Abstract
Background
To prospectively examine the association between diabetes and risk of prostate cancer defined by clinical and molecular features.
Methods
A total of 49,392 men from the Health Professionals Follow-up Study (HPFS) were followed from 1986 to 2014. Data on self-reported diabetes were collected at baseline and updated biennially. Clinical features of prostate cancer included localised, advanced, lethal, low-grade, intermediate-grade, and high-grade. Molecular features included TMPRSS2: ERG and PTEN subtypes. Cox proportional hazards regression models were used to evaluate the association between diabetes and incidence of subtype-specific prostate cancer.
Results
During 28 years of follow-up, we documented 6733 incident prostate cancer cases. Relative to men free from diabetes, men with diabetes had lower risks of total (HR: 0.82, 95% CI: 0.75–0.90), localised (HR: 0.82, 95% CI: 0.74–0.92), low-and intermediate-grade prostate cancer (HR: 0.77, 95% CI: 0.66–0.90; HR: 0.77, 95% CI: 0.65–0.91, respectively). For molecular subtypes, the HRs for ERG-negative and ERG-positive cases were 0.63 (0.42–0.95) and 0.72 (0.46–1.12); and for PTEN-intact and PTEN-loss cases were 0.69 (0.48–0.98) and 0.52 (0.19–1.41), respectively.
Conclusion
Besides providing advanced evidence for the inverse association between diabetes and prostate cancer, this study is the first to report associations between diabetes and ERG/PTEN defined prostate cancers.
Similar content being viewed by others
Background
Prostate cancer and type 2 diabetes mellitus are two of the most common chronic diseases that afflict the aging male population.1 The epidemiological findings of consistent inverse associations between diabetes and prostate cancer risk in multiple studies have represented an enigma.2,3,4,5 Although meta-analyses have reported similar inverse associations for diabetes with aggressive and nonaggressive prostate cancers,6 some studies have found a stronger inverse association for nonaggressive cancers, suggesting prostate-specific antigen (PSA) screening history and/or low PSA level among males with diabetes might lead to detection bias and underlie the inverse association.7,8
Additionally, it has been suggested that certain molecular subtypes of prostate cancer, including the TMPRSS2:ERG fusion and PTEN loss,9 are associated with biochemical recurrence or worse prognosis, even beyond that predicted by the Gleason score and tumour stage.10 Prior studies have reported a close biological relationship between ERG and PTEN,11,12 which together may delineate distinct prostate cancer subtypes with different prognosis; for example, relative to PTEN loss and ERG-negative prostate cancer patients, patients with PTEN intact and ERG positive/negative were observed to have better prognosis.13 Moreover, our group also found that risk factors associated with energy balance, such as high body mass index (BMI) and low physical activity are specifically associated with risk of TMPRSS2: ERG cancers.14,15 The association of diabetes with respect to the two molecular subtypes is of biological interest; however, no studies have been investigated to date.
Therefore, we examined the associations between diabetes and risk of developing prostate cancer defined by clinical features (stage, grade, and lethality) and molecular (TMPRSS2: ERG, PTEN) subtype taking into account screening patterns. We further examined whether the associations differed by diabetes lifestyle risk factors; and the potential effect of medication use on prostate cancer risk among men with diabetes.
Methods
Study population
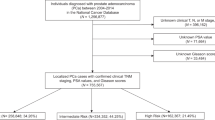
The Health Professionals Follow-up Study (HPFS) is an ongoing prospective cohort of men initiated in 1986 among 51,529 health professionals of age 40–75 years in the US at baseline. After excluding those who died, reported having cancers (excluding non-melanoma skin cancer) prior to baseline (n = 2092), or missing date of birth or prostate cancer diagnosis (n = 45), a total of 49,392 men were included in the current study.
Assessment of diabetes
On the baseline and subsequent follow-up biennial questionnaires, participants were asked if and when they had been diagnosed with diabetes by a physician. To confirm the self-reported cases of physician-diagnosed diabetes, a subsequent mailing was sent for ascertainment to obtain details about the date of diagnosis, symptoms, diagnostic tests and hypoglycaemic treatment. In addition, the information of regular use of insulin or oral hypoglycaemic medications was queried in the questionnaire. Diabetes cases identified before 1998 were defined according to the National Diabetes Data Group criteria,16 and the American Diabetes Association criteria was applied after 1998.17 The validity of the supplementary questionnaire for diabetes diagnosis has been confirmed in prior studies in HPFS, with 97% accuracy.18 Therefore, we took self-reported diabetes as the exposure. Duration of diabetes was calculated by subtracting the date of diagnosis from the date of the most recent completed questionnaire, and categorised as ≤1 year, 1.1–6 years, 6.1–15 years and >15 years.
