Abstract
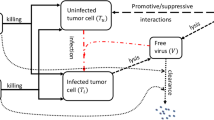
Poor tumor targeting of oncolytic adenoviruses (OAdv) after systemic administration is considered a major limitation for virotherapy of disseminated cancers. The benefit of using mesenchymal stem cells as cell carriers for OAdv tumor targeting is currently evaluated not only in preclinical models but also in clinical trials. In this context, we have previously demonstrated the enhanced antitumor efficacy of OAdv-loaded menstrual blood-derived mesenchymal stem cells (MenSCs). However, although significant, the antitumor efficacy obtained was modest, and we hypothesized that a greater antitumor efficacy could be obtained arming the OAdv with a therapeutic transgene. Here we show that combining MenSCs with ICOVIR15-cBiTE, an OAdv expressing an epidermal growth factor receptor (EGFR)-targeting bispecific T-cell engager (cBiTE), enhances the antitumor efficacy compared to MenSCs loaded with the unarmed virus ICOVIR15. We found that MenSCs properly produce cBiTE after viral infection leading to a greater antitumor potency both in vitro and in vivo. These findings indicate the mutual benefit of combining MenSCs and armed OAdv and support the combination of ICOVIR15-cBiTE and MenSCs as a cancer treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Rosewell Shaw A, Suzuki M. Recent advances in oncolytic adenovirus therapies for cancer. Curr Opin Virol. 2016;21:9–15.
Khare R, Chen CY, Weaver EA, Barry MA. Advances and future challenges in adenoviral vector pharmacology and targeting. Curr Gene Ther. 2011;11:241–58.
Rodriguez-Garcia A, Svensson E, Gil-Hoyos R, Fajardo CA, Rojas LA, Arias-Badia M, et al. Insertion of exogenous epitopes in the E3-19K of oncolytic adenoviruses to enhance TAP-independent presentation and immunogenicity. Gene Ther. 2015;22:596–601.
Uchibori R, Tsukahara T, Ohmine K, Ozawa K. Cancer gene therapy using mesenchymal stem cells. Int J Hematol. 2014;99:377–82.
Moreno R, Fajardo CA, Farrera-Sal M, Perise-Barrios AJ, Morales-Molina A, Al-Zaher AA, et al. Enhanced antitumor efficacy of oncolytic adenovirus-loaded menstrual blood-derived mesenchymal stem cells in combination with peripheral blood mononuclear cells. Mol Cancer Ther. 2019;18:127–38.
Fajardo CA, Guedan S, Rojas LA, Moreno R, Arias-Badia M, de Sostoa J, et al. Oncolytic adenoviral delivery of an EGFR-targeting T-cell engager improves antitumor efficacy. Cancer Res. 2017;77:2052–63.
Baeuerle PA, Kufer P, Bargou R. BiTE: teaching antibodies to engage T-cells for cancer therapy. Curr Opin Mol Ther. 2009;11:22–30.
Offner S, Hofmeister R, Romaniuk A, Kufer P, Baeuerle PA. Induction of regular cytolytic T cell synapses by bispecific single-chain antibody constructs on MHC class I-negative tumor cells. Mol Immunol. 2006;43:763–71.
Moreno R, Rojas LA, Villellas FV, Soriano VC, Garcia-Castro J, Fajardo CA, et al. Human menstrual blood-derived mesenchymal stem cells as potential cell carriers for oncolytic adenovirus. Stem cells Int. 2017;2017:3615729.
Melen GJ, Franco-Luzon L, Ruano D, Gonzalez-Murillo A, Alfranca A, Casco F, et al. Influence of carrier cells on the clinical outcome of children with neuroblastoma treated with high dose of oncolytic adenovirus delivered in mesenchymal stem cells. Cancer Lett. 2016;371:161–70.
Alfano AL, Nicola Candia A, Cuneo N, Guttlein LN, Soderini A, Rotondaro C, et al. Oncolytic adenovirus-loaded menstrual blood stem cells overcome the blockade of viral activity exerted by ovarian cancer ascites. Mol Ther Oncolytics. 2017;6:31–44.
Szoor A, Vaidya A, Velasquez MP, Mei Z, Galvan DL, Torres D, et al. T cell-activating mesenchymal stem cells as a biotherapeutic for HCC. Mol Ther Oncolytics. 2017;6:69–79.
Acknowledgements
We thank CERCA Program/Generalitat de Catalunya for their institutional support. The authors also thank Dolores Ramos and Silvia Torres for their lab technical support and Vanessa Cervera for samples processing. This work was supported by the Asociación Española Contra el Cáncer, BIO2017-897554-C2-1-R grant to R. Alemany from the Ministerio de Economía y Competitividad of Spain, Adenonet BIO2015-68990-REDT to R. Alemany from the Ministerio de Economía y Competitividad of Spain, Red ADVANCE(CAT) project COMRDI15-1-0013 to R. Alemany from Ris3CAT and 2017SGR449 research grant to RA from the ‘Generalitat de Catalunya’. Cofunded by the European Regional Development Fund, a way to Build Europe to RA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Barlabé, P., Sostoa, J.d., Fajardo, C.A. et al. Enhanced antitumor efficacy of an oncolytic adenovirus armed with an EGFR-targeted BiTE using menstrual blood-derived mesenchymal stem cells as carriers. Cancer Gene Ther 27, 383–388 (2020). https://doi.org/10.1038/s41417-019-0110-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-019-0110-1
This article is cited by
-
Immune landscape and response to oncolytic virus-based immunotherapy
Frontiers of Medicine (2024)
-
Mesenchymal stem cell-released oncolytic virus: an innovative strategy for cancer treatment
Cell Communication and Signaling (2023)
-
Oncolytic viruses encoding bispecific T cell engagers: a blueprint for emerging immunovirotherapies
Journal of Hematology & Oncology (2021)