Abstract
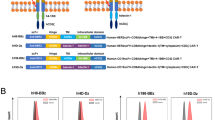
Chimeric antigen receptor T (CAR-T) cell therapy holds great promise as an innovative immunotherapeutic approach for cancer treatment. To optimize the production and application of CAR-T cells, we evaluated the in vivo stability and efficacy capacities of CAR-T cells developed under different conditions. In this study, CAR-T cells were activated using Phytohemagglutinin (PHA) or anti-CD3&anti-CD28 and were compared in an in vivo CD19+B-cell cancer model in mouse groups. Our results demonstrated that CAR-T cells activated with PHA exhibited higher stability and anti-cancer efficacy compared to those activated with anti-CD3&anti-CD28. Specifically, CAR19BB-T cells activated with PHA exhibited continuous proliferation and long-term persistence without compromising their anti-cancer efficacy. Kaplan–Meier survival analysis revealed prolonged overall survival in the CAR-T cell-treated groups compared to the only tumor group. Furthermore, specific LTR-targeted RT-PCR analysis confirmed the presence of CAR-T cells in the treated groups, with significantly higher levels observed in the CAR19BB-T (PHA) group compared to other groups. Histopathological analysis of spleen, kidney, and liver tissue sections indicated reduced inflammation and improved tissue integrity in the CAR-T cell-treated groups. Our findings highlight the potential benefits of using PHA as a co-stimulatory method for CAR-T cell production, offering a promising strategy to enhance their stability and persistence. These results provide valuable insights for the development of more effective and enduring immunotherapeutic approaches for cancer treatment. CAR-T cells activated with PHA may offer a compelling therapeutic option for advancing cancer immunotherapy in clinical applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Additional data beyond the data found in the article are available from the corresponding author on reasonable request.
References
Assessing national capacity for the prevention and control of noncommunicable diseases: report of the 2019 global survey. Geneva: World Health Organization; 2020;136.
American Cancer Society. Cancer facts & figures 2021–2022. The American Cancer Society journal, CA: A Cancer Journal for Clinicians. 2021. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2021.html.
JJaneway CA Jr, Travers P, Walport M, et al. Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science; 2001. Part V, The Immune System in Health and Disease. Available from: https://www.ncbi.nlm.nih.gov/books/NBK10775/.
Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ. Natural innate and adaptive immunity to cancer. Annu Rev Immunol. 2011;29:235–71. https://doi.org/10.1146/annurev-immunol-031210-101324.
Croft M. Co-stimulatory members of the TNFR family: keys to effective T-cell immunity? Nat Rev Immunol. 2003;3:609–20. https://doi.org/10.1038/nri1148.
Vinay DS, Kwon BS. 4-1BB (CD137), an inducible costimulatory receptor, as a specific target for cancer therapy. BMB Rep. 2012;45:5–12. https://doi.org/10.5483/bmbrep.2012.45.1.005.
Zhang C, Liu J, Zhong JF, Zhang X. Engineering CAR-T cells. Biomark Res. 2017a;5:22 https://doi.org/10.1186/s40364-017-0102-y.
Zhang L, Yu Z, Muranski P, Palmer DC, Restifo NP, Rosenberg SA, et al. Inhibition of TGF-β signaling in genetically engineered tumor antigen-reactive T cells significantly enhances tumor treatment efficacy. Gene Ther. 2017b;24:201–10. https://doi.org/10.1038/gt.2017.10.
Braendstrup P, Levine BL, Ruella M. The long road to the first FDA-approved gene therapy: chimeric antigen receptor T cells targeting CD19. Cytotherapy, 2020;22:57–69. https://doi.org/10.1016/j.jcyt.2019.12.004.
Patterson JD, Henson JC, Breese RO, Bielamowicz KJ, Rodriguez A. CAR T cell therapy for pediatric brain tumors. Front Oncol. 2020;10:1582 https://doi.org/10.3389/fonc.2020.01582.
Blaeschke F, Kaeuferle T, Feucht J, Weber D, Lotfi R, Kaiser A, et al. Defined central memory and stem memory T cell phenotype of CD4 and CD8 CAR T cells for the treatment of CD19+ acute lymphoblastic leukemia in an automated closed system. Blood. 2016;128:4558. https://doi.org/10.1182/blood.V128.22.4558.4558.
Blaeschke F, Stenger D, Kaeuferle T, Willier S, Lotfi R, Kaiser AD, et al. Induction of a central memory and stem cell memory phenotype in functionally active CD4+ and CD8+ CAR T cells produced in an automated good manufacturing practice system for the treatment of CD19+ acute lymphoblastic leukemia. Cancer Immunol Immunother. 2018;67:1053–66. https://doi.org/10.1007/s00262-018-2155-7.
López-Cantillo G, Urueña C, Camacho BA, Ramírez-Segura C. CAR-T cell performance: how to improve their persistence? Front Immunol. 2022;13:878209. https://doi.org/10.3389/fimmu.2022.878209.
Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA, et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 2015;385:517–28. https://doi.org/10.1016/S0140-6736(14)61403-3.