Assessment of covariates
Information on age, race and height was collected at baseline; aspirin use, weight and lifestyles were collected at baseline and on each biennial questionnaire; waist circumference was assessed in 1987; family history of prostate cancer in father or brother was collected in 1990; statins use was collected at 1990 and on each subsequent biennial questionnaire. Current BMI and BMI at age 21 were calculated as self-reported weight divided by the square of height reported (kg/m2). Information on PSA screening was first asked in 1994 when men were asked to report their most recent PSA test, and in subsequent biennial cycles, they were asked whether they had a PSA test in the past 2 years. Dietary and nutrient intakes were assessed by a validated food frequency questionnaire at baseline and every 4 years thereafter.
Ascertainment of prostate cancer cases
Incident prostate cancers were initially self-reported on questionnaires, followed by confirming cancer diagnosis and extracting clinical and treatment information through medical records and pathology reports.19 Deaths were reported by family members, or identified through the National Death Index, with >98% sensitivity;20 Prostate cancer-specific death was determined by review of death certificates and medical records by an endpoint committee of physicians. Archival prostate tumour tissue from about half of HPFS participants diagnosed with prostate cancer was retrieved and undergone central histopathologic reviewed by study pathologists for the standardised tumour grading.
Stage T1a prostate cancer cases (n = 295) were excluded from this analysis since these cases are incidentally diagnosed and prone to detection bias. We classified clinical subtypes of prostate cancer as localised (stage T1 or T2 and N0, M0), advanced (stage T3b, T4, N1, or M1), lethal (distant metastases or prostate cancer was the cause of death); low-grade (Gleason 2–6), intermediate-grade (Gleason 7), and high-grade (Gleason 8–10) prostate cancer using information from prostatectomy or biopsy pathology reports.
A total of 5932 prostate cancer cases were accumulated between 1986 and 2009, among 2509 prostate cancer patients who received radical prostatectomy (RP) or transurethral resection of the prostate (TURP), we leveraged tumour ERG and PTEN immunohistochemistry (IHC) data (available for 949 and 757 cases, respectively) from tissue microarrays (TMAs).21 Tumours were classified as ERG positive if the case had positive ERG staining within prostate cancer epithelial cells on at least one TMA core. Tumours were classified as PTEN-loss if PTEN immunohistochemistry expression was either markedly decreased or entirely lost across >10% of tumour cells compared with surrounding benign glands or stroma.13 Relative to cases without IHC data, cases with IHC data were diagnosed at a more localised stage, had tumours with lower Gleason scores, had lower PSA levels, and were more often diagnosed in earlier years (Supplementary Table 1).
Statistical analysis
Person-time for participants was calculated from the return of the baseline questionnaire until the date of prostate cancer diagnosis, death, loss to follow-up, or the end of the follow-up (January 2014), whichever came first. For molecular defined prostate cancer outcomes, follow-up ended on 31 December 2009 because this was the last year a case assayed for ERG and PTEN was diagnosed. Consistent with our previous study,2 we evaluated the association with status (no vs. yes) and duration of diabetes (≤1, 1.1–6, 6.1–15 and >15 years). Cox proportional hazards regression models were applied to calculate the multivariable hazard ratios (HRs) and 95% confidence intervals (95% CIs). Age in months and calendar year at start of follow-up of each 2-year questionnaire cycle were used as stratification variables in the model. In multivariable analyses, we adjusted for race (white, African American, Asian American, other), family history of prostate cancer in father/brother (yes, no), height (≤68, >68–70, >70–72, >72 inches), BMI at age 21 (<20, 20 to <22.5, 22.5 to <25, ≥25 kg/m2), current BMI (<21, 21 to <25, 25 to <30, ≥30 kg/m2), smoking (never, former/quit >10 years ago, former/quit ≤10 years ago, current), lagged PSA testing history (yes, no, lagged by one period to avoid counting diagnostic PSA tests), lagged PSA testing in >50% of possible time periods (yes, no, lagged by one period to avoid counting diagnostic PSA tests), physical activity (quintiles of metabolic equivalent of task (MET)-h/week), total calories (quintiles of kcal/day), calcium intake (quintiles of mg/day), tomato sauce intake (quintiles of servings/week), fish intake (quintiles of servings/week), and coffee intake (quintiles of cups/day). All covariates, except race, height, and BMI at age 21, were updated with each questionnaire. Information from the prior questionnaire was carried forward for missing values.
