Abstract
Objective
The aim of this study was to evaluate the perifoveal and peripapillary microvascular structure in patients with Behçet’s disease (BD) without clinically ocular involvement.
Methods
Fifty-six eyes of 28 patients with Behçet’s disease without clinically ocular involvement and 50 age-matched healthy eyes were included in this prospective study. Vessel densities (VD) of macula and optic nerve head, foveal avascular zone (FAZ), choroid capillary plexus (CCP) flow area, perifoveal capillary nonperfusion, capillary dilatation and/or telangiectasia and perifoveal capillary arcade disruption were analyzed with optical coherence tomography angiography (OCTA). The relationship between duration of disease and vessel densities was also evaluated.
Results
The FAZ was significantly higher in the BD group (p < 0.001). VD of the foveal and parafoveal region in deep capillary plexus (DCP) were significantly lower in the BD group than in the control group (p < 0.001, p < 0.001). Total disc, peripapillary and inside-disc VD were significantly lower in BD group (p = 0.001, p = 0.002, p = 0.004). Perifoveal morphological changes in DCP were significantly higher in nonocular BD (p < 0.001, p < 0.001, p < 0.001). There was a mild negative correlation between disease duration and whole VD in DCP, total disc VD, peripapillary VD was determined (For whole VD in DCP, r = −0.400, p = 0.035; for total disc VD r = −0.396, p = 0.037; for peripapillary VD r = −0.442, p = 0.018).
Discussion
Perifoveal and peripapillary microvascular changes that can be detected by OCTA may occur in Behçet’s patients without clinically ocular involvement.
Similar content being viewed by others
Introduction
Behçet’s disease (BD) is a chronic, relapsing, multisystem, autoimmune, idiopathic inflammatory disorder with high morbidity. Diagnosis is based on clinical findings. According to the International Study Group for Behcet’s Disease, the diagnosis is based on agreed clinical criteria that require recurrent oral ulcerations and two of the following: recurrent genital ulcerations, ocular involvement, defined skin lesions, or positive pathergy [1, 2].
Typical ocular involvements are non-granulomatous uveitis, panuveitis, diffuse vitritis, retinal vasculitis, and optic disc edema. Histopathological examinations have shown the obstructive vasculitis originating from the circulation of abnormal immune complexes. Although seen worldwide, it has been noticed that the prevalence of the disease is increasing from the Mediterranean and Central Asia to Japan. Ocular involvement has been seen in 50–70% of patients with BD [3,4,5]. In the presence of retinal vasculitis or macular involvement, multimodal imaging (fluorescein angiography, optical coherence tomography, indocyanine green angiography) is useful in the determination of macular edema, vascular blockages, peripheral ischaemic areas, neovascularisation and the inflammatory activity [2, 6, 7].
Optical coherence tomography angiography (OCTA) is a non-invasive, highly repeatable and high-resolution novel imaging technique that allows physicians to evaluate the foveal and peripapillary microvascular structure without contrast dye injection. It is based on the principle of decorrelation, in which the signals are red blood cells [7,8,9]. OCTA provides noninvasive and rapid evaluation and has the advantage of demonstrating both of retinal and choroidal vessels. The diagnostic efficacy of OCTA has been shown in several retinal vascular diseases such as age-related macular degeneration, serous chorioretinopathy, diabetic retinopathy, and polypoidal choroidal vascular disease [10, 11].
Recently, literature published on the role of OCTA in uveitis and retinal vasculitis is still limited. A few studies have utilized the macular OCTA findings in BD with or without ocular involvement. In the studies, the authors reported decrease in VD, especially in DCP in nonocular BD patients [12, 13]. However, the microvascular changes in optic nerve head (ONH) have not been studied. In this study, we aimed to evaluate the features of the microvasculature in ONH and macula using OCTA and the relationship between duration of disease and OCTA findings in BD patients without clinically ocular involvement.
