Abstract
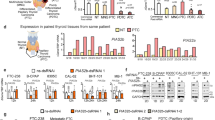
The Apoptosis Stimulating Protein of p53 2 (ASPP2) is a heterozygous insufficient tumor suppressor; however, its molecular mechanism(s) in tumor suppression is not completely understood. ASPP2 plays an essential role in cell growth, as shown by liver hepatocellular carcinoma (LIHC) RNA-seq assay using the Cancer Genome Atlas (TCGA) and High-Throughput-PCR assay using ASPP2 knockdown cells. These observations were further confirmed by in vivo and in vitro experiments. Mechanistically, N-terminus ASPP2 interacted with Keratin 18 (k18) in vivo and in vitro. Interestingly, the RDIVpSGP motif of ASPP2 associates with 14-3-3 and promotes ASPP2/k18/14-3-3 ternary-complex formation which promotes MEK/ERK signal activation by impairing 14-3-3 and BRAF association. Additionally, ASPP2-rAd injection promotes paclitaxel-suppressed tumor growth by suppressing cell proliferation in the BALB/c nude mice model. ASPP2 and k18 were preferentially downregulated in Hepatocellular Carcinoma (HCC), which predicted poor prognosis in HCC patients. Overall, these findings suggested that ASPP2 promoted BRAF/MEK/ERK signal activation by promoting the formation of an ASPP2/k18/14-3-3 ternary complex via the RDIVpSGP motif at the N terminus. Moreover, this study provides novel insights into the molecular mechanism of tumor suppression in HCC patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The authors declare that the data supporting our findings are included in the paper and its supplementary information files.
References
Kampa KM, Acoba JD, Chen DX, Gay J, Lee HJ, Beemer K, et al. Apoptosis-stimulating protein of p53 (ASPP2) heterozygous mice are tumor-prone and have attenuated cellular damage—response thresholds. Proc Natl Acad Sci USA. 2009;106:4390–5.
Rotem S, Katz C, Benyamini H, Lebendiker M, Veprintsev D, Rudiger S, et al. The structure and interactions of the proline-rich domain of ASPP2. J Biol Chem. 2008;283:18990–9.
Wang Z, Liu Y, Takahashi M, Hook KV, Kampa KM, Sheppard BC, et al. N terminus of ASPP2 binds to Ras and enhances Ras/Raf/MEK/ERK activation to promote oncogene-induced senescence. Proc Natl Acad Sci USA. 2013;110:312–7.
Kashyap D, Garg VK, Sandberg EN, Goel N, Bishayee A. Oncogenic and tumor suppressive components of the cell cycle in breast cancer progression and prognosis. Pharmaceutics. 2021;13:569.
Hinojosa JM, Lam-Chung CE, Ramos DC. Recent understanding and future directions of recurrent corticotroph tumors. Front Endocrinol. 2021;12:657382.
Obsilova V, Obsil T. The 14-3-3 proteins as important allosteric regulators of protein kinases. Int J Mol Sci. 2020;21:8824.
Mcgowan JE, Kratch J, Chattopadhyay S, Joe B, Conti HR, Chakravarti R. Bioinformatic analysis reveals new determinants of antigenic 14-3-3 proteins and a novel antifungal strategy. PLoS One. 2017;12:e0189503.
Stevers LM, Sijbesma E, Botta M, MacKintosh C, Obsil T, Landrieu I. Modulators of 14-3-3 protein–protein interactions. J Med Chem. 2018;61:3755–78.
Mariani RA, Paranjpe S, Dobrowolski R, Weber GF. 14-3-3 targets keratin intermediate filaments to mechanically sensitive cell–cell contacts. Mol Biol Cell. 2020;31:930–43.
He L, Liu XH, Yang JG, Li WJ, Liu SM, Liu XJ. Imbalance of the reciprocally inhibitory loop between the ubiquitin-specific protease USP43 and EGFR/PI3K/AKT drives breast carcinogenesis. Cell Res. 2018;28:934–51.
