Abstract
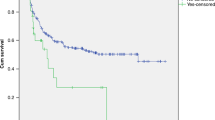
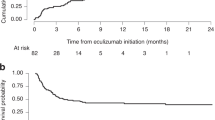
The efficacy and outcome of therapeutic plasma exchange (TPE) for transplant-associated thrombotic microangiopathy (TA-TMA) remain controversial. We therefore sought to evaluate the outcome and efficacy of TPE in patients with TA-TMA and to identify TA-TMA patients who would benefit from TPE management. Eighty-two patients with TA-TMA were treated with TPE. We reported a response rate of 52% and overall survival rates of 20% and 15% at 100 days and 1 year after TA-TMA, respectively, in TPE-treated patients, with a significantly lower survival in gastrointestinal (GI) bleeding patients (5% vs. 41% in non-GI bleeding patients, P = 0.003). Multivariate analysis revealed that patients with GI bleeding, grade III-IV aGVHD, severe anemia, and a lower cumulative volume of TPE were less likely to respond to TPE. GI bleeding, a lower initial volume of TPE, and elevated total bilirubin were independently associated with 100-day mortality. The leading causes of death were infection, active TA-TMA, and MODS. The results of this large cohort of real-world practice indicate that the efficacy and outcome of TPE for TA-TMA patients without GI bleeding are encouraging, and a higher volume of TPE is warranted to achieve favorable outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Laskin BL, Goebel J, Davies SM, Jodele S. Small vessels, big trouble in the kidneys and beyond: hematopoietic stem cell transplantation-associated thrombotic microangiopathy. Blood. 2011;118:1452–62. https://doi.org/10.1182/blood-2011-02-321315.
Jodele S, Davies SM, Lane A, Khoury J, Dandoy C, Goebel J, et al. Diagnostic and risk criteria for HSCT-associated thrombotic microangiopathy: a study in children and young adults. Blood. 2014;124:645–53. https://doi.org/10.1182/blood-2014-03-564997.
Ho VT, Cutler C, Carter S, Martin P, Adams R, Horowitz M, et al. Blood and marrow transplant clinical trials network toxicity committee consensus summary: thrombotic microangiopathy after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2005;11:571–5. https://doi.org/10.1016/j.bbmt.2005.06.001.
Jodele S, Licht C, Goebel J, Dixon BP, Zhang K, Sivakumaran TA, et al. Abnormalities in the alternative pathway of complement in children with hematopoietic stem cell transplant-associated thrombotic microangiopathy. Blood. 2013;122:2003–7. https://doi.org/10.1182/blood-2013-05-501445.
Jodele S, Zhang K, Zou F, Laskin B, Dandoy CE, Myers KC, et al. The genetic fingerprint of susceptibility for transplant-associated thrombotic microangiopathy. Blood. 2016;127:989–96. https://doi.org/10.1182/blood-2015-08-663435.
Ricklin D, Cines DB. TMA: beware of complements. Blood. 2013;122:1997–9. https://doi.org/10.1182/blood-2013-07-512707.
Seaby EG, Gilbert RD. Thrombotic microangiopathy following haematopoietic stem cell transplant. Pediatr Nephrol. 2018;33:1489–1500. https://doi.org/10.1007/s00467-017-3803-4.
Jodele S, Laskin BL, Dandoy CE, Myers KC, El-Bietar J, Davies SM, et al. A new paradigm: diagnosis and management of HSCT-associated thrombotic microangiopathy as multi-system endothelial injury. Blood Rev. 2015;29:191–204. https://doi.org/10.1016/j.blre.2014.11.001.
Jodele S, Laskin BL, Pinkard SL, Carey PM, Goebel J, Davies SM. Does early initiation of therapeutic plasma exchange affect outcome in pediatric stem cell transplant-associated thrombotic microangiopathy? Biol Blood Marrow Transplant. 2011;17:S258.
Kennedy GA, Kearey N, Bleakley S, Butler J, Mudie K, Durrant S. Transplantation-associated thrombotic microangiopathy: effect of concomitant GVHD on efficacy of therapeutic plasma exchange. Bone marrow Transplant. 2010;45:699–704. https://doi.org/10.1038/bmt.2009.233.
Worel N, Greinix HT, Leitner G, Mitterbauer M, Rabitsch W, Rosenmayr A, et al. ABO-incompatible allogeneic hematopoietic stem cell transplantation following reduced-intensity conditioning: close association with transplant-associated microangiopathy. Transfus Apheresis Sci. 2007;36:297–304. https://doi.org/10.1016/j.transci.2007.03.004.
Cho BS, Yahng SA, Lee SE, Eom KS, Kim YJ, Kim HJ, et al. Validation of recently proposed consensus criteria for thrombotic microangiopathy after allogeneic hematopoietic stem-cell transplantation. Transplantation. 2010;90:918–26. https://doi.org/10.1097/TP.0b013e3181f24e8d.
