Abstract
Background
We previously proposed an immune cell score (tumour node metastasis (TNM)-Immune cell score) classifier as an add-on to the existing TNM staging system for non-small cell lung cancer (NSCLC). Herein, we examined how to reliably assess a tertiary lymphoid structure (TLS) score to refine the TNM staging system.
Methods
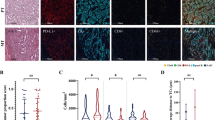
Using immunohistochemistry (CD8/cytokeratin), we quantified TLS in resected NSCLC whole-tumour tissue sections with three different scoring models on two independent collections (total of 553 patients). In a pilot setting, NanoString gene expression signatures were analysed for associations with TLS.
Results
The number of TLSs significantly decreased in stage III patients as compared to stage II. The TLS score was an independent positive prognostic factor, regardless of the type of (semi)-quantification strategy used (four-scale semi-quantitative; absolute count of total TLS; subpopulation of mature TLS) or the endpoint (disease-specific survival; overall survival; time to recurrence). Subgroup analyses revealed a significant prognostic impact of TLS score within each pathological stage, patient cohort and main histological subtype. Targeted gene expression analysis showed that high TLS levels were associated with the expression of B cell and adaptive immunity genes/metagenes including tumour inflammation signature.
Conclusions
The TLS score increases the prognostic power in each pathological stage and hence has the potential to refine TNM staging in resected NSCLC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Goldstraw, P., Crowley, J., Chansky, K., Giroux, D. J., Groome, P. A., Rami-Porta, R. et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM classification of malignant tumours. J. Thorac. Oncol. 2, 706–714 (2007).
Fridman, W. H., Zitvogel, L., Sautès–Fridman, C., Kroemer, G., Sautès-Fridman, C. & Kroemer, G. The immune contexture in cancer prognosis and treatment. Nat. Rev. Clin. Oncol. 25, 717–734 (2017).
Bremnes, R. M., Busund, L.-T., Kilvær, T. L., Andersen, S., Richardsen, E., Paulsen, E. E. et al. The role of tumor-infiltrating lymphocytes in development, progression, and prognosis of non-small cell lung cancer. J. Thorac. Oncol. 11, 789–800 (2016).
Pagès, F., Mlecnik, B., Marliot, F., Bindea, G., Ou, F. S., Bifulco, C. et al. International validation of the consensus Immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet 391, 2128–2139 (2018).
Donnem, T., Kilvaer, T. K., Andersen, S., Richardsen, E., Paulsen, E. E., Hald, S. M. et al. Strategies for clinical implementation of TNM-immunoscore in resected nonsmall-cell lung cancer. Ann. Oncol. 27, 225–232 (2016).
Donnem, T., Hald, S. M., Paulsen, E.-E., Richardsen, E., Al-Saad, S., Kilvaer, T. K. et al. Stromal CD8+ T-cell density—a promising supplement to TNM staging in non-small cell lung cancer. Clin. Cancer Res. 21, 2635–2643 (2015).
Paulsen, E.-E., Kilvaer, T., Khanehkenari, M. R., Maurseth, R. J., Al-Saad, S., Hald, S. M. et al. CD45RO(+) memory T lymphocytes-a candidate marker for TNM-immunoscore in squamous non-small cell lung cancer. Neoplasia 17, 839–848 (2015).
Rakaee, M., Kilvaer, T. K., Dalen, S. M., Richardsen, E., Paulsen, E.-E., Hald, S. M. et al. Evaluation of tumor-infiltrating lymphocytes using routine H&E slides predicts patient survival in resected non-small cell lung cancer. Hum. Pathol. 79, 188–198 (2018).
Rakaee, M., Busund, L. T. R., Jamaly, S., Paulsen, E. E., Richardsen, E., Andersen, S. et al. Prognostic value of macrophage phenotypes in resectable non–small cell lung cancer assessed by multiplex immunohistochemistry. Neoplasia 21, 282–293 (2019).
Paulsen, E.-E., Kilvaer, T. K., Rakaee, M., Richardsen, E., Hald, S. M., Andersen, S. et al. CTLA-4 expression in the non-small cell lung cancer patient tumor microenvironment: diverging prognostic impact in primary tumors and lymph node metastases. Cancer Immunol. Immunother. 66, 1449–1461 (2017).
Paulsen, E.-E., Kilvaer, T. K., Khanehkenari, M. R., Al-Saad, S., Hald, S. M., Andersen, S. et al. Assessing PDL-1 and PD-1 in non-small cell lung cancer: a novel immunoscore approach. Clin. Lung Cancer 18, 220–233.e8 (2017).
Rakaee, M., Busund, L.-T., Paulsen, E.-E., Richardsen, E., Al-Saad, S., Andersen, S. et al. Prognostic effect of intratumoral neutrophils across histological subtypes of non-small cell lung cancer. Oncotarget 7, 72184–72196 (2016).
Helmink, B. A., Reddy, S. M., Gao, J., Zhang, S., Basar, R., Thakur, R. et al. B cells and tertiary lymphoid structures promote immunotherapy response. Nature 577, 549–555 (2020).