An extension of Cox modelling that allows for exposure associations to vary by disease subtype was applied in the current study,22,23 and the details of this competing risks method have been described in our previous study.24 In brief, this model allowed for estimating HRs separately for the risk of diagnosis with ERG-positive cancer and ERG-negative cancer versus no cancer, and PTEN-intact and PTEN-loss versus no cancer. We tested heterogeneity across hazard ratios using likelihood ratio tests.25 In further, we applied inverse probability weights (IPW) to the competing risk model to validly estimate the association between history of diabetes and prostate cancer incidence by ERG and PTEN expression subtype. The method to create these weights have been described before.24 In brief, we first set weights to be 1 for subjects free from cancer and to be zero for patients who developed cancer but did not have RP or TURP tissue, second, we applied weights that accounted for clinical characteristics at and timing of diagnosis for patients who had tissue for IHC assay. To further investigate potential confounding by PSA screening, stratified analyses were applied for PSA screening history (yes, no), PSA screening intensity (>50% and ≤50% of reporting a PSA test in possible time periods) among PSA screened men, and PSA test level (normal, elevated) among PSA screened men.
We also conducted analyses of joint effect of diabetes and its high-risk lifestyle factors, including current BMI (≥30 vs. <30 kg/m2), BMI at 21 (≥23 vs. <23 kg/m2), waist circumference (≥40 vs. <40 inches), physical activity (<9 vs. ≥9 METS-h/week), and family history of diabetes (yes vs. no), with the risk of prostate cancer. We used a Wald test to examine whether the cross-product terms between these variables and diabetes status were statistically significant. Finally, we restricted to the diabetes population to test associations between use of aspirin, statins, and anti-diabetic medications and prostate risk.
All statistical analyses were conducted using the SAS software (Version 9.4; SAS Institute, Cary, NC, USA). All statistical tests were two-sided, and the significance level was set at P < 0.05.
Results
Participant characteristics by diabetes status and durations
During 1,078,832 person-years of follow-up, we documented a total of 6733 incident cases of prostate cancer. Among molecular defined prostate cancer cases, 452 (48%) prostate cancers were ERG fusion positive, 109 (14%) prostate cancers were PTEN loss. Midway through follow-up in 2002, 10% of men had reported a history of diabetes. Participants with diabetes were older, more likely to smoke and have a family history of diabetes. Moreover, participants with diabetes were more likely to have higher BMI and waist circumference and were less likely to be physically active. Patients with longer duration of diabetes generally tended to be more likely to have a family history of diabetes, and a higher proportion of aspirin and statins use (Table 1).
Diabetes and risk of prostate cancer, by clinical and pathologic tumour characteristics
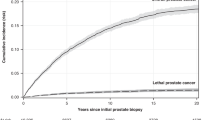
Table 2 shows that history of diabetes is inversely associated with prostate cancer risk (HR: 0.82, 95% CI: 0.75–0.90), particularly in localised (HR: 0.82, 95% CI: 0.74–0.92) and low-and intermediate-grade prostate cancer (HR: 0.77, 95% CI: 0.66–0.90; HR: 0.77, 95% CI: 0.65–0.91, respectively). For advanced (HR: 0.83, 95% CI: 0.61–1.14) and lethal (HR: 0.86, 95% CI: 0.68–1.08), the reduced risks were also observed but not statistically significant. Meanwhile, the magnitude of this association was stronger in men whose duration of diabetes was longer than 1 year (Ptrend = 0.0067); compared to men without diabetes history, the HRs (95% CI) for ≤ 1 year, 1.1–6 years, 6.1–15 years and >15 years duration of diabetes groups, for total prostate cancer were 1.23 (0.92–1.64), 0.79 (0.67–0.93), 0.83 (0.72–0.96) and 0.77 (0.64–0.92), respectively. Notably, a positive association with low-grade prostate cancer was observed for men with ≤1-year duration of diabetes (HR: 1.61, 95% CI: 1.07–2.42).
Diabetes and risk of prostate cancer, by molecular tumour characteristics
In Table 3, the HRs for diabetes and ERG-negative and ERG-positive prostate cancers were 0.63 (0.42–0.95) and 0.72 (0.46–1.12); and for PTEN-intact and PTEN-loss prostate cancer were 0.69 (0.48–0.98) and 0.52 (0.19–1.41), respectively. However, no significant interaction was observed between diabetes and ERG or PTEN subtypes (P-heterogeneity = 0.67 and 0.59, respectively).
Stratified analysis for diabetes and risk of prostate cancer, by PSA screening history
Table 4 presents the stratified analysis by PSA screening history. We found similar associations between diabetes and prostate cancer risk among men who received regular PSA screening (HR: 0.83, 95% CI: 0.74–0.92) or not (HR: 0.81, 95% CI: 0.67–0.98). In addition, we found an inverse association among PSA screened men with high intensity of screening (>50% of reporting a PSA test in possible time periods) (HR: 0.80, 95% CI: 0.71–0.90), and among screened men who had a normal PSA test value (HR: 0.85, 95% CI: 0.74–0.99). Similar inverse associations were observed for the risk of less aggressive prostate cancers (localised, or low-/intermediate-grade) but not for aggressive cases (advanced, lethal or high-grade).