Methods
Fifty-six eyes of 28 patients with BD who had no ocular involvement were evaluated this prospective, comparative study in the ophthalmology department of Muğla Sıtkı Koçman University, between November 2018-November 2019. The study was adhered to the principles of the Declaration of Helsinki and approved by the local ethics committee. All participants received written information about the study, and each participant provided written informed consent.
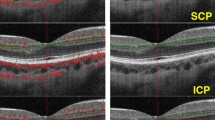
All patients received a complete ophthalmic examination. Applanation tonometer, biomicroscopy, fundus examination, axial length and fundus fluorescein angiography (Optos 200x, Dunfermline, United Kingdom) were performed. All eyes were imaged with the AngioVue Imaging System (RTVue XR Avanti; Optovue, Inc, Fremont, CA) by the same experienced examiner. OCTA data were analyzed by two of the authors who were masked to the findings. The AngioVue Imaging System is a spectral-domain optical coherence tomography (SD-OCT) device that enables simultaneous three-dimensional structural imaging of the retina and generation of en face maps of blood flow through a split spectrum amplitude decorrelation angiography (SSADA) algorithm. The AngioAnalytics software (Optovue, Inc.) allows measurement of foveal avascular zone (FAZ) and capillary vessel density (VD) from selected regions of the retina. Quantitative analysis of 6 × 6 mm OCT angiograms, was chosen for automated detection of flow, including FAZ (mm2), capillary VD (%) and choriocapillaris flow area (mm2) measurement. The device automatically inserted three fovea-centered circles on the macula via density assessment tool in both SCP and DCP (Fig. 1A). Foveal zone vessel density was defined by the area of the small circle with a diameter of 1 mm. The parafoveal zone vessel density was defined by the area of the middle circle with a diameter of 3 mm, and the perifoveal zone vessel density was defined by the area of the outer circle with a diameter of 6 mm. Flow area of the choriocapillaris layer was obtained at 1 mm radius areas, centered on the FAZ (Fig. 1B, C). Poor quality OCTA images due to blinking or fixation loss, were excluded from the study. Peripapillary flow was validated by total, peripapillary and inside disc VD was measured using a 4.5 × 4.5-mm scan that was centered on the ONH, as previously described [14]. The software automatically hangs a 2.0 mm diameter circle centered on the optic disc and determines the peripapillary area as a 1.0-mm-wide round annulus extending from the optic disc 2.0-mm circle (Fig. 1D). The peripapillary vessels were analyzed in the radial peripapillary capillary (RPC) section which prolongs from the inner limiting membrane (ILM) to the nerve fiber layer. The peripapillary vessel density is represented as the percentage area filled by vessels (microvasculature) in the peripapillary region. Vessel densities for the whole 4.5 × 4.5 mm scan region (whole image), optic disc area (inside disc), entire peripapillary area, as well as average RNFL thickness for the entire peripapillary area, were generated by applying an automated software algorithm. Peripapillary VD, defined as the total length of the perfused vasculature per unit area in the region of measurement (mm–1), were automatically measured in the average (Fig. 1). Additionally, in order to evaluate morphological alterations, we used the 3 × 3 mm OCTA scans for the following parameters: perifoveal capillary arcade disruption in the DCP (when extending over 1 quadrant of the entire length), capillary changes (capillary dilatation, telangiectasia), areas of capillary nonperfusion (presenting as irregular hypointense grayish areas), because of more detailed image quality (Fig. 2). OCTA images were accepted as Q8 or higher quality images and evaluated by two independent observers. Inter-investigator reliability (κ) was evaluated.
The study included 28 patients, diagnosed with BD according to the Behçet’s Syndrome International Study Group Criteria whereby oral ulcers, genital ulcers, and ocular lesions are assigned two points each, and skin lesions, vascular manifestations, and neurologic manifestations are assigned one point each. A patient with a score of ≥4 points is classified as having BD. Patients with previous surgery, signs of recent or previous anterior or posterior uveitis, history of red eye and systemic disease (hypertension, diabetes mellitus), pathology detected in biomicroscopic and fundus examination, eyes with axial length greater than 24 mm and shorter than 22 mm, intraocular pressure higher than 21 mmHg and history of topical or systemic medication were excluded. Patients with optic disc staining and early, late-stage peripheral leakage on fundus fluorescein angiography and patients with poor visual quality in OCTA were excluded from the study. The control group consisted of 50 age and sex matched healthy eyes with a refractive index of less than 2 D, no systemic or ocular disease and visual acuity in both eyes.
