Abstract
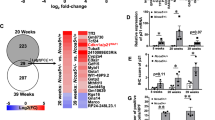
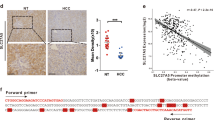
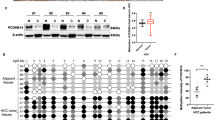
Long-term alcohol use is a confirmed risk factor of liver cancer tumorigenesis and metastasis. Multiple mechanisms responsible for alcohol related tumorigenesis have been proposed, including toxic reactive metabolite production, oxidative stress and fat accumulation. However, mechanisms underlying alcohol-mediated liver cancer metastasis remain largely unknown. We have previously demonstrated that SIRT7 regulates chemosensitivity by altering a p53-dependent pathway in human HCC. In the current study, we further revealed that SIRT7 is a critical factor in promoting liver cancer metastasis. SIRT7 expression is associated with disease stage and high SIRT7 predicts worse overall and disease-free survival. Overexpression of SIRT7 promotes HCC cell migration and EMT while knockdown of SIRT7 showed opposite effects. Mechanistically, we found that SIRT7 suppresses E-Cadherin expression through FOXO3-dependent promoter binding and H3K18 deacetylation. Knockdown of FOXO3 abolished the suppressive effect of SIRT7 on E-cadherin transcription. More importantly, we identified that alcohol treatment upregulates SIRT7 and suppresses E-cadherin expression via a CYP2E/ROS axis in hepatocytes both in vitro and in vivo. Antioxidant treatment in primary hepatocyte or CYP2E1−/− mice fed with alcohol impaired those effects. Reducing SIRT7 activity completely abolished alcohol-mediated promotion of liver cancer metastasis in vivo. Taken together, our data reveal that SIRT7 is a pivotal regulator of alcohol-mediated HCC metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data are within the manuscript and supporting information. Any additional information or data is available upon request.
References
Altekruse SF, McGlynn KA, Reichman ME. Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J Clin Oncol 2009;27:1485–91.
Aravalli RN, Steer CJ, Cressman EN. Molecular mechanisms of hepatocellular carcinoma. Hepatology 2008;48:2047–63.
Parker R, Kim SJ, Gao B. Alcohol, adipose tissue and liver disease: mechanistic links and clinical considerations. Nat Rev Gastroenterol Hepatol. 2018;15:50–9.
Huang CS, Ho CT, Tu SH, Pan MH, Chuang CH, Chang HW, et al. Long-term ethanol exposure-induced hepatocellular carcinoma cell migration and invasion through lysyl oxidase activation are attenuated by combined treatment with pterostilbene and curcumin analogues. J Agric Food Chem. 2013;61:4326–35.
Brandon-Warner E, Walling TL, Schrum LW, McKillop IH. Chronic ethanol feeding accelerates hepatocellular carcinoma progression in a sex-dependent manner in a mouse model of hepatocarcinogenesis. Alcohol-Clin Exp Res. 2012;36:641–53.
Szabo G. Alcoholic liver disease accelerates early hepatocellular cancer in a mouse model. Adv Exp Med Biol. 2018;1032:71–9.
McKillop IH, Schrum LW. Role of alcohol in liver carcinogenesis. Semin Liver Dis. 2009;29:222–32.
Lu Y, Cederbaum AI. CYP2E1 and oxidative liver injury by alcohol. Free Radic Biol Med. 2008;44:723–38.
Chen DL, Yu DD, Wang XY, Liu Y, He YJ, Deng RQ, et al. Epithelial to mesenchymal transition is involved in ethanol promoted hepatocellular carcinoma cells metastasis and stemness. Mol Carcinog. 2018;57:1358–70.
Chen D, Yan Y, Wang X, Li S, Liu Y, Yu D, et al. Chronic alcohol exposure promotes HCC stemness and metastasis through beta-catenin/miR-22-3p/TET2 axis. Aging 2021;13:14433–55.
McKillop IH, Schrum LW, Thompson KJ. Role of alcohol in the development and progression of hepatocellular carcinoma. Hepatic Oncol. 2016;3:29–43.