Diabetes and its lifestyle risk factors with risk of prostate cancer
The independent and joint associations of current high BMI (≥30 kg/m2), BMI at 21 (≥23 kg/m2), waist circumference (≥40 inches), physical activity (≥9 METS-h/week), family history of diabetes, and diabetes are showed in Table 5. In particular, there was a stronger inverse association between diabetes and risk of prostate cancer among those with high waist circumference, although no significant interaction was observed (P-interaction = 0.22).
Medications and risk of prostate cancer among patients with diabetes
In addition, we tested that whether use of oral anti-diabetic medications, insulin, aspirin, and statins affect prostate cancer risk among men with diabetes (Table 6). Results showed that there were no statistically significant associations (HR: 0.80, 95% CI: 0.58–1.12; HR: 0.96, 95% CI: 0.52–1.77; HR: 1.14, 95% CI: 0.94–1.39; HR: 1.14, 95% CI: 0.91–1.43, respectively).
Discussion
In this large, updated analysis within the HPFS cohort with up to 28 years of follow-up, we confirmed inverse associations in the risk of prostate cancer among men with long-term diabetes. The associations were particularly strong for localised and low-/intermediate-grade prostate cancer. Of note, the inverse associations remained even when controlling for PSA screening history and frequency. Additionally, for the first time, we present data on the association between diabetes and risk of prostate cancer based on two molecular subtypes.
Twenty years ago, findings from the HPFS were the first prospective data with more than one thousand incident prostate cancer cases to demonstrate a statistically significant inverse association between diabetes and risk of prostate cancer.26 This finding has since been replicated in several cohort studies among different populations, including the 2009 analysis in HPFS.2 However, the inverse association differed by disease aggressiveness,3,7,8 and was primarily observed in the localised, low-grade prostate cancer, which keeps in line with our updated results. Considering the aggressiveness of prostate cancer is defined based on subsequent outcomes after diagnosis, such as metastasis and death, we assumed that diabetes may not be inversely associated with the most clinically relevant outcomes of prostate cancer, and a meta-analysis study showed that pre-existing type-2 diabetes is non-significantly positively associated with prostate cancer-specific mortality (RR: 1.17, 95% CI: 0.96–1.42) in prostate cancer patients.27
Although a previous meta-analysis found no statistically significant departure from linearity between length of time being diabetic and prostate cancer risk (p < 0.34),28 we observed the trend of linear association when four groups of diabetes duration were analysed as continuous variable. And the inverse associations were more frequently observed in men with longer duration of diabetes;5,8,29,30,31 even a positive relation could be observed in some studies for the shorter diabetes duration.5,8,29,31
One possible mechanism to explain the inverse association between diabetes and prostate cancer is the relative insulin-deficient environment in long-term diabetes, resulting in lower plasma insulin (C-peptide) and insulin-like growth factor-1 (IGF-1) levels in diabetics compared to non-diabetics.32 This is important given consistent findings in prospective studies that higher circulating levels of IGF-1 are associated with an increased risk of prostate cancer, particularly for the non-aggressive and low-grade disease.33 Additionally, circulating levels of the insulin-like growth factor-binding protein 2 (IGFBP2) have been positively correlated with insulin sensitivity over prolonged periods,34 and the risk of developing diabetes was 5-fold lower for IGFBP2 levels in the top quintile versus the lowest quintile;35 however, IGFBP2 concentration was positively associated with prostate cancer risk.33 Another potential mechanism is the genetic link, several loci, especially hepatocyte nuclear factor-1 β gene (HNF1β), have been reported to be associated with the risk of both diabetes and prostate cancer;36,37 however, mediation analyses provided insufficient evidence for the inverse relationship between diabetes and prostate cancer risk is mediated through diabetes risk variants.38,39 Our previous prospective study indicated that ERG positive tumours were characterised by higher expression of insulin receptor and IGF-1 receptor, compared with ERG-negative tumours.40 In addition, experimental studies found that PTEN mutations may reduce the risk of type 2 diabetes owing to enhanced insulin sensitivity.41 Although our findings of inverse associations between diabetes and ERG-negative and PTEN-intact disease aligned with the hypothesis above, given the analyses by ERG and PTEN status used a smaller number of cases than analyses of prostate cancer overall, chance might have played a role in the different results across diabetes status, and the results need to be confirmed by larger studies.