The statistical analyses were performed using SPSS (Statistical Package for the Social Sciences, version 22, for Windows; SPSS Inc.). The conformity of the data to normal distribution was evaluated with the Kolmogorov–Smirnov and Shapiro–Wilk tests. In the group comparisons, the independent Student’s t test was applied to numerical data that met parametric assumptions and the Mann–Whitney U test was applied to numerical data that did not meet parametric assumptions. Categorical variables between the groups were analyzed using the χ2 test. The superficial and full retinal vasculature FAZ measurements, flow area of the choriocapillaris layer, and vessel density measurements of both SCP and DCP in the foveal, parafoveal, and perifoveal zones of the groups were compared by independent t-test. To determine the relationship between duration of disease and vessel densities, Pearson’s correlation coefficients were calculated. A p-value < 0.05 was considered statistically significant.
Results
The mean patient age was 38.50 ± 14.30 (20–57) in the study group and 40.20 ± 14.10 (19–60) in the control group. All eyes were phakic in the study group. There was no significant difference between the groups in terms of age, gender distribution, lens status, central macular thickness, axial length and intraocular pressure (p > 0.05 for all). The peripapillary RNFL thickness was lower in patients with preclinical BD compared to healthy controls, but this was not statistically significant (Table 1). Mean disease duration was 7.4 ± 3.6 (3–12) years. 28 patients had oral and genital ulceration, 12 had positive pathergy test, 10 had skin lesions such as erythema nodosum and pseudofolliculitis, and 2 had intestinal involvement.
The mean area of the FAZ was 0.351 mm2 in the BD eyes and 0.183 mm2 in the control group, and the FAZ was significantly higher in the BD group (p < 0.001). VD of the foveal and parafoveal region in DCP were significantly lower in the BD group than in the control group (p < 0.001). However, VD of the foveal and parafoveal region in SCP were not significantly different from healty eyes (p = 0.324 and p = 0.471, respectively). There was no significant difference between the two groups in the mean CCP flow area (p = 0.867). Total disc, peripapillary and inside-disc VD were significantly lower in BD group (p = 0.001, p = 0.002, p = 0.004) (Table 2).
In the morphological examination of OCTA images, HD quality images were obtained and evaluated by two independent observers. Inter-investigator reliability (κ) was evaluated, and κ value was 0.857 (p < 0.001). A clear and organized microvascular network was observed in the control group and the foveal avascular arcade was regular. Perifoveal morphological changes (perifoveal capillary nonperfusion, perifoveal capillary arcade disruption and capillary dilatation and/or telangiectasia) in DCP were significantly higher in BD (p < 0.001, p < 0.001, p < 0.001) (Table 3 and Fig. 2).
We evaluated the relation between duration of disease and microvascular parameters detected by OCTA. No obvious correlation between disease duration and foveal, parafoveal density in DCP, inside disc VD was noted in the study (For foveal VD, r = 0.213, p = 0.277; for parafoveal VD r = −0,252, p = 0.196; for inside disc VD r = −0.216, p = 0.292). However, we found mild negative correlation between disease duration and whole VD in DCP, total disc VD, peripapillary VD (For whole VD in DCP, r = −0.400, p = 0.035; for total disc VD r = −0.396, p = 0.037; for peripapillary VD r = −0.442, p = 0.018).
Discussion
It is known that OCTA has advantages such as providing the possibility of separate and more detailed evaluation of the internal and external retinal circulation and the choriocapillaris. OCTA allows quantitative measurement of the capillary perfusion of the macula and optic nerve [15,16,17]. Considering that Behçet’s disease is one of the vascular occlusive syndromes [18], we assumed that ocular ischemia may be present in Behçet’s patients without ocular involvement. In this study, we reported that preclinical ocular features in patients with Behçet’s disease on OCTA. We found the VD in macula and OHN were lower and the mean area of FAZ was significantly larger in the BD group than in control group.