Leung TM, Nieto N. CYP2E1 and oxidant stress in alcoholic and non-alcoholic fatty liver disease. J Hepatol. 2013;58:395–8.
Zhou J, Wen Q, Li SF, Zhang YF, Gao N, Tian X, et al. Significant change of cytochrome P450s activities in patients with hepatocellular carcinoma. Oncotarget 2016;7:50612–23.
Gao J, Wang GJ, Wang Z, Gao N, Li J, Zhang YF, et al. High CYP2E1 activity correlates with hepatofibrogenesis induced by nitrosamines. Oncotarget 2017;8:112199–210.
Leung T, Rajendran R, Singh S, Garva R, Krstic-Demonacos M, Demonacos C. Cytochrome P450 2E1 (CYP2E1) regulates the response to oxidative stress and migration of breast cancer cells. Breast cancer Res: BCR. 2013;15:R107.
Mohr AM, Gould JJ, Kubik JL, Talmon GA, Casey CA, Thomas P, et al. Enhanced colorectal cancer metastases in the alcohol-injured liver. Clin Exp metastasis. 2017;34:171–84.
Wang RY, Chen XW, Zhang WW, Jiang F, Liu MQ, Shen XB. CYP2E1 changes the biological function of gastric cancer cells via the PI3K/Akt/mTOR signaling pathway. Mol Med Rep. 2020;21:842–50.
Acloque H, Adams MS, Fishwick K, Bronner-Fraser M, Nieto MA. Epithelial-mesenchymal transitions: the importance of changing cell state in development and disease. J Clin Investig. 2009;119:1438–49.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell 2009;139:871–90.
Larue L, Bellacosa A. Epithelial-mesenchymal transition in development and cancer: role of phosphatidylinositol 3’ kinase/AKT pathways. Oncogene 2005;24:7443–54.
Yoshida J, Horiuchi A, Kikuchi N, Hayashi A, Osada R, Ohira S, et al. Changes in the expression of E-cadherin repressors, Snail, Slug, SIP1, and Twist, in the development and progression of ovarian carcinoma: the important role of Snail in ovarian tumorigenesis and progression. Med Mol Morphol. 2009;42:82–91.
Ford E, Voit R, Liszt G, Magin C, Grummt I, Guarente L. Mammalian Sir2 homolog SIRT7 is an activator of RNA polymerase I transcription. Genes Dev. 2006;20:1075–80.
Vakhrusheva O, Smolka C, Gajawada P, Kostin S, Boettger T, Kubin T, et al. Sirt7 increases stress resistance of cardiomyocytes and prevents apoptosis and inflammatory cardiomyopathy in mice. Circ Res. 2008;102:703–10.
Ryu D, Jo YS, Lo Sasso G, Stein S, Zhang H, Perino A, et al. A SIRT7-dependent acetylation switch of GABPbeta1 controls mitochondrial function. Cell Metab. 2014;20:856–69.
Li Z, Bridges B, Olson J, Weinman SA. The interaction between acetylation and serine-574 phosphorylation regulates the apoptotic function of FOXO3. Oncogene 2017;36:1887–98.
Chen S, Blank MF, Iyer A, Huang B, Wang L, Grummt I, et al. SIRT7-dependent deacetylation of the U3-55k protein controls pre-rRNA processing. Nat Commun. 2016;7:10734.
Barber MF, Michishita-Kioi E, Xi Y, Tasselli L, Kioi M, Moqtaderi Z, et al. SIRT7 links H3K18 deacetylation to maintenance of oncogenic transformation. Nature 2012;487:114–8.
Kim JK, Noh JH, Jung KH, Eun JW, Bae HJ, Kim MG, et al. Sirtuin7 oncogenic potential in human hepatocellular carcinoma and its regulation by the tumor suppressors MiR-125a-5p and MiR-125b. Hepatol (Baltim, Md). 2013;57:1055–67.
Malik S, Villanova L, Tanaka S, Aonuma M, Roy N, Berber E, et al. SIRT7 inactivation reverses metastatic phenotypes in epithelial and mesenchymal tumors. Sci Rep. 2015;5:9841.