Heybeli C, Sridharan M, Alkhateeb HB, Villasboas Bisneto JC, Buadi FK, Chen D et al. Characteristics of late transplant-associated thrombotic microangiopathy in patients who underwent allogeneic hematopoietic stem cell transplantation. Am J Hematol. 2020. https://doi.org/10.1002/ajh.25922.
Popat U, Mehta RS, Rezvani K, Fox P, Kondo K, Marin D, et al. Enforced fucosylation of cord blood hematopoietic cells accelerates neutrophil and platelet engraftment after transplantation. Blood. 2015;125:2885–92. https://doi.org/10.1182/blood-2015-01-607366.
Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, et al. American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med. 1992;20:864–74.
Kroschinsky F, Weise M, Illmer T, Haenel M, Bornhaeuser M, Hoeffken G, et al. Outcome and prognostic features of intensive care unit treatment in patients with hematological malignancies. Intensive Care Med. 2002;28:1294–1300. https://doi.org/10.1007/s00134-002-1420-5.
Jodele S, Dandoy CE, Lane A, Laskin BL, Teusink-Cross A, Myers KC, et al. Complement blockade for TA-TMA: lessons learned from a large pediatric cohort treated with eculizumab. Blood. 2020;135:1049–57. https://doi.org/10.1182/blood.2019004218.
Vasu S, Wu H, Satoskar A, Puto M, Roddy J, Blum W, et al. Eculizumab therapy in adults with allogeneic hematopoietic cell transplant-associated thrombotic microangiopathy. Bone Marrow Transplant. 2016;51:1241–4. https://doi.org/10.1038/bmt.2016.87.
Konar M, Granoff DM. Eculizumab treatment and impaired opsonophagocytic killing of meningococci by whole blood from immunized adults. Blood. 2017;130:891–9. https://doi.org/10.1182/blood-2017-05-781450.
Peffault de Latour R, Xhaard A, Fremeaux-Bacchi V, Coppo P, Fischer AM, Helley D, et al. Successful use of eculizumab in a patient with post-transplant thrombotic microangiopathy. Br J Haematol. 2013;161:279–80. https://doi.org/10.1111/bjh.12202.
Yang LP, Liu X, Zhang XH. Advances in the diagnosis and management of transplant-associated thrombotic microangiopathy. Zhonghua xueyexue zazhi. 2021;42:693–9. https://doi.org/10.3760/cma.j.issn.0253-2727.2021.08.018.
Jodele S, Laskin BL, Goebel J, Khoury JC, Pinkard SL, Carey PM, et al. Does early initiation of therapeutic plasma exchange improve outcome in pediatric stem cell transplant-associated thrombotic microangiopathy? Transfusion. 2013;53:661–7. https://doi.org/10.1111/j.1537-2995.2012.03776.x.
Mulay S, Kreuter JD, Bryant SC, Elliott MA, Hogan WJ, Winters JL, et al. Outcomes of plasma exchange in patients with transplant-associated thrombotic microangiopathy based on time of presentation since transplant. J Clin Apheresis. 2015;30:147–53. https://doi.org/10.1002/jca.21352.
Uderzo C, Bonanomi S, Busca A, Renoldi M, Ferrari P, Iacobelli M, et al. Risk factors and severe outcome in thrombotic microangiopathy after allogeneic hematopoietic stem cell. Transplant Transplant. 2006;82:638–44. https://doi.org/10.1097/01.tp.0000230373.82376.46.
Zhao P, Wu Y, He Y, Chong S, Qu Q, Deng R et al. A prognostic model (BATAP) with external validation for patients with transplant-associated thrombotic microangiopathy. Blood Adv. 2021. https://doi.org/10.1182/bloodadvances.2021004530.
Schoettler M, Lehmann LE, Margossian S, Lee M, Kean LS, Kao PC, et al. Risk factors for transplant-associated thrombotic microangiopathy and mortality in a pediatric cohort. Blood Adv. 2020;4:2536–47. https://doi.org/10.1182/bloodadvances.2019001242.
Siami K, Kojouri K, Swisher KK, Selby GB, George JN, Laszik ZG. Thrombotic microangiopathy after allogeneic hematopoietic stem cell transplantation: an autopsy study. Transplantation. 2008;85:22–28. https://doi.org/10.1097/01.tp.0000297998.33418.7e.
Elfeky R, Lucchini G, Lum SH, Ottaviano G, Builes N, Nademi Z, et al. New insights into risk factors for transplant-associated thrombotic microangiopathy in pediatric HSCT. Blood Adv. 2020;4:2418–29. https://doi.org/10.1182/bloodadvances.2019001315.
Postalcioglu M, Kim HT, Obut F, Yilmam OA, Yang J, Byun BC, et al. Impact of thrombotic microangiopathy on renal outcomes and survival after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2018;24:2344–53. https://doi.org/10.1016/j.bbmt.2018.05.010.
