Abstract
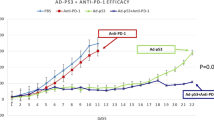
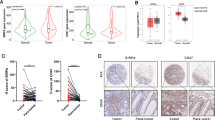
Targeted therapy results in objective responses in cervical cancer. However, the responses are short. In contrast, treatment with immune checkpoint inhibitors results in a lower responses rate, but the responses tend to be more durable. Based on these findings, we hypothesized that HPV16 E6/E7-targeted therapy may synergize with the PD-1 pathway blockade to enhance antitumor activity. To test hypothesis, we described for the first time the effects of the CRISPR/Cas9 that was targeted to the HPV and PD1 in vitro and in vivo. Our data showed that gRNA/cas9 targeted HPV16 E6/E7 induced cervical cancer cell SiHa apoptosis, and suggested that overexpression of PD-L1, induced by HPV16 E6/E7, may be responsible for lymphocyte dysfunction. In established SiHa cell- xenografted humanized SCID mice, Administration of gRNA-PD-1 together with gRNA-HPV16 E6/E7 treatment improved the survival and suppressed the tumor growth obviously. In addition, combination treatment increased the population of dendritic cells, CD8+ and CD4+ T lymphocyte cells. According, it enhanced the expression of Th1-associated immune-stimulating genes while reducing the transcription of regulatory/suppressive immune genes, reshaping tumor microenvironment from an immunosuppressive to a stimulatory state. These results demonstrate potent synergistic effects of combination therapy using HPV16 E6/E7-targeted therapy and immune checkpoint blockade PD1, supporting a direct translation of this combination strategy in clinic for the treatment of cervical cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Parkin DM, Pisani P, Ferlay J. Estimates of the worldwide incidence of 25 major cancers in 1990. Int J Cancer. 1999;80:827–41.
Moody CA, Laimins LA. Human papillomavirus oncoproteins: pathways to transformation. Nat Rev Cancer. 2010;10:550–60.
Chaturvedi AK. Beyond cervical cancer: burden of other HPV-related cancers among men and women. J Adolesc Health. 2010;46:S20–6.
Brinkman JA, Hughes SH, Stone P, Caffrey AS, Muderspach LI, Roman LD, et al. Therapeutic vaccination for HPV induced cervical cancers. Dis Markers. 2017;23:337–52.
Scheffner M, Werness BA, Huibregtse JM, Levine AJ, Howley PM. The E6 oncoprotein encoded by human papilloma virus types 16 and 18 promotes the degradation of p53. Cell. 1990;63:1129–36.
Dyson N, Howley PM, Kand Münger. The human papilloma virus16 E7 oncoprotein is able to bind to the retino-blastoma gene product. Science. 1989;243:934–7.
McConkey D, Choi W, Dinney C. Reply to Mattias Aine, Fredrik Liedberg, Gottfrid Sjödahl, and Mattias Höglund’s Letter to the Editor re: David J.McConkey, Woonyoung Choi, Colin P.N. Dinney. New insights into subtypes of invasive bladder cancer: considerations of the clinician. Eur Urol. 2014;66:609–10.
Carosella ED, Ploussard G, LeMaoult J, Desgrandchamps F. A systematic review of immunotherapy in urologic cancer: evolving roles for targeting of CTLA-4, PD-1/PD-L1, and HLA-G. Eur Urol. 2015;68:267–79.
Lyford-Pike S, Peng S, Young GD, Taube JM, Westra WH, Akpeng B, et al. Evidence for a role of the PD-1:PD-L1 pathway in immune resistance of HPV-associated head and neck squamous cell carcinoma. Cancer Res. 2013;73:1733–41.
Menderes G, Black J, Schwab CL, Santin AD. Immunotherapy and targeted therapy for cervical cancer: An update. Expert Rev Anticancer Ther. 2016;16:83–98.
Cairns RA, Hill RP. Acute hypoxia enhances spontaneous lymph node metastasis in an orthotopic murine model of human cervical carcinoma. Cancer Res. 2004;64:2054–61.
Zhen S, Hua L, Takahashi Y, Narita S, Liu YH, Li Y. In vitro and in vivo growth suppression of human papillomavirus 16-positive cervical cancer cells by CRISPR/Cas9. Biochem Biophys Res Commun. 2014;450:1422–6.
Yang W, Song Y, Lu YL, Sun JZ, Wang HW. Increased expression of programmed death (PD)-1 and its ligand PD-L1 correlates with impaired cell-mediated immunity in high-risk human papillomavirus-related cervical intraepithelial neoplasia. Immunology. 2013;139:513–22.
Lyford-Pike S, Peng S, Young GD, Taube JM, Westra WH, Akpeng B. Evidence for role of the PD-1: PD-L1 pathway in immune resistance of HPV-associated head and neck squamous cell carcinoma. Cancer Res. 2013;73:1733–41.
Ebina Hirotaka, Misawa Naoko, Kanemura Yuka, Koyanagi Y. Harnessing the CRISPR/Cas9 system to disrupt latent HIV-1 provirus. Sci Rep. 2013;3:2510.
Fujii Wataru, Kawasaki Kurenai, Sugiura Koji, Naito K. Efficient generation of large-scale genome-modified mice using gRNA and CAS9 endonuclease. Nucleic Acids Res. 2013;41:e187.
Zhen S, Hua L, Liu YH, Gao LC, Fu J, Wan DY. Harnessing the clustered regularly interspaced short palindromic repeat (CRISPR)/CRISPR-associated Cas9 system to disrupt the hepatitis B virus. Gene Ther. 2015;22:404–12.
Muñoz N. Human papillomavirus and cancer: the epidemiological evidence. J Clin Virol. 2000;19:15.
Isaacson Wechsler E, Wang Q, Roberts I, Pagliarulo E, Jackson D, Untersperger C. Reconstruction of human papillomavirus type 16 mediated early stage neoplasia implicates E6/E7 deregulation and the loss of contact inhibition in neoplastic progression. J Virol. 2012;86:6358–64.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (Grant nos. 81602295 to Shuai Zhen). This study was supported by grants from Key Research and Development Program of Shaanxi Province of China (2017ZDXM‐SF‐24‐1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The study was consistent with the principles used to relieve the pains of animals, as well as using the least number of animals as possible.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhen, S., Lu, J., Liu, YH. et al. Synergistic antitumor effect on cervical cancer by rational combination of PD1 blockade and CRISPR-Cas9-mediated HPV knockout. Cancer Gene Ther 27, 168–178 (2020). https://doi.org/10.1038/s41417-019-0131-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-019-0131-9
This article is cited by
-
Harnessing the evolving CRISPR/Cas9 for precision oncology
Journal of Translational Medicine (2024)
-
TNF-α-induced down-regulation of type I interferon receptor contributes to acquired resistance of cervical squamous cancer to Cisplatin
The Journal of Antibiotics (2024)
-
Activating STING/TBK1 suppresses tumor growth via degrading HPV16/18 E7 oncoproteins in cervical cancer
Cell Death & Differentiation (2024)
-
CRISPR/Cas9-mediated silencing of CD44: unveiling the role of hyaluronic acid-mediated interactions in cancer drug resistance
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
Understanding the HPV associated cancers: A comprehensive review
Molecular Biology Reports (2024)