Given the stronger inverse associations for diabetes with less aggressive prostate cancer, there are lingering concerns that PSA screening could lead to detection bias for the relation. First, prior studies indicated that the participation rate for PSA test could be higher8,42,43 or lower44 for men with diabetes compared to men without. Second, in men without cancer, PSA levels in diabetics are lower than in men without diabetes, which could contribute to reduced detection rates of prostate cancer, particularly the localised.
To address these two issues, similar with the study conducted in Israel8 and our cohort,14 we first adjusted for lagged PSA testing and intensity in the main analysis, and we additionally undertook several stratified analyses. The inverse association for diabetes remained in the subgroup of men with regular PSA testing, which were consistent with our previous results2 and findings from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Screening Trial cohort.7 Moreover, when stratified on men with normal PSA levels at testing, the reduced risk of overall and nonaggressive prostate cancer among males with diabetes persisted. Therefore, similar with previous studies considering PSA level and screening frequency,3 our results suggested that detection bias might contribute to part of the inverse association but is unlikely to fully explain the link between diabetes and prostate cancer. Studies have consistently reported an inverse association between obesity and the risk of less aggressive prostate cancer.45,46 Obesity has broad systemic effects including lower circulating testosterone levels.47 The slightly stronger inverse associations between diabetes and prostate cancer among obese males, especially those with central obesity, and males with low physical activity level suggests a potential modified effect of obesity on the association. Meanwhile, the slightly stronger association has also been observed in males with a family history of diabetes, and a nationwide study from Sweden has reported that family history of type 2 diabetes mellitus was associated with a lower incidence of prostate cancer, and the risk was even lower for those with more than one affected relatives.48 The potential mechanisms may be attributed to the genetic factors or shared familial factors, such as obesity.
Medications such as aspirin and statins have been recommended to be used in patients with diabetes for the prevention of cardiovascular events;49 together with anti-diabetic medications, they have shown a decreased risk of prostate cancer.50 Data from this prospective study showed that there may be no association between these medications and overall prostate cancer risk in men with diabetes. Although there may be misclassification due to self-report, this is expected to be nondifferential in HPFS, where medical professionals repeatedly reported on medication use before cancer diagnosis.
There are several potential strengths and limitations in our study to consider in interpreting the findings. First, we relied on self-reported diabetes, which may lead to the misclassification of exposure. However, the cohort is comprised of male health professionals, and we have shown the accuracy of self-reported cases with physician-diagnosed diabetes in our cohort was very high (97%). Second, our results might be influenced by detection bias. However, the detailed information available on PSA testing history allowed us adjusted and stratified the potential confounding by PSA test in our results. Moreover, the molecular subtype of prostate cancer may be less susceptible to screening and detection biases, which offered stronger evidence for the association between diabetes and prostate cancer. Third, the ERG and PTEN-featured prostate cancer cases were derived from males who received RP or TURP, but when the inverse probability weighting method was used to balance the potential bias, the results were similar with the unweighted analysis (Supplementary Table 2). The strengths of our study include the prospective study design, the high follow-up rates on questionnaires, with >90% follow-up in each cycle,51 and the 28 years of follow-up for cancer incidence and mortality, which enabled us to examine the association between long-term diabetes and different clinical featured prostate cancers with considerable statistical power. Moreover, we have detailed covariate data to control for potential confounding and undertook the sub-analysis to assess potential for bias.
In summary, the updated results from this large prospective male cohort provided converging evidence for the inverse association between diabetes and prostate cancer, particularly for the nonaggressive prostate cancer, suggesting that the presence of diabetes may influence the frequency and interpretation of screening tests for prostate cancer. In addition, this is the first study to our knowledge to report the association between diabetes and molecular defined prostate cancer, which might contribute to the interpretation of the inverse association between diabetes and prostate cancer and may help researchers to follow-up with potential mechanisms underlying the association for future targets of intervention.
References
Prince, M. J., Wu, F., Guo, Y., Gutierrez Robledo, L. M., O’Donnell, M., Sullivan, R. et al. The burden of disease in older people and implications for health policy and practice. Lancet 385, 549–562 (2015).
Kasper, J. S., Liu, Y. & Giovannucci, E. Diabetes mellitus and risk of prostate cancer in the health professionals follow-up study. Int. J. Cancer 124, 1398–1403 (2009).
Waters, K. M., Henderson, B. E., Stram, D. O., Wan, P., Kolonel, L. N. & Haiman, C. A. Association of diabetes with prostate cancer risk in the multiethnic cohort. Am. J. Epidemiol. 169, 937–945 (2009).
Tsilidis, K. K., Allen, N. E., Appleby, P. N., Rohrmann, S., Nothlings, U., Arriola, L. et al. Diabetes mellitus and risk of prostate cancer in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 136, 372–381 (2015).