Martinez MT, Bucher C, Stussi G, Heim D, Buser A, Tsakiris DA, et al. Transplant-associated microangiopathy (TAM) in recipients of allogeneic hematopoietic stem cell transplants. Bone Marrow Transplant. 2005;36:993–1000. https://doi.org/10.1038/sj.bmt.1705160.
Li A, Wu Q, Davis C, Kirtane KS, Pham PD, Sorror ML, et al. Transplant-associated thrombotic microangiopathy is a multifactorial disease unresponsive to immunosuppressant withdrawal. Biol Blood Marrow Transplant. 2019;25:570–6. https://doi.org/10.1016/j.bbmt.2018.10.015.
Matsui H, Arai Y, Imoto H, Mitsuyoshi T, Tamura N, Kondo T, et al. Risk factors and appropriate therapeutic strategies for thrombotic microangiopathy after allogeneic HSCT. Blood Adv. 2020;4:3169–79. https://doi.org/10.1182/bloodadvances.2020002007.
Epperla N, Li A, Logan B, Fretham C, Chhabra S, Aljurf M, et al. Incidence, risk factors for and outcomes of transplant-associated thrombotic microangiopathy. Br J Haematol. 2020;189:1171–81. https://doi.org/10.1111/bjh.16457.
Kraft S, Bollinger N, Bodenmann B, Heim D, Bucher C, Lengerke C, et al. High mortality in hematopoietic stem cell transplant-associated thrombotic microangiopathy with and without concomitant acute graft-versus-host disease. Bone Marrow Transplant. 2019;54:540–8. https://doi.org/10.1038/s41409-018-0293-3.
Biedermann BC. Vascular endothelium and graft-versus-host disease. Best Pract Res Clin Haematol. 2008;21:129–38. https://doi.org/10.1016/j.beha.2008.02.003.
Naymagon S, Naymagon L, Wong S, Ko H, Renteria A, Levine J, et al. Acute graft-versus-host disease of the gut: considerations for the gastroenterologist. Nat Rev Gastroenterol Hepatol. 2017;14:711–26. https://doi.org/10.1038/nrgastro.2017.126
Chinese consensus on the diagnosis and management of transplant-associated thrombotic microangiopathy (2021). Zhonghua xue ye xue za zhi = Zhonghua xueyexue zazhi 2021;42:177–84. https://doi.org/10.3760/cma.j.issn.0253-2727.2021.03.001.
Howard MA, Williams LA, Terrell DR, Duvall D, Vesely SK, George JN. Complications of plasma exchange in patients treated for clinically suspected thrombotic thrombocytopenic purpura-hemolytic uremic syndrome. Transfusion. 2006;46:154–6. https://doi.org/10.1111/j.1537-2995.2006.00687.x.
Batts ED, Lazarus HM. Diagnosis and treatment of transplantation-associated thrombotic microangiopathy: real progress or are we still waiting? Bone Marrow Transplant. 2007;40:709–19. https://doi.org/10.1038/sj.bmt.1705758.
Gavriilaki E, Sakellari I, Anagnostopoulos A, Brodsky RA. Transplant-associated thrombotic microangiopathy: opening Pandora’s box. Bone Marrow Transplant. 2017;52:1355–60. https://doi.org/10.1038/bmt.2017.39.
Acknowledgements
This work was supported by National Key Research and Development Program of China (No. 2017YFA0105503), National Natural Science Foundation of China (Nos. 81970113, 81800116), Key Program of National Natural Science Foundation of China (No. 81730004), Beijing Natural Science Foundation (No. H2018206423), and Beijing Municipal Science and Technology Commission (No. Z171100001017084). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
L-PY and X-HZ contributed to the concept and design. L-PY, PZ, Y-JW, H-XF, YH, X-DM, ML, F-RW, C-HY, and Y-HC collected the data. L-PY, PZ, and Y-JW contributed to the data analysis and interpretation. Y-JC, L-PX, K-YL, X-JH revised the manuscript critically. L-PY wrote the manuscript. X-HZ designed, conceived and supervised the study, analyzed the data, and drafted and revised the manuscript. All authors critically reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study protocol was approved by the central institutional review board of the Peking University People’s Hospital, Beijing, China.
Informed consent
Written informed consent was obtained for each patient in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Yang, LP., Zhao, P., Wu, YJ. et al. Treatment outcome and efficacy of therapeutic plasma exchange for transplant-associated thrombotic microangiopathy in a large real-world cohort study. Bone Marrow Transplant 57, 554–561 (2022). https://doi.org/10.1038/s41409-022-01581-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01581-1
This article is cited by
-
Clinical characteristics of pediatric allogeneic hematopoietic stem cell transplantation-associated thrombotic microangiopathy (TA-TMA): a retrospective single-center analysis
Clinical and Translational Oncology (2023)
-
Comparison of clinical characteristics of first-episode thrombotic thrombocytopenic purpura and TTP-like syndrome: a retrospective cohort study in a level I hematology center in China
Annals of Hematology (2023)