Cabrita, R., Lauss, M., Sanna, A., Donia, M., Skaarup Larsen, M., Mitra, S. et al. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature 577, 561–565 (2020).
Petitprez, F., de Reyniès, A., Keung, E. Z., Chen, T. W. W., Sun, C. M., Calderaro, J. et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature 577, 556–560 (2020).
Moyron-Quiroz, J. E., Rangel-Moreno, J., Kusser, K., Hartson, L., Sprague, F., Goodrich, S. et al. Role of inducible bronchus associated lymphoid tissue (iBALT) in respiratory immunity. Nat. Med. 10, 927–934 (2004).
Randall, T. D. Bronchus-associated lymphoid tissue (BALT). Structure and function. Adv. Immunol. 107, 187–241 (2010).
Pitzalis, C., Jones, G. W., Bombardieri, M. & Jones S. A. Ectopic lymphoid-like structures in infection, cancer and autoimmunity. Nat. Rev. Immunol. 14, 447–462 (2014).
Victora, G. D. & Nussenzweig, M. C. Germinal centers. Annu. Rev. Immunol. 30, 429–457 (2012).
Bruno, T. C. New predictors for immunotherapy responses sharpen our view of the tumour microenvironment. Nat. Res. 577, 474–476 (2020).
Teillaud, J. L. & Dieu-Nosjean, M. C. Tertiary lymphoid structures: an anti-tumor school for adaptive immune cells and an antibody factory to fight cancer? Front. Immunol. 8, 830 (2017).
Sautès-Fridman, C., Petitprez, F., Calderaro, J. & Fridman, W. H. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat. Rev. Cancer 19, 307–325 (2019).
Sautès-Fridman, C., Lawand, M., Giraldo, N. A., Kaplon, H., Germain, C., Fridman, W. H. et al. Tertiary lymphoid structures in cancers: prognostic value, regulation, and manipulation for therapeutic intervention. Front. Immunol. 7, 407 (2016).
Silina, K., Soltermann, A., Attar, F. M., Casanova, R., Uckeley, Z. M., Thut, H. et al. Germinal centers determine the prognostic relevance of tertiary lymphoid structures and are impaired by corticosteroids in lung squamous cell carcinoma. Cancer Res. 78, 1308–1320 (2018).
Goc, J., Germain, C., Vo-Bourgais, T. K. D., Lupo, A., Klein, C., Knockaert, S. et al. Dendritic cells in tumor-associated tertiary lymphoid structures signal a th1 cytotoxic immune contexture and license the positive prognostic value of infiltrating CD8+ t cells. Cancer Res. 74, 705–715 (2014).
Goldstraw, P., Chansky, K., Crowley, J., Rami-Porta, R., Asamura, H., Eberhardt, W. E. E. et al. The IASLC lung cancer staging project: proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM Classification for lung cancer. J. Thorac. Oncol. 11, 39–51 (2016).
Travis, W. D., Brambilla, E., Nicholson, A. G., Yatabe, Y., Austin, J. H. M., Beasley, M. B. et al. The 2015 World Health Organization Classification of lung tumors. J. Thorac. Oncol. 10, 1243–1260 (2015).
McShane, L. M., Altman, D. G., Sauerbrei, W., Taube, S. E., Gion, M. & Clark, G. M. REporting recommendations for tumor MARKer prognostic studies (REMARK). Breast Cancer Res. Treat. 100, 229–235 (2006).
Eustace, A., Smyth, L. J. C., Mitchell, L., Williamson, K., Plumb, J. & Singh, D. Identification of cells expressing IL-17A and IL-17F in the lungs of patients with COPD. Chest 139, 1089–1100 (2011).
Kuroda, E., Ozasa, K., Temizoz, B., Ohata, K., Koo, C. X., Kanuma, T. et al. Inhaled fine particles induce alveolar macrophage death and interleukin-1α release to promote inducible bronchus-associated lymphoid tissue formation. Immunity 45, 1299–1310 (2016).
Schaadt, N. S., Schönmeyer, R., Forestier, G., Brieu, N., Braubach, P., Nekolla, K. et al. Graph-based description of tertiary lymphoid organs at single-cell level. PLoS Comput. Biol. 16, e1007385 (2020).
Ritchie, M. E., Phipson, B., Wu, D., Hu, Y., Law, C. W., Shi, W. et al. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Damotte, D., Warren, S., Arrondeau, J., Boudou-Rouquette, P., Mansuet-Lupo, A., Biton, J. et al. The tumor inflammation signature (TIS) is associated with anti-PD-1 treatment benefit in the CERTIM pan-cancer cohort. J. Transl. Med. 17, 1–10 (2019).
Asleh, K., Brauer, H. A., Sullivan, A., Lauttia, S., Lindman, H., Nielsen, T. O. et al. Predictive biomarkers for adjuvant capecitabine benefit in early-stage triple-negative breast cancer in the FinXX Clinical Trial. Clin. Cancer Res. 26, 2603–2614 (2020).
Ayers, M., Lunceford, J., Nebozhyn, M., Murphy, E., Loboda, A., Kaufman, D. R. et al. IFN-γ-related mRNA profile predicts clinical response to PD-1 blockade. J. Clin. Invest. 127, 2930–2940 (2017).