Haggstrom, C., Van Hemelrijck, M., Zethelius, B., Robinson, D., Grundmark, B., Holmberg, L. et al. Prospective study of Type 2 diabetes mellitus, anti-diabetic drugs and risk of prostate cancer. Int. J. Cancer 140, 611–617 (2017).
Xu, H., Jiang, H. W., Ding, G. X., Zhang, H., Zhang, L. M., Mao, S. H. et al. Diabetes mellitus and prostate cancer risk of different grade or stage: a systematic review and meta-analysis. Diabetes Res. Clin. Pract. 99, 241–249 (2013).
Miller, E. A., Pinsky, P. F. & Pierre-Victor, D. The relationship between diabetes, prostate-specific antigen screening tests, and prostate cancer. Cancer Causes. Control 29, 907–914 (2018).
Dankner, R., Boffetta, P., Keinan-Boker, L., Balicer, R. D., Berlin, A., Olmer, L. et al. Diabetes, prostate cancer screening and risk of low- and high-grade prostate cancer: an 11 year historical population follow-up study of more than 1 million men. Diabetologia 59, 1683–1691 (2016).
Cancer Genome Atlas Research N. The molecular taxonomy of primary prostate cancer. Cell 163, 1011–1025 (2015).
Carneiro, A., Barbosa, A. R. G., Takemura, L. S., Kayano, P. P., Moran, N. K. S., Chen, C. K. et al. The role of immunohistochemical analysis as a tool for the diagnosis, prognostic evaluation and treatment of prostate cancer: a systematic review of the literature. Front. Oncol. 8, 377 (2018).
Squire, J. A. TMPRSS2-ERG and PTEN loss in prostate cancer. Nat. Genet. 41, 509–510 (2009).
Yoshimoto, M., Joshua, A. M., Cunha, I. W., Coudry, R. A., Fonseca, F. P., Ludkovski, O. et al. Absence of TMPRSS2:ERG fusions and PTEN losses in prostate cancer is associated with a favorable outcome. Mod. Pathol. 21, 1451–1460 (2008).
Ahearn, T. U., Pettersson, A., Ebot, E. M., Gerke, T., Graff, R. E., Morais, C. L. et al. A prospective investigation of PTEN loss and ERG expression in lethal prostate cancer. J. Natl. Cancer. Inst. 108, 1–9 (2016).
Pernar, C. H., Ebot, E. M., Pettersson, A., Graff, R. E., Giunchi, F., Ahearn, T. U. et al. A prospective study of the association between physical activity and risk of prostate cancer defined by clinical features and TMPRSS2:ERG. Eur. Urol. 76, 33–40 (2019).
Graff, R. E., Ahearn, T. U., Pettersson, A., Ebot, E. M., Gerke, T., Penney, K. L. et al. Height, obesity, and the risk of TMPRSS2:ERG-defined prostate cancer. Cancer Epidemiol. Biomark. Prev. 27, 193–200 (2018).
National Diabetes Data Group. Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. National Diabetes Data Group. Diabetes 28, 1039–1057 (1979).
Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 20, 1183–1197 (1997).
Hu, F. B., Leitzmann, M. F., Stampfer, M. J., Colditz, G. A., Willett, W. C. & Rimm, E. B. Physical activity and television watching in relation to risk for type 2 diabetes mellitus in men. Arch. Intern. Med. 161, 1542–1548 (2001).
Wilson, K. M., Kasperzyk, J. L., Rider, J. R., Kenfield, S., van Dam, R. M., Stampfer, M. J. et al. Coffee consumption and prostate cancer risk and progression in the Health Professionals Follow-up Study. J. Natl. Cancer Inst. 103, 876–884 (2011).
Stampfer, M. J., Willett, W. C., Speizer, F. E., Dysert, D. C., Lipnick, R., Rosner, B. et al. Test of the National Death Index. Am. J. Epidemiol. 119, 837–839 (1984).
Pettersson, A., Graff, R. E., Bauer, S. R., Pitt, M. J., Lis, R. T., Stack, E. C. et al. The TMPRSS2:ERG rearrangement, ERG expression, and prostate cancer outcomes: a cohort study and meta-analysis. Cancer Epidemiol. Biomark. Prev. 21, 1497–1509 (2012).
Lunn, M. & McNeil, D. Applying Cox regression to competing risks. Biometrics 51, 524–532 (1995).
Rosner, B., Glynn, R. J., Tamimi, R. M., Chen, W. Y., Colditz, G. A., Willett, W. C. et al. Breast cancer risk prediction with heterogeneous risk profiles according to breast cancer tumor markers. Am. J. Epidemiol. 178, 296–308 (2013).
Graff, R. E., Pettersson, A., Lis, R. T., Ahearn, T. U., Markt, S. C., Wilson, K. M. et al. Dietary lycopene intake and risk of prostate cancer defined by ERG protein expression. Am. J. Clin. Nutr. 103, 851–860 (2016).
