ghanchi, F., Kelly, S.P. et al. Evaluation of standard of care intravitreal aflibercept treatment of diabetic macular oedema treatment-naive patients in the UK: DRAKO study 12-month outcomes. Eye 36, 64–71 (2022). https://doi.org/10.1038/s41433-021-01624-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01624-9
This article is cited by
-
Suboptimal outcomes and treatment burden of anti-vascular endothelial growth factor treatment for diabetic macular oedema in phakic patients
Eye (2024)
-
Intravitreal Aflibercept for the Treatment of Diabetic Macular Edema in Routine Clinical Practice: Results from the 24-Month AURIGA Observational Study
Ophthalmology and Therapy (2024)
-
Predictive factors for treatment outcomes with intravitreal anti-vascular endothelial growth factor injections in diabetic macular edema in clinical practice
International Journal of Retina and Vitreous (2023)
-
Evaluation of care with intravitreal aflibercept treatment for UK patients with diabetic macular oedema: DRAKO study 24-month real-world outcomes
Eye (2023)
-
Evaluation of standard-of-care intravitreal aflibercept treatment practices in patients with diabetic macular oedema in the UK: DRAKO study outcomes
Eye (2023)