Zhao J, Wozniak A, Adams A, Cox J, Vittal A, Voss J, et al. SIRT7 regulates hepatocellular carcinoma response to therapy by altering the p53-dependent cell death pathway. J Exp Clin Cancer Res. 2019;38:252.
Li Z, Zhao J, Tikhanovich I, Kuravi S, Helzberg J, Dorko K, et al. Serine 574 phosphorylation alters transcriptional programming of FOXO3 by selectively enhancing apoptotic gene expression. Cell Death Differ. 2016;23:583–95.
Li Z, Zhao J, Zhang S, Weinman SA. FOXO3-dependent apoptosis limits alcohol-induced liver inflammation by promoting infiltrating macrophage differentiation. Cell Death Disco. 2018;4:16.
Sebastian BM, Roychowdhury S, Tang H, Hillian AD, Feldstein AE, Stahl GL, et al. Identification of a cytochrome P4502E1/Bid/C1q-dependent axis mediating inflammation in adipose tissue after chronic ethanol feeding to mice. J Biol Chem. 2011;286:35989–97.
Xie Y, McGill MR, Dorko K, Kumer SC, Schmitt TM, Forster J, et al. Mechanisms of acetaminophen-induced cell death in primary human hepatocytes. Toxicol Appl Pharm. 2014;279:266–74.
Yu HY, Ye W, Wu JX, Meng XQ, Liu RY, Ying XF, et al. Overexpression of Sirt7 exhibits oncogenic property and serves as a prognostic factor in colorectal cancer. Clin Cancer Res. 2014;20:3434–45.
Yin H, Hu M, Liang X, Ajmo JM, Li X, Bataller R, et al. Deletion of SIRT1 from hepatocytes in mice disrupts lipin-1 signaling and aggravates alcoholic fatty liver. Gastroenterology 2014;146:801–11.
Tumurbaatar B, Tikhanovich I, Li Z, Ren J, Ralston R, Kuravi S, et al. Hepatitis C and alcohol exacerbate liver injury by suppression of FOXO3. Am J Pathol. 2013;183:1803–14.
Parira T, Figueroa G, Laverde A, Casteleiro G, Gomez Hernandez ME, Fernandez-Lima F, et al. Novel detection of post-translational modifications in human monocyte-derived dendritic cells after chronic alcohol exposure: role of inflammation regulator H4K12ac. Sci Rep. 2017;7:11236.
Tan HK, Yates E, Lilly K, Dhanda AD. Oxidative stress in alcohol-related liver disease. World J Hepatol. 2020;12:332–49.
Wang HL, Lu RQ, Xie SH, Zheng H, Wen XM, Gao X, et al. SIRT7 exhibits oncogenic potential in human ovarian cancer cells. Asian Pac J Cancer Prev. 2015;16:3573–7.
Zhang S, Chen P, Huang Z, Hu X, Chen M, Hu S, et al. Sirt7 promotes gastric cancer growth and inhibits apoptosis by epigenetically inhibiting miR-34a. Sci Rep. 2015;5:9787.
Paredes S, Villanova L, Chua KF. Molecular pathways: emerging roles of mammalian Sirtuin SIRT7 in cancer. Clin Cancer Res. 2014;20:1741–6.
Li H, Tian Z, Qu Y, Yang Q, Guan H, Shi B, et al. SIRT7 promotes thyroid tumorigenesis through phosphorylation and activation of Akt and p70S6K1 via DBC1/SIRT1 axis. Oncogene 2019;38:345–59.
Loh CY, Chai JY, Tang TF, Wong WF, Sethi G, Shanmugam MK, et al. The E-cadherin and N-cadherin switch in epithelial-to-mesenchymal transition: signaling, therapeutic implications, and challenges. Cells-Basel. 2019;8.
Han LL, Jia L, Wu F, Huang C. Sirtuin6 (SIRT6) Promotes the EMT of hepatocellular carcinoma by stimulating autophagic degradation of E-cadherin. Mol Cancer Res: MCR. 2019;17:2267–80.
Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y, et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Sci (NY). 2004;303:2011–5.
Ghosh AP, Klocke BJ, Ballestas ME, Roth KA. CHOP potentially co-operates with FOXO3a in neuronal cells to regulate PUMA and BIM expression in response to ER stress. PLoS ONE. 2012;7:e39586.