Previous studies demonstrated that VD reduction and higher perifoveal capillary hypoperfusion, perifoveal capillary network disorganization, and FAZ irregularity in Behçet’s [14, 19, 20]. Khairallah et al. investigated that the perifoveal microvascular changes in 25 patients with Behçet’s uveitis and reported that OCTA is more sensitive than FA in detecting these changes. They observed these microvascular changes more frequently in the DCP than in the SCP [14]. Furthermore, the capillary vessel density was lower in the Behçet uveitis group than in the control group. In our study, the presence of grayish nonperfused/hypoperfused areas was common in terms of morphological perifoveal findings of the OCTA in DCP and the differences were statistically significant. According to the degree of decreased VD, these regions may appear well-defined black, roundish or oblong areas completely devoid of flow. The other morphological perifoveal findings also determined as rarefied, dilated or shunting perifoveal capillary vessels. Both quantitative and qualitative analyses of FAZ findings reported by Rafaat et al. was similar to the findings of a recent in our results [13].
The average frequency of non-ocular behçet disease is 30% [3]. In the literature, there are few recent studies about OCTA findings in non-ocular BD [12, 13, 21]. In an observational study by Rafaat et al. which included 20 eyes of 10 patients with non-ocular Behçet’s disease, detected microvascular anomalies such as capillary nonperfussion area and perifoveal capillary arcade disruption, capillary telangiectasia and perifoveal capillary dilatation in 80% eyes of nonocular BD group in SCP and 85% eyes of nonocular BD group in DCP. They reported no significant difference in the mean FAZ compared to the control group. They found a significantly lower mean capillary density in both SCP and DCP in OCTA. They performed central 10-2 perimetry to check for macular scotomata that could appear in response to the areas of capillary nonperfusion seen on OCTA. They reported that only 60% eyes of study group had relative central scotomata in the pattern deviation map and found no significant correlation between the mean deviation and average capillary density [13]. In another prospective study in 22 eyes with nonocular Behçet’s disease reported that the mean FAZ was reported to be significantly higher than the control group, and superficial and deep capillary plexus density were significantly lower than the control group [12]. Unlike their results, in our study, there was no significant difference in vessel density in SCP when compared to the control group.
On the other hand, Değirmenci et al. reported that VD measurements in SCP and DCP were lower in BD patients without ocular involvement, but these differences were not statistically significant [21]. In another study, no difference between any OCTA measurements in BD and healty eyes was obtained by Koca et al. [19]. The different outcomes may be linked to the different number of patients between the studies and the duration of BD.
Although the exact mechanism remains unclear, the reduction in VD in BD probably results from diffuse oclusive vasculitis which is caused by the circulation of abnormal immunocomplexes and endothelial cell proliferation [15, 22]. The OCTA assessment of SCP and DCP allowed us to identify the different involvement of the two plexuses; DCP was more severely involved than SCP with retinal capillary nonperfusion/hypoperfusion and other capillary changes. A similar predominance of microvascular changes on OCTA in the DCP has been observed in retinal vein occlusion, diabetic retinopathy and sickle cell retinopathy [3, 14, 23,24,25,26,27,28]. Opposite to SCP, capillaries in DCP are not directly connected to arterioles, and therefore may be weaker for ischemia [24]. Decreases in DCP which is particularly vulnerable to tissue hypoxia and increased inflammatory cytokine levels may predispose to future ocular involvement in BD. Furthermore, the other hypothesis is the outer retina could become ischemic because of decrease of blood supply from the choroid. VD in DCP can serve as a quantitative measure to obtain flow changes caused by pathological processes. In BD the outer retina may suffer from hypoxia, retinal capillary dilatation, hyperpermeability. Decreased vessel densities might be observed before diffuse microvascular occlusions and clinical involvement.