Maude SL, Barrett D, Teachey DT, Grupp SA. Managing cytokine release syndrome associated with novel T cell-engaging therapies. Cancer J. 2014a;20:119–22. https://doi.org/10.1097/PPO.0000000000000035.
Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N. Engl J Med. 2014b;371:1507–17. https://doi.org/10.1056/NEJMoa1407222.
Sadelain M, Brentjens R, Rivière I. The basic principles of chimeric antigen receptor design. Cancer Discov. 2013a;3:388–98. https://doi.org/10.1158/2159-8290.CD-12-0548.
Sadelain M, Brentjens R, Rivière I. The promise and potential pitfalls of chimeric antigen receptors. Curr Opin Immunol. 2013b;25:267–76. https://doi.org/10.1016/j.coi.2009.02.009.
Duarte RF, Chen FE, Lowdell MW, Potter MN, Lamana ML, Prentice HG, et al. Functional impairment of human T-lymphocytes following PHA-induced expansion and retroviral transduction: implications for gene therapy. Gene Ther. 2002;9:1359–68. https://doi.org/10.1038/sj.gt.3301807.
Gulden G, Sert B, Teymur T, Ay Y, Tiryaki NN, Mishra AK, et al. CAR-T cells with phytohemagglutinin (PHA) provide anti-cancer capacity with better proliferation, rejuvenated effector memory, and reduced exhausted T Cell frequencies. Vaccines. 2023;11:313. https://doi.org/10.3390/vaccines11020313.
Longo PA, Kavran JM, Kim MS, Leahy DJ. Transient mammalian cell transfection with polyethylenimine (PEI). Methods Enzymol. 2013;529:227–40. https://doi.org/10.1016/B978-0-12-418687-3.00018-5.
Morgan LR, Krementz ET, Fan SW, Fan D. Adoptive immunotherapy of advanced renal cell cancer using PHA-stimulated autologous lymphocytes. Anticancer Res. 1993;13:1763–7.
Gross G, Waks T, Eshhar Z. Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci USA. 1989;86:10024–8. https://doi.org/10.1073/pnas.86.24.10024.
Wang X, Popplewell LL, Wagner JR, Naranjo A, Blanchard MS, Mott MR, et al. Phase 1 studies of central memory derived CD19 CAR T-cell therapy following autologous HSCT in patients with B-cell NHL. Blood. 2016;127:2980–90. https://doi.org/10.1182/blood-2016-01-695122.
Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat Rev Immunol. 2013;13:227–42. https://doi.org/10.1038/nri3405.
Curran MA, Montalvo W, Yagita H, Allison JP. PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci USA. 2010;107:4275–80. https://doi.org/10.1073/pnas.0915174107.
Kinter AL, Godbout EJ, McNally JP, Riggin L, Sereti I, Roby G, O’Shea M, Fauci AS. The common γ-chain cytokines IL-2, IL-7, IL-15, and IL-21 induce the expression of programmed death-1 and its ligands. J Immunol. 2008;181:6738–46. https://doi.org/10.4049/jimmunol.181.10.6738.
Movafagh A, Heydary H, Mortazavi-Tabatabaei SA, Azargashb E. The significance application of indigenous phytohemagglutinin (PHA) mitogen on metaphase and cell culture procedure. Iran J Pharm Res. 2011;10:895.
Zhang Q, Vignali DAA. Co-stimulatory and co-inhibitory pathways in autoimmunity. Immunity. 2016;44:1034–51. https://doi.org/10.1016/j.immuni.2016.04.017.
Funding
All funding for the project was supported by the Presidency of Turkish Health Institutes (TUSEB) with grant number 4293.
Author information
Authors and Affiliations
Contributions
BS, GG, TT, YA, CT designed experiments. BS, GG, TT, YA, RDT, OMU, EG, HEE, CT performed experiments and acquired data. BS, GG, TT, YA, RDT, CT analyzed data and interpreted results. BS, GG, TT, YA, RDT, SO, CT performed animal treatments. BS, GG, TT, YA, RDT, OMU, EG, HEE, SI, PO, TA, OK, CT performed the histopathological analysis of tissues. IB, OMU performed clinical specimen collection and data following. CT supervised the work, acquired funds and performed the administration of the project. CT supervised the statistical analysis of the data. All authors wrote, reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
CT, BS, GG, TT, and YA are inventors of patent applications (pending) including “Allogeneic CAR-T cells expressing bispecific chimeric antigen receptors (CARs) targeting CD19 along with CD20 or CD22” (PCT/TR2023/050541 and 2022/009703) at Turkish Patent and Trademark Office. No other author has a competing interest except for these authors.
Ethics approval and consent to participate
In vitro and in vivo studies were approved by the Acibadem University and Acibadem Healthcare Institutions Medical Research Ethics Committee (ATADEK-2019-17/31). All animal studies have been approved by Acıbadem University Animal Experiments Local Ethics Committee (ACU-HADYEK; 11.13.2019).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sert, B., Gulden, G., Teymur, T. et al. Enhancing CAR-T cells: unleashing lasting impact potential with phytohemagglutinin activation in in vivo leukemia model. Cancer Gene Ther 31, 387–396 (2024). https://doi.org/10.1038/s41417-023-00709-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-023-00709-9