Qiu J, Wilkens C, Barrett K, Meyer AS. Microbial enzymes catalyzing keratin degradation: Classification, structure, function. Biotechnol Adv. 2020;44:107607.
Jacob JT, Coulombe PA, Kwan R, Omary MB. Types I and II keratin intermediate filaments. Cold Spring Harb Perspect Biol. 2018;10:a018275.
Posada I, Serulla M, Yong Z, Oetken-Lindholm C, Lectez B. ASPP2 is a novel pan-ras nanocluster scaffold. PLoS One. 2016;11:e0159677.
Meek S, Lane WS, Piwnica-Worms H. Comprehensive proteomic analysis of interphase and mitotic 14-3-3-binding proteins. J Biol Chem. 2004;279:32046–54.
Liau N, Venkatanarayan A, Quinn JG, Phung W, Malek S, Hymowitz SG, et al. Dimerization induced by C-terminal 14-3-3 binding is sufficient for BRAF kinase activation. Biochemistry. 2020;59:3982–92.
Xu WH, Xi B, Wu JY, An HY, Zhu J, Abassi YM, et al. Natural product derivative Bis(4-fluorobenzyl)trisulfide inhibits tumor growth by modification of beta-tubulin at Cys 12 and suppression of microtubule dynamics. Mol Cancer Ther. 2009;8:3318–30.
Yoneda K, Nakagawa T, Lawrence OT, Huard J, Demitsu T, Kubota Y, et al. Interaction of the profilaggrin N-terminal domain with loricrin in human cultured keratinocytes and epidermis. J Invest Dermatol. 2012;132:1206–14.
Boesze-Battaglia K, Song H, Sokolov M, Lillo C, Pankoski-Walker L, Gretzula C, et al. The tetraspanin protein peripherin-2 forms a complex with melanoregulin, a putative membrane fusion regulator. Biochemistry. 2007;46:1256–72.
Yang TW, Ouyang YB, Gao YX, Liu DJ, Zang YJ, Chen D. Enriched highthroughput reverse transcriptionquantitative PCR template preparation without preamplification. Mol Med Rep. 2020;22:3541–8.
Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012;16:284–7.
Yang TW, Gao YX, Liu DJ, Wang Y, Wu J, Liu XN, et al. ASPP2 enhances chemotherapeutic sensitivity through the down-regulation of XIAP expression in a p53 independent manner in hepatocellular carcinoma. Biochem Biophys Res Commun. 2019;508:769–74.
Wang Y, Godin-Heymann N, Wang XD, Bergamaschi D, Llanos S, Lu X. ASPP1 and ASPP2 bind active RAS, potentiate RAS signalling and enhance p53 activity in cancer cells. Cell Death Differ. 2013;20:525–34.
Shi H, Zhang Y, Ji J, Xu P, Shi H, Yue X, et al. Deficiency of apoptosis-stimulating protein two of p53 promotes liver regeneration in mice by activating mammalian target of rapamycin. Sci Rep. 2018;8:17927.
Roake CM, Artandi SE. Control of cellular aging, tissue function, and cancer by p53 downstream of telomeres. Cold Spring Harb Perspect Med. 2017;7:a026088.
Kai L, Tao J, Ouyang Y, Ying S, Zang YJ, Li N, et al. Nuclear EGFR impairs ASPP2-p53 complex-induced apoptosis by inducing SOS1 expression in hepatocellular carcinoma. Oncotarget. 2015;6:16507–16.
Mo XL, Fu H. BRET: NanoLuc-based bioluminescence resonance energy transfer platform to monitor protein–protein interactions in live cells. Methods Mol Biol. 2016;1439:263–71.
Toivola DM, Nieminen MI, Hesse M, Tao H, Baribault H, Magin TM, et al. Disturbances in hepatic cell-cycle regulation in mice with assembly-deficient keratins 8/18. Hepatology. 2001;34:1174–83.
Reinhardt HC, Yaffe MB. Phospho-Ser/Thr-binding domains: Navigating the cell cycle and DNA damage response. Nat Rev Mol Cell Biol. 2013;14:563–80.