In healthy optic disc, the blood supply to its superficial layers (nerve fiber layer on the surface of the optic disc) is derived from the central retinal artery circulation, and that to the deeper layers (the prelaminar, lamina cribrosa, and retrolaminar regions) is originated from the posterior ciliary artery circulation [29, 30]. The radial peripapillary capillaries, of which lower density in the current study, are the most superficial layer of capillaries located in the inner part of the nerve fiber layer. The radial peripapillary capillaries may be more prone to ischemic damage and inflammation, such as the deep capillary plexus in the macula in Behçet disease. Therefore, decreased peripapillary VD could provide insight into ONH circulatory disorder which may be component of BD pathophysiology.
ONH parameters (retinal nerve fibre layer thickness, densities of peripapillary, inside disc, whole disc) and the factors influencing its signal were investigated in previous similar studies. These studies found that older age, longer diabetes duration and lower OCTA signal strength were associated with lower vessel densities [31,32,33]. For the clinical use of OCTA, it is necessary to identify the factors influencing its signal. Yun et al. also demonstrated that peripapillary VD (although not representing ocular perfusion pressure) is significantly associated with optic nerve head parameters [34]. Our study also revealed an another factor (Behçet Disease) that may affect ONH parameters.
Mittal et al. supported that subfoveal choroidal thickness in eyes of Behçet’s patients without ocular involvement was significantly thinner than control group [35]. In another study, they reported that choroidal thickness was significantly thicker than the control group during an acute attack [17]. Previous studies suggested that the choroid of the fellow eyes might be equally affected despite the absence of any obvious inflammation and hypothesized that patients with BD without ocular involvement could still have subclinical choroidal involvement [12, 17]. In our study, we evaluated the choriocapillaris flow area and found no statistical difference among the groups. This difference may be described by limitations of OCTA in terms of choroidal circulation/flow measurements using a SD platform.
The main limitation of this study was small sample size. However, we compared a particular subgroup of BD with healty population and obtained significant results. No reports addressing ONH microvasculature evaluated by OCTA in nonocular BD have been published. Therefore, in addition to the perifoveal microvascular structure in non-ocular BD, we also investigated the peripapillary microvascular structure. The total disc, peripapillary and inside-disc VD were observed lower in nonocular BD group. To the best of our knowledge, this is the first study evaluating ONH microvascular parameters by OCTA in nonocular BD.
We concluded that perifoveal and peripapillary microvascular changes that can be detected by OCTA may occur in Behçet’s patients without ocular involvement. Disorganization of the normal architecture of the capillary network was also more common in the DCP. Enlarged FAZ could be considered a form of ocular ischemia at the capillary level. Changes in these parameters may indicate preclinical ocular involvement. More reliable results can be obtained with a greater number of patients and longer follow-up.
Summary
What was known before
-
OCTA has advantages such as providing the possibility of separate and more detailed evaluation of the internal and external retinal circulation and the choriocapillaris. OCTA allows quantitative measurement of the capillary perfusion of the macula and optic nerve.
What this study adds
-
The perifoveal and peripapillary microvascular changes that can be detected by OCTA may occur in Behcet’s patients without ocular involvement.
References
Criteria for diagnosis of Behcet’s disease. International Study Group for Behcet’s Disease. Lancet. 1990;335:1078–80.
Hatemi G, Christensen R, Bang D, Bodaghi B, Celik AF, Fortune F, et al. 2018 update of the EULAR recommendations for the management of Behcet’s syndrome. Ann Rheum Dis. 2018;77:808–18.
Tugal-Tutkun I, Onal S, Altan-Yaycioglu R, Huseyin Altunbas H, Urgancioglu M. Uveitis in Behcet disease: an analysis of 880 patients. Am J Ophthalmol. 2004;138:373–80.
Yazici H, Tuzun Y, Pazarli H, Yurdakul S, Ozyazgan Y, Ozdogan H, et al. Influence of age of onset and patient’s sex on the prevalence and severity of manifestations of Behcet’s syndrome. Ann Rheum Dis. 1984;43:783–9.