Glynn, R. J. & Rosner, B. Comparison of risk factors for the competing risks of coronary heart disease, stroke, and venous thromboembolism. Am. J. Epidemiol. 162, 975–982 (2005).
Giovannucci, E., Rimm, E. B., Stampfer, M. J., Colditz, G. A. & Willett, W. C. Diabetes mellitus and risk of prostate cancer (United States). Cancer Causes. Control 9, 3–9 (1998).
Lee, J., Giovannucci, E. & Jeon, J. Y. Diabetes and mortality in patients with prostate cancer: a meta-analysis. Springerplus 5, 1548 (2016). eCollection 42016.
Xu, H., Mao, S. H., Ding, G. X., Ding, Q. & Jiang, H. W. Diabetes mellitus reduces prostate cancer risk—no function of age at diagnosis or duration of disease. Asian Pac. J. Cancer Prev. 14, 441–447 (2013).
Johnson, J. A., Bowker, S. L., Richardson, K. & Marra, C. A. Time-varying incidence of cancer after the onset of type 2 diabetes: evidence of potential detection bias. Diabetologia 54, 2263–2271 (2011).
Baradaran, N., Ahmadi, H., Salem, S., Lotfi, M., Jahani, Y., Baradaran, N. et al. The protective effect of diabetes mellitus against prostate cancer: role of sex hormones. Prostate 69, 1744–1750 (2009).
Carstensen, B., Witte, D. R. & Friis, S. Cancer occurrence in Danish diabetic patients: duration and insulin effects. Diabetologia 55, 948–958 (2012).
Kasper, J. S., Liu, Y., Pollak, M. N., Rifai, N. & Giovannucci, E. Hormonal profile of diabetic men and the potential link to prostate cancer. Cancer Causes. Control 19, 703–710 (2008).
Travis, R. C., Appleby, P. N., Martin, R. M., Holly, J. M. P., Albanes, D., Black, A. et al. A meta-analysis of individual participant data reveals an association between circulating levels of IGF-I and prostate cancer risk. Cancer Res. 76, 2288–2300 (2016).
Clemmons, D. R. Role of IGF-binding proteins in regulating IGF responses to changes in metabolism. J. Mol. Endocrinol. 61, T139–T169 (2018).
Rajpathak, S. N., He, M., Sun, Q., Kaplan, R. C., Muzumdar, R., Rohan, T. E. et al. Insulin-like growth factor axis and risk of type 2 diabetes in women. Diabetes 61, 2248–2254 (2012).
Frayling, T. M., Colhoun, H. & Florez, J. C. A genetic link between type 2 diabetes and prostate cancer. Diabetologia 51, 1757–1760 (2008).
Xiang, Y. Z., Jiang, S. B., Zhao, J., Xiong, H., Cui, Z. L., Li, G. B. et al. Racial disparities in the association between diabetes mellitus-associated polymorphic locus rs4430796 of the HNF1beta gene and prostate cancer: a systematic review and meta-analysis. Genet. Mol. Res. 13, 6582–6592 (2014).
Waters, K. M., Wilkens, L. R., Monroe, K. R., Stram, D. O., Kolonel, L. N., Henderson, B. E. et al. No association of type 2 diabetes risk variants and prostate cancer risk: the multiethnic cohort and PAGE. Cancer Epidemiol. Biomark. Prev. 20, 1979–1981 (2011).
Machiela, M. J., Lindstrom, S., Allen, N. E., Haiman, C. A., Albanes, D., Barricarte, A. et al. Association of type 2 diabetes susceptibility variants with advanced prostate cancer risk in the Breast and Prostate Cancer Cohort Consortium. Am. J. Epidemiol. 176, 1121–1129 (2012).
Pettersson, A., Lis, R. T., Meisner, A., Flavin, R., Stack, E. C., Fiorentino, M. et al. Modification of the association between obesity and lethal prostate cancer by TMPRSS2:ERG. J. Natl. Cancer Inst. 105, 1881–1890 (2013).
Pal, A., Barber, T. M., Van de Bunt, M., Rudge, S. A., Zhang, Q., Lachlan, K. L. et al. PTEN mutations as a cause of constitutive insulin sensitivity and obesity. N. Engl. J. Med. 367, 1002–1011 (2012).
Hamoen, E. H., Reukers, D. F., Numans, M. E., Barentsz, J. O., Witjes, J. A. & Rovers, M. M. Discrepancies between guidelines and clinical practice regarding prostate-specific antigen testing. Fam. Pract. 30, 648–654 (2013).