Dieu-Nosjean, M.-C., Giraldo, N. A., Kaplon, H., Germain, C., Fridman, W. H. & Sautès-Fridman, C. Tertiary lymphoid structures, drivers of the anti-tumor responses in human cancers. Immunol. Rev. 271, 260–275 (2016).
Buisseret, L., Desmedt, C., Garaud, S., Fornili, M., Wang, X., Van Den Eyden, G. et al. Reliability of tumor-infiltrating lymphocyte and tertiary lymphoid structure assessment in human breast cancer. Mod. Pathol. 30, 1204–1212 (2017).
Dieu-Nosjean, M. C., Goc, J., Giraldo, N. A., Sautès-Fridman, C. & Fridman W. H. Tertiary lymphoid structures in cancer and beyond. Trends Immunol. 35, 571–580 (2014).
Remark, R., Lupo, A., Alifano, M., Biton, J., Ouakrim, H., Stefani, A. et al. Immune contexture and histological response after neoadjuvant chemotherapy predict clinical outcome of lung cancer patients. Oncoimmunology 5, e1255394 (2016).
Galon, J., Mlecnik, B., Bindea, G., Angell, H. K., Berger, A., Lagorce, C. et al. Towards the introduction of the “immunoscore” in the classification of malignant tumours. J. Pathol. 232, 199–209 (2014).
Bera, K., Schalper, K. A., Rimm, D. L., Velcheti, V. & Madabhushi, A. Artificial intelligence in digital pathology - new tools for diagnosis and precision oncology. Nat. Rev. Clin. Oncol. 16, 703–715 (2019).
Pimenta, E. & Barnes, B. Role of tertiary lymphoid structures (TLS) in anti-tumor immunity: potential tumor-induced cytokines/chemokines that regulate TLS formation in epithelial-derived cancers. Cancers 6, 969–997 (2014).
Hennequin, A., Derangère, V., Boidot, R., Apetoh, L., Vincent, J., Orry, D. et al. Tumor infiltration by Tbet+ effector T cells and CD20+ B cells is associated with survival in gastric cancer patients. Oncoimmunology 5, e1054598 (2016).
Danaher, P., Warren, S., Lu, R., Samayoa, J., Sullivan, A., Pekker, I. et al. Pan-cancer adaptive immune resistance as defined by the tumor inflammation signature (TIS): results from The Cancer Genome Atlas (TCGA). J. Immunother. Cancer 6, 63 (2018).
Acknowledgements
We thank Dr. Wenjie Xu at NanoString Data Analysis Service (DAS) and Anette Skogstad at Molecular Pathology Lab (UNN) for NanoString analysis assistance.
Author information
Authors and Affiliations
Contributions
Conception and design: M.R. and T.D. Methodology (tissue processing, mIHC and NanoString): M.R., T.B. and M.B. Methodology (histopathology and development of immune scoring): M.R., L.-T.R.B., S.A.-S., F.P., E.R. and S.J. Clinical annotations including follow-up and tumour restaging: S.A., T.D., M.P., R.M.B. and E.-E.P. Analysis and interpretation of data: M.R. and T.K.K. Data visualisation: M.R., D.J.K. and T.K.K. Writing, review and editing: all authors. Project administration, ethical approval and funding acquisition: T.D., L.-T.R.B. and R.M.B.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was performed in accordance with the Declaration of Helsinki. The study was ethically approved by the Regional Committee (REK) and Norwegian Data Protection Organization (REK 2016/714). The ethics committee waived the need for patient’s consent in this retrospective study.
Consent to publish
This manuscript does not contain any individual person’s data.
Data availability
The datasets used and analysed during this study are available from the corresponding author on reasonable request, and most of the original data are included within the article and its Supplementary Materials.
Competing interests
The authors declare no competing interests.
Funding information
This work was financially supported by Norwegian Cancer Society and the Northern Norway Health Region Authority. The funders had no role in study design, data collection/analysis, decision to publish and preparation of the manuscript.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Rakaee, M., Kilvaer, T.K., Jamaly, S. et al. Tertiary lymphoid structure score: a promising approach to refine the TNM staging in resected non-small cell lung cancer. Br J Cancer 124, 1680–1689 (2021). https://doi.org/10.1038/s41416-021-01307-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01307-y
This article is cited by
-
Immature central tumor tertiary lymphoid structures are associated with better prognosis in non-small cell lung cancer
BMC Pulmonary Medicine (2024)
-
Tertiary lymphoid structural heterogeneity determines tumour immunity and prospects for clinical application
Molecular Cancer (2024)
-
Multi-resolution deep learning characterizes tertiary lymphoid structures and their prognostic relevance in solid tumors
Communications Medicine (2024)
-
Analyzing the associations between tertiary lymphoid structures and postoperative prognosis, along with immunotherapy response in gastric cancer: findings from pooled cohort studies
Journal of Cancer Research and Clinical Oncology (2024)
-
Intratumoral tertiary lymphoid structures promote patient survival and immunotherapy response in head neck squamous cell carcinoma
Cancer Immunology, Immunotherapy (2023)