Mishima S, Masuda K, Izawa Y, Mochizuki M, Namba K. The eighth Frederick H. Verhoeff Lecture. presented by saiichi mishima, MD Behcet’s disease in Japan: ophthalmologic aspects. Trans Am Ophthalmol Soc. 1979;77:225–79.
Ozyazgan Y, Ucar D, Hatemi G, Yazici Y. Ocular involvement of Behcet’s syndrome: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:298–306.
Ozdal PC, Ortac S, Taskintuna I, Firat E. Posterior segment involvement in ocular Behcet’s disease. Eur J Ophthalmol. 2002;12:424–31.
Pei M, Zhao C, Gao F, Qu Y, Liang A, Xiao J, et al. Analysis of parafoveal microvascular abnormalities in Behcet’s uveitis using projection-resolved optical coherence tomographic angiography. Ocul Immunol Inflamm 2019;1–6. E-pub ahead of print.
Bae K, Nam SW, Kang SW, Kim ES, Yu SY, Kim KT, et al. Central serous chorioretinopathy in elderly subjects: angiographic and tomographic characteristics. Graefes Arch Clin Exp Ophthalmol. 2019;257:279–88.
Sambhav K, Grover S, Chalam KV. The application of optical coherence tomography angiography in retinal diseases. Surv Ophthalmol. 2017;62:838–66.
Chung YR, Cho EH, Jang S, Lee SY, Lee ES, Lee K. Choroidal thickness indicates subclinical ocular and systemic inflammation in eyes with Behcet disease without active inflammation. Korean J Ophthalmol. 2018;32:290–5.
Goker YS, Yilmaz S, Kiziltoprak H, Tekin K, Demir G. Quantitative analysis of optical coherence tomography angiography features in patients with nonocular Behcet’s disease. Curr Eye Res. 2019;44:212–8.
Raafat KA, Allam R, Medhat BM. Optical coherence tomography angiography findings in patients with nonocular Behcet disease. Retina. 2019;39:1607–12.
Khairallah M, Abroug N, Khochtali S, Mahmoud A, Jelliti B, Coscas G, et al. Optical coherence tomography angiography in patients with Behcet uveitis. Retina. 2017;37:1678–91.
Atmaca LS. Fundus changes associated with Behcet’s disease. Graefes Arch Clin Exp Ophthalmol. 1989;227:340–4.
Cunningham ET Jr, Tugal-Tutkun I, Khairallah M, Okada AA, Bodaghi B, Zierhut M. Behcet uveitis. Ocul Immunol Inflamm. 2017;25:2–6.
Kim M, Kim H, Kwon HJ, Kim SS, Koh HJ, Lee SC. Choroidal thickness in Behcet’s uveitis: an enhanced depth imaging-optical coherence tomography and its association with angiographic changes. Invest Ophthalmol Vis Sci. 2013;54:6033–9.
Flammer J, Pache M, Resink T. Vasospasm, its role in the pathogenesis of diseases with particular reference to the eye. Prog Retin Eye Res. 2001;20:319–49.
Koca S, Onan D, Kalayci D, Alli, N. Comparison of optical coherence tomography angiography findings in patients with Behcet’s disease and healthy controls. Ocul Immunol Inflamm. 2019;28:806–13.
Cheng D, Shen M, Zhuang X, Lin D, Dai M, Chen S, et al. Inner retinal microvasculature damage correlates with outer retinal disruption during remission in Behcet’s posterior uveitis by optical coherence tomography angiography. Invest Ophthalmol Vis Sci. 2018;59:1295–304.
Degirmenci MFK, Temel E, Yalcindag FN. Quantitative evaluation of the retinal vascular parameters with OCTA in patients with behcet disease without ocular involvement. Ophthalmic Surg Lasers Imaging Retin. 2019;51:31–4.