Sanderson, M., Fowke, J. H., Lipworth, L., Han, X., Ukoli, F., Coker, A. L. et al. Diabetes and prostate cancer screening in black and white men. Cancer Causes. Control 24, 1893–1899 (2013).
Littlejohns, T. J., Travis, R. C., Key, T. J. & Allen, N. E. Lifestyle factors and prostate-specific antigen (PSA) testing in UK Biobank: implications for epidemiological research. Cancer Epidemiol. 45, 40–46 (2016).
Xie, B., Zhang, G., Wang, X. & Xu, X. Body mass index and incidence of nonaggressive and aggressive prostate cancer: a dose-response meta-analysis of cohort studies. Oncotarget 8, 97584–97592 (2017).
Discacciati, A., Orsini, N. & Wolk, A. Body mass index and incidence of localized and advanced prostate cancer–a dose-response meta-analysis of prospective studies. Ann. Oncol. 23, 1665–1671 (2012).
Garcia-Cruz, E., Piqueras, M., Huguet, J., Perez-Marquez, M., Gosalbez, D., Peri, L. et al. Hypertension, dyslipidemia and overweight are related to lower testosterone levels in a cohort of men undergoing prostate biopsy. Int. J. Impot. Res 24, 110–113 (2012).
Ji, J., Sundquist, J., Sundquist, K. Association of family history of type 2 diabetes with prostate cancer: A National Cohort Study. Front. Oncol. 6, 194 (2016).
Wienbergen, H., Senges, J. & Gitt, A. K. Should we prescribe statin and aspirin for every diabetic patient? Is it time for a polypill? Diabetes Care 31, dc08–s253 (2008).
Lehman, D. M., Lorenzo, C., Hernandez, J. & Wang, C. P. Statin use as a moderator of metformin effect on risk for prostate cancer among type 2 diabetic patients. Diabetes Care 35, 1002–1007 (2012).
Rimm, E. B., Stampfer, M. J., Colditz, G. A., Giovannucci, E. & Willett, W. C. Effectiveness of various mailing strategies among nonrespondents in a prospective cohort study. Am. J. Epidemiol. 131, 1068–1071 (1990).
Acknowledgements
We would like to thank the following state cancer registries for their help: AL, AZ, AR, CA, CO, CT, DE, FL, GA, ID, IL, IN, IA, KY, LA, ME, MD, MA, MI, NE, NH, NJ, NY, NC, ND, OH, OK, OR, PA, RI, SC, TN, TX, VA, WA, WY; We assume full responsibility for analyses and interpretation of these data.
Author information
Authors and Affiliations
Contributions
X.F. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study conception and design: L.M. and E.G. Acquisition of data: L.M., E.G., X.F., M.S., L.L., Y.H., W.M. Analysis and interpretation of data: all authors. Drafting of the paper: X.F. Critical revision of the paper for important intellectual content: all authors. Statistical analysis: X.F., M.S., L.L. Administrative, technical, or material support: none. Supervision: L.M. and E.G.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study protocol was approved by the institutional review boards of the Brigham and Women’s Hospital and Harvard T.H. Chan School of Public Health, and those of participating registries as required.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to confidentiality reasons, but anonymised versions may be available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Funding information
This work was supported by the National Institutes of Health (T32 CA09001 to C.H.P.; R03CA226942 to E.L.G; R00 CA215314 to M.S.) and American Cancer Society (MRSG-17-220-01-NEC to M.S.). The Health Professionals Follow-up Study is supported by U01 CA 167552 from the National Cancer Institute. The TMAs were constructed by the Tissue Microarray Core Facility at the Dana-Farber/Harvard Cancer Center (P30 CA 06516). X.F. was supported by the program of China Scholarships Council (No.201806210455).
Additional information
Note This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution 4.0 International (CC BY 4.0).
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Feng, X., Song, M., Preston, M.A. et al. The association of diabetes with risk of prostate cancer defined by clinical and molecular features. Br J Cancer 123, 657–665 (2020). https://doi.org/10.1038/s41416-020-0910-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-020-0910-y
This article is cited by
-
Understanding Cardiovascular Risk in Prostate Cancer: Role of Disparities, Diabetes, and Aging
Current Treatment Options in Cardiovascular Medicine (2024)
-
Global, regional, and national time trends in cancer mortality attributable to high fasting plasma glucose: an age-period cohort analysis
BMC Public Health (2023)
-
Clinical biomarker-based biological aging and risk of cancer in the UK Biobank
British Journal of Cancer (2023)
-
Relationship between exposure to parabens and benzophenones and prostate cancer risk in the EPIC-Spain cohort
Environmental Science and Pollution Research (2023)
-
Family history, obesity, urological factors and diabetic medications and their associations with risk of prostate cancer diagnosis in a large prospective study
British Journal of Cancer (2022)