Shen CH, Yuan P, Perez-Lorenzo R, Zhang Y, Lee SX, Yang O, et al. Phosphorylation of BRAF by AMPK impairs BRAF-KSR1 association and cell proliferation. Mol Cell. 2013;52:161–72.
Hevia LG, Fanarraga ML. Microtubule cytoskeleton-disrupting activity of MWCNTs: Applications in cancer treatment. J Nanobiotechnol. 2020;18:181.
Cao Y, Hamada T, Matsui T, Date T, Iwabuchi K. Communications BR: Hepatitis C virus core protein interacts with p53-binding protein, 53BP2/Bbp/ASPP2, and inhibits p53-mediated apoptosis. Biochem Biophys Res Commun. 2004;315:788–95.
Sivaramakrishnan S, Schneider JL, Sitikov A, Goldman RD, Ridge KM. Shear stress induced reorganization of the keratin intermediate filament network requires phosphorylation by protein kinase C zeta. Mol Biol Cell. 2009;20:2755–65.
Chou CC, Buehler MJ. Structure and mechanical properties of human trichocyte keratin intermediate filament protein. Biomacromolecules. 2012;13:3522–32.
Hallberg B. Exoenzyme S binds its cofactor 14-3-3 through a non-phosphorylated motif. Biochem Soc Trans. 2002;30:401–5.
Bocca C, Gabriel L, Miglietta A. Cytoskeleton-interacting activity of geiparvarin, diethylstilbestrol and conjugates. Chem Biol Interact. 2001;137:285–305.
Mauri G, Chiodoni C, Parenza M, Arioli I, Colombo MP. Ultrasound-guided intra-tumor injection of combined immunotherapy cures mice from orthotopic prostate cancer. Cancer Immunol Immunother. 2013;62:1811–9.
Kawauchi K, Watanabe M, Kaku H, Huang P, Sasaki K, Sakaguchi M, et al. Preclinical safety and efficacy of in situ REIC/Dkk-3 gene therapy for prostate cancer. Acta Med Okayama. 2012;66:7–16.
Gao D, Jin L, Ge J, Wang K, Qi A, Ping WX, et al. Construction of recombinant baculovirus vectors started by EF1alpha. Wei Sheng Wu Xue Bao. 2014;54:688–95.
Kao WY. Keratin expression by corneal and limbal stem cells during development. Exp Eye Res. 2020;200:108206.
Acknowledgements
We are very grateful to TCGA project organizers as well as all study participants. This work was supported by the National Natural Science Foundation of China (81470098, 82073676, 81900575), the Beijing Municipal Natural Science Foundation and Beijing Municipal Education Commission (KZ202010025037) and the Beijing Municipal Commission of Science and Technology (Z191100006619064).
Author information
Authors and Affiliations
Contributions
YTW, ZCL, SY, SYT, and GYX designed the study, researched data, and wrote the paper. ZBW, JRF, LDJ, and OYB researched the data, helped design experiments, and helped write the paper. LXN, WWJ, and YPX researched data and helped design experiments. XQG, CJZ, and CDX designed the study, helped write the paper, and oversaw the research. All authors read and approved the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Ethical approval
This study was carried out under the guiding principles of the Declaration of Helsinki. The specimens recruited have been informed to the patients and their families that the biological samples collected for this study will not cause additional economic and health burdens and have been approved by the patients and their families. BALB/c nude mice were housed in cages under specific pathogen-free conditions and carried out under the Qingdao University Animal Care Facility and the National Institutes of Health guidelines. The ethics committee of Affiliated Hospital of Qingdao University approved these studies (QYFYWZLL26433).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, T., zhu, C., Shi, Y. et al. RDIVpSGP motif of ASPP2 binds to 14-3-3 and enhances ASPP2/k18/14-3-3 ternary complex formulation to promote BRAF/MEK/ERK signal inhibited cell proliferation in hepatocellular carcinoma. Cancer Gene Ther 29, 1616–1627 (2022). https://doi.org/10.1038/s41417-022-00474-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-022-00474-1