Mullaney J, Collum LM. Ocular vasculitis in Behcet’s disease. A pathological and immunohistochemical study. Int Ophthalmol. 1985;7:183–91.
Pathanapitoon K, Kunavisarut P, Saravuttikul FA, Rothova A. Ocular manifestations and visual outcomes of Behcet’s Uveitis in a Thai population. Ocul Immunol Inflamm. 2019;27:2–6.
Scarinci F, Nesper PL, Fawzi AA. Deep retinal capillary nonperfusion is associated with photoreceptor disruption in diabetic macular ischemia. Am J Ophthalmol. 2016;168:129–38.
Hsu YR, Huang JC, Tao Y, Kaburaki T, Lee CS, Lin TC, et al. Noninfectious uveitis in the Asia-Pacific region. Eye (Lond). 2019;33:66–77.
Coscas F, Glacet-Bernard A, Miere A, Caillaux V, Uzzan J, Lupidi M, et al. Optical coherence tomography angiography in retinal vein occlusion: evaluation of superficial and deep capillary plexa. Am J Ophthalmol. 2016;161:160–71.
Samara WA, Shahlaee A, Sridhar J, Khan MA, Ho AC, Hsu J. Quantitative optical coherence tomography angiography features and visual function in eyes with branch retinal vein occlusion. Am J Ophthalmol. 2016;166:76–83.
Takase N, Nozaki M, Kato A, Ozeki H, Yoshida M, Ogura Y. Enlargement of foveal avascular zone in diabetic eyes evaluated by en face optical coherence tomography angiography. Retina. 2015;35:2377–83.
Hayreh SS. Blood supply of the optic nerve head and its role in optic atrophy, glaucoma, and oedema of the optic disc. Br J Ophthalmol. 1969;53:721–48.
Hayreh SS. Posterior ciliary artery circulation in health and disease: the Weisenfeld lecture. Invest Ophthalmol Vis Sci. 2004;45:749–57.
Spaide RF, Fujimoto JG, Waheed NK, Sadda SR, Staurenghi G. Optical coherence tomography angiography. Prog Retin Eye Res. 2018;64:1–55.
Cao D, Yang D, Yu H, Xie J, Zeng Y, Wang J, et al. Optic nerve head perfusion changes preceding peripapillary retinal nerve fibre layer thinning in preclinical diabetic retinopathy. Clin Exp Ophthalmol. 2019;47:219–25.
Jo YH, Sung KR, Shin JW. Effects of age on peripapillary and macular vessel density determined using optical coherence tomography angiography in healthy eyes. Invest Ophthalmol Vis Sci. 2019;60:3492–8.
Yun YI, Kim YW, Lim HB, Lee DH, Kim JH, Oh BL, et al. Peripapillary vessel parameters and mean ocular perfusion pressure in young healthy eyes: OCT angiography study. Br J Ophthalmol. 2020. E-pub ahead of print.
Mittal A, Velaga SB, Falavarjani KG, Nittala MG, Sadda SR. Choroidal thickness in non-ocular Behcet’s disease—a spectral-domain OCT study. J Curr Ophthalmol. 2017;29:210–3.
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Financial disclosure
The authors have no proprietary or commercial interest in any of the materials discussed in this article. The manuscript has been read and approved by all the authors, that the requirements for authorship have been met, and that each author believes that the manuscript represents honest work.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Karalezli, A., Kaderli, S.T., Sul, S. et al. Preclinical ocular features in patients with Behçet’s disease detected by optical coherence tomography angiography. Eye 35, 2719–2726 (2021). https://doi.org/10.1038/s41433-020-01294-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-020-01294-z
This article is cited by
-
Comparative analysis of optical coherence tomography angiography (OCTA) results between Behçet’s disease patients and a healthy control group
Clinical Rheumatology (2024)
-
Retinal microvasculature features in patients with Behcet’s disease: a systematic review and meta-analysis
Scientific Reports (2022)
-
Evaluation of subclinical ocular involvement in patients with deficiency of adenosine deaminase 2 (DADA2)
Clinical Rheumatology (2022)