Singh A, Ye M, Bucur O, Zhu S, Tanya Santos M, Rabinovitz I, et al. Protein phosphatase 2A reactivates FOXO3a through a dynamic interplay with 14-3-3 and AKT. Mol Biol Cell. 2010;21:1140–52.
Miyaguchi Y, Tsuchiya K, Sakamoto K. P53 negatively regulates the transcriptional activity of FOXO3a under oxidative stress. Cell Biol Int. 2009;33:853–60.
Chandramohan V, Mineva ND, Burke B, Jeay S, Wu M, Shen J, et al. c-Myc represses FOXO3a-mediated transcription of the gene encoding the p27(Kip1) cyclin dependent kinase inhibitor. J Cell Biochem. 2008;104:2091–106.
Blank MF, Grummt I. The seven faces of SIRT7. Transcription 2017;8:67–74.
Tang X, Shi L, Xie N, Liu Z, Qian M, Meng F, et al. SIRT7 antagonizes TGF-beta signaling and inhibits breast cancer metastasis. Nat Commun. 2017;8:318.
Zhou D, Shao L, Spitz DR. Reactive oxygen species in normal and tumor stem cells. Adv Cancer Res. 2014;122:1–67.
Diebold L, Chandel NS. Mitochondrial ROS regulation of proliferating cells. Free Radic Biol Med. 2016;100:86–93.
Klotz LO, Sanchez-Ramos C, Prieto-Arroyo I, Urbanek P, Steinbrenner H, Monsalve M. Redox regulation of FoxO transcription factors. Redox Biol. 2015;6:51–72.
Kovac S, Angelova PR, Holmstrom KM, Zhang Y, Dinkova-Kostova AT, Abramov AY. Nrf2 regulates ROS production by mitochondria and NADPH oxidase. Biochim Biophys Acta. 2015;1850:794–801.
Acknowledgements
This study was supported by grant 81974458, 82170607 from the National Natural Science Foundation of China, grant 2021JJ30463 from Hunan Provincial Natural Science Foundation of China, grant 2019RS1042, 2018RS3072, 2019TP1035 from the China Hunan Provincial Science/Technology Department, grant P30 GM118247 from the National Institute of General Medical Sciences from the National Institutes of Health (USA), grants 2022XKQ0205, KF2022001 and a startup grant from Hunan Normal University, and grants AA026025 and AA012863 to SAW from the National Institute on Alcohol Abuse and Alcoholism of the National Institutes of Health (USA). JQZ was supported by grant CSTC2020JCYJ-MSXMX0224 from Chongqing Natural Science Foundation. The specimens used in this study were provided, in part, by the University of Kansas Liver Center Biorepository. The authors acknowledge Dr Laura Nagy (Cleveland Clinic) who provided key materials for this study.
Author information
Authors and Affiliations
Contributions
ZL, CZ, and JQZ performed experiments and acquired data. BHL, WBT, YL, and WXH collected clinical samples. BHL performed the statistical analysis. ZL and SW conducted the experiments, analyzed data, and wrote the paper. All authors edited and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All animal handling procedures were approved by the Institutional Animal Care and Use. Committee at The University of Kansas Medical Center. Liver specimens were obtained from the University of Kansas Medical Center, the First Affiliated Hospital of Chongqing Medical University, and the Affiliated Hospital of Hunan Normal University. All studies using human tissue samples were approved by the Human Subjects Committee of the University of Kansas Medical Center. Written informed consent was obtained from all patients and all studies using human tissue samples were approved by the Human Subjects Committee of the University of Kansas Medical Center, Chongqing Medical University and Hunan Normal University School of Medicine.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, C., Zhao, J., Zhao, J. et al. CYP2E1-dependent upregulation of SIRT7 is response to alcohol mediated metastasis in hepatocellular carcinoma. Cancer Gene Ther 29, 1961–1974 (2022). https://doi.org/10.1038/s41417-022-00512-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-022-00512-y
This article is cited by
-
DDX3X interacts with SIRT7 to promote PD-L1 expression to facilitate PDAC progression
Oncogenesis (2024)
-
The dark side of SIRT7
Molecular and Cellular Biochemistry (2023)